Abstract
Keywords
Introduction
Breast cancer is the most common cancer diagnosed in women worldwide, 1 and also is the most common cancer in Chinese women nowadays; cases in China account for 12.2% of all newly diagnosed breast cancers and 9.6% of all deaths from breast cancer worldwide. 2 According to statistics, 60% to 70% of metastatic breast cancer (MBC) are hormone receptor-positive (HR+) and human epidermal growth factor receptor 2-negative (HER2−).3,4 And these relapsed patients usually have endocrine resistance and visceral metastasis, indicating a poor prognosis. These highlights the need to reform the therapeutical patterns of these patients, thereby enhancing the sensitivity to endocrine therapy (ET) and overcoming endocrine resistance.
Previous standard ET often focused on blocking the estrogen receptor signaling pathway, but researchers gradually shifted their attention to the proliferation process of breast cancer cells. Cyclin-dependent kinase 4/6 (CDK4/6) is a key activation mediator in cancer cell hyperproliferation. It prevents DNA synthesis by hyper-phosphorylating and inactivating retinoblastoma (Rb) by complexing with cyclin D, thereby preventing the cell cycle from G1 to S phase. 5 At present, it is known that the phosphorylation inactivation of the Rb gene is related to endocrine drug resistance. 6 The first CDK4/6 inhibitor approved for marketing in China is palbociclib, which combined with AI as initial ET or fulvestrant following ET has been approved in HR+/HER2− patients with MBC.
Palbociclib combined with ET has shown excellent clinical efficacy and safety in both RCT and the multi-country Ibrance Real World Insights.7–9 However, the application pattern of palbociclib in China is mostly based on the data of the Western population. Chinese breast cancer is different from Western breast cancer by earlier age at onset (60 vs 49), affecting more premenopausal women. 2 Also due to a lack of emphasis on early diagnosis and detection, the majority of breast cancer patients in China are diagnosed with higher stage of disease. 2 Meanwhile, a multicenter study showed that 77.01% of HR+/HER2− Chinese MBC patients received chemotherapy instead of ET in the first-line treatment, while the proportion was only 25% to 56% of MBC in Europe and the United States. 10 This may imply that a larger proportion of pretreatment of palliative chemotherapy in HR+/HER2− Chinese MBC patients than those in the West. PALOMA-4 is currently the largest clinical trial of CDK 4/6 inhibitors of advanced breast cancer (ABC) patients in Asia, but all women in the study were postmenopausal. 11 Therefore, real-world study (RWS) based on Chinese clinical practice is indispensable.
Most of real-world studies on Chinese patients had the disadvantage of small sample size and lack of distinction between types of endocrine drugs (tamoxifen, AI, and fulvestrant) combined with palbociclib,10,12–15 which may influence the interpretation of the results. Aiming to add another single-center experience from the southeastern area of China, herein we did this report to conduct more patterns of clinical efficacy and tolerances of palbociclib from the perspective of breast cancer epidemiology in China.
Material and Method
Patient Eligibility
This retrospective study was conducted in accordance with the Declaration of Helsinki. We have de-identified all patients' details. The reporting of this study conforms to STROBE guidelines. 16 The study was approved by the Ethics Committee of our center (XMFHIIT-2022SL018). All of the patients gave written informed consent before their participation in the study. From February 2019 to December 2021, eligible patients who were treated with palbociclib (specifications: 125, 100, 75 mg) combined with ET in our center were retrospectively analyzed. Inclusion criteria were defined as follows: the primary or metastatic sites of breast cancer were confirmed to be HR+ and HER2− in pathology; all patients had Eastern Cooperative Oncology Group (ECOG) performance status of 0 to 2; all patients received at least 1 cycle of palbociclib, and safety follow-up data were available after every treatment; life expectancy is not less than 12 weeks. HER2 negativity was defined as HER2 immunohistochemistry (invasive carcinoma) interpreted as 0/1+ or 2+ negative in situ hybridization (Dual-probe HER2/CEP17 ratio < 2.0, mean HER2 copy number < 4.0 signals/cell). Luminal A subtype was defined as ER ≥ 1%, PR ≥ 20%, HER2 negative, and Ki-67 ≤ 14%, others are Luminal B subtype. Exclusion criteria: the primary or metastatic sites were confirmed to be HER2 positive or triple-negative; patients who had inadequate efficacy and safety records. We minimized potential selection bias by consecutively choosing patients treated with palbociclib who met inclusion and exclusion criteria in the order of visiting our hospital.
