Abstract
Purpose
To comprehensively evaluate the efficacy and safety of combining poly (ADP-ribose) polymerase (PARP) inhibitors with chemotherapy in patients with advanced breast cancer.
Methods
A systematic literature search was conducted in PubMed, Embase, Cochrane Library, Web of Science, and ClinicalTrials.gov to identify randomized controlled trials (RCTs) evaluating PARP inhibitor-chemotherapy combinations. Studies reporting progression-free survival (PFS), overall survival (OS), overall response rate (ORR), and safety outcomes were included. Data extraction and quality assessment were performed independently by two reviewers, and a meta-analysis was conducted using random-effects models.
Results
Of 970 studies retrieved, four RCTs involving 1064 patients met the inclusion criteria. PARP inhibitors combined with chemotherapy significantly improved PFS (hazard ratio [HR] 0.73, 95% confidence interval [CI] 0.63-0.84,
Conclusion
PARP inhibitor combined with chemotherapy is an effective option for the treatment of patients with advanced breast cancer, but its potential increased risks of specific AEs need to be weighed. Clinicians should make individualized treatment plans according to the specific conditions of patients, comprehensive consideration of efficacy and safety.
Keywords
Introduction
Breast cancer remains the most common malignancy affecting women worldwide, with persistently high morbidity and mortality rates. It is estimated that there will be approximately 300 590 new cases of breast cancer and 43 700 deaths in 2023. 1 Distant metastasis continues to be the primary cause of death in breast cancer patients. Despite advancements in novel drug therapies over the past two decades, the 5-year survival rate for patients with metastatic breast cancer remains below 30%. 2 This highlights the ongoing challenge for healthcare providers in managing patients with advanced breast cancer. In the subtypes of breast cancer, triple negative breast cancer (TNBC) lacks the expression of estrogen receptor (ER), progesterone receptor (PR) and human epidermal growth factor receptor-2 (HER-2), which makes traditional endocrine therapy and anti-HER2-targeted therapy ineffective, and chemotherapy has become the main systemic treatment. TNBC is particularly aggressive and has a worse prognosis compared to other subtypes, with higher mortality rates in patients with advanced, recurrent, or metastatic disease.3,4 In the molecular characteristics of TNBC, the presence of BRCA gene mutations has significant biological importance and clinical value. The incidence of BRCA gene mutations in TNBC patients is approximately 20%, leading to a more aggressive disease phenotype. 5 About 85% of breast cancer patients with BRCA1 mutation are TNBC6,7 The BRCA gene is involved in the homologous recombination repair (HRR) pathway, which is responsible for repairing DNA double-strand breaks. Mutations in the BRCA gene can lead to HRR deficiency, making tumor cells more sensitive to DNA damage. This biological characteristic provides a theoretical basis for the application of poly(ADP-ribose) polymerase (PARP) inhibitors, as PARP inhibitors can further block the repair of DNA single-strand breaks, thereby leading to synthetic lethality in tumor cells.8,9 The efficacy of PARP inhibitors in BRCA-mutated breast cancer has been validated in multiple clinical trials, leading to FDA approval for olaparib and talazoparib in advanced breast cancer patients with germline BRCA mutations.10,11
Despite these advances, the overall therapeutic efficacy of PARP inhibitors in combination with chemotherapy remains controversial due to heterogeneous study designs and patient populations. To address this gap, we conducted a systematic review and meta-analysis of randomized controlled trials (RCTs) to evaluate the efficacy and safety of PARP inhibitors combined with chemotherapy in the treatment of advanced breast cancer.
In recent years, PARP inhibitors have demonstrated substantial advancements in the treatment of BRCA-mutated breast cancer and are being increasingly investigated for their potential in treating TNBC. However, despite promising findings on the efficacy and safety of PARP inhibitors in combination with chemotherapy, existing literature exhibits certain limitations that hinder broader clinical application. Primarily, current studies predominantly focus on specific patient subpopulations, such as those with BRCA mutations or TNBC, without adequately evaluating the overall therapeutic efficacy in advanced breast cancer.12,13 Secondly, different studies have differences in chemotherapy regimens, PARP inhibitors, and efficacy evaluation criteria, which increases the complexity of data integration.12,14,15 Finally, although some studies have shown potential advantages of combination therapy, a full assessment of long-term survival benefits and adverse events (AEs) remains unclear. 13 Therefore, we conducted a systematic review and meta-analysis of RCTs to evaluate the efficacy and safety of PARP inhibitors combined with chemotherapy in the treatment of advanced breast cancer.
Material and Methods
This systematic review and meta-analysis was conducted based on the PRISMA checklist, with the research being registered on PROSPERO (CRD42023453143). 16
Search Strategy
A systematic and comprehensive search was conducted across PubMed, EMBASE, the Cochrane Library, Web of Science, and the ClinicalTrials.gov website, utilizing a search method that combined subject words with free words. The keywords retrieved for this search were “PARP Inhibitor”, “Breast Neoplasms”, “Chemotherapy”, and “randomized controlled trial”. The retrieval period extended from the inception of each database to August 9, 2023, resulting in the identification of qualified studies. Furthermore, the references cited in the articles and related reviews were also searched to uncover additional potentially eligible studies (Detailed in Supplementary File 1: Search Strategy).
Inclusion and Exclusion Criteria
The following inclusion criteria were needed for eligible studies: (1) Patients with locally advanced, recurrent or metastatic breast cancer. (2) Patients were treated with PARP inhibitors combined with chemotherapy in the intervention arm and the same chemotherapy in the control arm. (3) Phase II or III RCTs. The following are the exclusion criteria: (1) Case reports or series, letters, editorials, commentaries, reviews, meta-analyses, conference abstracts, and basic or preclinical trials. (2) Phase I, single-arm trials or nonrandomized controlled trials. (3) PARP inhibitors were used for neoadjuvant therapy. (4) The study lacked available data on relevant outcomes. (5) The study is ongoing or ended early. (6) The outcome indicators of the study did not match.
Outcomes
The primary endpoints of this study were progression-free survival (PFS), overall survival (OS) and overall response rate (ORR). Safety profiles (all grades AEs, grade 3-4 AEs, PARPi discontinuation due to AEs, drug dose reductions due to AEs, drug interruption due to AEs, death due to AEs) were the secondary endpoints.
Data Extraction and Risk of Bias Assessment
Two researchers independently extracted the following basic information from the study: first author, year of publication, clinical phase, median age, number of patients, experimental treatment regimen (detailed in Table S3), control treatment regimen, BRCA status, homologous recombination deficiency-positive (HRD+), TNBC, PFS, OS, ORR, all-grade AEs, and grade 3-4 AEs. Disagreements were resolved by a third investigator.
The potential risks of bias in the trials were assessed using the Cochrane Collaboration Risk of Bias Assessment Tool 17 (Figure 1). The evaluation report mentioned six biases, namely, selection bias, performance bias, detection bias, attrition bias, reporting bias and other bias, and at the same time divided the risk into three levels: high, unclear and low. Similarly, if there were disagreements, the third reviewer would decide.

