Abstract
Background:
The aim of this study was to evaluate the effectiveness of lapatinib in human epidermal growth factor receptor 2 (HER2)-positive metastatic breast cancer.
Methods:
We retrospectively reviewed the medical records of patients who received lapatinib for salvage treatment at any line setting from January 1, 2007 to August 31, 2019 at Shandong Cancer Hospital and Institute.
Results:
A total of 115 (89.1%) patients were included in the study. In the overall cohort, the median disease-free survival (DFS) was 19.0 months; the median progression-free survival (PFS), 6.3 months; and median overall survival (OS), 88.0 months, with 32.2% of patients alive at 5 years. In the second line setting, the median PFS among trastuzumab, lapatinib, and trastuzumab plus lapatinib were 4.2 months, 5.2 months, and 7.3 months, respectively (P = 0.004). No significant differences between the median PFSs and OSs of the different line salvage treatments with lapatinib was observed (all P > 0.05). For brain metastasis patients, the median PFSs in first line, second line, and more than 3 lines were 7.2 months, 4.5 months, and 6.3 months, respectively.
Conclusions:
Our findings suggest that patients would benefit more from trastuzumab plus lapatinib than from lapatinib or trastuzumab alone for second line treatment in the advanced stages of the disease. Lapatinib could be used as an alternative selection for HER2-positive metastasic breast cancer patients when there is disease progression after trastuzumab or pyrotinib treatment, which is used as part of China’s national health insurance.
Introduction
Overexpression of the human epidermal growth factor receptor 2 (HER2) occurs in approximately 15% of newly diagnosed early-stage breast cancer patients and approximately 20% of advanced breast cancer patients. 1 As a potent cancer-driving gene, it was first recognized in the 1980s. 2 Pharmacological targeting of HER2 has dramatically improved the outcomes of HER2-positive breast cancer regardless of staging. A growing number of novel anti-HER2 drugs have been developed, including monoclonal antibodies, such as trastuzumab and pertuzumab, the antibody-drug conjugated T-DM1, and tyrosine kinase inhibitors, such as lapatinib and neratinib. 3 First-line anti-HER2 therapy offers an overall response rate of 80% after dual target treatment and 69% after trastuzumab monotherapy. 4,5 Despite the addition of trastuzumab and/or pertuzumab to chemotherapy to improve the disease-free survival (DFS) and overall survival (OS) of early-stage breast cancer patients, 15% to 24% of HER2-positive patients will eventually develop recurrence or metastasic breast cancer (MBC) during the long-term follow-up period. 6,7
Lapatinib, first approved in 2007, is an oral reversible HER2 tyrosine kinase inhibitor that blocks the phosphorylation of HER1 and HER2. The combination therapy of capecitabine and lapatinib offers an enormous increase in the survival rate of HER2-positive MBC patients who are resistant to previous trastuzumab-containing therapy. 8,9 According to the National Comprehensive Cancer Network guidelines, lapatinib is a valid third-line treatment option, though it is inferior to T-DM1 in trastuzumab-resistant MBC patients.
A clinical trial proved that pyrotinib plus capecitabine yielded a better overall response rate and progression-free survival (PFS) than lapatinib plus capecitabine in HER2-positive breast cancer patients previously treated with anthracyclines, taxanes, and/or trastuzumab. 10 Pertuzumab, which was approved on December 17, 2018 by the National Medical Products of Administration in China, was recommended as the first choice for first line salvage treatment in HER2-positive advanced breast cancer patients. Meanwhile, T-DM1 is not yet listed in China at the time of writing. Although the patients received the aforementioned medication, they eventually progressed.
