Abstract
Purpose:
A meta-analysis was conducted to evaluate the diagnostic performance of contrast-enhanced ultrasonography using the contrast agent SonoVue to differentiate benign from malignant breast lesions.
Method:
A comprehensive search of the literature was performed using the Embase, PubMed, and Web of Science databases to retrieve studies published before February 2020. Data were extracted, and pooled sensitivity, specificity, and diagnostic odds ratios were calculated with meta-analysis software. Heterogeneity was evaluated via the Q test and I2 statistic. Meta-regression and subgroup analyses were applied to evaluate potential sources of heterogeneity. Publication bias was assessed using the Deeks’ funnel plot asymmetry test. A summary receiver operating characteristic curve (SROC) was constructed.
Results:
A total of 27 studies including 5378 breast lesions subjected to CEUS examination with SonoVue were included in the meta-analysis. The pooled sensitivity and specificity values were 0.90 (95% confidence interval [CI], 0.88–0.91; inconsistency index [I2 ] = 75.7%) and 0.83 (95% CI, 0.82–0.85; I2 = 91.0%), respectively. The pooled diagnostic odds ratio was 48.35% (95% CI, 31.22–74.89; I2 = 77.6%). The area under the summary receiver operating characteristic curve (AUC) was 0.9354. Meta-regression analysis revealed the region of patient residence and dose of contrast agent as potential sources of heterogeneity (P < .01). Subgroup analysis showed a higher area under the summary receiver operating characteristic curve for European and higher contrast agent dose subgroups (P < .05).
Conclusion:
Contrast-enhanced ultrasonography with SonoVue displays high sensitivity, specificity, and accuracy when differentiating benign from malignant breast lesions. Despite its current limitations, this technique presents a promising tool for diagnosing breast lesions in clinical practice.
Introduction
Breast cancer is the most commonly diagnosed type of cancer and the leading cause of tumor-related mortality in women worldwide, accounting for 24.2% of all new cases and 15.0% of overall cancer deaths, as reported by the Global Cancer Statistics in 2018. 1 Breast cancer survival is dependent on stage at diagnosis, and early diagnosis is associated with distinctly higher 5-year KM survival probability (62.5%) than diagnosis at a late stage (35.8%), 2 highlighting the vital importance of early disease detection in clinical prognosis. 3 Mammography plays a central role in early detection of breast cancers as changes in the breast can be visualized using this technique before physical observation by the patient or physician. Newly developed technologies, including contrast mammography and computer-aided detection (CAD) systems, have facilitated the optimization of screening performance. Breast ultrasound and magnetic resonance imaging (MRI) are also used as supplemental screening modalities, particularly for patients who should not be exposed to X-rays or with a family history of breast cancer. Ultrasound can be effectively applied to screen dense breast tissue containing abundant ducts, glands, fibrous tissue, and less fat. Such tissues are challenging when identifying cancers with traditional mammography. 4,5 As a radiation-free, cost-effective, and widely available option, ultrasound technologies, including conventional ultrasound, elastography, and contrast-enhanced ultrasound (CEUS), are commonly used as a supplemental breast cancer screening modality. Elastography measures the consistency or hardness of tissues to differentiate benign from malignant breast lesions. CEUS presents a considerable advantage over conventional ultrasound and elastography in terms of visualization of microvascular distribution and perfusion features within lesions (Figure 1). 6

Conventional ultrasound and CEUS results of 2 breast lesions. A, Conventional ultrasound showed a regular-shaped lesion classified as BI-RADS category 4A (left); CEUS showed hyperenhancement and enlarged scope of the lesion, which was classified as BI-RADS category 4B (right). This lesion finally proved to be an invasive ductal carcinoma. B, Conventional ultrasound showed an irregular-shaped lesion with heterogeneous echo classified as BI-RADS category 4A (left); CEUS showed no internal blood perfusion, nor enlarged scope of the lesion, which was then classified as BI-RADS category 3 (right). This lesion finally proved to be a fibrocystic breast disease.
