Abstract
Purpose:
Induction chemotherapy plus concurrent chemoradiotherapy and concurrent chemoradiotherapy alone are both standard treatment regimens for managing locally advanced nasopharyngeal carcinoma. However, the results of comparisons between them in clinical trials vary. Therefore, we designed this meta-analysis to illustrate their advantages and disadvantages in patients with locally advanced nasopharyngeal carcinoma.
Methods:
We thoroughly searched the PubMed, EMBASE, and Cochrane Library databases and then merged the effect indicators of hazard ratios and risk ratios using RevMan 5.1.
Results:
Seven randomized controlled trials totaling 2,319 patients were included in our research. The synthesized results showed that induction chemotherapy plus concurrent chemoradiotherapy improved overall survival (HR = 0.75, 95% CI: 0.63-0.89, P = 0.001), progression-free survival (HR = 0.69, 95% CI: 0.60-0.80, P < 0.001), distant metastasis-free survival (HR = 0.65, 95% CI: 0.53-0.80, P < 0.001) and locoregional recurrence-free survival (HR = 0.68 95%, CI: 0.54-0.86, P = 0.001) versus concurrent chemoradiotherapy alone. It also increased the risk of anemia, thrombocytopenia, and neutropenia during concurrent chemoradiotherapy. However, the incidence of leukopenia and mucositis was similar in induction chemotherapy and induction chemotherapy plus concurrent chemoradiotherapy. Furthermore, the subgroup analysis showed better survival outcomes with induction chemotherapy plus concurrent chemoradiotherapy than with concurrent chemoradiotherapy alone in the triweekly cisplatin subgroup (all P < 0.01), whereas induction chemotherapy plus concurrent chemoradiotherapy could only improve progression-free survival and locoregional recurrence-free survival in the weekly cisplatin subgroup (HR = 0.78, P = 0.02; and HR = 0.66, P = 0.03, respectively).
Conclusions:
Induction chemotherapy plus concurrent chemoradiotherapy improved survival outcomes in patients with locally advanced nasopharyngeal carcinoma versus concurrent chemoradiotherapy. For the weekly cisplatin regimen subgroup, it did not improve remote control or overall survival versus concurrent chemoradiotherapy alone, warranting further clarification.
Introduction
Nasopharyngeal carcinoma (NPC) is a malignant tumor that originates in the epithelium and glands of the nasopharynx. According to global cancer statistics from 2018, the number of new cases was 1,290,79 (0.7%), and the number of deaths was 72,987 (0.8%). 1 NPC has an unbalanced geographical distribution and is mainly distributed in Asia (81%); 38% of new NPC cases occur in China and are concentrated in Guangxi, Guangdong, Hunan, Jiangxi, and Hainan. 2 -4 The occurrence of NPC is closely related to Epstein-Barr virus (EBV) infection. Due to its special anatomical relationship and sensitivity to radiation, radiotherapy is preferred. As most patients are diagnosed with a locally advanced stage at the time of initial diagnosis, platinum-based concurrent chemoradiotherapy (CCRT) has been considered to be a standard treatment for these patients after the 0099 clinical trial, but the main cause of treatment failure is still distant and local recurrence. 5 -8
Therefore, the strategy of introducing induction chemotherapy (IC) based on CCRT to reduce micrometastases and thereby improve the overall survival (OS) of patients was proposed. In recent years, many teams have conducted a series of explorations and upgraded the treatment regimen of IC plus CCRT without adjuvant chemotherapy to the category IIA recommendation evidence of the National Comprehensive Cancer Network (NCCN version 2.2020) guidelines. 9
However, the efficacy and toxicity of the IC-plus-CCRT regimen is still controversial. Hence, the purpose of this meta-analysis was to verify whether IC plus CCRT can prolong the survival outcomes of patients with locally advanced NPC and assess its associated treatment-related grade 3 or 4 adverse events compared with the CCRT regimen.
