Abstract
Introduction
Colorectal cancer (CRC) is a classic solid tumor that causes many deaths worldwide, 1 including in China. 2 Although early diagnosed cases could be cured by surgery, the majority of advanced cases are still incurable. 3 Identifying simple and reliable prognostic factors in the disease have become increasingly popular in recent years.
Albumin (ALB) acts as a carrier for practically all metabolites, drugs, hormones, and products of toxic degradation in the human body, 4 and its metabolism is affected by many exogenous and endogenous factors, including cancer cells. 5 Previously, several studies demonstrated the usefulness of individual ALB as a prognostic factor in many malignancies, including gastric cancer, 6 non-small cell lung cancer, 7 ovarian cancer, 8 head and neck cancer 9 and, in particular, CRC.10–13 Notably, the prognostic efficacy of ALB alone was inferior when compared with other markers. 11 In recent years, some innovative markers based on ALB, such as the ALB-to-globulin ratio,14,15 ALB-to-fibrinogen ratio, 16 C-reactive protein-to-ALB ratio (CAR),17–19 and others,20–22 have been frequently reported in cancer studies, including CRC. However, some of these parameters are not conventional prognostic factors and are not included in routine clinical tests, and more common markers are still needed.
Carcinoembryonic antigen (CEA) is a member of a family of glycoproteins anchored to the cell membrane of colonic epithelial cells and up to approximately 90% of CRC cells can release CEA.23,24 CEA is also a long-term established marker in the diagnosis, surveillance, and prognosis of CRC. Regrettably, only approximately one-third of patients have an abnormally elevated CEA at the time of diagnosis, 25 which largely impairs its application in clinical practice. Interestingly, some investigators have attempted to further explore the prognostic value of CEA in patients whose levels fall into the normal range before surgery;26,27 however, the cut-off points were not consistent, and its value in postoperative surveillance in such a scenario was still mainly unclear. These studies also indicated that a single CEA assay was still insufficient for predicting patients’ prognosis. Taking into consideration these results, it was plausible that combining ALB and CEA would have potential prognostic value in CRC, but studies addressing this topic have not been published.
In this study, we aimed to explore the prognostic value of the ALB to CEA ratio (ACR) as well as its prognostic efficacy when compared with individual ALB, CEA, and other systemic inflammation markers in CRC.
Methods
Patient Enrollment
A retrospective study with CRC patients treated in Hainan Hospital of Chinese PLA General Hospital from January 2011 to January 2018 was performed. Those meeting the following criteria were included: (1) age>18 years; (2) radical recession of the primary lesion; and patients were excluded based on any one of the following criteria: (1) neoadjuvant therapies; (2) missing laboratory tests within a week before surgery or key information in postoperative pathological reports; (3) multiple or recurrent malignancies or in situ lesions; (4) abnormal aminotransferase or bilirubin by liver function tests; and (5) loss to follow-up of less than 36 months. Other clinicopathological parameters were collected as previously reported,28,29 and the body mass index was calculated as body weight (kg) divided by height (m2). Tumor size was cut off at 4 cm according to a previous report. 30 The study was approved by the ethics committee of Hainan Hospital of Chinese PLA General Hospital (ID: 301HLFYLS15), and written informed consent was received from the patients or their authorized relatives. The reporting of this study conforms to STROBE guidelines 31 and all the details of the patients were de-identified.
Determination of ACR and Other Systemic Inflammation Markers
Laboratory tests were performed between 6:00 and 9:00 am on peripheral venous blood within 1 week to 1 month before the surgery, and the latest results were used when more than one result was found. The ACR was calculated as ALB (Ref: 35-50 g/L) divided by CEA (Ref: 0-5.0 ng/mL) and then by 106 to make the results easier to read. In addition, other systemic inflammation markers, including NLR, 32 LMR 33 and PLR, 34 were also determined according to previous reports.
Definition of PFS and OS
The follow-up was conducted by telephone, WeChat, or by visit to the medical records at the hospital with an interval of 3 to 6 months for the first 2 years and 6 to 12 months for the following years. Progression-free survival (PFS) was defined as the point from the date of surgery until the date of first recurrence at any location, disease progression by imaging examinations according to the RECIST (version 1.1), 35 or death from any cause. Overall survival (OS) was defined as the date of surgery until the date of death by any cause. The latest follow-up point was June 2021.
Statistical Analysis
The statistical analyses were performed using SPSS 20.0 (SPSS Inc.). A receiver operating characteristic (ROC) curve analysis was used to determine the optimal discriminator value of ACR, and comparison of the areas under the curve (AUCs) was achieved by MedCalc v19.0.7 (MedCalc Software Ltd). The differences in clinicopathological parameters in the ACR subgroups were calculated by chi-square test or Fisher's exact test and Student's t-test when appropriate. Survival differences for ACR-low or high groups were conducted by Kaplan–Meier (K-M) analysis followed by log-rank tests. Risk factors for survival were estimated by a Cox proportional hazards model. Double-sided P < .05 was considered statistically significant.
