Abstract
Objective
This real-world study aimed to investigate the efficacy and safety of palbociclib plus endocrine therapy in patients with hormone receptor-positive/human epidermal growth factor receptor 2-negative metastatic breast cancer in the real world in a Chinese population.
Methods
The clinical data of consecutively enrolled patients from the Cancer Hospital Chinese Academy of Medical Sciences, Shenzhen Center, and the University of Hong Kong - Shenzhen Hospital were collected. Progression-free survival curves were generated using log-rank tests with the Kaplan-Meier method. Univariate and multivariate logistic regression analyses were performed to identify the factors affecting progression-free survival.
Results
In total, 118 patients were enrolled, including 6 patients with brain metastases. At the last follow-up date, the median progression-free survival was 16.8 months (95% confidence interval, 11.1-22.5), with the 6-month and 12-month progression-free survival rates of 77.1% and 57.6%, respectively. The disease control rate and the intracranial disease control rate were 82.2% and 50%, respectively. A longer progression-free survival was observed for patients with the following characteristics: treatment-naive; without hepatic metastasis; sensitive to previous endocrine therapy and harboring fewer metastatic sites. The multivariate logistic regression analysis demonstrated that treatment lines and exposure to palliative chemotherapy were independent influencing factors of progression-free survival.
Conclusions
Palbociclib plus endocrine therapy in patients with hormone receptor-positive/human epidermal growth factor receptor 2-negative metastatic breast cancer was effective and well-tolerated, even in patients with brain metastases. More benefits were observed in frontline therapy, chemotherapy-naive, and endocrine therapy–sensitive patients with fewer metastatic sites.
Introduction
In recent years, breast cancer has replaced lung cancer as the most common cancer in women. 1 According to the latest data published by the World Health Organization International Agency for Research on Cancer, published in 2020, there were up to 2.26 million new cases of breast cancer worldwide. 2 Metastatic breast cancer (MBC) is an incurable disease with a 5-year survival rate of nearly 25% and a mean overall survival (OS) of only 2 to 3 years. 3 In approximately 60% to 65% of breast cancer patients, tumor cells are positive for a hormone receptor (HR), 4 suggesting that HR signaling pathways play an important role in the pathogenesis of breast cancer. Therefore, HR signaling pathways have become important targets in the treatment of HR+ breast cancer. Although endocrine therapy (ET) is an effective treatment for HR+ breast cancer, most patients with advanced disease will eventually develop resistance to individual ETs.
In the process of carcinogenesis, the cyclin D/CDK4/6/p16INK4a/retinoblastoma (Rb) protein pathway is frequently disrupted. Clinical studies have shown that cyclin D is overexpressed in more than 50% of breast cancers, most of which are also positive for estrogen receptor (ER). 5 The activated cyclin D-CDK4/6 complex phosphorylates Rb-associated protein, which depolymerizes the transcriptional repressor complex Rb-E2F and releases E2F transcription factors. Then, free E2F activates cells into S phase, and cells begin DNA replication. 6 Thus, CDK4 and CDK6 are representative targets for treatment, especially for ER+ patients. To date, 4 kinds of CDK 4/6 inhibitors have been studied for the treatment of ER+ breast cancer, including palbociclib, ribociclib, abemaciclib, and dalpiciclib, while palbociclib is the first one marketed worldwide and in China. The PALOMA clinical studies clinical practice showed that palbociclib combined with ET could significantly improve the survival of patients with ER+/human epidermal growth factor receptor 2 (HER2)- MBC.7–10 Our real-world study aimed to investigate the efficacy and safety of palbociclib-ET in Chinese patients with ER+/HER2- MBC.
Materials and Methods
Case Screening
The clinical data of consecutive patients eligible for inclusion were retrospectively collected at the Cancer Hospital Chinese Academy of Medical Sciences, Shenzhen Center (between January 2018 and November 2021) and the University of Hong Kong-Shenzhen Hospital (between January 2017 and August 2021). Palbociclib was administered orally at 125 mg/d in 3 weeks of treatment followed by 1-week off, for every 28 days. Concurrently, patients received an aromatase inhibitor (AI) or fulvestrant. The inclusion criteria were as follows: (1) pathologically confirmed invasive breast cancer; (2) the presence of unresectable local recurring lesions or metastatic lesions; (3) with HR+/HER2- primary/metastatic sites; (4) a performance score of 0 to 2 according to the Eastern Cooperative Oncology Group scoring criteria; (5) the reception of at least one cycle (21 days) of palbociclib combined with ET; and (6) life expectancy not less than 12 weeks. The exclusion criteria were as follows: (1) no follow-up data after medication administration; (2) a diagnosis of a secondary tumor; and (3) the reception of less than one cycle of medication. The screening of the patient eligibility is shown in Figure 1. All patient details had been de-identified. The reporting of this study conforms to STROBE guidelines. 11