Observations on the ultrasound contrast effect were initially reported in the 1960s and microbubbles were primarily used in contrast echocardiography. 7 Since then, CEUS has been widely applied for the diagnosis of tumors in the liver, kidney, breast, and other human organs. 8 Previous studies have shown that CEUS, particularly qualitative analysis, provides useful information on the course and distribution of internal vessels that can aid in distinguishing between benign and malignant breast lesions. 9,10 SonoVue (Bracco Imaging S.p.A., Milan, Italy), a second-generation ultrasound contrast agent, is currently used for diagnosing suspicious lesions in different human organs. 11 The pooled diagnostic performance of CEUS in breast lesions is extensively documented. Hu et al and Ma et al reported good sensitivity and specificity of CEUS for characterizing breast lesions. 12,13 Hu et al. showed that the use of Perfluoro containing microbubbles (Sonovue or Optison) significantly increased diagnostic precision, compared with Levovist. 12 In 2018, Li et al demonstrated better diagnostic performance of CEUS than conventional ultrasound in differentiation of breast lesions. 14 Previously, CEUS examinations using different contrast agents were conducted for breast cancer screening in different populations. While diagnostic performance is clearly variable with different contrast agents, the efficacy of specific contrast agents remains to be established. In recent years, CEUS with SonoVue has been increasingly employed to diagnose breast lesions in Europe and China. The current meta-analysis aimed to review relevant studies and evaluate the overall performance of CEUS with the contrast agent SonoVue when differentiating benign from malignant breast lesions.
Materials and Methods
Literature Search Strategy
Studies published before February 2020 were identified by comprehensively searching the Embase, PubMed, and Web of Science databases. Both MeSH terms and free words were adopted. The following terms were included in the search strategy: “breast,” “contrast-enhanced (or contrast enhanced),” “neoplasm (or carcinoma, cancer, tumor, mass, lesion),” “ultrasonography (or ultrasound, sonography),” and “SonoVue.” We additionally conducted a manual search of references of the relevant literature to expand the included studies.
Study Selection
Two reviewers (J. Lu, with 3 years of experience in breast CEUS and meta-analysis, and C. Jin, with 3 years of experience in breast CEUS) independently evaluated the eligible literature. Disagreements between individual judgments were resolved by discussion or consultation with a third author (P. Zhou, with 8 years of breast CEUS and 3 years of meta-analysis experience). The following selection criteria were applied: (1) true-positive (TP), false-positive (FP), false-negative (FN), and true-negative (TN) rates could be extracted directly or indirectly from 2-by-2 tabulated data in the study; (2) patients with suspicious malignant breast lesions screened using other techniques, such as breast MRI, mammography or conventional ultrasonography, were further examined using CEUS with SonoVue; and (3) the nature of suspicious breast lesions was determined by pathological examination.
There was no limit on patient age or region but the language of publication was restricted to English and the number of lesions in each enrolled study had to be greater than 10. Reviews, case reports, meta-analyses, letters, conference abstracts, and duplicate publications were excluded.
Data Extraction
The following data were extracted independently by 2 reviewers: (1) general study characteristics including the first author’s name, country, publication year, number of lesions, mean age of patients, mechanical index and dose of contrast agent; and (2) TP, FP, FN, and TN rates obtained directly or calculated according to the sensitivity and specificity values in every selected study. We contacted the original authors for any additional information needed for analysis.
Quality Assessment
The quality of all included studies was assessed by 2 independent reviewers using the Quality Assessment of Diagnostic Accuracy Studies checklist version 2 (QUADAS-2) in Review Manager 5.3 (Cochrane Collaboration, London, England). QUADAS-2 included 4 domains: (1) patient selection, (2) index test, (3) reference standard, and (4) flow and timing. Each item within the domains of QUADAS-2 was classified as low, high, and unclear risk.