Treatment and Follow-up
All patients were treated with palbociclib combined ET. The ET in this study included AI (including letrozole and exemestane) and selective estrogen receptor degradation (fulvestrant). The initial dosage of palbociclib had 3 options including 125/100/75 mg (a treatment cycle including once daily for 3 weeks, followed by 1 week-off treatment), and the recommended initial dose of palbociclib was 125 mg. All adverse events (AEs), dose reductions and discontinuations were recorded. AEs were assessed according to Common Terminology Criteria for AEs version 5.0 developed by the US-National Cancer Institute. Dosage adjustment or discontinuation was assessed in accordance with the palbociclib capsule instruction, and any reductions in use were fully documented. Baseline characteristics of patients of interest (age, menstrual status, molecular subtypes, previous ET, and metastases in the initial stage of treatment) were retrospectively collected through the hospital's electronic medical record system. Follow-up is achieved through outpatient reviews, inpatient examinations, and patients’ feedback received by phone.
Endpoints
The primary endpoint is progression-free survival (PFS), which was defined as the range from initiation of palbociclib combined with ET to disease progression, toxicity intolerance, or death. Ultrasound, Computerized tomography and other imaging tools were used to evaluate the efficacy of tumor sites every 2 treatment cycles, and the treatment efficacy was in accordance with the Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1. Objective response rate (ORR) = (CR + PR)/number of evaluable cases × 100%; disease control rate (DCR) = (CR + PR + SD)/ number of evaluable cases × 100%.
Statistical Analyses
The count data were described by frequency, and the chi-square test was used to compare the distribution differences among groups to be compared. Survival was estimated by the Kaplan–Meier method, and any differences in survival were evaluated with a stratified log-rank test. The Cox proportional-hazards model was used to estimate the simultaneous effects of prognostic factors on survival. The IBM SPSS Statistics 25.0 statistical software package was used for all calculations. Differences were considered to be statistically significant when the P-value was .05 or less. All statistical tests were two-sided.
Results
Patient Characteristics and Treatment Patterns
A total of 101 patients received treatment of palbociclib, of which 6 patients did not meet the inclusion criteria and were excluded. Finally, a total of 95 women patients were assigned for this analysis (Figure 1). The median follow-up period was 15 months (range, 2-37). The baseline characteristics of the included patients can be seen in Table 1. The mean age of the total population was 56.3 years (32-86 years). The 68 cases (71.6%) were postmenopause, including 62 cases (65.3%) were natural menopause and 6 patients (6.3%) who underwent bilateral salphingo-oophorectomy (BSO) for artificial menopause before starting palbociclib. Thirty-one (32.6%) patients were initially diagnosed at stage IV. There were 35 patients (36.8%) with bone metastases only, and 57 patients (60.0%) with visceral metastases.
