Abstract
Keywords
Introduction
Thymic epithelial tumors (TETs), including thymoma, thymic carcinoma, and thymic neuroendocrine neoplasms, are rare tumors that mainly occur in the mediastinum.1,2 Their incidence in Europe is only 1.7/million people per year, and the data registered in the United States is 0.13/100000 person-years.3,4 According to the World Health Organization (WHO) histological classification, TETs can be divided into A, AB, B1-3, and other categories. 5 The current standard treatment for TETs is surgery.6–9 A meta-analysis by Tateishi et al showed that postoperative adjuvant therapy can improve the overall survival (OS) of Masaoka stage II/III patients, 10 but the role of postoperative adjuvant therapy remains to be confirmed by prospective studies. A variety of clinical factors have been confirmed to be related to prognosis including tumor size, 11 T stage, 12 Masaoka stage, 13 WHO histologic types, 14 and tumor vascular invasion. 15 However, molecular biomarkers with prognostic value have not yet been identified or confirmed for this type of malignancy.
Since carcinoembryonic antigen (CEA) was discovered in intestinal adenocarcinoma in 1965, 16 it has gradually become an important diagnostic and prognostic indicator for patients with colorectal cancer. With the deepening of research, serum CEA levels have been found to increase in various cancers, 17 including pancreatic cancer, colorectal cancer, lung cancer, 18 gastric cancer, 19 and others. Meanwhile, CEA has been established as a prognostic marker in a variety of cancers, including colorectal cancer, 20 lung cancer, 21 and breast cancer. 22 However, its prognostic value in TETs has not yet been fully studied.
In a similar fashion, neuron-specific enolase (NSE) is often expressed at high levels in the brain and is elevated in some tumors of neuroendocrine cell origin, and it can be used as a prognostic biomarker for small cell lung cancer, 23 non-small cell lung cancer, 24 esophageal neuroendocrine cell carcinoma, 25 prostate cancer, 26 and other diseases. Some studies have shown that the expression of NSE may also be abnormal in TETs, 27 but its relationship with OS and recurrence-free survival (RFS) in this type of tumors remains to be elucidated.
Although a variety of tumor markers have prognostic roles in various tumors, given the rarity of TETs, research on their specific markers has not attracted a lot of interest from researchers. Therefore, this study explored the prognostic value of tumor markers CEA and NSE in these rare tumors by collecting patients’ data for more than 10 years.
Materials and Methods
Study Design
This study was approved by the ethics review board of the Sun Yat-sen University Cancer Center (No. B2020-353-01), and we obtained the subject's informed consent exemption, the original dataset was uploaded to the Sun Yat-sen University Cancer Center database (RDDA2021002090). Our study has de-identified all patient details.
We retrospectively reviewed the medical records of patients with TETs who underwent thymoma resection at the Sun Yat-sen University Cancer Center between May 2004 and August 2015. The inclusion criteria were as follows: (1) age > 18 years, (2) presence of histopathologically confirmed TETs, including thymomas and thymic carcinomas (TCs), (3) complete surgical resection (R0, no residual disease), and (4) complete relevant laboratory tests (serum tumor marker examination) within 7 days before surgery. Patients were excluded if (1) they were administered radiotherapy or chemotherapy prior to surgery, before and after surgery, or in an unknown sequence of treatment with surgery, (2) they were followed-up for less than 5 years, (3) they had more than 1 malignancy or a history of other malignancies, (4) they had a postoperative survival time of less than 3 months, (5) they only underwent thymoma biopsy, (6) they were treated by cryoablation, and (7) or their follow-up information is incomplete (Figure 1).
