Abstract
Keywords
Introduction
Gastric and gastroesophageal junction (G/GEJ) cancer is one of the most common malignancies worldwide, with an estimated 1,089,103 new cases and 768,793 deaths worldwide in 2020. 1 The symptoms are not obvious and difficult to find in the early stage of G/GEJ cancer, so most patients are diagnosed at an advanced stage. Chemotherapy is conventional treatment for advanced G/GEJ cancer. The combination of platinum compounds plus fluoropyrimidines chemotherapy is currently the global standard for the first-line treatment of advanced G/GEJ cancer.2,3 However, chemotherapy has reached a plateau, due to limited clinical benefit, and the prognosis of advanced G/GEJ cancer is still poor. The median OS for advanced-stage GC treated with conventional chemotherapy is less than one year. 4 Programmed death 1 (PD-1), programmed death ligand 1 (PD-L1) and cytotoxic T lymphocyte associated antigen 4 (CTLA-4) represent three targetable immune checkpoints. Recently, immune checkpoint inhibitor (ICI) has been shown to provide antitumor activity with a manageable safety profile in patients with G/GEJ cancer,5–7 and has revolutionized the treatment of some solid and hematological cancers. ICI works by blocking the binding of ligands to checkpoint receptors and re-activating the human cellular immune response. 8 Some evidence suggested that chemotherapy can enhance tumor immunogenicity and increase susceptibility to immune attack, potentially increasing the efficacy of ICI. 9
Previously, pembrolizumab was approved in the U.S. as second-line or later therapy for the treatment of solid tumors with microsatellite instability-high or mismatch repair deficient or tumor mutational burden-high (≥ ten mutations / megabase). 10 Nivolumab has been approved in China for the treatment of advanced or recurrent gastric cancer in patients who have previously received two or more systemic treatment regiments. 11 Also, the U.S Food and Drug Administration (FDA) approved nivolumab in combination with fluoropyrimidine- and platinum-containing chemotherapy for treating advanced or metastatic G/GEJ cancer and EC. 7 Even more remarkably, on May 5, 2021, the FDA granted accelerated approval to pembrolizumab in combination with trastuzumab, fluoropyrimidine- and platinum-containing chemotherapy for the first-line treatment of patients with locally advanced unresectable or metastatic HER2-positive G/GEJ cancer. 12 Approval was based on the prespecified interim analysis of the first 264 patients of the ongoing KEYNOTE-811 trial. 13 The main efficacy measure for this analysis was overall response rate (ORR) assessed by blinded independent review committee. The ORR was 74% in the pembrolizumab arm and 52% in the placebo arm. 13 The adverse reaction profile observed in patients receiving pembrolizumab was consistent with the known pembrolizumab safety profile. 13 Patients’ survival data were currently not available for this study.
Although the combination of ICI and chemotherapy is promising, there remain some unanswered questions. For example, will combined regimen improve patient outcomes at the expense of increased toxicity? Will the combined regimen prolong patient progression free survival (PFS) or overall survival (OS)? The dimension of immunotherapy plus chemotherapy benefits remains controversial. Therefore, we performed a meta-analysis to evaluate efficacy and safety of PD-1/PD-L1 inhibitor plus chemotherapy as first-line treatment for advanced G/GEJ cancer.
Material and Methods
This systematic review and meta-analysis were conducted in accordance with Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. 14 The protocol was registered in PROSPERO (No. CRD42022300907).
Search Strategy
PubMed, Embase, Cochrane Library, and Web of Science were searched by two independent researchers from the inception of the databases to December 2021. The search strategy included key search terms: (“gastric cancer” or “gastric carcinoma” or “stomach cancer” or “gastric adenocarcinoma” or “cancer of the stomach” or “adenocarcinoma of the stomach” or “stomach neoplasm” or “gastric neoplasm” or “gastroesophageal junction cancer” or “gastroesophageal junction carcinoma” or “adenocarcinoma of the gastroesophageal junction”) and (“nivolumab” or “pembrolizumab” or “toripalimab” or “sintilimab” or “camrelizumab” or “tislelizumab” or “penpulimab” or “zimberelimab” or “cemiplimab” or “dostarlimab” or “durvalumab” or “avelumab” or “atezolizumab” or “lambrolizumab” or “pidilizumab” or “PD-1” or “PD-L1” or “programmed cell death 1 receptor” or “programmed cell death ligand-1” or “immune checkpoint inhibitor” or “immune checkpoint blockade” or “immunotherapy”) and (“first line” or “first-line” or “1 line” or “untreated” or “previously untreated”).
