Abstract
Background:
Triple-negative breast cancer constitutes approximately 12%-17% of all breast cancer cases, and >33% of patients develop distant metastases. Systemic cytotoxic chemotherapy is the primary treatment for patients with metastatic triple-negative breast cancer; however, the role of first-line platinum-based chemotherapy in these patients remains controversial. This meta-analysis evaluated the efficacy and safety of platinum-based first-line chemotherapy for patients with metastatic triple-negative breast cancer.
Methods:
We systematically searched the PubMed, Embase, Cochrane, and Clinical Trials registry databases up to June 1, 2020 to identify randomized controlled trials that investigated platinum-based vs. first-line platinum-free chemotherapy in patients with metastatic triple-negative breast cancer. We used fixed and random effects models to calculate pooled hazard ratios and odds ratios with 95% confidence intervals for progression-free and overall survival, objective response rates, and grade 3 and 4 adverse events.
Results:
Four randomized controlled trials (N = 590 patients) were included. Platinum-based chemotherapy significantly increased the objective response rates from 43.1% to 62.7% (odds ratio 2.34, 95% confidence interval 1.66-3.28, P < 0.001). Three randomized controlled trials (N = 414 patients) reported survival outcomes. Patients administered platinum-based regimens showed significantly longer progression-free survival (hazard ratio 0.55, 95% confidence interval 0.37-0.82, P = 0.004) and a nonsignificant trend toward improved overall survival (hazard ratio 0.76, 95% confidence interval 0.57-1.00, P = 0.05). Only 2 studies reported the rates of grade 3 and 4 adverse events; grade 3-4 thrombocytopenia was more commonly associated with platinum-based chemotherapy (odds ratio 7.54, 95% confidence interval 1.37-41.60, P = 0.02) and grade 3-4 fatigue with platinum-free chemotherapy (odds ratio 0.23, 95% confidence interval 0.08-0.68, P = 0.008).
Conclusions:
First-line platinum-based chemotherapy was associated with significantly increased objective response rates, longer progression-free survival, and a nonsignificant trend toward improved overall survival in patients with metastatic triple-negative breast cancer at the high risk of grade 3-4 thrombocytopenia.
Introduction
Triple-negative breast cancer (TNBC), which constitutes approximately 12%-17% of all breast cancers cases is a subtype of breast cancer that shows lack of expression of estrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor receptor-2 (HER2). 1 Despite overall improvements in the management of breast cancer, the recurrence rates are higher, disease-free survival is shorter, and overall survival (OS) is poorer in patients with TNBC than in those with other breast cancer subtypes. 2 Notably, >33% of patients with TNBC present with distant metastases, either at the time of diagnosis or as recurrent disease. 3 The median survival period is approximately 1 year in those with metastases. 4
Systemic cytotoxic chemotherapy is the mainstay of treatment for patients with metastatic TNBC (mTNBC) owing to the lack of therapeutic targets. Although previous studies suggest that patients with TNBC show higher rates of pathological complete response (pCR) after neoadjuvant chemotherapy, 5 a significant number of patients show a high risk of local relapse and distant metastases during the first 3-5 years after treatment. 3,6 TNBC is characterized by genomic instability secondary to abnormal DNA repair systems induced by mutations in the BRCA1/2 and/or other genes involved in the homologous recombination (HR) repair mechanism. 7 Gonzalez-Angulo et al 8 reported that up to 20% of patients with TNBC presented with a BRCA1/2 gene mutation. Deficiencies in BRCA1/2 confer hypersensitivity to interstrand crosslinking agents, such as platinum and mitomycin C because these compounds cause DNA inter- and intra-strand crosslinking that cannot be recognized and subsequently repaired by the HR system. 9 -11 Some studies have evaluated the efficacy of platinum-based neoadjuvant or adjuvant chemotherapy in patients with TNBC. A meta-analysis that included 9 randomized controlled trials (RCTs) (N = 2109 patients) reported that platinum-based neoadjuvant chemotherapy significantly increased the pCR rate from 37.0% to 52.1% (odds ratio [OR] 1.96, 95% confidence interval [CI] 1.46-2.62, P < 0.001) in patients with TNBC, and it also significantly increased the risk of grade 3 and 4 hematological adverse events (AEs). 12 However, a randomized phase II study has recently reported that neoadjuvant single-agent cisplatin did not increase the pCR rate than doxorubicin plus cyclophosphamide in germline BRCA mutation carriers with stage I-III HER2-negative breast cancer. 13 The role of platinum salts in the treatment of patients with stage I-III TNBC remains uncertain and platinum salts may increase the risk of grade 3 and 4 hematological AEs, so taxanes and anthracyclines remain standard therapeutic agents for patients with non-mTNBC. 14,15 Pretreated tumors that metastasize are usually refractory to previous therapy, and platinum-based regimens may be a better option in patients with mTNBC.