Materials and Methods
Information Retrieval Strategy
Published studies were searched in PubMed, EMBASE, and the Cochrane Library, with the search time from inception to December 1, 2019. The search terms, which were connected using “AND” and “OR,” included “nasopharyngeal carcinoma,” “concurrent chemoradiotherapy,” “neoadjuvant therapy,” “induction chemotherapy” and “randomized controlled trial (RCT).” Two team members (Mr Xu and Ms Wang) independently searched for eligible articles in the above databases.
Inclusion Criteria
The included clinical trials were required to meet the conditions of the participants, interventions, comparisons, outcomes, and study design (PICOS) principles. The participants needed to satisfy the following conditions: (1) age at least 18 years old; (2) newly diagnosed locally advanced NPC (clinical stage III-IV); and (3) no distant metastasis. Regarding the interventions, patients received IC plus CCRT without adjuvant chemotherapy. Interventions should be explained as patients were divided into experimental and control groups, and those in the experimental group received IC plus CCRT while the others received CCRT alone without adjuvant chemotherapy. The trials were designed to compare the efficacy and safety between the 2 treatment regimens. The outcomes observed in the selected articles should include at least one of the following: overall survival (OS), progression-free survival (PFS), distant metastasis-free survival (DMFS), locoregional recurrence–free survival (LRFS), and treatment-related grade 3 or 4 acute adverse events during CCRT. The study design must be a randomized controlled trial (RCT).
Exclusion Criteria
Patients should be removed from the trial if any of the following occurred: (1) palliative treatment; (2) lactation or pregnancy; (3) severe coexisting illness; or (4) recurrent NPC.
Data Extractions
The basic characteristics of the available articles were sorted by 2 researchers (Mr. Xu and Ms. Wang) by reading the full texts of the included literature. The items collected were the main author, year of publication, country initiating the trial, median follow-up time, sample size, clinical stage of the tumor, Karnofsky Performance Status (KPS) or Eastern Cooperative Oncology Group (ECOG) score of the patient, specific IC scheme, chemotherapy scheme during CCRT, radiotherapy method and dose. Two types of outcomes were extracted. One was survival outcomes including OS (defined as the time from patient enrollment to death), PFS (defined as the time from patient enrollment to local recurrence, distant metastasis or death of any event), DMFS (defined as the time from patient enrollment to distant metastasis), and LRFS (defined as the time from patient enrollment to local recurrence). If the survival data could not be extracted directly from the article, Engauge software was employed for extraction according to the method applied by Tierney. 10 The other outcome was acute grade 3 or 4 treatment-related adverse events during CCRT including leukopenia, anemia, thrombocytopenia, mucositis, and neutropenia.
Statistical Analysis
The outcomes were merged by RevMan software (version 5.1). Time-to-event data were indicated as hazard ratios (HRs) through the inverse variance (IV) statistical method. The combined effect of dichotomous variables was assessed as risk ratios (RRs). The χ2 and I 2 tests were applied to test the heterogeneity of the combined effects. If the P value of the χ2test was less than 0.10 and I 2 was greater than 50%, then the heterogeneity was significant and a random effects model was used; otherwise, a fixed effects model was employed. In addition, a subgroup analysis was performed according to the different cisplatin regimens during CCRT. The triweekly and weekly regimens of cisplatin during CCRT were analyzed in the subgroup analysis.
Quality Assessment and Risk of Bias
The Cochrane Collaboration’s risk of bias assessment tool was employed to evaluate the quality of the included RCTs. The assessment processes were performed independently by 2 researchers (Mr. Xu and Ms. Wang). 11 If different opinions arose, they would be resolved by the third team member (Ms. Cao). In addition, the Guideline Development Tool (GRADEpro GDT) was used to grade the quality of evidence for the combined data (such as OS, PFS, DMFS and LRFS). 12
Results
Study Selection
By thoroughly searching the PubMed, EMBASE, and Cochrane Library databases, a total of 249 articles of RCTs were obtained. Of these, 78 were duplicates. In addition, 139 out of 171 articles were excluded after reading the titles and abstracts and determining that they were irrelevant to the topic. Then, 12 out of the remaining 32 articles were excluded because the full texts were not available. After reading the full texts of the remaining 20 articles, 13 articles were excluded for the following reasons: 5 articles were meta-analyses, 5 articles were duplicated data, and 3 articles were published in Chinese. Finally, 7 articles with a total of 2,319 patients were included in our meta-analysis. The flow diagram is shown in Figure 1.