The Cochrane Risk of Bias Diagram.
Statistical Analysis
The meta-analysis was conducted using Review Manager 5.4, with hazard ratios (HR) employed to compare the PFS and OS between the PARP inhibitor arm and the control arm. The confidence interval was set at 95% (95% CI). If HR > 1, the summary results favored the PARP inhibitor groups, and vice versa. The ORR and AEs of both arms were measured using risk ratios (RR) at 95% CI. An RR > 1 suggested that the PARP inhibitor arm demonstrated a superior ORR compared to the control arm. Conversely, for AEs, an RR > 1 indicated that the control group exhibited a better safety profile than the PARP inhibitor group. Statistical heterogeneity was assessed using the I2 statistic and Cochrane's Q test. To mitigate the effects of heterogeneity, random effect models were applied to pool the summary effect estimates in all analyses. Publication bias was evaluated through Begg's and Egger's tests, while sensitivity bias was analyzed using Stata 12.0 Software. A
Ethical Statement
This study is a systematic review and meta-analysis and does not involve human or animal experiments. Therefore, ethical approval from an Institutional Review Board (IRB) or ethics committee was not required. All data were obtained from published literature and do not involve any personal privacy or sensitive information.
Certainty Assessment
The certainty of evidence for each outcome (PFS, OS, ORR, AEs) was assessed using the GRADE (Grading of Recommendations Assessment, Development and Evaluation) framework. 18 The GRADE criteria evaluated five domains: 1. Risk of bias: Based on the Cochrane risk of bias tool. 2. Inconsistency: Assessed via heterogeneity (I² statistic and Cochrane's Q test). 3. Indirectness: Judged by population, intervention, and outcome applicability. 4. Imprecision: Determined by confidence interval width and sample size. 5. Publication bias: Evaluated through Begg's and Egger's tests. The overall certainty was categorized as High, Moderate, Low, or Very Low.
Results
Study Selection and Inclusion Characteristics
Initially, a total of 970 articles were selected through systematic literature retrieval. After layer-by-layer screening and strict evaluation, 28 studies assessed the potential eligibility by screening the full-text, and 4 articles were finally included.19-22 The detailed flow chart of the retrieval and selection process is shown in Figure 2. Specifically, the related research on inipalib was ruled out because it was later proven that inipalib was not a PARP inhibitor.23-25 The BRAVO trial was excluded because it ended early, the relevant data were incomplete and the risk of bias was unclear. 26 The ISPY-2 trial was excluded because it was a neoadjuvant therapy for patients with early breast cancer, which did not meet the inclusion criteria. 27 The rest were not included either because the outcome indicators did not match or because the clinical trial had not yet been completed.