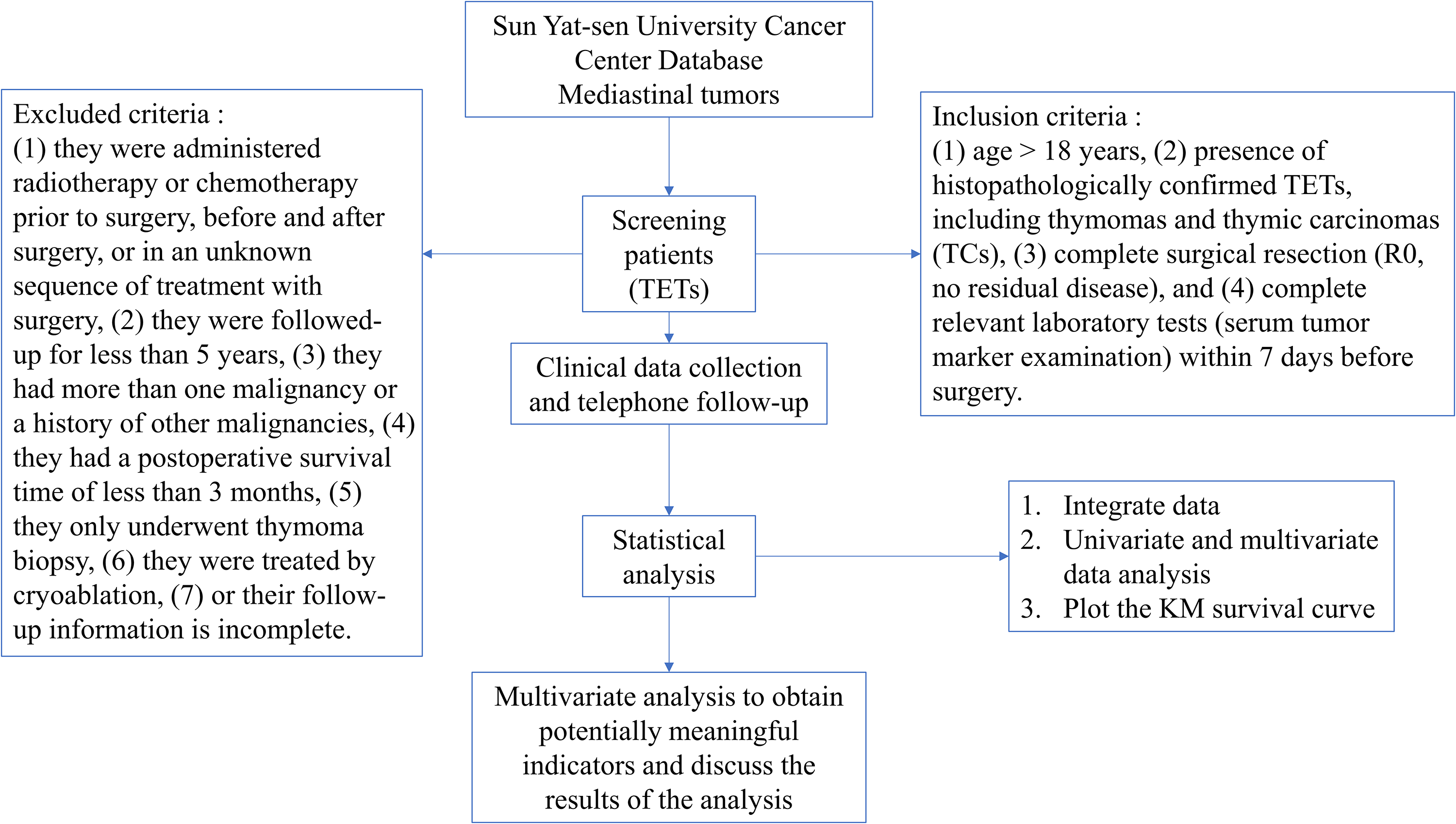
Flowchart.