Flowchart of study selection.
Characteristics of the Included Studies
The basic information of the included literature is shown in Tables 1 and 2. The 7 included studies were evaluated by the Cochrane bias assessment tool, and the results are shown in Figures 2 and 3. The random sequence generation and allocation concealment of the study by Fountzilas and colleagues 13 were assessed as unclear risk. The blinding of participants and personnel and blinding of outcome assessment of study by Frikha and colleagues 14 were assessed as unclear risk. The allocation concealment and blinding of participants and personnel of the studies of Hong and Tan were assessed as unclear risk. 15,16 The remaining studies were assessed as low risk. In addition, the quality of Grading of Recommendations Assessment, Development, and Evaluation (GRADE) evidence for OS, PFS, DMFS and LRFS was high (Figure 4).
Basic Information of the Included Studies.

IC: induction chemotherapy; CCRT: concurrent chemoradiotherapy; AJCC: American Joint Committee on Cancer; UICC: The Union for International Cancer Control; KPS: Karnofsky Performance Status; ECOG: Eastern Cooperative Oncology Group.
Treatment Characteristics of the Included Studies.

IC: induction chemotherapy; CCRT: concurrent chemoradiotherapy; 5FU: fluorouracil; IMRT: intensity modulated radiotherapy; 2D-RT: 2 Dimentional Radiotherapy; 3DCRT: 3-Dimensional conformalradiation therapy; OS: overall survival; DRFS: distant recurrence–free survival; LRFS: locoregional recurrence–free durvival; DFS: disease-free survival; DMFS: distant metastasis-free survival; LRFFS: locoregional relapse-free survival; QOL: the European Organization for Research and Treatment of Cancer QOL questionnaire modules QLQ-30 version 3.0; FFS: failure-free survival; DFFS: distant failure-free survival; PFS: progression free survival; LRF: locoregional progression; DF: metastasis; ORR: overall response rate.
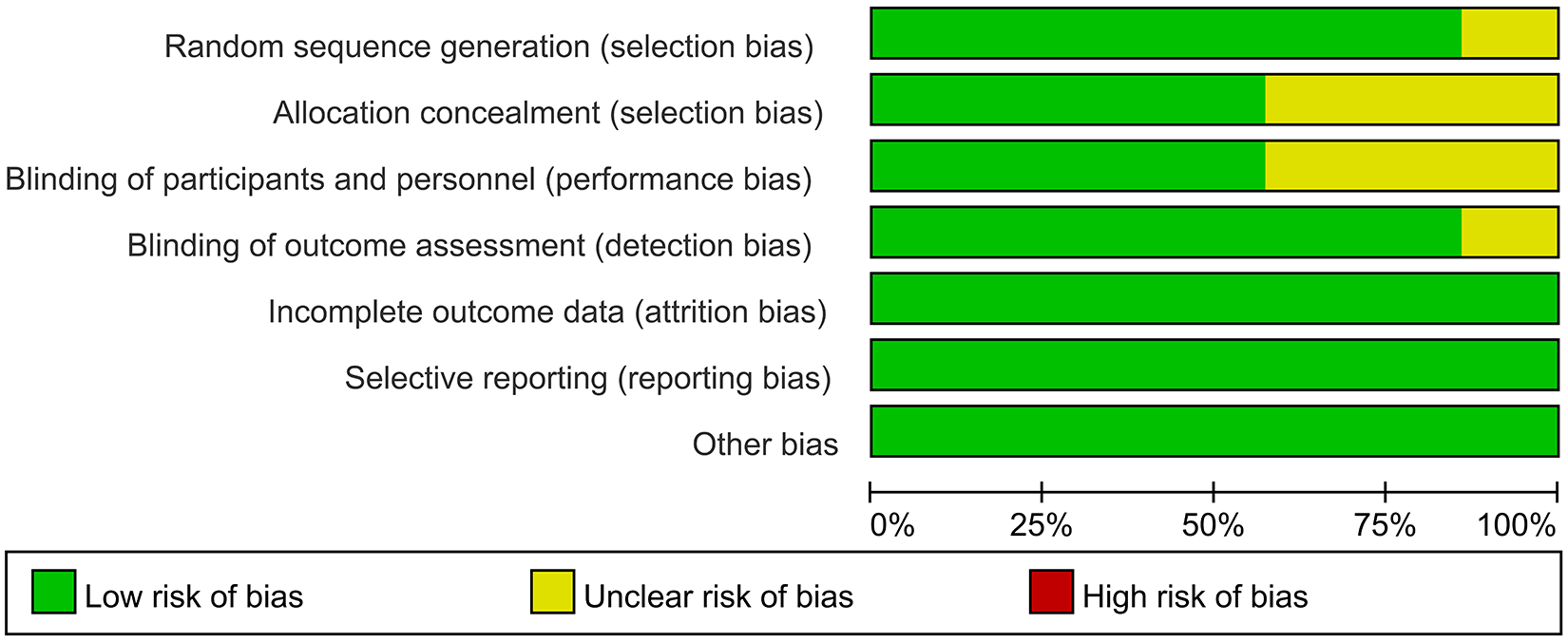
Risk of bias summary of RCTs.

Risk of bias graph of RCTs.
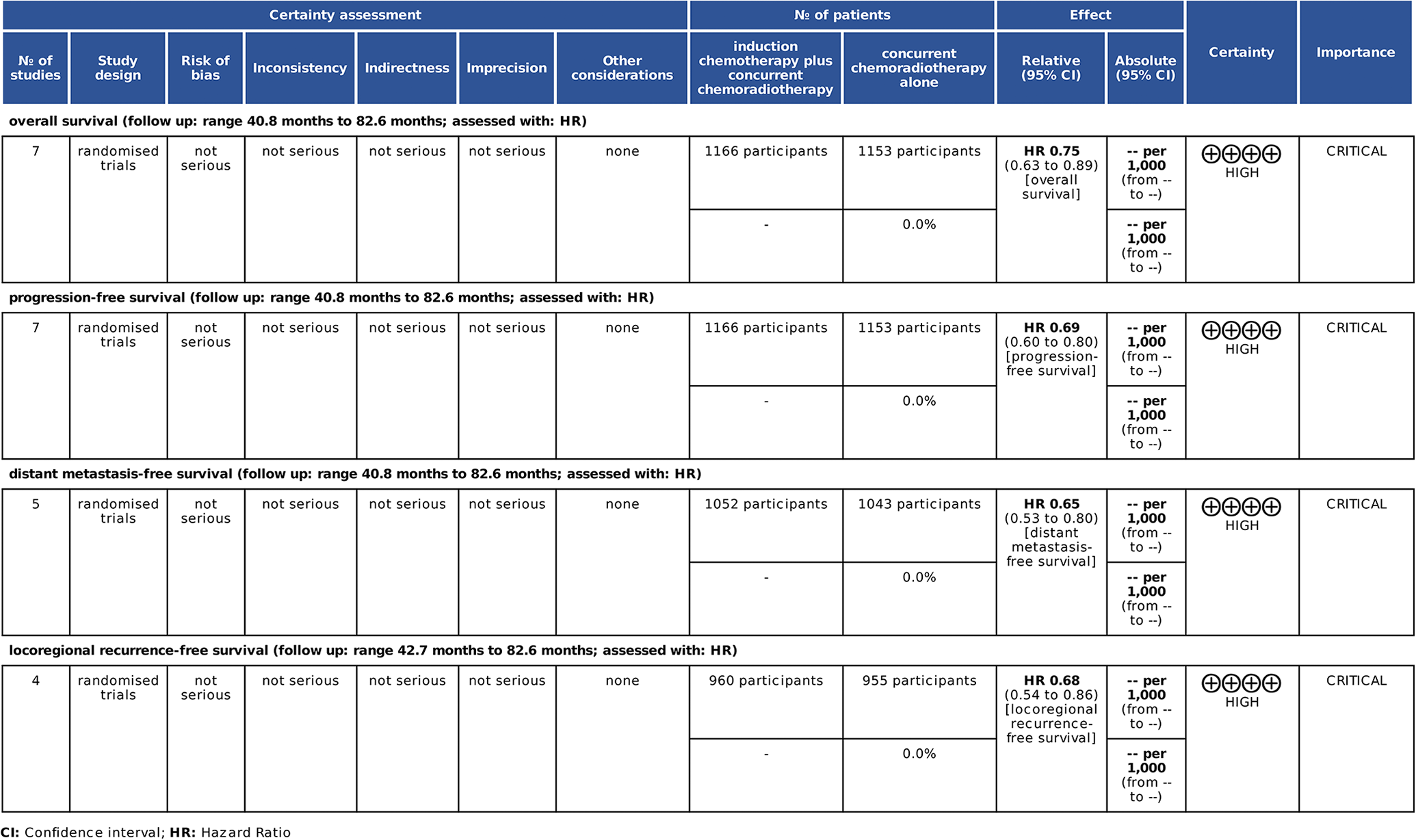
The GRADE quality of the summarized evidence.