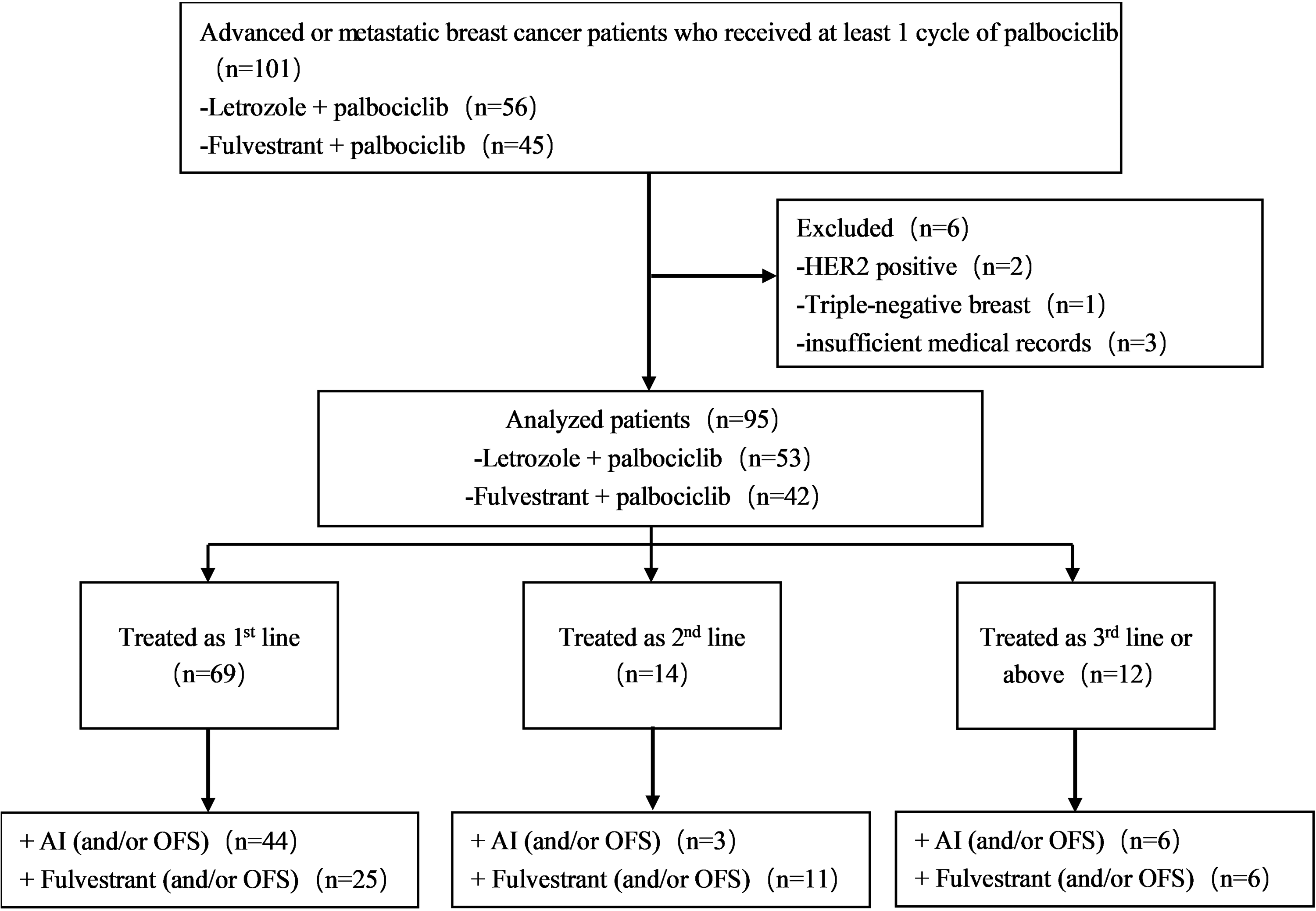
Flowchart of the treatment pattern of Palbociclib plus endocrine therapy of 95 patients with HR+/HER2− ABC/MBC. Abbreviations: ABC, advanced breast cancer; OFS, ovarian function suppression; HR+, hormone receptor-positive; HER2−, human epidermal growth factor receptor 2 negative; MBC, metastatic breast cancer.
Patient Characteristics at Palbociclib Initiation in Different ET Drug Cohort Settings.