Flow Chart of the Study Selection Process.
A total of 1064 patients with advanced breast cancer were encompassed in four studies, with 617 patients receiving PARP inhibitors in combination with chemotherapy and 447 patients receiving chemotherapy alone. The patients’ median age spanned from 44 to 56 years. Regarding molecular typing, 1054 patients (99.06%) were HER-2 negative (with the exception of 10 HER-2 positive patients in the BROCADE 2 study), 669 patients (62.88%) had TNBC, and 754 patients (70.86%) possessed BRCA mutations. All four studies utilized veliparib as a low-toxicity chemotherapy combination drug. Three of the studies focused on assessing the efficacy of PARP inhibitors added to platinum-based chemotherapy, while the remaining study examined their efficacy when combined with cyclophosphamide chemotherapy. A pooled analysis disclosed that four studies reported on PFS and ORR, and three studies on OS and AEs. The key characteristics of these studies are summarized in Table 1.
Characteristics of Included Studies.

Abbreviations: qd, once daily; bid, twice daily; E, experimental arm; C, control arm; HRD+, homologous recombination deficiency-positive; TNBC, triple-negative breast cancer.
PARP Inhibitors + Chemo versus Chemo: Antitumor Efficacy
The consolidated analysis of PFS results from the four studies revealed that the integration of PARP inhibitors into chemotherapy significantly prolonged PFS in patients with advanced breast cancer (HR 0.73, 95% CI [0.63, 0.84],

Forest Plots of PARP Inhibitor + Chemo Versus Chemo on PFS.
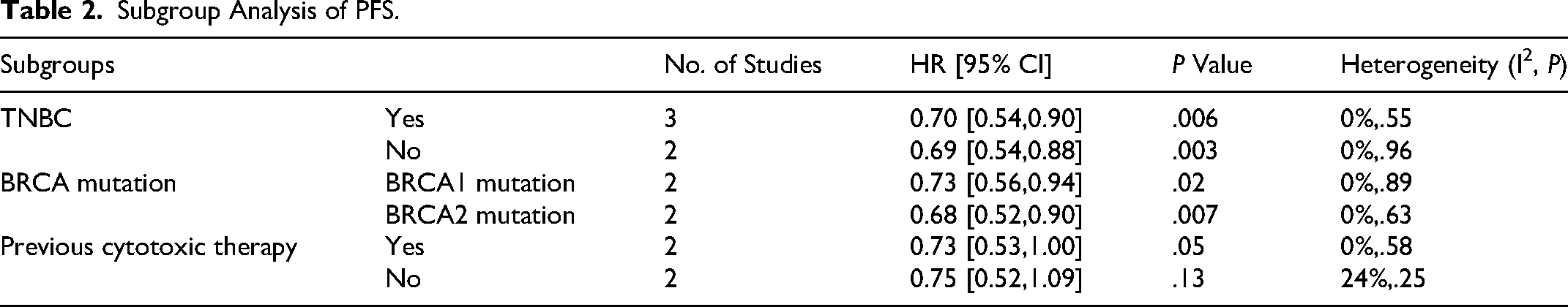
Subgroup Analysis of PFS.
The analysis of OS results from the three studies indicated that there is a trend toward improved OS in advanced breast cancer patients receiving PARP inhibitors in combination with chemotherapy. However, this finding did not reach statistical significance (HR 0.93, 95% CI [0.79, 1.09],

Forest Plots of PARP Inhibitor + Chemo Versus Chemo on OS.