Data Collection
Data were collected on the following clinical variables: CEA (ng/mL) and NSE (ng/mL) levels (recorded within 1 week before surgery), sex, age, smoking history, drinking history (alcohol consumption per day, specific alcohol consumption is not limited and described), family history of tumors, tumor size, myasthenia gravis symptoms, T stage, histological subtype, Masaoka stage, comorbidities (hypertension and/or diabetes), tumor capsule status, and other clinical information.
Follow-up
The follow-up strategy was specific for the first 2 years, with follow-up every 6 to 12 months for all postoperative patients, every 12 months for the third to fifth years, and then every year. The follow-up examination items were chest computed tomography plain scan and hematological examination (eg, blood routine, biochemical routine, tumor markers, etc.). The last follow-up encounter was in August 2020. The primary endpoints of this study were OS and RFS.
Data Analysis
In this study, the X-Tile was used to obtain the best cut-off values for CEA and NSE (http://www.tissuearray.org/rimmlab). Statistical analyses were performed using SPSS (version 25.0; IBM) and R software (version 4.0.3; https://www.r-project.org/). Univariate and multivariate regression analyses were performed using Cox proportional hazards regression models in SPSS and relying on hazard ratios (HRs) and 95% confidence intervals (CIs) to assess the risk value for each factor. Survival curves were drawn using the Kaplan–Meier method, and the log-rank test was used to compare the survival and recurrence of patients in different groups. All tests were two-way, with a significance level of
Results
Patient Characteristics
A total of 111 patients with TETs were included in this study, including 58 men and 53 women, with an average age of 50.72 ± 12.09 years and an average tumor size of 6.63 ± 3.15 cm. Table 1 summarizes the patients’ WHO histologic types, T staging, smoking history, drinking history, myasthenia gravis, and other relevant clinical information.
Basic Demographic Data, Disease Specific Characteristics (n = 111).

Abbreviations: CEA, carcinoembryonic antigen; NSE, neuron-specific enolase; pT stage, pathological T stage; WHO, World Health Organization.
Optimal cut-off Values for Preoperative CEA and NSE
Taking OS as the endpoint, the optimal cut-off value for preoperative CEA was 2.3 (
X`
Association of CEA and NSE With Survival Outcomes
Using OS and RFS as endpoints, we compared the OS (Figure 2C;

KM analysis of T stage (A), WHO (B), CEA (C), and tumor size (D) based on overall survival (OS).

KM analysis of T stage (A), WHO (B), CEA(C), and drinking history (D) based on relapse-free survival (RFS).
Univariate and Multivariate Survival Analyses Based on OS
Univariate Cox regression analysis showed that 7 variables were significantly associated with OS: WHO histologic types, T stage, Masaoka stage, drinking history, tumor size, CEA levels, and tumor capsule status (Table 2). Four parameters were defined as independent prognostic factors for OS by multivariate Cox regression analysis: WHO histologic types (A-B1 vs B2-B3, HR = 0.456, 95% CI [0.122-1.706]; A-B1 vs C, HR = 6.938, 95% CI [1.814-26.538]), T stage (HR = 12.418, 95% CI [2.610-59.085]), tumor size (HR = 4.995, 95% CI [1.410-17.697]), and CEA (HR = 5.421, 95% CI [1.344-21.858]) (Table 2) and finally the Kaplan-Meier(KM) survival curve was used to show all the significant Cox factors (Figures 2 and 3).

KM analysis of Masaoka stage (A), tumor capsule status (B), and drinking history (C) based on overall survival (OS).
Univariate and Multivariate Analysis Results in Thymic Epithelial Tumor (TET) Based on Overall Survival (OS) (n = 111).

Abbreviations: CI, confidence interval; CEA, carcinoembryonic antigen; HR, hazard ratio; NSE, neuron-specific enolase; pT stage, pathological T stage; WHO, World Health Organization.