The flow diagram of screening for patient eligibility. Abbreviations: HR, hormone receptor; HER-2, human epidermal growth factor receptor 2.
Efficacy and Toxicity Assessment
Imaging, including computed tomography scans, magnetic resonance imaging, or bone scans, was performed every 2 months during treatment until disease progression or intolerable toxicity. The efficacy was evaluated in accordance with the Response Evaluation Criteria in Solid Tumors version 1.1, while the adverse events (AEs) were recorded and graded according to the Common Terminology Criteria for Adverse Events, version 5.0.
Statistical Analysis
Progression-free survival (PFS) was defined as the time from palbociclib administration to progressive disease or death, whichever occurred first. Median PFS was generated using the Kaplan-Meier method. Both univariate and multivariate analyses were performed using the Cox regression model. Significance was defined as a P value of <.05. Parameters with P values <.05 in the univariate analysis were included in the multivariate analysis. All statistical analyses were performed using IBM SPSS version 18.0.
Results
Clinical Characteristics
In total, 118 patients were eligible for the efficacy analysis (excluded patients: 2 patients with HER-2 conversion, 6 patients who received less than one cycle of medication, one patient with a secondary tumor, and 4 patients who were lost to follow-up). Of these 118 patients, 103 were analyzed for AEs (15 patients with no in-hospital hematological assessment records were excluded).
The clinical and demographic characteristics of the enrolled patients are detailed in Table 1. In this study, all patients were female with a median patient age of 49.5 years (29-88). Premenopausal women accounted for 56.8% (n = 67) of the study sample, and they received drug-induced ovarian suppression therapy during treatment. Among all the patients, 47.5% (n = 56) received palbociclib combined with an AI, while 52.5% (n = 62) received palbociclib combined with fulvestrant. Sixty-eight patients (57.6%) received palbociclib in the first-line setting, 29.7% (n = 35) in the second-line setting, and 12.7% (n = 15) in subsequent lines. Sixty-one patients (51.7%) had previously received palliative chemotherapy. In total, 62 patients (52.5%) were diagnosed with visceral metastasis, and 43.5% (n = 27) had hepatic metastasis.
Patient Demographic and Clinical Characteristics.

Abbreviations: AI, aromatase inhibitor; ET, endocrine therapy.
Efficacy
At the time of the last follow-up (April 25, 2023), the median PFS was 16.8 months (95% confidence interval [CI] 11.1-22.5), and 39 patients were still on medication (disease progression was not reached). The PFS rate at 6 months was 77.1%, and 57.6% of patients remained progression-free at 12 months. Overall, the disease control rate (DCR) was 82.2%. The median PFS of first-line and subsequent-line treatment was 30.3 months (95% CI, 21.4-39.2) and 7.6 months (95% CI, 6.7-8.3), respectively (Figure 2B). The univariate analysis showed that a longer PFS was observed for patients in the first-line setting, those who were chemotherapy-naive, sensitive to previous ET, without hepatic metastasis and had fewer than 3 metastatic sites (Table 2 and Figure 2B, 2E, 2F, 2H and 2I). The multivariate analysis showed that treatment lines and exposure to palliative chemotherapy were independent factors affecting PFS (Table 3). For 6 patients with brain metastasis (BM), the intracranial DCR (iDCR) was 50%, and all of them received first-line palbociclib-ET therapy (details are shown in Table 4). The intracranial lesion of one patient decreased in size from 2.1 cm to 1.4 cm after 16 months of medication, and the partial response of this lesion persisted despite extracranial progressive disease (intracranial magnetic resonance imaging is shown in Figure 3).

Progression-free survival of patients receiving palbociclib plus endocrine therapy stratified by patient characteristics. Abbreviations: AI, aromatase inhibitor; Ful, Fulvestrant; ET, endocrine therapy.

Brain MRI showing the largest lesion in one patient with a partial response: at baseline (A), 2 months (B), 5 months (C), 8 months (D), 12 months (E), and 16 months (F) after the initiation of palbociclib-ET therapy.
Univariate Analysis of PFS Stratified by Patient Characteristics.