Study Eligibility and Identification
Inclusion criteria: (1) randomized controlled trials (RCTs); (2) previously untreated, advanced or metastatic GC/GEJC patients; (3) a PD-1/PD-L1 inhibitor plus chemotherapy as the experimental arm (ICI-chemotherapy); (4) chemotherapy plus placebo or chemotherapy alone as the control arm for comparison (chemotherapy); (5) inclusion of one or more of the following outcomes: overall survival (OS), progression-free survival (PFS), objective response rate (ORR) or adverse events (AEs) were available; (6) published articles were reported in English.
Exclusion criteria: (1) case, review or editorial etc; (2) insufficient data available to estimate the outcomes; (3) studies that involved ICI in both the experimental and control arms. If trials had multiple publications, the latest and most complete report was adopted.
Data Extraction and Risk of Bias Assessment
All citations were imported into Endnote X9, and duplicate records were removed electronically or manually. Two authors screened the remaining articles at the title and abstract level followed by full text. The reference list of relevant systematic reviews and meta-analysis was scanned to identify potentially eligible studies. The following information was extracted from the eligible studies by two independent authors: abbreviation of study, publication year, age, gender, phase, cancer type, ICI, and follow-up. Duplicates were removed before screening the titles and abstracts. Then, the full text of remaining studies was assessed according to the inclusion and exclusion criteria. The Cochrane risk of bias assessment tool was used to determine the methodological quality of included RCTs. 15 The judgments for each domain were made strictly by following the Cochrane Hand-book. Disagreements in the above parts were resolved through group discussions.
Statistical Analysis
For the assessment of efficacy, the primary outcome were OS, PFS and ORR. The safety outcome was adverse events (AEs), which included all AEs, all AEs, treatment-related adverse event (TRAE), TRAE of grade 3 or higher (grade ≥3 TRAE) and TRAE leading to death. The meta-analysis was conducted to compare the efficacy and safety of ICI-chemotherapy with chemotherapy. The pooled estimates for PFS and OS were presented with HRs, 95% CIs and P values, while the measures for dichotomous data (ORR and frequency of adverse events) were pooled with the risk ratios (RRs), 95% CIs and P values. If there was no significant heterogeneity (P < .05 was considered heterogeneous) between studies, a fixed-effects model was used; otherwise (I2 > 50% was considered heterogeneous), a random-effects model was used.
Subgroup analysis were done to determine associations between OS, PFS and ORR outcomes and some patient characteristics, including CPS, Tumor cell proportion score (TPS), HER2 expression, microsatellite instability, which may be possible sources of heterogeneity. Publication bias was evaluated by examining the funnel plot of the effect size for each trial against the reciprocal of its standard error, together with the Egger's test.16,17 We defined significant publication bias as P < .05. We used Stata version 14.0 (Stata Corporation, College Station, TX) for all of the analyses. The nominal level of significance was set at 0.05.
Results
Of the 1, 115 studies identified from database searches, five trials13,18–21 involving 2, 814 untreated, advanced or metastatic G/GEJ cancer patients were included for data analysis. The study protocol was registered with PROSPERO, number CRD42022300907. The characteristics reported in the included RCTs were presented in Table 1. The selection process for included studies was shown in a flow diagram (Figure 1). The overall quality of the included studies was high and the assessment of risk of bias was provided in Table 2.

Studies selection process.
Characteristics of Included Trials.