Univariate and Multivariate Survival Analysis Based on RFS
In the univariate analysis, 5 variables were significantly associated with RFS: WHO histologic types, T stage, drinking history, Masaoka stage, and tumor capsule status (Table 3). However, multivariate Cox regression analysis showed that 4 parameters were defined as independent prognostic factors for RFS: WHO histologic types (A-B1 vs B2-B3, HR = 1.669, 95% CI [.485-5.743]; A-B1 vs C, HR = 6.431, 95% CI [1.675-24.693]), T stage (HR = 4.722, 95% CI [1.580-14.114]), drinking history (HR = 4.227, 95% CI [1.431-12.484]) and NSE(HR = .305, 95% CI [.105–.887]) (Table 3) and finally the KM survival curve was used to show all the significant Cox factors (Figures 4 and 5).

KM analysis of Masaoka (A) and tumor capsule status (B) based on relapse-free survival (RFS).
Univariate and Multivariate Analysis Results in Thymic Epithelial Tumor (TET) Based on Relapse-Free Survival (RFS) (n = 111)

Abbreviations: CI, confidence interval; CEA, carcinoembryonic antigen; HR, hazard ratio; NSE, neuron-specific enolase; pT stage, pathological T stage; WHO, World Health Organization.
Discussion
This study confirmed the predictive effect of the tumor marker CEA on OS of patients with TETs through a rigorous Cox regression analysis. Interestingly, we also found that tumor size was an independent predictive factor of OS, and drinking history was an independent predictive factor of postoperative recurrence. However, NSE did not show any prognostic power for patients in our cohort.
It is well known that T stage, 12 WHO histologic types, 28 and Masaoka stage 13 have significant effects on the survival of patients with TETs and the recurrence of these tumors, which was also reflected in our study. In addition, the Masaoka-Koga staging system is widely accepted for the staging of TETs. However, Yanagiya et al found in their cohort that WHO histological type and age were important prognostic factors without including the Masaoka-Koga staging system. 29 Similarly, Fukui et al showed that a new classification system had a better prognostic effect than Masaoka-Koga staging for TETs.30,31 Combined with our findings, T staging may be more accurate in prognostic prediction than Masaoka-Koga staging. This needs to be validated in a larger sample size database. Eriksson et al showed that drinking history may be a risk factor for thymic tumors, 32 but due to the rarity of TETs, few studies have investigated the relationship between drinking history and prognosis. Therefore, this may be an interesting research direction, which needs to be further confirmed by studies with larger sample sizes.
Whether tumor size affects the prognosis of TETs remains controversial. Okumura et al analyzed the clinical information of 2083 postoperative patients with thymomas by integrating multiinstitutional data and concluded that tumor size has an important impact on prognosis. 33 In addition, Fukui et al found that patients with tumors larger than 4 cm had worse RFS results, and this relationship was found even in patients with stage I disease. 9 There are other related studies supporting this conclusion.34–36 However, Nicholson et al analyzed more than 8000 patients and found that tumor size did not have a prognostic significance in thymic malignancies. 37 Similarly, Tseng et al also found that tumor size did not improve the prognostic predictive power of the TNM staging. 38 Using the data of more than 2000 patients in the European Society of Thoracic Surgeons (ESTS) database, Ruffini et al found that tumor size did not predict OS and RFS but could predict incomplete resection and recurrence risk. 39 Finally, in this study, our data support an independent association between tumor size and OS after R0 resection in patients with TETs.
As a tumor marker first discovered in intestinal adenocarcinoma, CEA has shown a strong prognostic role in lung cancer, breast cancer, colorectal cancer, and other tumors.20–22 In TETs, Savino et al found that CEA may play a role as a thymic epithelial cell growth factor, 40 and Tomita et al found that the expression of CEA in thymoma was correlated with clinical stage. 41 Additionally, Tomita et al found that CEA is highly expressed in thymic carcinomas, possibly indicating the aggressiveness of these carcinomas. 42 Due to the rarity of TETs, the sample size of previous studies was extremely limited. Based on previous research and considering that CEA may affect the malignant potential and invasiveness of TETs, the team further explored the value of CEA as a tumor biomarker in the prognosis prediction of patients with these tumors. Although CEA was not significantly associated with postoperative RFS, our team first found that CEA was an independent predictor of postoperative R0 resection of TETs.