Forest Plots of PARP Inhibitor + Chemo Versus Chemo on ORR.
PARP Inhibitors + Chemo versus Chemo: Safety and Toxicity
AEs were reported across all four included studies, with three studies providing quantitative data on the incidence of AEs of all grades and grades 3-4. The pooled analysis indicated that the incidence of grade 3-4 AEs in the combined group was generally elevated compared to the control group, though this elevation was not statistically significant (RR 1.10, 95% CI [0.87, 1.38],

Forest Plots of PARP Inhibitor + Chemo Versus Chemo on Grade 3-4 AEs.

Forest Plots of PARP Inhibitor + Chemo Versus Chemo on All Grades AEs.
An initial aggregation of grade 3-4 AEs was performed, with the top 10 AEs presented in Table S1, and Table 3 specifically highlighting the five most prevalent hematological toxic AEs observed in both the combined and control groups. Specifically, the combined group experienced neutropenia (65.45%), anemia (31.90%), thrombocytopenia (31.90%), leukopenia (25.79%), and lymphocyte count decrease (8.52%) as the most common grade 3-4 AEs, while the control group reported neutropenia (52.08%), anemia (22.69%), thrombocytopenia (17.82%), leukopenia (15.97%), and lymphocyte count decrease (6.06%). A meta-analysis further revealed a trend towards increased thrombocytopenia in the combined group (RR 1.86, 95% CI [1.01, 3.43],
Top 10 Grade 3-4 AEs.

Top 10 All-Grade AEs and Some Safety Profiles.

Sensitivity Analysis
The results of sensitivity analysis on PFS data were presented in Figure 8, through the sequential exclusion of individual studies and subsequent re-aggregation of effect sizes. The resulting analysis confirmed the stability and reliability of PFS findings.

Sensitivity Analysis of PFS.
Certainty of Evidence
The GRADE assessments for each outcome are summarized as follows: - PFS: High certainty (⊕⊕⊕⊕), supported by consistent results across studies and precise effect estimates. - OS: Moderate certainty (⊕⊕⊕◯), due to wide confidence intervals and non-significant trends. - ORR: High certainty (⊕⊕⊕⊕), despite non-significant pooled results. - Adverse events (AEs): Moderate certainty (⊕⊕⊕◯), influenced by heterogeneity in AE reporting and dose variations.
These ratings indicate that the PFS and ORR findings are robust for clinical decision-making, while OS and AE results require cautious interpretation. (Detailed in Supplementary File 2: Outcome Evaluation Based on GRADE Scales)
Publication Bias
Publication bias for both PFS and ORR was assessed using Begg's and Egger's tests. The results indicated no evidence of publication bias for PFS (Begg's test:

The Publication Bias of PFS (A) and ORR (B).
Discussion
This meta-analysis critically evaluated the efficacy and safety of PARP inhibitors supplementation to chemotherapy in patients with advanced breast cancer and showed that the combination of PARP inhibitors with chemotherapy is an attractive treatment option for these patients, which can improve patient survival (PFS), but has the risk of enhancing potential specific AEs. The results demonstrated a significant improvement in PFS and a modest benefit in OS with the combination therapy, despite a non-significant enhancement in ORR. In terms of safety, the combination therapy did not significantly increase toxicity compared to the control arm in this patient cohort. However, the incidence of AEs was generally elevated in the combination therapy group. Notably, hematological toxicities comprised the top 5 most frequently reported grade 3-4 AEs in both groups, with neutropenia being the most prevalent AE across all grades. Additionally, the combination therapy significantly augmented the risk of anemia, nausea, and diarrhea across all AE grades, and demonstrated a tendency towards an elevated risk of grade 3-4 thrombocytopenia.
Recently, the FDA has approved PARP inhibitors, olaparib and talazoparib, for the management of germline BRCA-mutated advanced breast cancer based on promising outcomes from two phase III clinical trials.10,11 Given that chemotherapy serves as the cornerstone of treatment for advanced breast cancer and its potential synergy with PARP inhibitors through shared DNA damage mechanisms, clinical trials exploring their combined administration have emerged. However, early studies of combination strategies were hindered by significant adverse effects, limiting their medical advancement. 28 To mitigate safety concerns while optimizing the benefits of this combination, researchers subsequently incorporated PARP inhibitors with lower capture capacity or doses into chemotherapy regimens. Nevertheless, the conclusions from various studies remain inconsistent. Consequently, this meta-analysis was undertaken, encompassing four clinical trials of combined therapy, all of which utilized veliparib—a PARP inhibitor with low toxicity—in conjunction with chemotherapy 29 to evaluate the overall therapeutic efficacy of PARP inhibitors in combination with chemotherapy in advanced breast cancer.
Platinum chemotherapy was compared and analyzed as the standard of care for advanced breast cancer patients in three of the studies included in this meta-analysis, echoing the notable outcomes reported in the CBCSG 006 and TNT trials among TNBC and BRCA1/2-mutated breast cancer patients.30,31 The consolidated analysis revealed a significant improvement in PFS and a modest benefit in OS for the combination therapy group, while ORR enhancements were not statistically significant. Notably, a pooled analysis of ORR highlighted that only non-BRCA-like patients in the S1416 study failed to demonstrate an improvement, potentially attributed to a non-BRCA-like phenotype have a more intact DNA repair machinery, with low sensitivity to PARP inhibitors, consistent with previous studies. 32 Although the improvement of OS was less significant than that of PFS, it still showed a certain trend. This may be related to the complexity and heterogeneity of advanced breast cancer, some patients may be more sensitive to the response to combination therapy, while others may benefit less due to the biological characteristics of the tumor or other factors. In the absence of formal head-to-head comparison of PARP inhibitors and platinum-based agents, this meta-analysis focuses on PARP inhibitors in combination with chemotherapy, particularly platinum chemotherapy, in advanced breast cancer. The GeparOLA study provides preliminary insights, demonstrating higher pCR rates with olaparib-paclitaxel over platinum-paclitaxel, particularly in younger and HR+ patients, alongside reduced AE burden in the olaparib arm. 33 While promising, the applicability of these findings to advanced recurrent and metastatic breast cancer warrants further investigation. In conclusion, the integration of PARP inhibitors with chemotherapy emerges as a promising therapeutic strategy, expanding treatment options and enhancing survival outcomes for patients with advanced breast cancer.
The meta-analysis encompassed four studies, comprising 1054 patients (99.06%) identified as HER-2 negative (excluding 10 HER-2 positive patients from the BROCADE 2 study), 669 patients (62.88%) classified as TNBC, and 754 patients (70.86%) diagnosed with BRCA-mutated breast cancer. Based on these findings, it can be inferred that combination therapy may confer benefits to patients with HER-2-negative, BRCA-mutated, or TNBC breast cancer. Notably, TNBC is prevalent among BRCA1 mutation carriers, 34 explaining the focus of prior clinical investigations on these patient populations. Prior meta-analyses have established the efficacy of PARP inhibitor monotherapy or combination therapy in BRCA-mutated or TNBC patients.12,14,35,36 This meta-analysis mainly focuses on the patient group of locally advanced or metastatic breast cancer, further confirming the benefits of combination therapy in these cohorts, and analyzes the adverse events in the treatment process in detail, so as to provide reference for the safety of follow-up treatment of this group. Compared with other meta-analyses, we strictly implemented the inclusion and exclusion criteria, and did not include the studies on inipalib or neoadjuvant therapy. At the same time, we added the latest research, the S1416 trial, to the analysis, which enriched the sample size. In the follow-up analysis, it is further put forward to the point that the combination therapy can expand the beneficiary groups. At present, the potential for extending the beneficiary group through combination therapy remains an area of active exploration. Notably, the GeparOLA study broadened the scope of neoadjuvant therapy exploration in early breast cancer to include patients with systemic mutations excluding BRCA germline mutations but having high HRD scores, 33 yielding improved pCR rates in the HR+ subgroup. Similarly, the S1416 study introduced the concept of a “BRCA-like” population in advanced breast cancer combination