Statistical Analysis
A summary of sensitivity, specificity, positive and negative likelihood ratios (LR), and diagnostic odds ratios (OR) with corresponding 95% confidence intervals (95% CI) was obtained using TPs, FPs, FNs and TNs extracted from all studies. The heterogeneity of sensitivity and specificity among studies was evaluated using Q test and I 2 statistic. P values < 0.01 for Q test or I 2 values > 50% were considered significant for heterogeneity and a random-effects model applied under conditions of heterogeneity. Univariate meta-regression and subgroup analyses were performed to explore potential sources of heterogeneity. A summary receiver operating characteristic curve (SROC) was conducted using the Moses–Shapiro–Littenberg method. Publication bias was evaluated with the Deeks’ funnel plot asymmetry test. All statistical analyses were performed using Stata version 15.1 (StataCorp, College Station, TX) and Meta-Disc version 1.4 software.
Results
Study Selection
A total of 885 articles were identified after the initial search, and 786 were retained after removing duplicates. Another 734 studies were further excluded by reviewing titles and abstracts. After full-text assessment of the literature, 28 articles were considered eligible based on the inclusion and exclusion criteria. Three authors had published more than one article and one of the studies was removed after careful review of patient samples. Ultimately, 27 studies were included for analysis. 10,15 -40 The complete literature screening and selection process is presented in Figure 2.
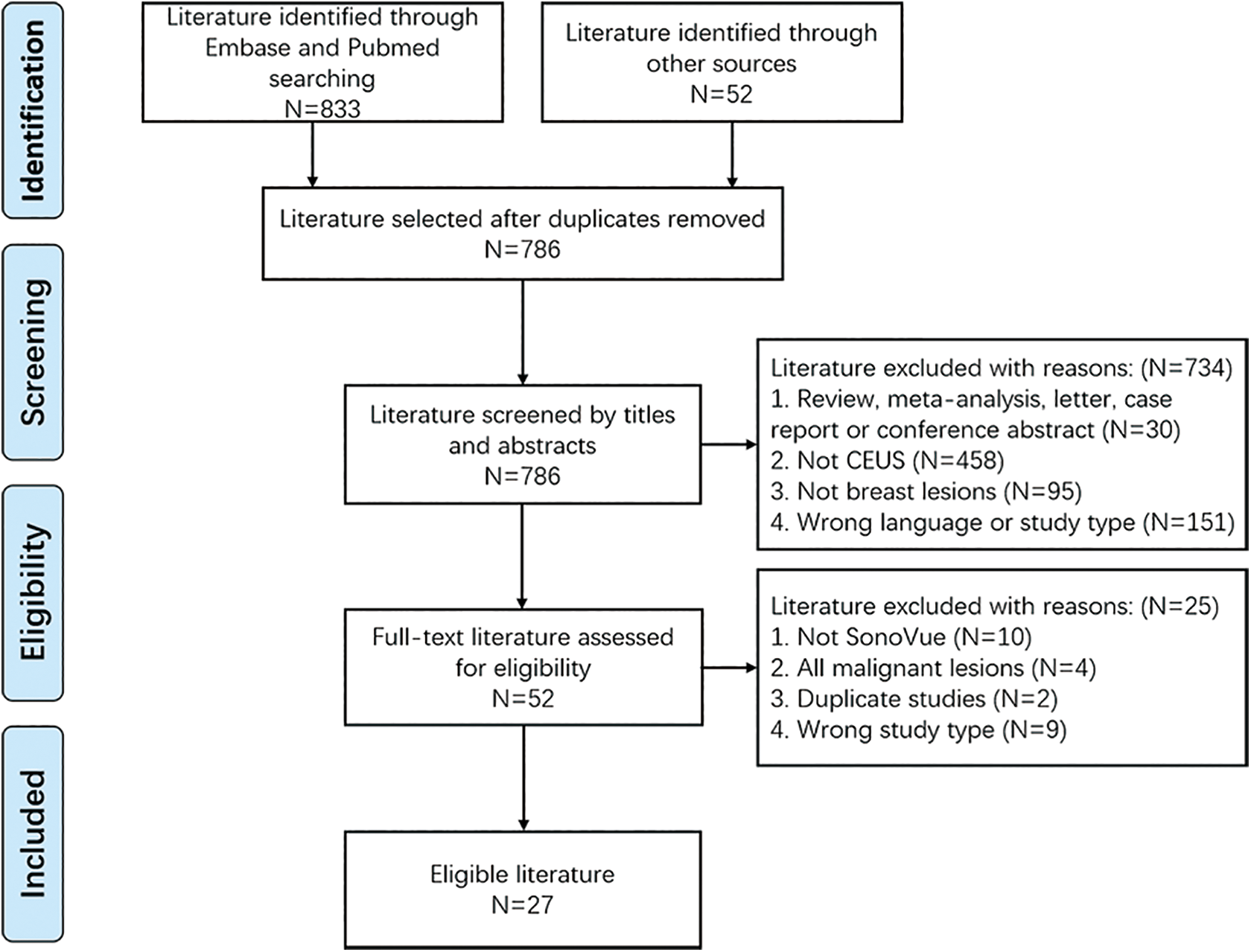
Flow chart of the literature search and selection.
Characteristics of Eligible Studies
The included studies were published from 2006 to 2019. Overall, 5378 lesions in 5276 patients were analyzed. The mean age of patients was 47.8 years and the mean number of lesions in each study was 199 (range, 15–1023). All 27 studies used SonoVue as the contrast agent in CEUS examinations using pathological results as the reference standards. Other main characteristics, including country, mean age, mechanical index (MI), and dose of contrast agent used, are presented in Table 1.