The Neoadjuvant Lapatinib and/or Trastuzumab Treatment Optimization (known as “NeoALTTO”) trial showed that combined lapatinib and trastuzumab in neoadjuvant therapy settings could achieve higher pathological complete remission (pCR) and survival benefits. 11 The EGF100151 trial, 12 EGF104900 trial, 13 and ALTERNATIVE trial 14 indicated that lapatinib could be used as a valuable selection in salvage settings. However, lapatinib has been excluded from the national medical insurance in China since January 1, 2020. At the time of writing, the cost-effectiveness of lapatinib is not known; this is an important aspect since lapatinib is expensive in China. In this study, we performed a retrospective study to evaluate the effectiveness of lapatinib in HER2-positive MBC patients from a single center under the specific conditions of China.
Materials and Methods
We retrospectively reviewed the medical records of all HER2-positive MBC patients who received lapatinib for anti-HER2 treatment from January 1, 2007 to August 31, 2019 at Shandong Cancer Hospital and Institute. This study was conducted in accordance with the Declaration of Helsinki and approved by the Ethics Committee at Shandong Cancer Hospital and Institute (SDTHEC201703010). All of the patients gave written informed consent before their participation in the study.
The characteristics of patients at the time of initial diagnosis (including age, performance status, and menstrual status), tumor characteristics (including tumor size, lymph nodes involved, grade, histology, and receptor status), and treatment regimen in the adjuvant and salvage settings (including chemotherapy, anti-HER2, endocrine regimen, dose reductions or delays, treatment for central nervous system metastasis, anti-bone destruction, and outcome of treatments in the adjuvant setting) were extracted from electronic medical records. Clinical response data were according to Response Evaluation Criteria in Solid Tumors, version 1.1. Adverse events (AEs) were graded based on the National Cancer Institute—Common Terminology Criteria for AEs, version 4.0.
The estrogen receptor (ER) and progesterone receptor (PR) were determined by at least 10% of nuclei being positively stained. HER2 positivity was defined as either an immunohistochemistry score of 3+ or 2+ together with HER2 gene amplification by fluorescence in situ hybridization. DFS was defined as the interval from surgery to the first occurrence resulting in inoperability, distant metastases, or death of any cause. 15 PFS after metastasis was defined as the time from treatment commencement until disease progression or death. Overall survival (OS) was defined as the time from the beginning of treatment until death from any cause or until the last follow-up date. Clinical benefit rate (CBR) was defined as the proportion of patients who achieved complete response (CR), partial response (PR) or stable disease (SD). Overall response rate (ORR) was defined as the proportion of patients who achieved CR or PR on anti-HER2 therapy.
The enrollment criteria were as follow: 1) female patients aged ≥ 18 years; 2) HER2-positive MBC; and 3) receiving lapatinib regardless of how many lines of treatment they have received. The exclusion criteria were as follow: 1) patients without complete medical records; 2) MBC at first diagnosis; and 3) non-measurable or non-evaluable lesions. All of the patients should have accepted lapatinib 1,250 mg once daily as an initial dose, administered orally in combination with chemotherapeutic drugs or other anti-HER2 target drugs of the appropriate dose and method of administration.
Tumor assessments were evaluated by 2 independent imaging physicians with computed tomography or magnetic resonance imaging. Throughout the study, each patient accepted the same method to evaluate therapeutic effects. Assessments were carried out at baseline every 2 or 3 months, and at the final visit.
Descriptive analysis was performed to summarize the medical records by using counts and percentages for categorical variables. The chi-squared test was used to compare the differences between the various patient groups. The Kaplan-Meier method was used to assess the DFS, PFS and OS. Until the last follow-up date, patients who became progression-free and/or were lost to follow-up were considered censored data. The median time of the event was registered with a 95% confidence interval (CI), and different subgroups were compared using the log-rank test.