However, studies have reported conflicting results regarding the use of platinum-based regimens as first-line treatment for patients with mTNBC, and this remains a controversial issue. 15 A retrospective cohort study performed by Zhang et al 16 reported that patients with mTNBC who received first-line platinum-based chemotherapy showed longer progression-free survival (PFS) than those who received platinum-free chemotherapy (7.8 months vs. 4.9 months, P < 0.001). Similarly, the CBCSG006 trial performed by Hu et al 17 reported longer PFS in patients with mTNBC who received cisplatin plus gemcitabine (GP) than those who received paclitaxel plus gemcitabine (GT) (7.73 months vs. 6.47 months, hazard ratio [HR] 0.629, 95% CI 0.523-0.915, P = 0.009); however, no OS benefit was observed. Another RCT reported that the response rate was only <20% in patients with mTNBC treated with cetuximab plus carboplatin. 18 In a retrospective study, Staudacher et al 19 observed that compared with other subtypes, patients with mTNBC treated with platinum-based chemotherapy tended to show a higher response rate but without significant improvement in PFS or OS. Overall, evidence suggests that platinum-based chemotherapy may be useful as alternative first-line chemotherapy in patients with mTNBC, although this remains controversial. We performed a meta-analysis of RCTs to evaluate the efficacy and safety of platinum-based regimens as first-line chemotherapy for patients with mTNBC, to gain a deeper understanding of this debatable topic.
Materials and Methods
Study Objectives
This meta-analysis involved a quantitative synthesis of available RCTs that investigated the efficacy and safety of platinum-based (experimental arm) vs. platinum-free (control arm) chemotherapy as first-line chemotherapy in patients with mTNBC.
Literature Search Strategy and Study Identification
We performed a systematic literature search of the PubMed, Embase, Cochrane, and the Clinical Trials registry databases without any language or date restriction up to June 1, 2020. Additionally, we manually searched the reference lists of all relevant original and review articles to identify any additional eligible studies. We used the following keywords for our search: “triple-negative,” “breast,” “mammary,” “carcinoma,” “neoplasm,” “cancer,” “tumor,” “metastatic,” “advanced”, “platinum,” “carboplatin,” “cisplatin,” and “chemotherapy.” Specific keywords and free-text terms were combined using Boolean operators. Abstracts of all available studies were reviewed, and full-text manuscripts were subsequently retrieved. The literature search and study screening were independently performed by 2 coauthors, and disagreements were resolved through discussion with a third author to reach a consensus.
This meta-analysis was performed according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines 20 and was registered with PROSPERO (registration number CRD42020190209).
Selection Criteria
Eligible studies had to satisfy all the following inclusion criteria: (a) phase II or III RCTs, (b) adult (18 years and above) patients with mTNBC, (c) treatment with first-line platinum-based chemotherapy in the experimental arm and first-line platinum-free chemotherapy in the control arm (for RCTs including patients with metastatic breast cancer subtypes other than TNBC, only those with available results in the TNBC cohort were also included), (d) studies with available information on data for time to tumor progression (TTP) or PFS, OS, objective response rates (ORRs) and side-effects in the experimental and control groups. Exclusion criteria were: (a) incomplete data on treatment and ER/PR/HER2 status, (b) non-RCTs, (c) non-first-line chemotherapy in patients with mTNBC, (d) and ongoing studies without results presented or published at the time of the literature search.