Main Characteristics of the Included Studies.

FP, false positive; FN, false negative; MI, mechanical index; TP, true positive; TN, true negative; NA, not available.
Quality Assessment
All eligible studies were assessed by 2 independent reviewers using the QUADAS-2 checklist (Figure 3). Among the included studies, 5 were judged to have unclear risk of bias in the “patient selection” domain due to lack of information on whether a consecutive or random sample of patients was enrolled. Two studies had unclear risk of bias in the “index test” domain, since they did not state whether a threshold was used or pre-specified. Two studies were judged to have unclear risk of bias in the “reference standard” domain, as no statement was included on whether the reference standard results were interpreted without knowledge of the index test results. For some studies, concerns regarding applicability were rated as unclear, as the relevant information was not clearly displayed in the article. Overall, the quality of the included studies was satisfactory.

Quality assessment of eligible studies. A, Risk of bias and applicability concerns: author judgments on the included studies in each domain presented as percentages. B, Summarized risk of bias and applicability concerns: author judgments on the included studies in each domain.
Diagnostic Performance in the Differentiation of Benign and Malignant Breast Lesions
A random-effects model was used for our meta-analysis due to the observed heterogeneity in the included studies (P < .01, I 2 > 50%). The pooled sensitivity and specificity of all eligible studies were 0.90 (95% CI, 0.88–0.91; I 2 = 75.7%) and 0.83 (95% CI, 0.82–0.85; I 2 = 91.0%), respectively (Figure 4). The pooled diagnostic OR for CEUS with SonoVue was 48.35 (95% CI, 31.22–74.89; I 2 = 77.6%). SROC for the diagnostic value of CEUS with SonoVue in the differentiation of benign and malignant breast lesions is shown in Figure 5. The area under SROC (AUC) was 0.9354 (SE, 0.0106).

Forest plots of sensitivities and specificities. A, Forest plot of sensitivity of CEUS with SonoVue for diagnosis of breast lesions. B, Forest plot of specificity of CEUS with SonoVue for diagnosis of breast lesions.