Results
Patients and Treatment Regimens
A total of 129 patients with HER2-positive MBC who treated with lapatinib were recruited. A final total of 115 (89.1%) patients were included in the research after excluding the patients with incomplete medical records (Figure 1). The median age for first treatment was 45 years (ranging between 27 and 77 years). Baseline characteristics of the patients included are reported in Table 1. Of these, 55 patients (47.8%) had biopsy results indicative of metastatic disease, and 25 patients (45.5%) had ER-positive cancer, which was confirmed by the metastatic specimens. The Eastern Cooperative Oncology Group performance status was 0-1 in 108 patients (93.9%). Before commencing anti-HER2 therapy for MBC, the metastatic sites involved were found to be the liver in 45 (39.1%), the brain in 42 (36.5%), the lung in 47 (40.9%), the bone in 49 (42.6%), the lymph nodes in 46 (40.0%), and the other organs, such as the spleen, adrenal gland, pancreas, and pleura, in 36 patients (31.3%). For the patients with brain metastasis, 25 (59.5%) accepted brain radiotherapy. The bone metastasis patients were given bisphosphonates every month for the first 2 years and every 3 months for follow-up treatment. The involved organs for advanced diseases were only one organ in 52 (45.2%), 2 organs in 41 (35.7%), 3 organs in 13 (11.3%), and more than 3 organs in 9 patients (7.8%). Lapatinib was used as the first line treatment in 20 (17.4%), second line in 62 (53.9%), third line in 24 (20.9%), and more than 3 lines in 9 patients (7.8%). Meanwhile, as dual target therapy, lapatinib and trastuzumab in combination were used as the first line treatment in 6 (30.0%), second line in 17 (27.4%), and 3 or more lines in 7 patients (21.2%) (Table 2). The baseline characteristics of patients with or without anti-HER2 treatment in the adjuvant setting are summarized in Table 3. Lapatinib was used earlier in the salvage setting for patients who accepted trastuzumab than for trastuzumab-free patients in the curative setting (P = 0.001).

Patient’s follow-up profile.
Baseline Characteristics (N = 115).

a Anti-HER2 treatment only included trastuzumab rather than pertuzumab or combination.
Characteristics of the Patients Receiving Target Therapy in Curative Setting.

a Dual target therapy only referred to trastuzumab and lapatinib.
Characteristics of the Patients at Baseline.

Efficacy
As shown in Figure 2A, in the overall cohort, the median DFS was 19.0 months (95% CI 15.2-22.8). A total of 79 patients (68.7%) achieved the clinical benefit criteria: with 5 CR (4.3%), 27 PR (23.5%), 47 SD (40.9%), and 36 PD (31.3%) (Table 4). In the overall cohort, ORR was 27.8% (32/115). In the subgroup analysis, the CBRs in first line, second line, third line, and more than 3 lines were 85.0%, 69.4%, 58.3%, and 55.6%, respectively. Similarly, the ORRs were 40.0%, 25.8%, 29.2% and 11.1%, respectively. We further investigated the median PFS and OS in the overall cohort, and we found that the median PFS was 6.3 months (95% CI 5.1-7.5) (Figure 2B) and the median OS was 88.0 months (95% CI 64.6-111.5) (Figure 2C), with 32.2% of patients alive at 5 years. For the subgroup analysis, in the salvage treatment setting, patients who received anti-HER2 treatment for early-stage tumors achieved a higher median PFS than patients without anti-HER2 treatment (8.3 months versus 5.1 months, respectively; P = 0.002) (Figure 2D). Between the median OSs between both groups were not significantly different (65.6 months versus 104.1 months, respectively; P = 0.317) (Figure 2E).

Kaplan-Meier estimates of disease-free survival (DFS), progression-free survival (PFS), and overall survival (OS), with 95% confidence interval (CI). In the overall cohort, the DFS (A), PFS (B) and OS (C) were illustrated. The PFS (D) and OS (E) were illustrated between trastuzumab in curative setting and no trastuzumab in curative setting.
Responses for Different Treatment Lines.