Primary Endpoint
Overall survival (OS)
OS was reported in 7 articles. 13 -19 Two studies conducted by Li et al. 18 and Yang et al. 19 reported the results of the same population at different times, so we extracted the long-term follow-up data. In addition, Engauge software was applied to calculate HRs that were not directly displayed in the research conducted by Fountzilas et al. 13 The quality of all of the included articles was high for OS, and no articles were excluded. The combined data showed that patients who received IC plus CCRT had higher OS rates than those who received CCRT alone (HR = 0.75, 95% confidence interval (CI): 0.63-0.89). The differences were significant, and no significant heterogeneity was found (P = 0.12, I 2 = 41%). In the subgroup analysis, patients treated with IC plus CCRT had higher OS rates than those who received CCRT alone in the triweekly cisplatin regimen subgroup (HR = 0.62, 95% CI: 0.49-0.79, P = 0.0001). Interestingly, the comparison result between IC plus CCRT and CCRT alone was similar in the weekly cisplatin regimen subgroup (HR = 0.92, 95% CI 0.71-1.19, P = 0.54). Notably, the subgroup analysis reduced the heterogeneity, and the homogeneity within each subgroup was improved (I 2 = 16% and I 2 = 0%, respectively, Figure 5).

Forest plot for the OS comparing IC+CCRT and CCRT.
Secondary Endpoints
Progression-free survival (PFS)
The HRs and 95% CIs of PFS could be extracted directly or indirectly from the 7 studies. 13 -19 The statistics in the research conducted by Fountzilas et al. 13 were extracted by Engauge software. The synthesized data showed that patients in the IC plus CCRT group had a lower risk of disease progression than patients in the CCRT group (HR = 0.69, 95% CI: 0.60-0.80). The difference was significant (P < 0.001). A fixed-effects model was employed due to low heterogeneity (I 2 = 35%). The subgroup analysis showed that the differences in the triweekly cisplatin regimen subgroup and the weekly cisplatin regimen subgroup were significant. IC plus CCRT resulted in higher PFS rates in both subgroups compared with CCRT alone (HR = 0.62, P < 0.0001and HR = 0.78, P = 0.002, respectively, Figure 6).

Forest plot for the PFS comparing IC+CCRT and CCR.
Distant metastasis-free survival (DMFS)
For DMFS, 5 studies 15 -19 were available from which we could extract HRs and 95% CIs. The synthesized data showed that IC plus CCRT was more effective than CCRT, and the difference was significant without heterogeneity (HR = 0.65, 95% CI: 0.53-0.80, P = 0.001, I 2 = 11%). In the subgroup analysis, the results of the triweekly cisplatin regimen showed that the IC plus CCRT had higher DMFS rates than CCRT (HR = 0.56, P < 0.0001). The DMFS rates of IC plus CCRT and CCRT were similar in the subgroup receiving the weekly cisplatin regimen (HR = 0.82, P = 0.22, Figure 7).

Forest plot for the DMFS comparing IC+CCRT and CCRT.