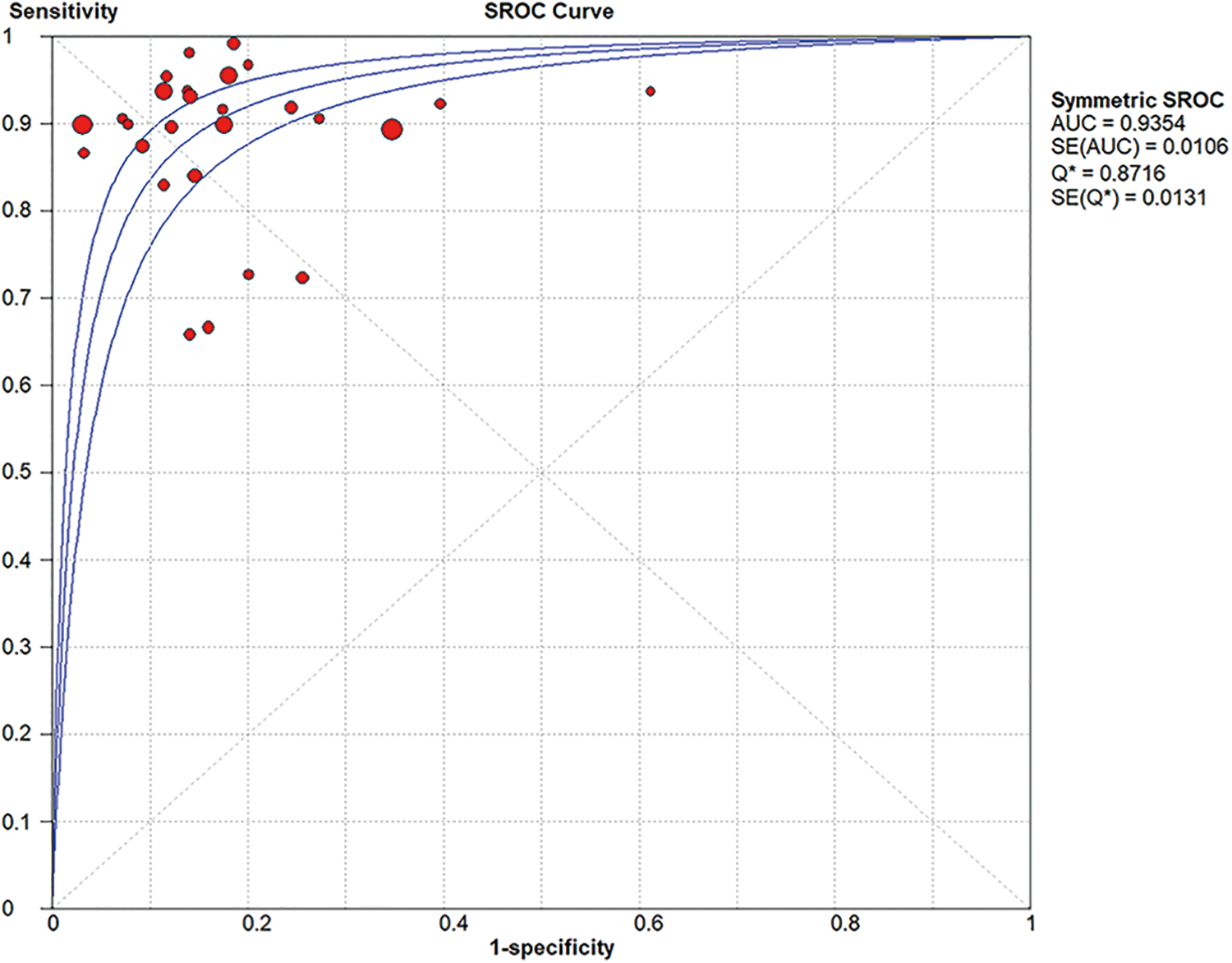
Summary receiver operating characteristic curve (SROC) and area under SROC (AUC).