Results
Demographic Characteristics and the Prognostic Efficacy of ACR
As shown in Figure 1, a total of 294 patients were registered, and 156 patients (100 males, 56 females) were ultimately included in the study. The mean age of the patients was 60.36 years (range: 24-85 years), and the mean follow-up was 47.61 months (range: 1-102 months). According to the ROC tests, ACR was significant for predicting PFS (AUC = 0.75, 95% CI: 0.368−0.83, P < .01) and OS (AUC = 0.74, 95% CI: 0.65−0.83, P < .01) (Figure 2), and when 5.98 (according to the Youden index) was used as the cut-off point, it had a sensitivity of 58.50% and 61.50% and a specificity of 83.50% and 80.50% for PFS and OS, respectively. Next, further comparison of the prognostic efficacy of ACR with other markers indicated that ACR had a superior efficacy than individual ALB (Z = 2.58), CEA (Z = 2.17) and NLR (Z = 2.63), LMR (Z = 2.08), and PLR (Z = 3.03) in PFS (all P < .05) (Figure 3A); and it also had a similar advantage over single ALB (Z = 2.05), CEA (Z = 2.04) NLR (Z = 2.29), and PLR (Z = 2.40) in OS (all P < .05) except LMR (Z = 1.72, P = .09) (Figure 3B).

Flow diagram of the study.

ROC tests of ACR for PFS and OS.

Comparison of AUCs for ACR with other markers in predicting PFS (A) and OS (B).
Differences in Clinicopathological Parameters in the ACR-low or ACR-High Subgroups
By ROC tests, the patients were subsequently divided into ACR-low (<5.98) or ACR-high (≥5.98) subgroups, and it was found that 31.41% (49 of 156) of cases were ACR-low. As shown in Table 1, these patients apparently presented with advanced T stages (T3 + 4) (P <.01), larger tumor diameters (P < .01) and distant metastases (P = .01), and a relatively lower LMR (P < .01).
Different ACR Among Varied Clinicopathological Parameters.
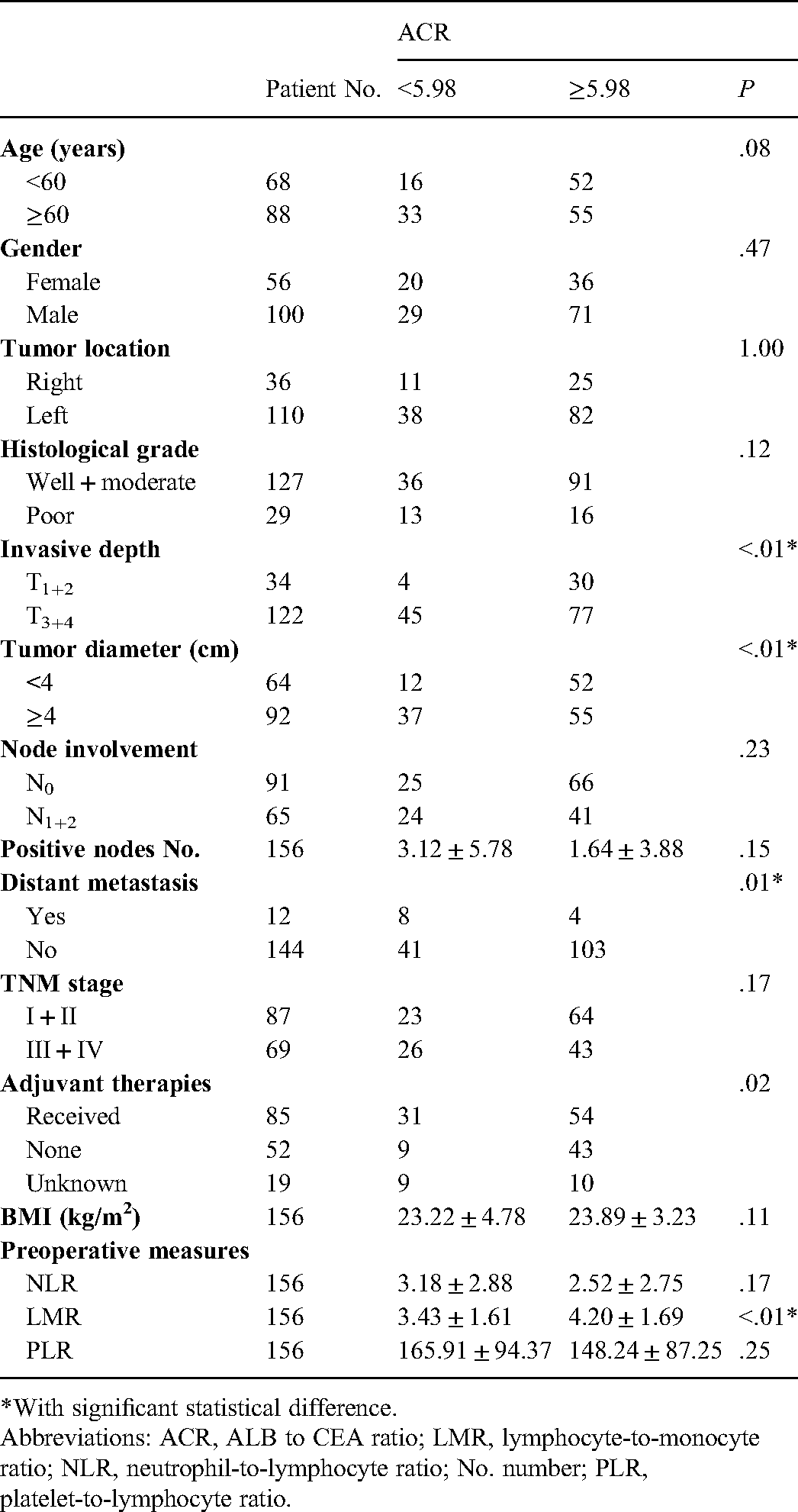
*With significant statistical difference.
Abbreviations: ACR, ALB to CEA ratio; LMR, lymphocyte-to-monocyte ratio; NLR, neutrophil-to-lymphocyte ratio; No. number; PLR, platelet-to-lymphocyte ratio.
Survival Differences of ACR-low or ACR-High Groups in PFS and OS