Values are presented as numbers (%). Abbreviations: BSO, bilateral salphingo-oophorectomy; NA, not available; ET, endocrine therapy.
In total, 53 patients were treated with palbociclib combined with letrozole or exemestane and 6 of them were treated with drugs-induced ovarian function suppressing (hereinafter referred to as OFS) simultaneously. In total, 42 patients were treated with albociclib combined with fulvestrant, of which 5 cases were combined with OFS. The treatment line was defined as the application line of albociclib for the first time after diagnosis as MBC or initially diagnosed stage IV disease. Until February 2022, 50 patients were still under the administration of albociclib, including 31 patients combined with AI and 19 patients combined with fulvestrant.
Efficacy
In total population, the ORR of patients receiving albociclib in the first-, second-, and later-line settings were 36.2%, 7.1% and 0 (sample size limitation), respectively. And DCR was 88.4%, 85.7%, and 66.7%, respectively (see Figure 2A). Meanwhile, the median PFS (mPFS) of the three cohorts above was 34.0 months, not available (NA) and 5.0 months, respectively. Patients who received first- and second-line therapy benefited more than patients who entered treatment procedure later (P = .002) (see Figure 2B).

Response to palbociclib combined with endocrine therapy of 95 patients with HR+/HER2- ABC/MBC patients(A); the relationship between the number of lines of palbociclib plus endocrine therapy and PFS of HR+/HER2− ABC patients(B). Abbreviations: HR, hazard ratio; HR+, hormone receptor-positive; HER2−, human epidermal growth factor receptor 2 negative; mPFS, median progression-free survival; ABC, advanced breast cancer; MBC, metastatic breast cancer; CI, confidence interval; mPFS, median progression-free survival.
The subgroup analyses based on the whole patients can be seen in Figure 4A for details. Patients who didn’t have the previous history of salvage chemotherapy howed better mPFS compared to patients who were pretreated in advanced stage (hazard ratio [HR] = 2.383; 95% confidence interval [CI] = 1.275-4.454; P = .007). To analyze the influence of distant metastasis, patients with bone metastasis only had a better mPFS performance than patients who had visceral metastasis (HR = 2.470; 95% CI = 1.202-5.075; P = .014). Liver metastasis is a significant predictor of poor mPFS (HR = 2.867; 95%CI = 1.549-5.307; P < .001). It is worth noting that when patients with liver metastases received the administration of palbociclib in the first line, the mPFS can still reach 13 months (95%CI = 11.49-56.51). However, when the application of palbociclib entered the second and later lines in patients with liver metastasis, the efficacy was significantly reduced, whose mPFS only reached 5.5 months (see Figure 3); The relative K-M survival curves are presented in Supplemental Files in Figure 1A to D.

Kaplan–Meier curves of patients with liver metastases administrated with palbociclib plus endocrine therapy in total population by lines.

Forest plot of subgroup analysis. Palbociclib plus endocrine therapy in total population (A); AI plus palbociclib (B); fulvestrant plus palbociclib (C).
The subgroup analyses based on the patients received palbociclib plus AI can be seen in Figure 4B for details. The mPFS of palbociclib combined with AI was 34 months (95%CI = 6.87-61.13) (Supplement file: Figure 2A). No prior palliative chemotherapy was associated with better performance of mPFS (HR = 2.946; 95%CI = 1.235-7.027; P = .015). And the presence of liver metastases still significantly associated with the progression of the disease (HR = 4.301; 95%CI = 1.811-10.212; P < .001). It can be seen that liver metastasis is an important reference factor for poor prognosis. The relative K-M survival curves are presented in Supplemental Files in Figure 3A and B.
The subgroup analyses based on the patients received palbociclib plus fulvestrant can be seen in Figure 4C for details. The mPFS of palbociclib plus fulvestrant was 12 months (95%CI = 7.76-16.24) (Supplement file: Figure 2B). The mPFS benefit of luminal A exhibited better mPFS relative to the luminal B (HR = 2.602; 95%CI = 1.032-6.561; P = .043) (Supplement file: Figure 3C).