Meta-Regression and Subgroup Analysis
Meta-regression analyses for sensitivity and specificity regarding patient number, mean age, and lesion number revealed no significant differences (Figure 6). Patient region was non-significant in the meta-regression analysis for specificity but significant for sensitivity (P < .01) while dose of contrast agent was significant for both sensitivity and specificity (P < .001). Pooled sensitivities, specificities, diagnostic ORs, AUCs of studies grouped by patient region, and the dose of the contrast agent used in CEUS are presented in Table 2. The pooled diagnostic OR and AUC values of the European group were higher than those of the Asian group (P < .05). The pooled sensitivity, diagnostic OR, and AUC of the higher-dose (>3 mL) group were higher than those of the lower-dose (≤ 3 mL) group (P < .05).
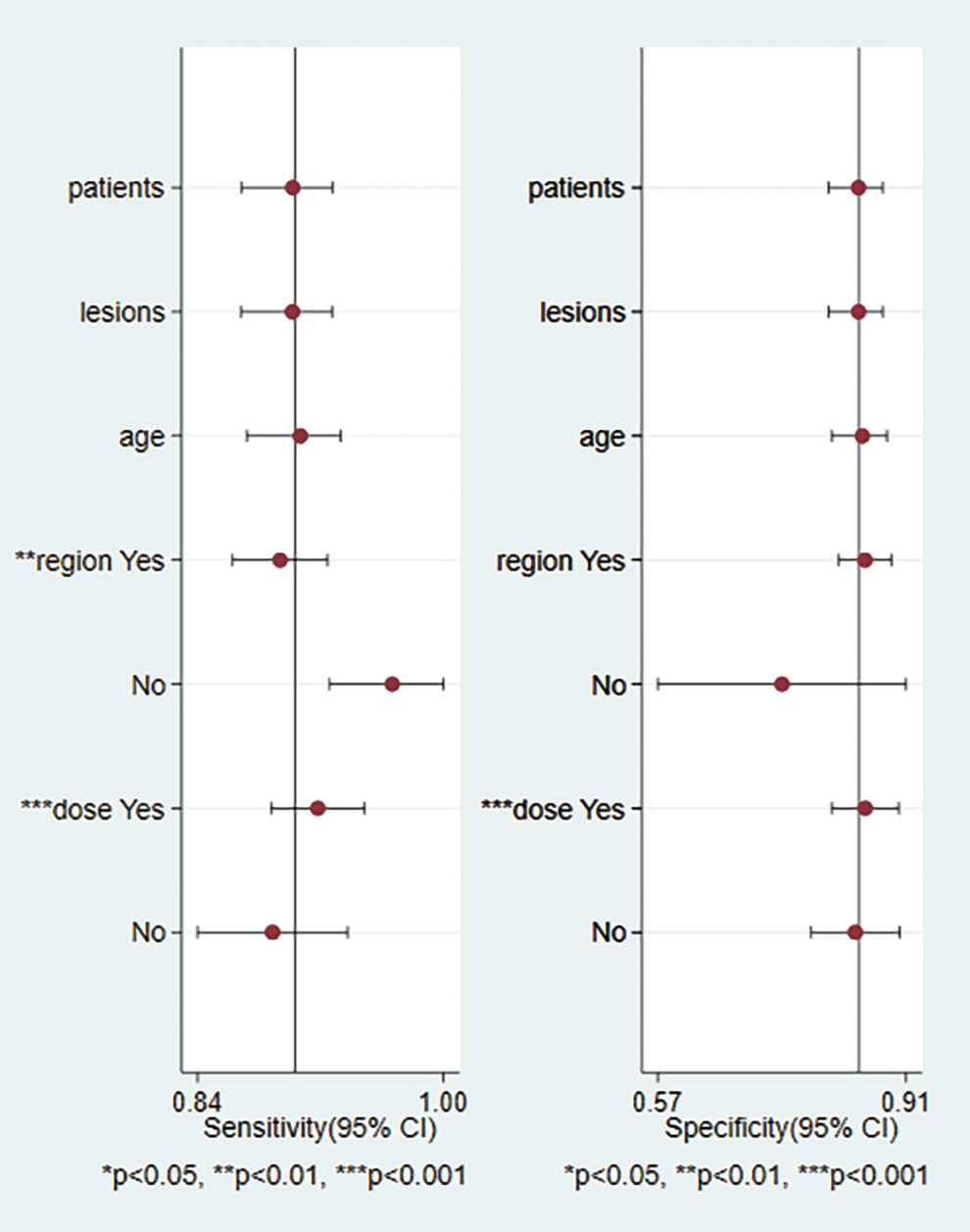
Meta-regression of included studies showing that patient region and dose of contrast agent serve as potential sources of heterogeneity.