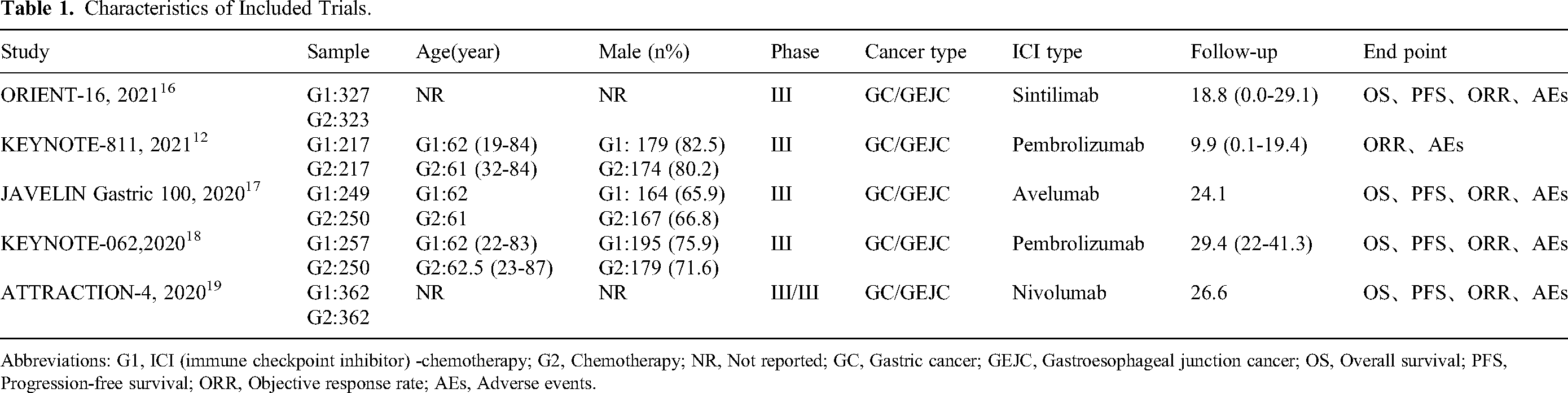
Abbreviations: G1, ICI (immune checkpoint inhibitor) -chemotherapy; G2, Chemotherapy; NR, Not reported; GC, Gastric cancer; GEJC, Gastroesophageal junction cancer; OS, Overall survival; PFS, Progression-free survival; ORR, Objective response rate; AEs, Adverse events.
Quality Assessment: Risk of Bias by Cochrane Collaboration Tool.
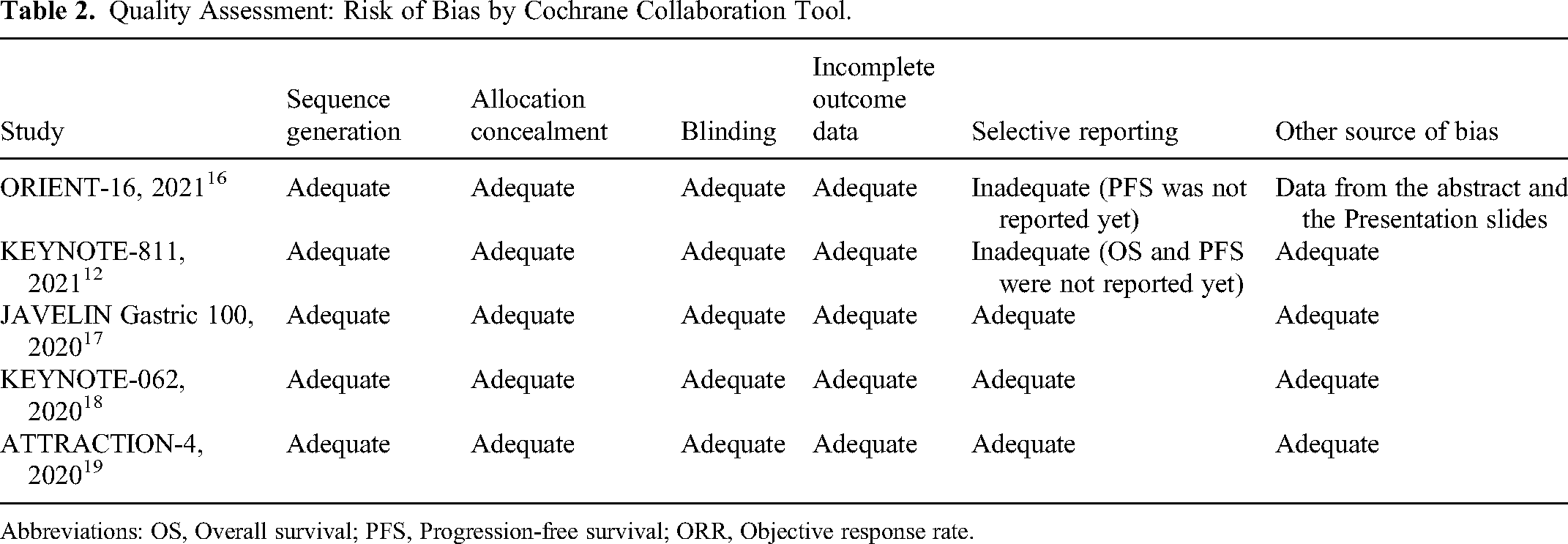
Abbreviations: OS, Overall survival; PFS, Progression-free survival; ORR, Objective response rate.
Efficacy of ICI-Chemotherapy
Data of OS were available in 4 studies, and the pooled result showed that ICI-chemotherapy significantly improved OS compared with chemotherapy (HR, 0.86; 95% CI 0.78-0.94; P = .002) (Figure 2a). There was no significant heterogeneity in the over-all treatment effect in terms of OS across the four trials (I2 = 0%, P = .611). The funnel plot for the OS revealed no asymmetry (Supplementary Figure 1a; Egger's test P = .676), indicating no obvious publication bias regarding OS.

Forest plot of hazard ratio and risk ratio comparing (2a) overall survival (OS), (2b) progression-free survival (PFS), and (2c) objective response rate (ORR) in patients who received ICI-chemotherapy versus chemotherapy.