There were significant differences in the ACR-low or ACR-high subgroups in the 3-year PFS (63.27% vs 20.56%, P < .01) and OS (48.98% vs 14.02%, P < .01) rates. By K-M analyses, it was found that patients with ACR-low had a significantly worse PFS (ACR-low vs high: 29.35 ± 24.73 m vs 49.81 ± 21.43 m, log-rank = 35.75, P < .01) and OS (ACR-low vs high: 35.22 ± 21.94 m vs 53.28 ± 18.98 m, log-rank = 29.68, P < .01) than those with ACR-high (Figure 4).

Impact of ACR-low or -high on PFS and OS in the patients.
Univariate and Multivariate Analyses of the Prognostic Factors for PFS and OS
Univariate tests indicated that invasive depth, tumor diameter, node involvement, positive node number, distant metastasis, TNM stage, NLR, LMR, and ACR were significant prognostic factors for PFS and OS (additional plus age and histological grade) (Table 2), and when all these factors were included in multivariate tests, the results indicated that the ACR was an independent prognostic factor for both PFS (HR = 0.31, 95% CI: 0.17−0.56, P < .01) and OS (HR = 0.33, 95% CI: 0.16−0.66, P < .01) (Table 3).
Univariate and Multivariate Analyses of Different Parameters for PFS.

*With significant statistical difference.
Abbreviations: ACR, ALB to CEA ratio; LMR, lymphocyte-to-monocyte ratio; NLR, neutrophil-to-lymphocyte ratio; No. number; PFS, progression-free survival; PLR, platelet-to-lymphocyte ratio.
Univariate and Multivariate Analyses of Different parameters for OS.

*With significant statistical difference.
Abbreviations: ACR, ALB to CEA ratio; LMR, lymphocyte-to-monocyte ratio; NLR, neutrophil-to-lymphocyte ratio; No. number; OS, overall survival; PLR, platelet-to-lymphocyte ratio.
Discussion
In the present study, we found that ACR was a useful prognostic marker in CRC, and patients with a relatively low preoperative ACR had a worse prognosis than those with a high preoperative ACR. The prognostic efficacy of ACR was significantly better than that of individual ALB, CEA, and other systemic inflammation markers and was an independent risk factor for survival. To the best of our knowledge, this is the first report concerning the role of ACR in cancer.