Effects of Previous Endocrine Therapy
Fourty-nine patients (40%) developed metastatic recurrence during or after the postoperative adjuvant ET. The ABC5 guideline 17 categorizes endocrine resistance as primary or secondary resistance. Most patients (24.2%) showed secondary endocrine resistance in this study, and there was no difference in mPFS according to the presence of endocrine sensitivity in the cohort treated palbociclib plus AI (log-rank P = 0.133) (see Figure 4A).
In the cohort of palbociclib combined with fulvestrant, patients who were classified as primary endocrine resistance showed no statistical differences in mPFS compared to secondary endocrine-resistant patients during analysis (mPFS reached 12 months in both groups) (see Figure 4B), which improved significantly compared to PALOMA-3 (primary resistance: 7.4 months; secondary resistance: 10.2 months). There was no difference in mPFS between the two cohorts with secondary endocrine resistance. The results of the subgroup analysis combined with fulvestrant are different from those of PALOMA-3, which may be related to larger proportion of first-line treatment of patients enrolled in the cohort of palbociclib combined with fulvestrant, but this result should be interpreted cautiously due to the small sample size, with the possibility of selection bias.

Kaplan–Meier curves of progression-free survival (PFS) of subgroup analysis according to the presence of endocrine resistance in AI (A) and primary or secondary endocrine resistance in fulvestrant plus palbociclib group (B).
Tolerance and Dose Reduction
Among the 95 patients, 78 (82.1%) patients took 125 mg/d as the initial dose. In total, 16 patients and 1 patient started at 100 mg/d and 75 mg/d, respectively. The proportion of patients who experienced any AEs during the treatment of palbociclib plus ET was shown in Table 2. The most common hematological AE was neutropenia occurred in 19 patients (20%), including 12 patients (12.6%) with grade 3 or higher. Thrombocytopenia occurred in 3 cases (3.2%). Other nonhematological toxicities included fatigue in 38 cases (40%), nausea (7.4%), rash (6.3%), mouth ulcer (4.2%), insomnia (4.2%), etc. In our study, 2 patients were permanently discontinued due to fourth-grade neutropenia and 3-grade fatigue, respectively. Among all cases, 7 (7.4%) patients discontinued medication for economic reasons.
Adverse Events in the Population Treated With Palbociclib Plus Endocrine Therapy.

*Pain includes low back pain and headache. Value n(%) represents its proportion of the total population.
Adverse events in the population treated with palbociclib plus endocrine therapy n(%)].
Furthermore, 6 patients reduced the dose of palbociclib from 125 to 100 mg due to intolerable AEs, including 3 elderly patients (≥age 65) and 3 patients with visceral metastasis. There was no mPFS difference according to dosage reduction of palbociclib (P = .361). However, the comparison should be interpreted with caution because the number of drugs-reducted patients in this study was only 6 cases.
Discussion
The clinical trials are often based on the preset inclusion criteria and strictly controlled environments, so it may be controversial to directly extend the results of clinical trials to the real world. The current strategy for HR+/HER2− MBC patients have transitioned from single ET to endocrine targeted therapy.18,19 Palbociclib is the longest-listed CDK4/6 inhibitor in China, however, reports of its first-line application after diagnosis of stage IV disease are still lacking. Since the epidemiology of Chinese breast cancer patients is different from those in Western, the indications obtained from PALOMA-2/3 for reference may deviate from the real-world clinical practice in China. To the best of our knowledge, our study is one of the few studies hitherto that has further distinguished the type of concomitant endocrine drugs in Chinese MBC patients. We aimed to provide more first-line application experience of palbociclib in China to select suitable patients.