Subgroup Analysis.

AUC, area under the curve; CI, confidence interval; I 2 , inconsistency index; OR, odds ratio; SE, standard error. *: P < 0.05.
Publication Bias Assessment
Publication bias was assessed using the Deeks’ funnel plot asymmetry test. No significant publication bias was found in the diagnostic odds ratio of CEUS with SonoVue in the differentiation of benign and malignant breast lesions (P = .62) (Figure 7).
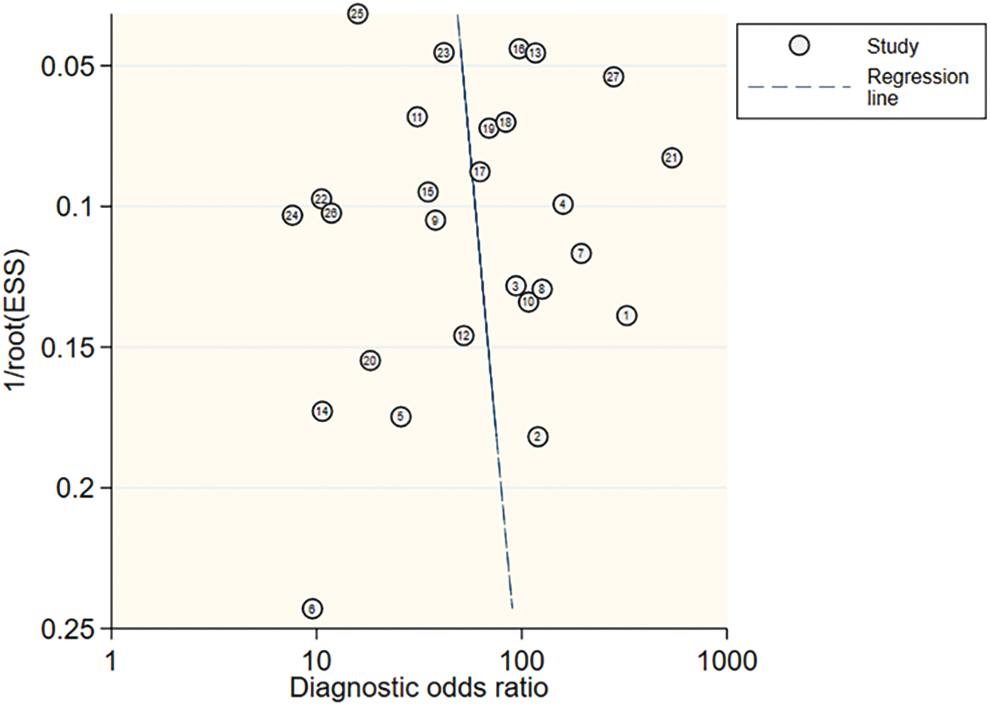
Deeks’ funnel plot of publication bias. No potential publication bias for all eligible studies was observed (P = 0.62).