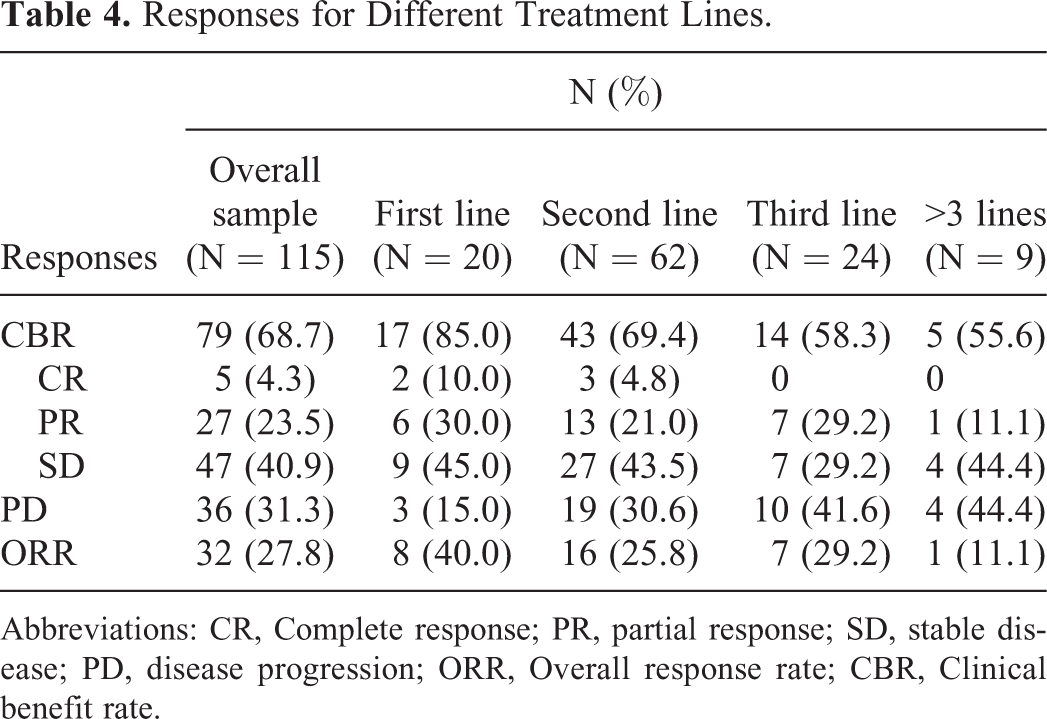
Abbreviations: CR, Complete response; PR, partial response; SD, stable disease; PD, disease progression; ORR, Overall response rate; CBR, Clinical benefit rate.
To determine the best combination for lapatinib, we investigated the second line anti-HER2 treatment in MBC. There were 95 patients who were eligible to be analyzed, and the median PFSs among trastuzumab, lapatinib, and trastuzumab plus lapatinib were 4.2 months, 5.2 months, and 7.3 months, respectively. There were obvious statistical differences among these 3 groups (P = 0.004) (Figure 3A), but no significant differences were observed between the median OSs of trastuzumab, lapatinib, and trastuzumab plus lapatinib (P = 0.773) (Figure 3B).

Kaplan-Meier estimates of progress-free survival (PFS), and overall survival (OS), with 95% confidence interval (CI) for those patients with different drug combinations. For second line anti-HER2 treatment in MBC, the PFS (A) and OS (B) were compared among trastuzumab group, lapatinib group and trastuzumab plus lapatinib group.
Previous findings indicated that trastuzumab and lapatinib could be a better combination in second line anti-HER2 therapy. In order to research the best line for lapatinib in salvage treatment, we performed this study to compare the efficacy for various line treatments. No significant differences were found between the median PFSs and OSs of the different line salvage treatments (all P > 0.05, as shown in Figures 4A and 4B). The median PFS in first line was 8.5 months; second line, 5.3 months; third line, 7.0 months; and more than 3 lines, 5.8 months. The median OS in first line was 65.6 months; second line, 56.2 months; third line, 88.5 months; and more than 3 lines, 88.4 months.