Locoregional recurrence-free survival (LRFS)
Four studies 16 -19 reported the outcomes of LRFS. The pooled data indicated that the risk of locoregional recurrence was reduced by 32% in the IC plus CCRT group in terms of LRFS compared with that in the CCRT group (HR = 0.68, 95% CI: 0.54-0.86). The statistical difference was significant, and no heterogeneity was found (P = 0.001, I 2 = 0%). The results of the subgroup analysis showed that IC plus CCRT was superior to CCRT alone regardless of the type of cisplatin regimen used during the CCRT period (HR = 0.66, P = 0.003 and HR = 0.69, P = 0.002, respectively, Figure 8).

Forest plot for the LRFS comparing IC+CCRT and CCRT.
Treatment-related grade 3 or 4 adverse events during CCRT
The grade 3 or 4 acute adverse events related to treatment during CCRT, including leukopenia, anemia, thrombocytopenia, mucositis, and neutropenia, are shown in Table 3. Grade 3 or 4 acute adverse events of leukopenia, anemia, thrombocytopenia and mucositis were extracted from 6 studies 13,15 -19 while neutropenia could be obtained from only 5 studies. 13,15,17 -19 In addition, the acute adverse events published by Cao et al. 20 who reported the results of the short-term follow-up of the same participants in the study by Yang, 19 were extracted.
Risk Ratios (RRs) of Treatment-Related Grade 3 or 4 Adverse Events During CCRT.

IC: induction chemotherapy; CCRT: concurrent chemoradiotherapy; RR: risk ratio; CI: confidence interval.
For the occurrence of leukopenia, the synthesized results showed no significant difference between IC plus CCRT and CCRT (P = 0.56). However, the subgroup analysis of the triweekly cisplatin regimen showed that the incidence of leukopenia with IC plus CCRT was 1.62 times that of CCRT, and the difference was significant (RR = 1.62, 95% CI: 1.07-2.43, P = 0.02). However, no significant difference was found between IC plus CCRT and CCRT in the subgroup analysis of the weekly cisplatin regimen (P = 0.59).
Similarly, we found that compared with CCRT, the occurrence of mucositis with IC plus CCRT was not significantly different in the total synthesized effect (P = 0.35). However, IC plus CCRT reduced the risk of mucositis during CCRT by a striking 18% compared with CCRT in the weekly cisplatin regimen subgroup analysis (RR = 0.82, 95% CI: 0.69-0.98, P = 0.03). Nevertheless, the subgroup analysis of the triweekly cisplatin regimen showed no difference between IC plus CCRT and CCRT (P = 0.58).
In addition, IC plus CCRT increased the risk of anemia compared with CCRT alone (RR = 3.52, 95% CI: 1.31-9.46, P = 0.01). Interestingly, the risk of anemia between CCRT and IC plus CCRT was similar in both the weekly and the triweekly cisplatin regimen subgroups (P = 0.08 and P = 0.12, respectively).
For thrombocytopenia, compared with CCRT, IC plus CCRT increased the incidence of thrombocytopenia to 8.63 times (RR = 8.63, 95% CI: 3.14-23.70, P < 0.0001). In addition, IC plus CCRT increased the incidence of thrombocytopenia both in the weekly and triweekly cisplatin subgroup analysis (P < 0.0001 and P = 0.002, respectively).
Overall, IC plus CCRT had a higher incidence of grade 3 or 4 anemia, thrombocytopenia, neutropenia than CCRT, but the incidence of grade 3 or 4 leukopenia and mucositis in IC plus CCRT was similar to that in CCRT.
Discussion
Our results were in accordance with the results of 2 meta-analyses, both of which confirmed that IC plus CCRT improved OS and PFS. 21,22 The reason for these results may be that IC plays an important role in eliminating micrometastases at the initial treatment while local control is improved by the combination of concurrent chemotherapy with radiotherapy. Notably, the level of evidence of our research is higher than that of published articles because all studies included in our meta-analysis were evidence-based RCTs with the latest results, and the included data extracted from the studies came from long-term follow-up times (ranging from 40.8 months to 82.6 months). Thus, the timing of distant metastasis was delayed, which may lead to favorable OS and PFS.