Data Extraction
The following data was extracted, if possible, from all the included RCTs independently by 2 authors: name of the study, first authors’ names, year of publication, study design, number of randomized patients, type and dose of chemotherapy administered, and ORRs, PFS, OS and grade 3 and 4 adverse events (AEs) in the experimental and control arms.
Risk of Bias and Grading Quality of Evidence
The risk of bias and grading quality among included studies were assessed by 2 independent investigators in accordance with the Cochrane Handbook for RCTs based on the following 7 criteria: random sequence generation, allocation concealment, blinding of outcome participants and personnel, blinding of outcome assessment, incomplete outcome data, selective reporting and other bias. 21 Each study was regarded as high, low or unclear of risk of bias based on the criteria mentioned above.
Statistical Analysis
We used the Review Manager software (RevMan, version 5.3 for Windows, Cochrane Collaboration, Oxford, UK) for this meta-analysis. We calculated the ORs with 95% CIs for ORRs, and grade 3 and 4 AEs for an intergroup comparison of these rates. HRs with 95% CIs were used as the summary statistic for time-to-event variables (PFS, OS); an HR value <1 represented a survival benefit in favor of platinum-based chemotherapy.
Before data synthesis was performed, the Cochrane Q test and I 2 statistic were used to test heterogeneity across the included studies, and a P value <0.10 or I 2 >50% was considered statistically significant heterogeneity. 22 A fixed-effects model was used in the absence of significant heterogeneity, and significant heterogeneity necessitated the use of a random-effects model. For significant heterogeneity identified across studies, we performed sensitivity analysis by removing individual studies from the meta-analysis to determine potential sources of heterogeneity. A few studies provided Kaplan-Meier curves rather than HRs and 95% CIs; in such cases, we extracted data from the Kaplan-Meier curves using the method described by Tierney. 23 Funnel plots were generated using RevMan to detect publication bias, which was tested using the Begg’s 24 and Egger’s tests. 25 A P value <0.05 was considered statistically significant.
Results
Selection and Characteristics of Eligible Literature
We initially identified 2163 records as follows: 715 from PubMed, 1226 from Embase, 143 from the Cochrane Library, and 79 from the Clinical Trials registry database. Of these, 186 were excluded as duplicate studies. Based on the aforementioned inclusion and exclusion criteria, we excluded 1955 studies after screening the title or abstract (reviews, case reports, conference abstracts, or editorials, non-RCTs, studies that described other breast cancer subtypes, and non-first-line chemotherapy in mTNBC), and the remaining 22 studies were further analyzed. After reviewing the full text, 4 RCTs 17,26 -28 (N = 590 patients) met the inclusion criteria and were included in this meta-analysis (Figure 1).

Flow chart summarizing the process for the identification of eligible randomized controlled trials. mTNBC, metastatic triple-negative breast cancer; RCTs, randomized controlled trials.