Previously, both individual ALB and CEA were reported as conventional prognostic markers in CRC. In a study that included 69 nonmetastatic and 57 metastatic CRC patients, Wei et al. found that ALB was significantly reduced in metastatic patients and was an independent prognostic factor for PFS. 10 Li et al. studied 312 staged I to III CRC patients and found that low ALB correlated with shortened PFS and OS. 11 In line with these results, Almasaudi et al. performed a study on 795 CRC patients and found that hypoalbuminemia was associated with poorer cancer-specific survival and OS. 36 Lakemeyer et al. conducted a study with 1487 staged I to IV CRC and found that an elevated CEA (>5 ng/mL) could predict significantly worse OS. 37 Additionally, Egenvall et al. carried out a study on 2509 staged II-III CRC and found that an elevation of CEA (>5 ng/mL) either before or after treatment could predict an increased risk of recurrence as well as cancer-specific mortality and OS. 38 In addition, Beom et al. studied 2021 staged I to III CRC with a normal preoperative CEA level (<5 ng/mL) and demonstrated that a relatively high CEA (>2.1 ng/mL) correlated with significantly poor disease-free survival (DFS). 27 Interestingly, all these studies consistently indicated that a lower ALB or a relatively higher CEA could be associated with poor survival in patients, which could be partially interpreted as equal to a lower ACR in our study. Additionally, as indicated in previous studies, a lower ALB was more commonly found in cases with metastasis10,39 or large tumor diameter and advanced T stages.39–41 The significant differences in invasive depth, tumor diameter, and distant metastasis in the ACR-low and ACR-high subgroups in our study were also in line with these results to some extent.
Furthermore, serum ALB levels have long been regarded as a reflection of nutritional status in patients, and serum ALB metabolism could be significantly altered in the context of cancer. Except for those who underwent surgery that presented with a decreased synthesis rate of ALB, 42 the generation of ALB could be purely suppressed by the activation of a series of inflammatory cytokines, including IL-1, IL-6, and TNFα, in cancer patients.18,36,43 Notably, CEA was found to be positively associated with IL-6.44,45 Based on these results, it was plausible that patients with a high preoperative CEA could have a low ALB (equal to a low ACR in our study) in CRC. Additionally, since a high CEA was produced by drug-resistant cancer cells in CRC, 46 these patients were also likely to have a poor prognosis.
Notably, individual ALB or CEA was insufficient for their prognostic efficacy in CRC, particularly when compared with other markers. For example, in Li et al.'s study, the AUCs for PFS and OS of ALB were 0.644 and 0.611, respectively, and ALB alone was not an independent prognostic factor when compared to PLR; 11 similarly, Artaç M et al. studied 90 metastatic CRC patients treated with the bevacizumab plus FOLFIRI regimen and found that ALB was not a risk factor for PFS when compared with NLR. 47 For CEA, Björkman K et al. investigated 322 staged I to IV CRC and found that CA125 was superior to CEA in predicting disease-specific survival (DSS). 48 In recent years, some authors have tried to combine ALB or CEA with other markers to further improve their prognostic efficacy. For ALB, Matsuoka et al. collected 133 stage III CRC and explored the prognostic value of pre- and postoperative CAR, which was found to be an independent prognostic factor for recurrence-free survival and OS, but its AUC was only 0.63 and was the highest of the assessed markers (NLR, PLR). 49 Yamamoto et al. studied 523 staged I to IV CRC and combined ALB with cholinesterase as a new prognostic indicator. The results suggested that patients in both ALB and cholinesterase high groups would have a better DSS than others; however, the AUCs of the combined groups were not compared with individual ALB (0.71) and cholinesterase (0.69) in their study. 22 Some authors tried to combine CEA with Ki-67, 50 p53, 51 tumor budding, 52 CD44v6, 53 peritoneal carcinomatosis index, 54 D-dimer, 55 and NLR 56 to generate new prognostic indicators, but these studies did not compare the AUC of the new indicators with individual CEA. In our study, we found that ACR displayed the largest AUC when compared with individual ALB, CEA and NLR, LMR, and PLR both for PFS and OS, which supports its priority in prognosis prediction in CRC.
The present study has some limitations. First, it was retrospectively conducted at a single center, and the relatively small sample size may attenuate the statistical power and lead to biased findings. Second, other factors could also influence PFS and OS. For example, some of the patients, including those with stage II disease with high recurrence or metastasis risks or those with stages III to IV disease, will accept subsequent therapies after surgery. Nonetheless, additional studies with increased cases could resolve these drawbacks and confirm the results of our study in the future.
Conclusion
Overall, our results indicated for the first time that ACR was a robust prognostic factor in CRC, and patients with a relatively low preoperative ACR would have significantly worse survival.
Footnotes
Abbreviations
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The study was conducted with grants from the Chinese Natural Science Foundation (grant Nos. 81873215 and 81503391).
Ethical Approval
The study was approved by the ethics committee of Hainan Hospital of Chinese PLA General Hospital (ID: 301HLFYLS15), and written informed consent was received from the patients or their authorized relatives.
Author Contribution
MX, YL, TX, QY, and JX contributed to data collection, statistical analysis, and drafting of the manuscript. LL and BY contributed to manuscript revision and study analysis. All authors read and approved the final version.
Data Availability
The data used to support the findings of this study are available from the corresponding author (Dr Bing Yan) upon request.