In addition, our study found that the IC plus CCRT regimen was not always superior to CCRT in the weekly cisplatin and triweekly cisplatin subgroups. When the CCRT regimen was applied to patients with locally advanced NPC, should weekly cisplatin or triweekly cisplatin be chosen? Some studies have been designed to directly explore the comparison of triweekly cisplatin regimens and weekly cisplatin regimens during CCRT. Two retrospective analyses 23,24 found no differences in PFS and OS between the weekly cisplatin protocol and the triweekly cisplatin protocol in locally advanced NPC patients who were treated with CCRT. Similar results were observed in 2 RCTs. 25,26 However, a retrospective study conducted by Su et al. 27 found opposite results showing that the triweekly cisplatin regimen improved OS and DMFS compared with the weekly cisplatin regimen when CCRT was applied to the all patients. Unfortunately, the patients recruited in this study were in clinical stage II. Hence, patients at different levels of risk may lead to different results from the same treatment regimen. A study showed that IC could only improve DMFS and OS in high-risk patients and constructed a nomogram. 28 In addition, Yang et al. 29 established a model of Sequestosome-1 expression and N-stage and verified that NPC patients with a high expression of Sequestosome-1 and advanced N-stage could benefit more from IC. Therefore, IC plus CCRT for the triweekly cisplatin regimen might be considered in patients with locally advanced NPC who are at high risk of metastasis.
Moreover, our study included 3 cycles of IC. The optimal number of IC cycles has been controversial in recent years. Yan et al. 30 found that compared with the 3 cycles of IC, the 4 cycles of IC achieved no advantage of survival outcomes while 2 cycles of IC resulted in similar survival outcomes and a lower incidence of treatment-related adverse events. The possible reason for these results may be that the participants in this study were in clinical stage II. Another study showed that 2 cycles of IC resulted in higher DMFS and OS rates than 3 to 4 cycles of IC in patients with locally advanced NPC (93.3% vs. 88.5%, P = 0.043; and 88.7% vs. 81.7%, P = 0.037, respectively). 31 Otherwise, for patients in stage N2-3 (lymph node stage) with a high potential for metastatic risk 32 whether 2 or 3 cycles of IC are sufficient is unknown. A study conducted by Wei et al. 33 found that 4 cycles of IC appeared to be more efficacious than 2 cycles of IC in patients with T1-4N2-3M0 NPC. A clinical trial of a 4-cycle protocol in patients with stage N2-3 NPC is ongoing (NCT04061278).
Overall, the advantage of IC in different subgroups was not absolutely stable, and personalized treatment strategies may be proposed to treat patients with different risks. Based on our research, a weekly cisplatin regimen during CCRT followed by 2-cycle IC may be mostly recommended to those who are at low risk of distant metastasis (N0-1) because of its acceptable severe toxicities and better local control. For young patients with a high risk of distant metastasis (N2-3), 3 cycles of IC following CCRT with triweekly cisplatin regimen may be considered first to achieve better distant control. Although IC plus CCRT certainly led to better survival outcomes than CCRT, balancing the survival benefits with the toxic effects is strongly necessary when weekly cisplatin or triweekly cisplatin during CCRT is applied.
In addition, CCRT plus adjuvant chemotherapy (AC) is one of the treatments for locally advanced NPC. The recommended evidence category in the NCCN guidelines is the same as that for IC, and both are category IIA. 9 However, a phase 3 multicenter randomized controlled trial conducted by Chen et al. 34 showed that CCRT plus AC could not improve failure-free-survival (P = 0.13), OS (P = 0.32), distant fail-free survival (P = 0.12) or locoregional-failure-free-survival (P = 0.10) in patients with locally advanced NPC compared with CCRT alone. Similarly, Yang et al. 35 found that adding AC to patients with residual NPC after CCRT cannot improve the OS (P = 0.44), failure-free-survival (P = 0.19), LRFS (P = 0.51) or DMFS (P = 0.23). Moreover, a Bayesian network meta-analysis showed that CCRT plus AC did not improve survival outcomes compared with CCRT alone in NPC. 36 Hence, whether adding AC after IC plus CCRT can prolong the survival of patients with locally advanced NPC needs more multicenter clinical trials to prove.