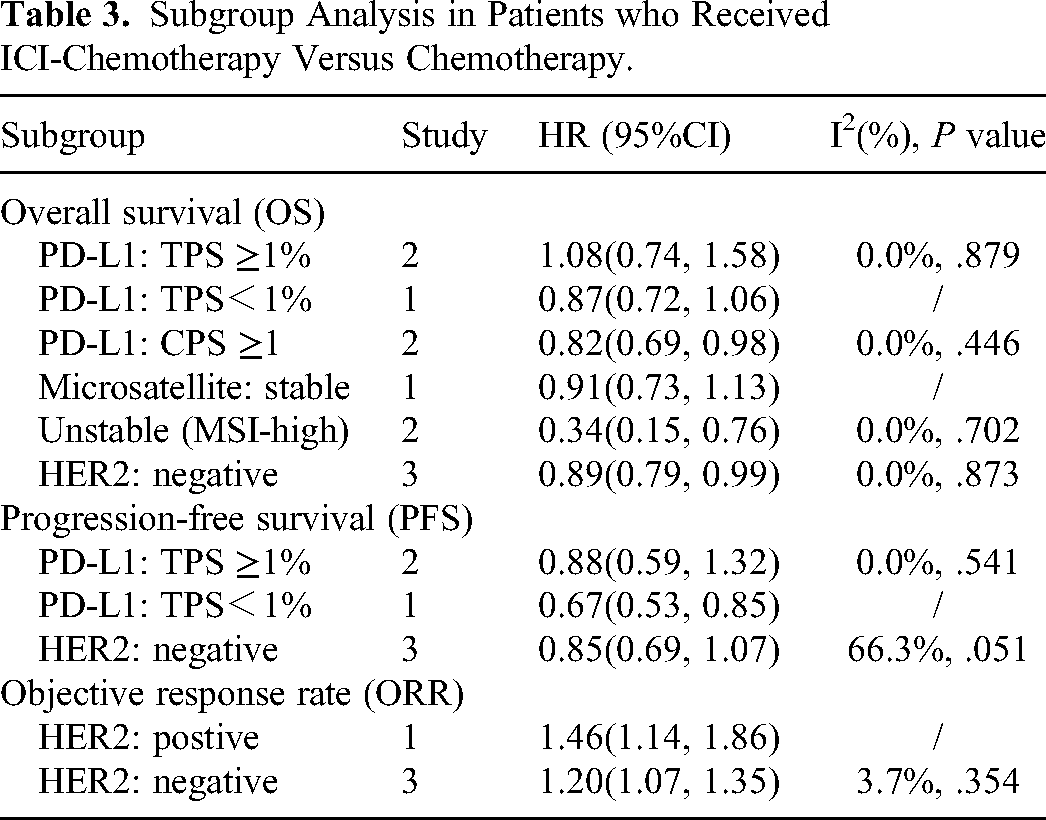
The subgroup analysis of PD-L1 expression showed that ICI-chemotherapy was found to prolong OS for patients with CPS ≥1 compared with chemotherapy (HR, 0.82; 95%CI, 0.69-0.98; P = .025). ICI-chemotherapy versus chemotherapy alone did not show a significant improvement in OS in patients with PD L1 TPS ≥1% (HR, 1.08; 95% CI, 0.74-1.58; P = .687) or <1% (HR, 0.87; 95% CI, 0.72-1.06; P = .158). ICI-chemotherapy had significantly better OS than chemotherapy in HER2-negative subgroup (HR, 0.89; 95% CI, 0.79-0.99; P = .033) and in microsatellite instability-high (MSI-H) subgroup (HR, 0.34; 95% CI, 0.15-0.76; P = .009), while ICI-chemotherapy did not show an OS advantage in microsatellite stable (MSS) subgroup (HR, 0.91; 95% CI, 0.73-1.13; P = .388). The results are showed in Table 3.
Subgroup Analysis in Patients who Received ICI-Chemotherapy Versus Chemotherapy.
In terms of PFS benefit, the ICI-chemotherapy combination led to a reduction in the risk of tumor progression compared with chemotherapy (HR, 0.79; 95% CI 0.63-0.99; P < .0001) (Figure 2b). The funnel plot revealed no asymmetry (Supplementary Figure 1b; Egger's