The main characteristics of the included studies are presented in Table 1. These RCTs were published between 2013 and 2019. The sample size ranged from 53 to 236. Among the included studies, 1 RCT 27 used carboplatin and three 17,26,28 used cisplatin as a platinum-based chemotherapeutic agent. Three of the included RCTs (N = 471 patients) compared gemcitabine plus platinum-based agents (cisplatin or carboplatin) with taxanes (paclitaxel or nab-paclitaxel [nab-P]) plus gemcitabine. 17,27,28 All RCTs reported ORRs in the 2 treatment arms; 3 studies 17,26,27 reported HRs and 95% CIs or survival curves for the PFS and OS, which can be extracted survival data used for pooled analysis of PFS and OS. Only 2 RCTs 17,27 reported specific rates of grade 3 and 4 AEs in the 2 treatment arms. A phase 2 trial performed by Yardley and Coleman 27 included patients who were randomly assigned to the following groups: nab-P plus carboplatin (arm 1, N = 64 patients), gemcitabine plus carboplatin (arm 2, N = 66 patients), and nab-P plus gemcitabine (arm 3, N = 61 patients). These authors only provided HRs and 95% CIs for the comparison of OS or PFS between arms 1 and 3; therefore, we extracted the data of arm 1 vs. arm 3 for pooled analysis of PFS and OS. For analyses of ORRs and safety, the data of arms 1 and 2 were integrated as the platinum-based group for subsequent analyses.
Main Characteristics of the Randomized Controlled Trials Included in the Present Meta-Analysis.

Abbreviations: ORR, objective response rate; PFS, progression-free survival; OS, overall survival; nab-P, nab-paclitaxel; AUC, area under the curve.
Quality Assessment
Quality assessment of the included RCTs is summarized in Figure 2. Three studies 17,26,27 did not use allocation concealment and blinding of participants, personnel, or outcome assessors; however, this is unlikely to have introduced an extensive bias because ORRs, PFS, OS, and safety are objective outcomes. A RCT performed by Mustafa et al 28 did not adequately describe the method for blinding and only reported the P value for PFS but without survival curves, HRs, and 95% CIs; therefore, this study was considered to show a high risk of bias.

Risk of bias summary using the Cochrane collaboration’s tool.
Objective Response Rates
All included studies (N = 590) reported the ORR, which was defined as the percentage of patients with complete or partial response between treatment initiation and disease progression or death. In the study performed by Yardley and Coleman, 27 we integrated the data of arms 1 and 2 as the platinum-based chemotherapy group for pooled analysis. A statistically significant improvement in ORRs was observed among patients treated with platinum-based regimens than among those treated with platinum-free regimens (62.7% vs. 43.1%, OR 2.34, 95% CI 1.66-3.28, P < 0.001), as shown in Figure 3. Moderate heterogeneity was observed among the included studies (I 2 = 40%, P = 0.17), and a random-effects model was used for analysis.

Forest plot showing pooled odds ratios for ORRs in first-line platinum vs non-platinum chemotherapy in patients with mTNBC. ORR, objective response rates; mTNBC, metastatic triple-negative breast cancer.
Progression-Free Survival