therapy, 22 encompassing patients without gBRCA mutations but exhibiting BRCA-like phenotypes akin to those associated with germline BRCA 1/2 mutations. Approximately 40% to 60% of TNBCs exhibit HRD or BRCA-like phenotypes31,37 and the meta-analysis revealed significant PFS prolongation in mTNBC patients with BRCA-like phenotypes. Although subgroup analysis was limited in some trials, existing studies suggest a potential benefit trend for combination therapy in the HRD population. Future studies are needed to explore potential biomarkers beyond BRCA mutations or HRD scores to better identify patients who may benefit from combination therapy. Nevertheless, the current meta-analysis findings provide valuable insights for clinicians, emphasizing the need to balance therapeutic efficacy and safety risks when selecting treatment regimens. Notably, while the overall incidence of AEs was comparable between the combination and control groups, grade 3-4 AEs were significantly more frequent in the combination group. Heterogeneity across studies, particularly in the S1416 study with cisplatin-based combination therapy, contributed to this observation. Hematological toxicities, particularly neutropenia, were prevalent in both groups, consistent with previous reports. 38 Moreover, anemia, nausea, and diarrhea significantly increased in all grades of AEs, while grade 3-4 thrombocytopenia showed an increasing trend in the combination group. These findings underscore the importance of proactive prevention and timely intervention to mitigate safety risks and ensure optimal treatment outcomes for patients.
Certain limitations of this meta-analysis ought to be acknowledged. Firstly, the study pool was constrained, encompassing a limited number of trials, including only a single level 3 clinical study, and the collective sample size was relatively modest. Secondly, notable variation existed in the PARP inhibitor dosages utilized across the studies, spanning a broad range from 60 mg to 300 mg. This variation could potentially impact the efficacy and toxicity profiles of PARP inhibitor-chemotherapy combinations. Furthermore, data constraints were encountered in the form of incomplete or absent subgroup analyses for specific outcome measures, hindering a more thorough examination of the potential beneficiary populations and optimal treatment strategies. Despite the above limitations, we believe that all the analyses performed may aid in presenting a brief assessment of the role of PARP inhibitors combined with chemotherapy in the management of patients with advanced breast cancer. A strength of this analysis is that it provides further confirmation of the benefit of combination therapy in patients with advanced disease and provides a detailed analysis of adverse events to inform the safety of subsequent therapy.
Conclusion
This meta-analysis demonstrates that the combination of PARP inhibitors with chemotherapy significantly improves progression-free survival in patients with advanced breast cancer, particularly those harboring BRCA mutations or triple-negative subtypes. However, the increased risk of anemia, nausea, and diarrhea necessitates careful monitoring. While our findings support the clinical utility of this combination, limitations such as heterogeneous dosing regimens and a modest sample size warrant further validation. Future studies should focus on identifying predictive biomarkers (eg, HRD scores) to optimize patient selection and explore long-term survival benefits in broader populations.
Supplemental Material
sj-docx-1-tct-10.1177_15330338251350630 - Supplemental material for Efficacy and Safety of Poly (ADP-Ribose) Polymerase Inhibitors in Combination with Chemotherapy for Advanced Breast Cancer: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-1-tct-10.1177_15330338251350630 for Efficacy and Safety of Poly (ADP-Ribose) Polymerase Inhibitors in Combination with Chemotherapy for Advanced Breast Cancer: A Systematic Review and Meta-Analysis by Qiuhua Duan, Yue Feng, Lichen Cao, Lijun Hu, Jianlin Wang, Fei Sun, Qinghong Meng, Mengyun Zhou, Jingping Yu and Haiyan Gao in Technology in Cancer Research & Treatment
Supplemental Material
sj-docx-2-tct-10.1177_15330338251350630 - Supplemental material for Efficacy and Safety of Poly (ADP-Ribose) Polymerase Inhibitors in Combination with Chemotherapy for Advanced Breast Cancer: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-2-tct-10.1177_15330338251350630 for Efficacy and Safety of Poly (ADP-Ribose) Polymerase Inhibitors in Combination with Chemotherapy for Advanced Breast Cancer: A Systematic Review and Meta-Analysis by Qiuhua Duan, Yue Feng, Lichen Cao, Lijun Hu, Jianlin Wang, Fei Sun, Qinghong Meng, Mengyun Zhou, Jingping Yu and Haiyan Gao in Technology in Cancer Research & Treatment
Footnotes
Abbreviations
Ethical Considerations
This systematic review and meta-analysis was conducted using data from published clinical trials and did not involve direct animal or human subjects. Therefore, it did not require approval from an ethics committee. All data analyzed in this study were publicly available and collected from RCTs that had already undergone ethical review and approval by the respective institutional review boards (IRBs) or ethics committees of the participating institutions. As such, no separate ethics approval was sought for this meta-analysis.
We confirm that this study complies with the ethical standards of scientific research and adheres to the PRISMA guidelines for systematic reviews and meta-analyses. All data sources were accessed in accordance with the guidelines and regulations set forth by the respective databases and publishers.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Shanghai Siming Medical Development Foundation (Grant No. SGKY-202413), the Scientific Research Project of Shanghai Municipal Health Commission (Grant No. 202340160), the 2024 Annual Clinical Research Project of Changzhou Medical Center, Nanjing Medical University (Project No. CMCC202412) and the Changzhou Science and Technology Program (Project No.ZD202225).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
In this published article, all data generated during this study are included, and references are provided.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