Whether for OS or RFS, NSE did not show statistical significance in univariate analysis in this study. However, in multivariate analysis with RFS as the end point of the study, NSE showed a certain prognostic value, but we think it cannot be used as one of the conclusions of this study. First, NSE did not show a prognostic value in univariate analysis, and its reference value in multivariate analysis was limited in this study. Second, due to the small number of NSE patients in this study, the results may have certain errors. Third, as a traditional oncological marker, NSE mainly reflects the diagnosis and differential diagnosis of meningitis, cerebral infarction, cerebral hemorrhage, islet cell tumor, melanoma, neuroblastoma, seminoma, small cell lung cancer, and other diseases, from a clinical point of view, it is not closely related to epithelial tumors. Fourth, it is well known that NSE is a risk factor in many benign and malignant diseases, and in our study, it is a protective factor, which violates the clinical common sense. In summary, we believe that the statistical significance of NSE in RFS should be held with skepticism. It is likely that this is just an accidental result and must be confirmed by a larger sample size or multiple centers to further determine its relationship with prognosis.
Although NSE did not show sufficient prognostic value in this study, it is important to consider the fact that this study mainly included thymoma and thymic carcinoma and did not consider other related thymic tumor types. Whereas NSE is mainly generated from neuroendocrine cells, and it can still show potential significance in future studies on thymic neuroendocrine neoplasms.
There is no doubt that the tumor capsule status is an important prognostic factor, but the prognostic value was not reflected in this study. The reason may be that, first, the tumor status in this study was relatively early, and all patients underwent complete surgical resection, so resection results are consistent regardless of tumor capsule status. Second, the data volume of this study is small, which may cause statistical errors.
This study had some limitations. First, this was a retrospective study conducted at a single center. In the future, multicenter, large sample studies are needed to confirm our results. Second, due to the extremely low incidence of thymic neuroendocrine neoplasms, this study did not include such patients, which may have affected the predictive effect of NSE on prognosis. At last, the dynamic changes in postoperative CEA and NSE levels were not included in the research scope, and their values need to be further explored.
Conclusion
Through retrospective analysis of more than 10 years of patient data, we found that preoperative serum CEA levels, WHO histologic types, and tumor size can independently predict OS in patients with TETs after R0 resection. On the other hand, patient drinking history and Masaoka stage were independently associated with postoperative RFS. Finally, the preoperative CEA level appears to be a powerful biomarker for the postoperative prognosis of TETs.
Footnotes
Acknowledgments
We are very grateful for the clinical data provided by the Follow-up Office and Information Section of Sun Yat-sen University Cancer Center. At the same time, I am very grateful to Professor Long Hao and Professor Zhang Xu for their selfless help in providing the thesis materials. Finally, we are very grateful to Editage (![]() ) for the meticulous work on document polishing.
) for the meticulous work on document polishing.
Author’s Contribution
Conception and design of the study: MGW and HYY. Provision of study materials or patients: MGW. Acquisition of data: HYY and LSH. Data analysis: HYY and LX. Interpretation of data: MGW and HYY. HYY drafted the manuscript alone. MGW, HY, LSH, LX, and HYY substantially revised the manuscript. All authors have read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Wu Jieping Medical Foundation (grant number 320.6750.2020-15-7).
Ethics Approval and Consent to Participate
This study was approved by the Medical Ethics Committee of Sun Yat-Sen University Cancer Center (B2020-353-01) and complied with the Declaration of Helsinki. Data were recorded at the Sun Yat-sen University Cancer Center under the record number: RDDA2021002090. At the same time, this study has obtained the exemption of informed consent application from the Ethics Committee of Sun Yat-sen University Cancer Center.