In our study, the mean age of patients treated with AI plus palbociclib was 55.3 years, younger than PALOMA-2. And about 30.2% of patients were premenopausal before starting it. These proportions are similar to the previous epidemiological reports in Chinese and western patients. 20 According to two multicenter studies from China and South Korea, more than half of Asian female breast cancer patients with HR+/HER2 have been treated with palliative chemotherapy before receiving palbociclib, 21 even 77.01% of HR+/HER2 Chinese MBC patients received chemotherapy in first-line treatment. 10 This means that a large proportion of Asian patients may have been pretreated with palliative chemotherapy before receiving palbociclib. Despite existing these unsatisfactory baseline characteristics, the mPFS of patients treated with AI plus palbociclib in our analysis was favorable to PALOMA-2. In addition to the small sample size, the proportion of patients with bone-only metastasis in this study was higher than that of PALOMA-2 (39.6% vs 23.2%), and less of patients with multiple metastases in our study. Meanwhile, the ORR of the first-line application of palbociclib in this study is slightly lower than that of PALOMA-2 (36.2% vs43%), but clinical benefit rate is better (88.4% vs81%). Our efficacy is also slightly better than that of other single-center retrospective studies in China .12,15–22
Likewise, the mean age of patients treated with fulvestrant plus palbociclib was 57.7 years, similar to PALOMA-3. However, patients in our study received palbociclib plus fulvestrant earlier than patients in PALOMA-3, and the proportion of patients with previous histories of salvage chemotherapy was significantly lower than that of PALOMA-3 (23.8% vs 37%). Therefore, although we had to consider the bias from the small sample size, the high proportion of liver metastases (66.7%) didn’t seem to affect the better performance of mPFS compared to PALOMA-3. It can be seen that visceral metastasis should not be an obstacle to the application of palbociclib in clinical practice. For HR+/HER2− MBC patients with visceral metastasis, palbociclib should be considered as soon as possible, rather than salvage chemotherapy, to maximize PFS improvement and ensure the quality of patients’ life.
Compared with young patients, elderly patients tend to consider that health-related quality of life is more important than survival improvement. 23 There was no mPFS difference between people aged ≥ 65 and those < 65. The rate of neutropenia events of grade 3 or above was 24.2% (8 of 33), which was much lower than that of PALOMA-2 (66.5%) and PALOMA-3 (65%). This may be related to the regular monitoring of the patients and accurate administration of preventive treatment of leukopenia. However, there was still a 75-year-old patient who discontinued permanently because of intolerable AE (4-degree neutropenia), which suggests that we still need to closely monitor the application of palbociclib in elderly patients.
In real-world practice, due to the lack of evidence on premenopausal Asian women and the inaccessibility of CDK4/6 inhibitors, the vast majority of Chinese premenopausal MBC patients first received palliative chemotherapy, resulting the late application of CDK4/6 inhibitors. The strategy of Asian HR+/HER2− premenopausal MBC is usually inferred from postmenopausal patients’ data. MONALEESA-7 24 is the first large phase III randomized clinical trial (RCT) dedicated to premenopausal HR+/HER2− ABC women, which included 30% of the Asian population. It is worth noting that among the concurrent phase III RCTs of CDK4/6 inhibitors, the MONALEESA-7 study was the first trial to obtain overall survival (OS) prolongation, which confirmed that premenopausal HR + ABC patients are worthy of further differentiation. Furthermore, it may be suggested that premenopausal women benefit more from it than postmenopausal patients. In our study, more than 85% (23 of 27) of premenopausal patients received palbociclib combined with ET as first-line ET. No mPFS difference was identified between the two menstrual status of the patients (NA vs18mo), and also there was no significant difference in mPFS between patients using OFS and patients with BSO (log-rank P = .101), which was consistent with an RWS in Korea. 21 Therefore, we cautiously believe that premenopausal and postmenopausal patients can be treated equally in the choice of CDK4/6 inhibitors. The addition of drugs-induced OFS will not affect the benefits of the Chinese population.