Abbreviations: PFS, progression-free survival; ET, endocrine therapy; AI, aromatase inhibitor; HR, hazard ratio.
Multivariate Analysis of PFS Stratified by Patient Characteristics.
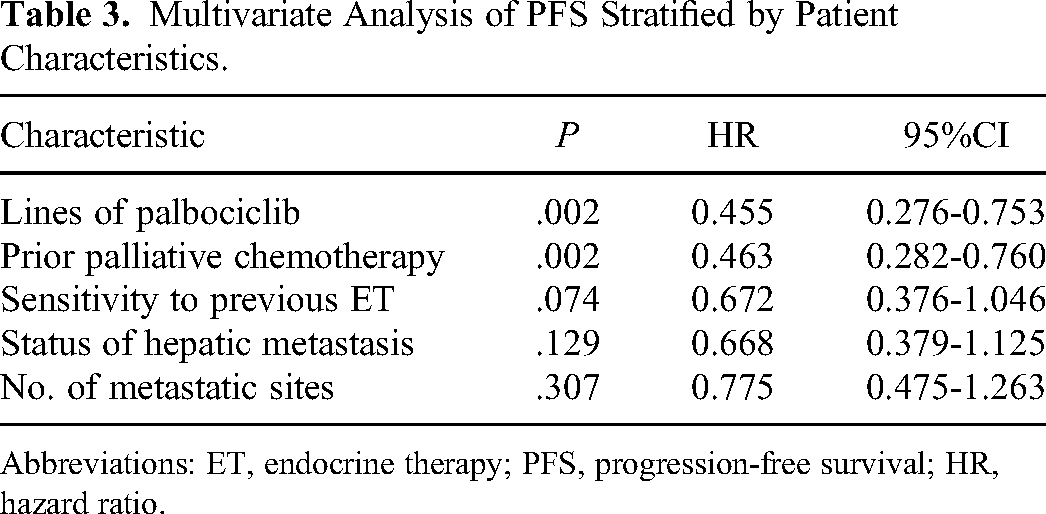
Abbreviations: ET, endocrine therapy; PFS, progression-free survival; HR, hazard ratio.
Characteristics of Patients With Brain Metastases.

Abbreviations: iPFS, intracranial progression-free survival; SBRT, stereotactic body radiation therapy; WBRT, whole brain radiation therapy; AI, aromatase inhibitor; PD, progressive disease; SD, stable disease; PR, partial response; NR, not reached.
Toxicity
Among the 103 patients with AE evaluation records, 86.4% of patients (n = 89) developed AEs of varying degrees (shown in Table 5). Neutropenia was the most common AE, with an overall incidence of 82.5% (n = 85) and a grade 3/4 incidence of 63.1% (n = 65). Grade 3/4 thrombocytopenia and anemia accounted for 10.7% (n = 11) and 4.9% (n = 5) of AEs, respectively. Other AEs, such as fatigue, hot flushes, decreased appetite, diarrhea, and stomatitis, were rarely observed and most of them presented as grade 1/2. In 4 patients, the medication dose was reduced due to neutropenia (a reduction to 100 mg/d in 3 patients and to 75 mg/d in one patient; one patient developed febrile neutropenia). One patient initiated treatment at a dose of 100 mg/d, while one patient discontinued medication due to intolerable AEs.
Adverse Events of Palbociclib-endocrine therapy.