Radiotherapy combined with chemotherapy has achieved satisfactory tumor control for the management of locally advanced NPC. The exploration of better treatment regimens is ongoing. What is the effectiveness of adding targeted therapy to traditional regimens? A study showed that the combination of CCRT alone with cetuximab resulted in no survival benefit for patients with head and neck cancer that overexpress epidermal growth factor receptor (EGFR). 37 In contrast, another study showed that the IC regimen of nituzumab combined with a cisplatin and fluorouracil regimen resulted in better treatment response in patients with positive EGFR. 38
In the era of precision radiotherapy, intensity-modulated radiotherapy (IMRT) has been widely used as an advanced radiotherapy technology. However, some of the included trials in our study focused on both traditional radiotherapy techniques such as 2-dimensional radiotherapy (2D-RT) and 3-dimensional conformal radiotherapy (3DCRT). 13 -16,19 Only the study conducted by Yang et al. 19 showed the outcomes of OS, PFS and DMFS were inconformity between the 2D-RT subgroup and the IMRT subgroup. The main reason is that the rates of patients who received these 2 radiotherapy techniques were not balanced in the experimental group and the control group. The rest of the studies had considered the balance of 2 different radiotherapy techniques in 2 comparison groups. In addition, a multivariate Cox analysis in 2 studies conducted by Fountzilas et al. 13 and Hong et al. 16 showed that different radiotherapy techniques were not significant variables that affected survival outcomes. The only one included article conducted by Yang et al. seem to ignore the influence of the radiation methods when we had a glance at the abstract, but the results stayed the same when we extracted the data of patients who were treated with 2D-RT (P < 0.05). And when we excluded the data of this trial, the results also showed that IC plus CCRT were superior to CCRT alone regarding OS, PFS and DMFS. In general, different radiotherapy techniques may have no significant impact on the combined results of our study. Innovations in radiotherapy technology may benefit patients from a variety of perspectives, including economic benefits, treatment time, dose distribution, safety and survival time, etc. Different tumors have different degrees of benefits from the new technology. Perhaps 2D-RT had reached the optimal state of radiotherapy effect for nasopharyngeal carcinoma, so the survival cannot be improved by improving radiotherapy technology. We look forward to that future studies will be more meaningful in exploring comprehensive therapies based on new technologies.
From the discussion above, we should be aware that many complicated problems await clarification in the treatment of NPC, such as the following: (1) whether a triweekly or weekly cisplatin regimen is preferred in CCRT after IC; (2) the optimal cycles of IC and the best drug combination of the IC scheme; (3) the efficacy of adding EGFR inhibitors to the IC plus CCRT regimen and the optimal time for this addition; and (4) how biomarkers guide clinically personalized therapy. We expect these problems to be solved soon.
Several limitations of our meta-analysis should be taken into account. First, all of the recruited patients were mostly from high-incidence areas in the Chinese population, and whether the results of our research are appropriate for patients of other races or non-endemic areas is unclear. Second, clinical heterogeneity among patients should be noted. The patients in our study were clinical stage III-IV, and the proportions of patients with different stages in each study was inconsistent; however, this did not affect the pooled results. The protocol for including IC varied among studies. In addition, our meta-analysis compared only a few treatment-related adverse events between IC plus CCRT and CCRT, ignoring the impact of late adverse events on patients’ quality of life. Finally, the age of the recruited patients in our study was at least 18 years old, so our findings may not be suitable for patients younger than 18 years old.
Footnotes
Abbreviations
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Statement
The review of the protocol did not require any ethical approval as it was based on published data.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: “Ten Thousand Plan” Youth Talent Project in Yunnan Province (no grant number is applicable). Scientific research fund project of Yunnan provincial department of education (Grant number: 2018JS222). Technology Innovation Team Construction Project Funding of Radiation Oncology of Kunming Medical University (Grant number: CXTD201806). Health science and technology program of Yunnan province (Grant number: 2018NS0066).