test P = .993), indicating no obvious publication bias regarding FPS. There was significant heterogeneity in the over-all treatment effect in terms of PFS across the four trials (I2 = 77.4%, P = .004).
ICI-chemotherapy had improved PFS compared with chemotherapy in patients with PD-L1 TPS <1% (HR, 0.67; 95% CI, 0.53-0.85; P = .001), but not in patients with TPS ≥1% (HR, 0.88; 95% CI, 0.59-1.32; P = 0.540). In HER2-negative subgroup, ICI-chemotherapy showed no PFS advantage compared to chemotherapy (HR, 0.85; 95% CI, 0.69-1.07; P = .163). The results are showed in Table 3.
Data of ORR were available in 5 studies, and the ORR of ICI-chemotherapy was also significantly improved (RR, 1.2; 95% CI, 1.11-1.30; P < .001) (Figure 2c). No significant heterogeneity was observed in the analysis of ORR (I2 = 35.3%, P = .186). The funnel plots of the ORR revealed no asymmetry (Supplementary Figure 1c; Egger's test P = .453).
AS for subgroup analysis of HER2 status, ORR of ICI-chemotherapy were also superior to chemotherapy in either HER2-negative subgroup (HR, 1.20; 95% CI, 1.07-1.35; P = 0.001) or in HER2-positive subgroup (HR, 1.46; 95% CI, 1.14-1.86; P = .003). The results are showed in Table 3.