A study performed by Mustafa et al 28 reported only the P value rather than HRs, 95% CIs, or survival curves for an intergroup comparison of the PFS; therefore, this RCT was not included in the pooled estimate of PFS. Finally, 3 studies (N = 414 patients), which only included patients of arms 1 vs. 3 in the RCT described by Yardley and Coleman 27 were evaluated to determine the differences in the PFS between the 2 chemotherapeutic regimens. The PFS was significantly longer in patients administered platinum-based regimens than in those administered platinum-free regimens (HR 0.55, 95% CI 0.37-0.82, P = 0.004, Figure 4). Significant heterogeneity was observed across the included studies (I 2 = 58%, P = 0.09); therefore, we performed sensitivity analysis by successive exclusion of individual studies. Following the exclusion of the study reported by Fan et al, 26 the heterogeneity among the remaining studies decreased to 0% (P = 0.55), and the remaining studies showed significant improvement in PFS in the platinum-based arm (HR 0.66, 95% CI 0.52-0.84, P = 0.0006). Heterogeneity could be attributed to a small sample size (N = 53 patients) and a greater number of grade 3 tumors observed in the platinum-based arm (P = 0.014) in this study. 26
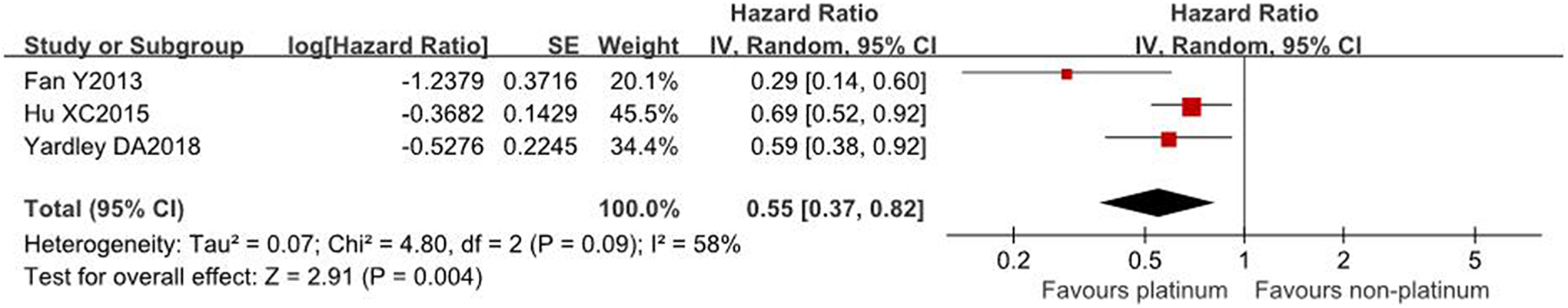
Forest plot showing pooled hazard ratios for PFS in first-line platinum vs. non-platinum chemotherapy in patients with mTNBC. PFS, progression-free survival; mTNBC, metastatic triple-negative breast cancer.
Overall Survival
Similar to the findings observed for the pooled estimate of PFS, 3 studies (N = 414 patients) estimated the pooled HRs of OS between the 2 arms. A trend toward better OS was observed among patients who received platinum-based regimens than among those who received platinum-free regimens; however, this difference was statistically nonsignificant (HR 0.76, 95% CI 0.57-1.00, P = 0.05, Figure 5), without significant heterogeneity observed across studies (I2 = 31%, P = 0.23).

Forest plot showing pooled hazard ratios for OS in first-line platinum vs non-platinum chemotherapy in patients with mTNBC. OS, overall survival; mTNBC, metastatic triple-negative breast cancer.
Grade 3 and 4 Adverse Events
Only 2 studies 17,27 reported the rates of grade 3 and 4 AEs, and similar to the pooled analysis of ORR, the data of arms 1 and 2 were integrated as the platinum-based group in the study performed by Yardley and Coleman. 27 Table 2 displayed the safety profile overview for grade 3 and 4 AEs in the platinum-based vs. platinum-free regimens group. In summary, there were no significant differences in grade 3 and 4 leucopenia, neutropenia, febrile neutropenia, anemia, peripheral neuropathy between the 2 groups. However, grade 3-4 thrombocytopenia was more common in platinum-based chemotherapy (OR 7.54, 95% CI 1.37-41.60, P = 0.02, I 2 = 77%), and grade 3-4 fatigue was more common in platinum-free chemotherapy (OR: 0.23, 95% CI: 0.08-0.68, P = 0.008, I 2 = 0%).
Summarized the Grade 3-4 Adverse Events.

Abbreviations: AEs, adverse events; OR, odds ratio; CI, confidence intervals.
Publication Bias
The publication biases were tested by Begg’s and Egger’s test. No significant publication biases were detected for OS (Begg’s test, P = 0.296; Egger’s test, P = 0.175) or PFS (Begg’s test, P = 0.296; Egger’s test, P = 0.160). However, the Egger’s test found significant publication bias for ORR (Begg’s test, P = 0.089; Egger’s test, P = 0.023).