Kaplan-Meier estimates of PFS (A), and OS (B), with 95% CI for those patients with different lines of anti-HER2 treatment.
For brain metastasis patients, as shown in Table 5, the ORRs for first line, second line, and 3 or more lines were 36.4%, 28.6%, and 40%, respectively. The CBRs for first line, second line, and more than 3 lines were 81.8%, 61.9%, and 60%, respectively. The median PFSs in first line, second line, and more than 3 lines were 7.2 months, 4.5 months, and 6.3 months, respectively.
Responses and Survival for Different Treatment Lines in HER2 Positive Breast Cancer.

Safety
Treatment was well-tolerated and no patients died of toxicity. Gastrointestinal (all grades 55.9%) and skin (all grades 23.5%) events were the common toxicities. One toxicity-related treatment interruption was attributed to gastrointestinal disorder. Grade 3/4 AEs occurred in 18 patients (15.7%) and were mostly connected with gastrointestinal disorders (7.8%), hand-foot syndrome (1.7%), nervous system disorders (2.6%), and neutropenia (3.5%). All of the AEs resolved with the appropriate measures. No cardiac events grade 3 or higher in severity were observed. In our cohort, no incidence of severe cardiac toxicity was observed in our MBC patients who received anti-HER2 therapy; thus, no treatment discontinuity occurred that was due to cardiac AEs. Only one patient receiving lapatinib and capecitabine was discontinued to anti-HER2 treatment for gastrointestinal side effects. Most cases were reversible following adequate medical therapy.
Discussion
Clinical guidelines provide us an essential strategy for persistently inhibiting the HER2 pathway using several lines of therapy for HER2-positive MBC. For HER2-positive MBC, standard strategy upon progression to trastuzumab regimens include T-DM1, reusing trastuzumab combined with other chemotherapy, or a combination of lapatinib and capecitabine. 16 -18 Trastuzumab and pertuzumab combined with a taxane are currently recommended as the optimal first-line treatment for HER2-positive breast cancer, at the time of writing. 19 For metastatic patients that have progressed to using trastuzumab, T-DM1 is a superior option, based on its advantages when compared to the standard regimen, which includes a combination of lapatinib and capecitabine. Despite a number of target choices in the treatment of HER2-positive breast cancer that have been proposed by randomized trials, the best anti-HER2 strategy is still under debate. In a real-world scenario, due to the limitations of various realistic conditions in China, such as a patient’s habits or drug accessibility, most MBC patients were compelled to receive lapatinib as their anti-HER2 treatment in any line condition in China before 2020. Herceptin and lapatinib as dual target drugs are the first combination to clinical application and were also the longest dual target drugs in medical use in China before 2020. Lapatinib has been excluded from the national medical insurance from January 1, 2020, and trastuzumab plus pertuzumab or pyrotinib has since become the main treatment selection for HER2-positive advanced breast cancer in China. Therefore, a retrospective analysis of its clinical value, especially in the salvage treatment of breast cancer, has a certain practical significance in proving it as a treatment option.
Phase III of Trial EGF100151 showed that lapatinib plus capecitabine for first line anti-HER2 treatment is superior to capecitabine monotherapy in HER2-positive MBC pretreated with anthracycline, taxanes, and trastuzumab, with a median PFS of 8.4 months versus 4.4 months, respectively. 12 Phase III of the EGF104900 study concluded that a regimen of trastuzumab plus lapatinib was superior to lapatinib alone, with a median PFS of 12 weeks versus 8.1 weeks and a median OS of 14 weeks versus 9.5 weeks, respectively. 13 the EGF100151 and EGF104900 trials indicated that a regimen of trastuzumab plus lapatinib>lapatinib>non-lapatinib for MBC anti-HER2 treatment, in terms of effectiveness. Interestingly, in our study, for second line anti-HER2 treatment, a similar phenomenon of PFS was observed, in that a regimen of trastuzumab plus lapatinib was superior to lapatinib or trastuzumab alone. However, although most patients enrolled in our study received post-progression treatment after lapatinib progression to achieve persistent suppression of the HER2 signaling pathway, no significant difference was observed in terms of the OSs. For those patients progressed from lapatinib, they may accept other anti-HER2 drugs such as pyrotinib or T-DM1, which could prolong the OS.