Discussion
The PALOMA studies showed that palbociclib-ET provides favorable benefits in HR+/HER2- MBC patients. In the PALOMA-2 study, the median PFS was 24.8 months in the palbociclib–letrozole group versus 14.5 months in the letrozole group in the first-line setting (P < .001), with a 66.4% incidence of grade 3/4 neutropenia; among patients with grade 3/4 neutropenia, the proportion of Asian patients was significantly higher than that of non-Asian patients (95.4% vs 76.8%). 8 The PALOMA-3 study in patients who had progressed on previous ET showed that the median PFS was 9.5 months in the palbociclib–fulvestrant group versus 4.6 months in the fulvestrant-placebo group (P < .0001), with 73% of patients developing grade 3/4 neutropenia. 9 In our study, palbociclib combined with an AI/fulvestrant showed favorable efficacy and good tolerance in patients with HR+/HER2- MBC, although up to 42.4% of the patients received palbociclib in subsequent-line settings. Moreover, the proportion of premenopausal patients was much higher than that in the PALOMA-2 and PALOMA-3 trials. The incidence of grade 3/4 neutropenia was 63.1% in our study, which was generally consistent with the results of the PALOMA-2 and PALOMA-3 trials.
Although high-quality clinical trials have provided convincing evidence for the efficacy/safety of the combination of palbociclib and ET in patients with HR+/HER2- MBC, real-world studies are still needed to validate these findings in unselected patients. The first real-world study with a large sample size (n = 1430) worldwide from the US Flatiron Database showed that first-line palbociclib plus letrozole could significantly prolong the median PFS and OS compared with letrozole alone in patients with HR+/HER2- MBC. 10 To our knowledge, there have been only a few real-world studies on CDK4/6 inhibitors in the Chinese population reported previously. A retrospective study by Professor Hu et al showed that the median PFS of palbociclib-ET was 9.2 months in 130 patients, and there was no significant difference between chemotherapy and continuous ET beyond progression on palbociclib. 12 Subsequently, the findings of another real-world study were reported by Zhang et al in 2021 and showed that the median PFS was 19.8 months in the first-line setting (n = 88), which was inferior to that observed in our study. 13
Breast cancer is the second most common solid cancer that metastasizes to the brain, and 15% to 30% of patients with MBC will eventually develop BMs. 14 Patients with BMs have a much poorer prognosis and the median OS was reported to be only 8.7 months. 15 The phase III PALOMA 1, 2, and 3 trials excluded patients with BMs. The retrospective observational analysis from the Flatiron Health Analysis Database mentioned above showed a significant benefit in terms of the real-world PFS and OS in patients with BMs and in patients overall in the palbociclib–letrozole cohort, 10 indicating potential intracranial activity of palbociclib. A phase II study of another CDK4/6 inhibitor, abemaciclib, in MBC patients with BMs showed that 3 patients out of 58 in cohort A (HR+/HER2- group) were confirmed responders, with an intracranial objective response rate of 5.2%, an intracranial clinical benefit rate of 24%, and a median OS of 12.5 months (95% CI, 9.3-16.4). 16 In another basket trial, palbociclib demonstrated a 53.3% intracranial benefit rate in patients with progressive BMs harboring CDK pathway alterations. 17 Michaud et al found that palbociclib could efficiently cross the blood–brain barrier and had high activity in suppressing the growth of glioblastoma multiforme intracranial xenografts. 18 However, another study conducted in a murine model showed that the blood–brain barrier efflux pump might decrease the brain penetration of palbociclib. 15 In our study, 6 patients with BMs were enrolled. The overall iDCR was 50%, and the responders showed a relatively long intracranial PFS. For these patients, it is possible that their blood–brain barrier had been disrupted by their breast cancer BMs or previous treatments; therefore, the brain distribution and hence the efficacy of palbociclib would be better in these patients. This is, to our knowledge, the first real-world study to reveal intracranial responses to palbociclib in a Chinese population.
However, our study has some intrinsic limitations, such as insufficient follow-up data, a small sample size, and the absence of a comparator arm. We are looking forward to a real-world study of CDK4/6 inhibitors among a larger sample size of Chinese patients in the near future, which will provide more real-world evidence for the clinical use of CDK4/6 inhibitors.
Conclusion
This real-world study showed that palbociclib-ET in HR+/HER2- MBC patients was effective and well tolerated, as well as in patients with BMs. More benefits could be achieved in patients in the first-line setting and those who were treatment-naive, sensitive to ET, and had fewer metastatic sites.
Footnotes
Abbreviations
Acknowledgments
The authors would like to thank Jiaqi Song from Pfizer Medical Affairs (China) for her scientific advice regarding this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics
This study protocol was reviewed and approved by the Ethics Committee of National Cancer Center/National Clinical Research Center for Cancer/Cancer Hospital & Shenzhen Hospital, Chinese Academy of Medical Sciences, and Peking Union Medical College (approval number: YW2023-2-1/2, date of approval: February 15, 2023) and the Ethics Committee of University of Hong Kong - Shenzhen Hospital (approval number: hkuszh2023144, date of approval: August 23, 2023). Due to the retrospective design of this study and patient anonymization, the ethical review board determined that informed consent was not required. All methods were performed in accordance with relevant guidelines and regulations.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by ShenZhen Science and Technology Program (JCYJ20180306171227129, 2018) and the National Natural Science Foundation of China (No. 81671750, 2016).