Discussion
TNBC is one of particularly aggressive diseases, and because of limited treatment options, it remains a therapeutic challenge. Moreover, due to the lack of phase 3 data, there are no recognized standards of care or guidelines for the treatment of advanced TNBC. The substantial overlap between TNBC and BRCA1-mutated breast cancer has been used to highlight the potential value of platinum salts. 14,29 Previous studies have demonstrated a significant improvement in pCR rate in patients with stage I-III TNBC to receive neoadjuvant chemotherapy with platinum agents, but the survival benefit is still uncertain, the current standard therapeutic agents for patients with non-mTNBC remain taxanes and anthracyclines. 14,15 Most metastatic breast cancer present as distant recurrence of initially localized invasive breast cancer (stage I-III) and just 6%-7% present with de novo metastatic disease, 30,31 so mTNBC is usually refractory to previous agents such as taxanes and anthracyclines. Researchers of Poland have observed a high frequency of BRCA1 mutations in metastatic breast cancer patients with primary resistance to docetaxel-based chemotherapy. 32 Platinum-based salvage chemotherapy has been reported in any subtype anthracyclines/taxanes-resistant metastatic breast cancer, with an OR rates of 26%-50% and a median OS of 8-13 months. 19,33 Given the above findings, platinum-based regimens may be a better choice in patients with mTNBC. In the present meta-analysis, we observed that first-line platinum-based chemotherapy was associated with significantly higher ORRs and improved PFS in patients with mTNBC; however, no significant difference was observed in OS. To our knowledge, this is the largest and the most current meta-analysis that evaluated the efficacy and safety of platinum-based chemotherapy as first-line treatment for patients with mTNBC.
In a meta-analysis that assessed platinum-based systemic treatment for patients with TNBC, 3 RCTs 17,18,26 (N = 531 patients) included in a subgroup analysis for patients with mTNBC. In contrast to our study, the results of this meta-analysis did not show better PFS in patients who received platinum-based systemic chemotherapy (HR 1.16, 95% CI 0.90-1.49, P = 0.24). 34 The discrepancy in results could be attributed to the inclusion of the study performed by Carey et al 18 in the meta-analysis, in which cetuximab alone was compared with cetuximab plus carboplatin as non-first-line treatment for patients with mTNBC. Their study reported that the response rate was only <20% in patients treated with cetuximab plus carboplatin. Another meta-analysis that assessed the effectiveness of platinum-based chemotherapy for patients with mTNBC regardless of the study design and administration of first or non-first-line chemotherapeutic regimens included 7 studies (3 retrospective trials, N = 1,571 patients) and confirmed that compared with platinum-free chemotherapy, platinum-based chemotherapy was associated with better OS and PFS rates. 35 Similar results were reported by a retrospective study in Canada; the authors compared the efficacy of platinum-based chemotherapy with conventional platinum-free regimens in patients with mTNBC and observed that the OS rates were better in patients treated with platinum-based chemotherapy than in those treated with conventional therapy (14.5 months vs. 10 months, P = 0.041). 36 However, only 58 patients received platinum-based chemotherapy in this study. Several studies did not support the role of platinum in OS benefit. In a recent multicenter real-world study in China, the authors retrospectively analyzed the medical records of 495 patients with advanced TNBC and observed that first-line platinum-based chemotherapy was superior to platinum-free chemotherapy with regard to ORRs (53.0% vs. 32.1%, P < 0.001) and the median PFS (8.4 months vs. 6.0 months, P = 0.022); however, OS rates was similar (19.2 months vs. 16.8 months, P = 0.439). 37 These results were consistent with our findings which observed longer PFS but no significant difference in OS in patients who received platinum-based chemotherapy.