Discussion
Vascular morphology is the best criterion to distinguish between benign and malignant tumors. CEUS contributes significantly to differential diagnosis of breast lesions by providing more detailed features of intratumoral microvascular distribution and blood flow compared with conventional ultrasonography or elastography. 41 Some patients may select CEUS in preference to mammography or MRI, since the technology is radiation-free, cost-effective, and widely available. However, CEUS diagnosis may be subjectively affected by operators or imaging features that exist in both benign and malignant lesions. Moreover, although the safety of the contrast agent has been validated, the risk of allergies cannot be overlooked. These limitations can be effectively resolved by other technologies, such as CAD algorithms or targeted contrast agent. 42 In the current meta-analysis, the diagnostic performance of CEUS with SonoVue when differentiating between benign and malignant breast lesions was systematically evaluated. Within the 27 included studies, sensitivity ranged from 0.66 to 1.00 and specificity from 0.38 to 0.97. The pooled sensitivity and specificity values were 0.90 and 0.83, respectively. The pooled diagnostic OR was 48.35 and overall diagnostic accuracy, represented by AUC, was 93.54%. These results indicate that CEUS with SonoVue present an effective modality in differentiation of benign and malignant breast lesions.
A similar meta-analysis was conducted in 2015, 13 which included 29 studies with 2296 lesions examined using CEUS. Their results supported the effectiveness of CEUS at diagnosing both benign and malignant breast lesions. However, only 14 studies enrolled in this earlier study used SonoVue as the contrast agent, and the diagnostic value of CEUS with SonoVue was not evaluated in subgroup analysis. In recent years, further studies relevant to this topic have been published, many of which used SonoVue as a contrast agent. Compared with previous similar reports, 6,13 our comprehensive meta-analysis included a larger number of studies (n = 27) and lesions (n = 5378) to assess on the value of CEUS with SonoVue in differentiating between benign and malignant breast lesions.
Meta-regression was conducted to investigate the potential sources of heterogeneity. In our investigation, 23 studies were located in Asia and 4 were located in Europe. In subgroup analysis, summarized diagnostic OR and AUC values were higher in the European than the Asian group. However, previous studies suggest that Asian women are more likely to have higher breast density and ultrasonography was used as a supplemental screening modality for breast cancer, since the diagnostic performance of mammography was not as satisfactory. 43,44 In contrast, our subgroup analysis indicated a better diagnostic performance of CEUS with SonoVue in breast tissue with lower-density breast tissue than higher-density breast tissue. These results require verification with follow-up studies owing to the limited sample of European patients in our study. Moreover, summarized diagnostic OR and AUC were higher in the higher-dose (>3 ml) than lower-dose (≤ 3 ml) group, consistent with the basic principle of contrast agents. 45 CEUS imaging is obtained by processing the echoes of microbubbles and reflectivity is proportional to the concentration of the microbubbles themselves. The dose of contrast agent used in CEUS examinations could affect diagnostic performance. Diagnosis of CEUS examinations may be more accurate if > 3 ml of SonoVue is bolus-injected once within the recommended dose range.
Our study has several potential limitations that should be acknowledged. First, among the 27 eligible studies, 22 were conducted in China and only 4 were conducted in Europe. The results of subgroup analysis may be more convincing with a larger European sample. Second, the influence of different operators and parameter settings was not evaluated since this information was not available. 46 Finally, although malignancies of breast lesions were all determined using CEUS with SonoVue, the image characteristics used for diagnosis were not exactly the same for individual studies. These limitations may be resolved in follow-up studies providing comprehensive results.
Conclusions
CEUS can be effectively applied to detect breast cancer owing to its specific advantages over other imaging modalities in some cases. Data from our meta-analysis suggest that CEUS conducted with the contrast agent SonoVue can effectively differentiate between benign and malignant breast lesions. Despite its current limitations, this technique presents a promising tool for diagnosing breast lesions in clinical practice. Within the recommended dose range, injection of more contrast agent may improve diagnostic performance, along with technologies, such as CAD algorithms or targeted contrast agents.
Footnotes
Abbreviations
Authors’ Note
Jianghao Lu, Peng Zhou, and Chunchun Jin contributed equally to this work.
Acknowledgments
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from Shenzhen Science and Technology Innovation Committee (JCYJ20170413161913429) and Sanming Project of Medicine in Shenzhen (SZSM201612027).