When the MBC patients were divided into 4 subgroups according to different lines of salvage treatment, CBRs in first line, second line, third line, and more were 85.0%, 69.4%, 58.3%, and 55.6%, respectively. ORRs were 40.0%, 25.8%, 29.2% and 11.1%, respectively. It may achieve a better CBR or ORR for first line treatment. However, we found that no matter how many lines of anti-HER2 therapy were utilized, the PFSs and OSs between these subgroups were not significantly different. The results suggested that lapatinib as salvage treatment could be used in any line setting and that lapatinib combined with trastuzumab may be an effective combination. However, according to survival results of the EGF104900 and EGF100151 studies, subgroup analyses indicated that patients accepting fewer lines of prior anti-HER2 agents obtained a more PFS benefit. The reason may attribute to that most of our enrolled patients receiving anti-HER2 treatment for second line or more and many patients receiving lapatinib as single target for salvage treatment. Our results suggest that treatment regimen containing lapatinib may be a therapeutic alternative for heavily treated HER2 positive breast cancer.
Lapatinib, as a small molecule tyrosine kinase inhibitor, can enter the central nervous system by passing through the blood-brain barrier. In our study, for the first line, the ORR of brain metastasis was 36.4% and the median PFS in first line treatment was 7.2 months. The LANDSCAPE trial. 20 has proved that the objective central nervous system response of lapatinib in first line treatment was 65.9%, and that the time to progression was 5.5 months. When compared with the results of the LANDSCAPE trial, our results showed a low ORR but long PFS. As such, we concluded that lapatinib is an alternative regimen for HER2-positive breast cancer with brain metastases.
Our results also indicated that continuation of targeted HER2 treatment remained safe and well-tolerated. Anti-HER2 treatment can increase the cardiotoxicity risk; therefore, regular monitoring of cardiac function is mandatory. 21 The incidence of cardiotoxicity varies depending on different patients’ conditions, such as previous chemotherapy regimens, comorbidities, and age. The event of gastrointestinal disorders was similar to that previously reported with lapatinib combined with trastuzumab, chemotherapy, or endocrine therapy. 13,22 In our study, most cases were reversible following adequate medical therapy.
Admittedly, there were several important limitations in our study. A retrospective design inevitably has problems, such as data selection bias and lack of useful information. Meanwhile, due to economic reasons, many patients received lapatinib more than 3 lines; as such, it is hard to draw firm conclusions because of only a few patients enrolled in some of the subgroups. In our study, the median follow-up period was 64.3 months, and 32.2% of the patients were still alive at 5 years. We will continue to follow up and to obtain more accurate data.
Conclusion
New agents are emerging rapidly in the management of HER2-positive breast cancer for any clinical stage. 23 In the setting of limited drug availability, methods to maximize the therapeutic effect is an important clinical issue. Lapatinib could be used as an alternative selection for HER2-positive MBC when progression occurs after trastuzumab or pyrotinib treatment, which is provided be the national health insurance in China.
Footnotes
Abbreviations
Authors’ Note
Xinzhao Wang, MD, and Lin Wang, MD, contributed equally to this work. This study was conducted in accordance with the Declaration of Helsinki and approved by the Ethics Committee at Shandong Cancer Hospital and Institute (SDTHEC201703010). All patients gave written informed consent before their participation in the study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by Natural Science Foundation of Shandong Province (ZR2017PH055, ZR2019MH109), Key Research and Development Program of Shandong Province (2018GSF118089) and Medical and Health Development Plan of Shandong Province (2016WS0556).