Safety Analyses
The additional characteristics of patients were summarized in Supplementary Table 1. and the number of patients included for each safety analysis was presented as following. Data of safety were available in 5 studies: all AEs (3 studies), TRAE (4 studies), grade ≥3 TRAE (2 studies), serious TRAE (2 studies), TRAE leading to death (5 studies). There was no significant difference in the frequency of All AEs (RR, 1.00, 95% CI 0.97-1.03, P = .994; I2 = 34.4%, P = .218), TRAE (RR, 0.99, 95% CI 0.92-1.07, P = .859; I2 = 80.4%, P = .002), Grade ≥3 TRAE (RR, 0.69, 95% CI 0.23-2.07, P = .965; I2 = 96.7%, P < .001), serious TRAE (RR, 1.25, 95% CI 0.61-2.54, P = .103; I2 = 78.7%, P = .030) and TRAE leading to death (RR, 1.01, 95% CI 0.54-1.89, P = 0.899; I2 = 0.0%, P = .497), respectively. Forest plot of risk ratio comparing adverse events (AEs) in patients who received ICI-chemotherapy versus chemotherapy was shown in Figure 3.

Forest plot of risk ratio comparing adverse events (AEs) in patients who received ICI-chemotherapy versus chemotherapy after sensitivity analysis.
Discussion
Although chemotherapy regimen has been improved over the past few decades, it is not enough to improve the prognosis of advanced G/GEJ cancer. Subsequently, targeted therapy has become a breakthrough direction in the treatment of G/GEJ cancer, but a large number of clinical research results are disappointing. Recently, with the development of the comprehensive treatment, PD-1/PD-L1 inhibitors become a hot topic in the field of G/GEJ cancer. Due to the high heterogeneity and poor prognosis of G/GEJ cancer, immunotherapy may bring hope for G/GEJ cancer patients. Immunotherapy has been previously shown to be effective for second-line and above treatment of G/GEJ cancer.5,6,11 Several ICIs in combination with chemotherapy have greatly enriched the choices for first-line treatment of G/GEJ cancer.11,13,22 A growing body of evidence has shown that drugs such as cisplatin, oxaliplatin, and paclitaxel, can up-regulate PD-L1 expression in tumor cells and immune cells, therefore providing an opportunity for the use of PD-1/PD-L1 inhibitors.23,24 However, the safety and efficacy of immunotherapy in the first-line treatment of advanced G/GEJ cancer lack sufficient evidence-based clinical evidence.
The results of KEYNOTE-062 study 20 found that pembrolizumab or pembrolizumab plus chemotherapy was not superior to chemotherapy for the OS and PFS end points tested. What's the future of chemotherapy combined with immunotherapy? Until 2020, the CheckMate-649 study 7 allowed the long-awaited immunotherapy to enter the first line of advanced gastric cancer. This study proved that nivolumab was the first PD-1 inhibitor to show superior OS, along with PFS benefit and an acceptable safety profile, in combination with chemotherapy versus chemotherapy alone in previously untreated patients with advanced G/GEJ cancer, or oesophageal adenocarcinoma. Based on the differences of the above two research results, the role of immunotherapy in first-line treatment of gastric cancer is still unclear, and some new studies continue to appear.
On this basis, we conducted this meta-analysis of these studies for the first time to evaluate the safety and efficacy of ICI plus chemotherapy. The main findings of our study were listed as follows: 1) ICI-chemotherapy was significantly associated with improvement of OS; 2) ICI-chemotherapy was significantly associated with improvement of PFS; 3) ICI-chemotherapy was significantly associated with improvement of ORR; 4) There were no significant differences in incidence of all AEs, TRAE, Grade ≥3 TRAE, serious TRAE and TRAE leading to death. These findings provide theoretical basis for ICI-chemotherapy in the first-line treatment of G/GEJ cancer without increasing TRAE.