In the guidelines for diagnosis and treatment of breast cancer of the Chinese Society of Clinical Oncology, experts generally agree that PR is an important prognostic indicator of breast cancer, and it is recommended that PR > 20% be used as the cut-off value for subtype luminal A and luminal B. According to this classification, the mPFS of luminal A is better than that of luminal B in our cohort of palbociclib plus fulvestrant, which is consistent with the result of PALOMA-3. 25 Some studies have found that the expression level of PR cannot be used as a biomarker to predict the efficacy of palbociclib, but when it was cross-analyzed with ki-67, high ki-67 combined with low PR expression seem to have a negative effect on mPFS in HR+/HER2− MBC patients treated with palbociclib. 26 Therefore, more studies are needed to fully understand the variable benefits of palbociclib on HR+ breast cancer patients in different molecular subtypes.
Based on the data from PALOMA-2/3, HR+/HER2− MBC patients showed a trend of full-line benefit treated with CDK4/6 inhibitor combined with AI/fulvestrant, regardless of the presence of endocrine resistance. In our study, there was no difference in mPFS according to the presence of endocrine sensitivity in the cohort treated AI plus palbociclib. Interestingly, in the cohort of palbociclib combined with fulvestrant, the mPFS of both groups with primary and secondary endocrine resistance reached 12 months, better than that of PALOMA-3 (primary endocrine resistance: 7.4 months and secondary endocrine resistance: 10.2 months). The causes for the differences are analyzed as follows. Among the patients with primary endocrine resistance treated with palbociclib combined with fulvestrant, the proportion of no previous history of palliative chemotherapy in the advanced stage was higher than that of PALOMA-3 (90% vs 66%). Meanwhile, the proportion of our patients who received palbociclib + fulvestrant without prior palliative therapy reached 60% (6 of 10), while 78.7% (273 of 347) of MBC patients in PALOMA-3 had received other advanced treatments (palliative chemotherapy or ET) before palbociclib combined with fulvestrant, so the patients with primary endocrine resistance in our study received earlier administration of palbociclib compared to PALOMA-3. Similar results can be seen in the subgroup analysis of patients with primary endocrine resistance in the MONARCH-2 study. 27 The MONARCH-2 study only included patients who progressed after first-line ET in the stage of advanced disease. And all patients were not allowed to have a previous history of salvage chemotherapy. Although the sample size of our study is limited, this result still suggested that we should pay attention to the multidrug resistance (MDR) that may evolve from primary endocrine resistance in later-line therapy. The earlier application of CDK4/6 inhibitors may further benefit the OS of people with primary endocrine resistance.
Liver metastasis is one of the most common patterns of metastasis in ABC. The proportion of ABC patients with liver metastasis is about 30.1%, which is often one of the predictive factors of poor prognosis. 28 In the PALOMA-2 study, the subgroup of liver metastases showed that the application of palbociclib decreased the risk of disease progression by 38%, and the mPFS of the liver metastases subgroup was 13.7 months. Among the first-line ET patients included in our study, mPFS of the liver metastasis subgroup of palbociclib combined with AI was 10 months, and that in subgroup which combined with fulvestrant was 13 months. However, after entering the second-and-later lines of palbociclib treatment, the mPFS of liver metastasis subgroups was significantly shortened. Comparing the patients’ characteristics in different therapeutic lines, only 6.3% (1 of 16) patients who received first-line palbociclib had the previous history of salvage chemotherapy, while the later-line patients with the history of salvage chemotherapy accounted for 69.2% (9 of 13) after the occurrence of liver metastasis. The history of salvage chemotherapy was associated with the benefit of palbociclib in different lines. Previous Young-pearl studies 29 have confirmed that for premenopausal HR+/HER2− ABC patients, palbociclib combined with ET showed better PFS benefit compared with chemotherapy and has better tolerance, so these patients should receive palbociclib treatment as soon as possible, which can not only maximize PFS improvement, but also ensure the quality of life.