TNBC is a heterogeneous disease, some studies have suggested that not all patients with TNBC are suitable for platinum-based chemotherapy. 38 The phase III TNT trial 39 compared docetaxel with carboplatin as first-line chemotherapy in women (N = 376 patients) with advanced TNBC or BRCA1/2 mutations. The results of the study showed that carboplatin was not more effective than docetaxel (ORR 31.4% vs. 34.0%, P = 0.66) in the unselected population, although patients with germline BRCA1/2 (gBRCA1/2) mutations showed a significantly better response to carboplatin than docetaxel (ORR 68.0% vs. 33.3%, P = 0.03). Furthermore, PFS was also improved with carboplatin treatment in patients with gBRCA1/2 mutations (median PFS 6.8 months vs. 4.4 months, P = 0.002), although no significant difference was observed in OS. The TBCRC009 40 and PrECOG0105 trials 41 also suggested that platinum-based chemotherapy showed better results in patients with TNBC diagnosed with gBRCA1/2 mutations. The National Comprehensive Cancer Network Panel has included platinum-based agents (cisplatin and carboplatin) as preferred therapeutic agents for patients with recurrent/stage IV TNBC and gBRCA1/2 mutations. 42
To our knowledge, the CBCSG006 trial 17 was the largest randomized, multicenter phase III clinical trial that investigated the efficacy of cisplatin combination therapy in patients with mTNBC and reported that GP treatment significantly prolonged PFS compared with GT treatment (median PFS 7.73 months vs. 6.47 months, HR 0·692, 95% CI 0·523-0·915, P = 0·009). As mentioned earlier, TNBC is characterized by abnormal DNA repair systems caused by HR deficiency (HRD). 7 For biomarker assessment in the CBCSG006 trial, patients diagnosed with HRD had significantly higher ORRs and longer PFS in the GP arm than in the GT arm (ORR 71.9% vs. 38.7%, P = 0.008, median PFS 10.37 months vs. 4.30 months, P = 0.011), whereas no significant differences in ORRs and PFS were observed in patients without HRD and no significant difference was observed in the OS between the 2 arms regardless of the HR status. 43 Recent genome-wide gene expression and DNA sequencing studies showed that TNBC is composed of a molecularly heterogeneous group of diseases with multiple somatic mutations and genomic structural changes, 7 which can be divided into at least 6 subtypes, including basal-like (BL1 and BL2) subtypes, immunomodulatory, mesenchymal, mesenchymal stem-like, luminal androgen receptor, and unstable types. 44 The BL1 subtype accounts for about 18% of TNBCs, 45 which is associated with a high expression of DNA damage response and cell cycle genes, and is known to respond to platinum-based agents. 4 Given the heterogeneity across patients, not all patients with TNBC might benefit from platinum-based chemotherapy. 38 Therefore, in order to identify new therapeutic targets and to recognize specific subgroups that might particularly benefit from the use of platinum, we need to have a better understanding of the potential biology of TNBC.
In addition, different platinum drugs may affect the efficacy of patients. In a retrospective study, investigators found when compared with other kinds of platinum drugs, cisplatin-based regimens as the first-line chemotherapy in patients with mTNBC showed better PFS (8.0 vs. 4.3 months, P = 0.03) and better ORR (59.6% vs. 35.0%, P = 0.03). 16 However, of the 218 patients enrolled in the platinum-based group, only 20 patients used other kinds of platinum drugs. Huang L et al 46 retrospectively analyzed 145 TNBC cases to compared the activity and tolerability of cisplatin and carboplatin in combination with paclitaxel as neoadjuvant regimen, and observed that there was no significant difference between the groups in pCR rates and survival. In our study, we cannot do further subgroup analysis to compare the efficacy of different platinum drugs due to the small number of studies included. An ongoing prospective phase 2 randomized trial (NCT02341911) has been designed to compare the efficacy and safety of gemcitabine plus cisplatin with gemcitabine plus carboplatin as the first-line treatment for patients with mTNBC and will hopefully provide a definitive answer to the question of the efficacy of different platinum drugs.
In this study, we also focused on the AEs of systemic therapy administered to patients with mTNBC and observed no significant differences in grade 3 and 4 leukopenia, neutropenia, febrile neutropenia, anemia, and peripheral neuropathy between platinum-based and platinum-free chemotherapy. However, grade 3-4 thrombocytopenia was more commonly associated with platinum-based chemotherapy (P = 0.02) and grade 3-4 fatigue with platinum-free chemotherapy (P = 0.008). The tnAcity trial 27 reported that AEs severe enough to warrant discontinuation or dose interruption of any study drug were more common in patients who received platinum-based regimens. Therefore, optimal patient selection, appropriate counseling regarding the merits and demerits of adding a platinum agent as first-line therapy and close clinical follow-up during treatment are essential in these patients.