PD-L1 overexpression has been observed in G/GEJ cancer tissues, and PD-L1 has become one of the more mature biomarkers for immunotherapy. TPS and CPS are commonly used to calculate the expression level of PD-L1 in tumor tissues. 25 The difference between the two is that CPS reflects not only the expression of PD-L1 in tumor cells, but also the expression of PD-L1 in immune cells. PD-L1 expression has been shown to correlate with efficacy of pembrolizumab monotherapy in gastric cancer.20,26,27 It will be informative to see whether the relative benefit of ICI plus chemotherapy for overall survival will be impacted by PD-L1 expression. A greater difference was found in ORR in participants with PD-L1 CPS ≥1, although the 95% CI for the CPS ≥1 and <1 subgroups overlapped in KEYNOTE-811 study. 13 ICIs, including pembrolizumab, nivolumab and avelumab, in combination with chemotherapy showed no advantages in OS and PFS in patients with either PD-L1 TPS ≥1% or CPS ≥1 compared with chemotherapy.19–21 Sintilimab plus chemotherapy was found to prolong OS in G/GEJ cancer patients with PD-L1 CPS ≥1, 18 and the ATTRACTION-4 trial also demonstrated nivolumab plus chemotherapy can prolong PFS in G/GEJ cancer patients when PD-L1 TPS <1%. 21 Nonetheless, OS and PFS were shown to be also superior with ICI plus chemotherapy versus chemotherapy in G/GEJ cancer patients with PD-L1 CPS ≥5. 18 For patients with PD-L1 CPS ≥10, OS benefit of sintilimab plus chemotherapy was also observed, 18 whereas pembrolizumab plus chemotherapy did not show a significant improvement in OS. 20 Whether PD-L1 expression can effectively predict the efficacy of ICI plus chemotherapy is uncertain. Patients with high level of PD-L1 expression appear to have a greater survival benefit. Due to the high heterogeneity of gastric cancer, the expression of PD-L1 may be different in different locations of the same lesion and among different lesions. Currently, there is no unified standard for detection platform, detection method, detection threshold and staining system, which also limits the clinical application of PD-L1 as a predictor of ICIs efficacy. According to the results of clinical studies, the clinical significance of the CPS score and its optimal cutoff for immune checkpoint inhibitors, in combination with chemotherapy, are yet to be established.
ICI plus chemotherapy may be the optimal regimen for first-line unselected patients without increasing security risk. Although our systematic review was rigorously designed and conducted, there were still some limitations that should be taken into consideration when interpreting the findings. First, all the included studies were ICI combined with chemotherapy, except for The JAVELIN Gastric 100, which was switched from first-line chemotherapy to ICI maintenance therapy. However, sensitivity analysis did not affect the results of the study. Second, although this systematic review included as many eligible literatures as possible, the number of included trials still needs to be increased by future updates. Future studies are needed to explore reliable predictors of treatment efficacy and to determine which chemotherapeutic modality will improve patient's survival in combination with PD-1/PD-L1 inhibitor. However, more well-controlled randomized clinical trials are urgently needed in order to further research.
Conclusion
ICI plus chemotherapy can not only prolong OS, especially for patients with CPS ≥1, HER2-negative MSI-H, but also improve ORR and extend PFS in first-line treatment for advanced G/GEJ cancer in contrast to chemotherapy, without increasing TRAE. This study could redefine the role of ICI in combination with chemotherapy in the first-line setting for G/GEJ cancer, and provide effective and reliable information for clinical.
Supplemental Material
sj-docx-1-tct-10.1177_15330338241273286 - Supplemental material for Immune Checkpoint Inhibitor Plus Chemotherapy as First-Line Treatment for Advanced Gastric or Gastroesophageal Junction Cancer: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-1-tct-10.1177_15330338241273286 for Immune Checkpoint Inhibitor Plus Chemotherapy as First-Line Treatment for Advanced Gastric or Gastroesophageal Junction Cancer: A Systematic Review and Meta-Analysis by Lianghui Zhang, MS, Lingli Huang, MS, Zhixian Liu, PhD, and Tao Ling, MS in Technology in Cancer Research & Treatment
Footnotes
Availability of Data
The data that support the findings of this study are available from the corresponding author.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Research Project of Jiangsu Cancer Hospital, (grant number No. ZJ202222).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