There was no survival benefit according to bone-only disease in AI/fulvestrant plus palbociclib treated patients. However, in the total population with palbociclib treated, bone-only disease patients showed trends for superior mPFS, but this result should be interpreted with caution due to the small number of patient population.
Palbociclib combination therapy was well tolerated in our patients, consistent with other real-world studies in China.14–22 The most common AEs were neutron penia (19.8%) and fatigue (40%). A lower percentage of our patients receiving palbociclib combination therapy experienced grade ≥ 3 neutropenia but a higher percentage of fatigue compared with the PALOMA-2 or PALOMA-3, which may because some patients in our study were treated with dosage reduction at the initial usage. Due to intolerable AEs, 6 patients reduced the dosage of palbociclib from 125 to 100 mg. The population of dose adjustment was mainly over 65 years old and multiple visceral metastases were involved. Compared with the unadjusted patients, there was no significant difference in mPFS between the two cohorts, which was consistent with the Asian subgroups of PALOMA-2 and 3.30,31
However, there was still a patient who permanently discontinued medication due to fourth-degree neutropenia. The patient was a 75-year-old woman with multiple lung metastases who started palbociclib at a dose of 100 mg/d. Fourth-grade neutropenia occurred after the first cycle of palbociclib. With the symptomatic treatment, the patient's leukocyte level returned to normal, but the patient refused to continue the next cycle of palbociclib combination therapy. The other patient in the palbociclib plus fulvestrant cohort was permanently discontinued due to third-grade fatigue. Overall, in this study, the proportion of SAE-induced discontinuation was relatively low (2%), while 7 patients (7.4%) discontinued due to economic reasons. Therefore, aside from progressive disease, the economy is still the leading cause of the interruption of palbociclib treatment in the RWS of China.
There are some limitations in this study. Firstly, this study is a single-center study. In addition, the researchers retrospectively collected clinical baseline data of patients through electronic medical record systems, and supplement data were acquired by contacting patients by telephone, which may lead to recall bias. In the future, we will continue to expand the sample size and track the subsequent therapy of these patients.
Conclusion
In conclusion, our findings further confirmed the effective pattern and acceptable toxicity of palbociclib in Chinese MBC patients. Although there is no biomarker to assist clinicians in choosing the applicable patients of palbociclib, we can see the overall benefit of palbociclib combined with ET based on predesigned prognostic factors. Especially, the early application of palbociclib in patients with primary endocrine resistance and liver metastases has further enhanced patients’ mPFS. Therefore, we must consider the above-mentioned baseline factors related to disease treatment and prognosis in clinical practice, and continue to explore effective biomarkers to jointly formulate better treatment strategies.
Supplemental Material
sj-docx-1-tct-10.1177_15330338221132926 - Supplemental material for Early Application of Palbociclib Plus Endocrine Therapy in HR+/HER2− Metastatic Breast Cancer: A Better Choice Based on Data From the Chinese Population
Supplemental material, sj-docx-1-tct-10.1177_15330338221132926 for Early Application of Palbociclib Plus Endocrine Therapy in HR+/HER2− Metastatic Breast Cancer: A Better Choice Based on Data From the Chinese Population by Yusi Zhang, Wenlin Chen, Shuanglong Chen, Qingmo Yang and Zhong Ouyang in Technology in Cancer Research & Treatment
Footnotes
Abbreviations
Acknowledgements
Thanks to all authors for their joint efforts in the completion of the manuscript. And we would like to thank all editors and reviewers for their useful feedback that improved this paper.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics Statement
The study was approved by the Ethics Committee of the First Affiliated Hospital of Xiamen University (receipt number: XMFHIIT-2022SL018).
Informed Consent
All of the patients gave written informed consent before their participation in the study.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