Following are the limitations of our study that should be considered in the interpretation of our results: (a) This meta-analysis was based on abstracted data because individual patient data were unavailable; therefore, the association between treatment benefit and toxicity and other factors (including patient age, drug dose, and duration of chemotherapy) could not be evaluated. (b) This meta-analysis included only 4 RCTs, some of which were relatively small-sized studies. Although these RCTs were prospective well-conducted trials, the results should be interpreted with caution because overestimation of treatment effects is a known drawback of small-sized studies. (c) The platinum agents included cisplatin and carboplatin, lack of subgroup analyses compared the differences of cisplatin vs. carboplatin because just 4 studies were included in this meta-analysis. (d) Only 2 studies reported the rates of grade 3 and 4 AEs, and AEs were not uniform across studies; therefore, several AEs (such as gastrointestinal toxicities) were not analyzed. However, these limitations would not affect the overall interpretation of our results, which provide valuable information regarding the questionable role of platinum agents added to standard first-line chemotherapeutic regimens in patients with mTNBC. Specifically, these findings provide updated point estimates regarding the risk-benefit ratio of including a platinum agent for first-line systemic chemotherapy and may serve as guidelines for physicians and patients during treatment decision-making.
Conclusions
In conclusion, this meta-analysis highlights that first-line platinum-based chemotherapy was associated with significantly increased ORRs, PFS, and a nonsignificant trend toward improved OS in patients with mTNBC; however, this approach was associated with the high risk of grade 3-4 thrombocytopenia. Further studies are warranted to identify better predictive biomarkers to improve the efficacy of platinum-based therapy and to confirm the efficacy and safety of combinations of modern therapies such as targeted or immunotherapy in patients with mTNBC.
Supplemental Material
Supplemental Material, sj-pdf-1-tct-10.1177_15330338211016369 - Efficacy and Safety of Platinum-Based Chemotherapy as First-Line Therapy for Metastatic Triple-Negative Breast Cancer: A Meta-Analysis of Randomized Controlled Trials
Supplemental Material, sj-pdf-1-tct-10.1177_15330338211016369 for Efficacy and Safety of Platinum-Based Chemotherapy as First-Line Therapy for Metastatic Triple-Negative Breast Cancer: A Meta-Analysis of Randomized Controlled Trials by Fei Lu, Yu Hou, Zhengting Chen, Jie Jiang, Xi He, Yaoxiong Xia, Ke Cao, Li Chang and Wenhui Li in Technology in Cancer Research & Treatment
Footnotes
Abbreviations
Authors’ Note
Conception, design and data analysis: LF, CL, LW, HY and JJ. Interpretation of data: CL and HX. Initial manuscript writing: LF, CZ, XY and CK. Revision of manuscript: CL, HX, JJ and LW. All authors read and approve the final manuscript. Fei Lu, Yu Hou, Zhengting Chen and Jie Jiang contributed equally to this study and share first authorship. The need for ethics approval by an institutional board review was waived as this study does not directly involve human subjects.
Acknowledgments
We thank “SAGE Author Services” for language editing and correction.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Key Research and Development Program of China (No.2018YFC1311400/2018YFC1311402), Ten-thousand Talents Program of Yunnan Province (Yunling scholar, Youth talent), Yunnan Provincial Training Funds for Middle-Young Academic and Technical Leader candidate (202005AC160025), Yunnan Provincial Training Funds for High-level Health Technical Personnel (No.L-2018001), Cancer Research Program of National Cancer Center (No. NCC2017A32). The funding body participates in data collection and software support.
Supplemental Material
The supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
