Abstract
Introduction
We aimed to investigate the feasibility of abbreviated protocol (AP) breast magnetic resonance imaging (MRI) screening in women with dense breasts in a screening program in a middle-income country.
Methods
This prospective study included 649 women selected randomly from 1285 women with type C and D breasts in a screening program, of which 238 underwent an abbreviated breast MRI screening (AP-MRI). The AP-MRI consisted of dynamic series during the first and second post-contrast phases, in addition to axial T1W and axial T2W-fat sat (TRIM) sequences. The reference standard for the study was based on pathology results obtained from biopsies and normal screening mammography results within two years.
Results
MRI scans of 201 women (84.4%) displayed normal or benign findings (BIRADS-1 and 2). An additional 25 women (10.5%) were recommended for MRI or ultrasound (USG) follow-up (BI-RADS-3) and did not show any progression in the two-year follow-up. The remaining 12 women (5%) were recommended a biopsy (BIRADS-4), of which five were reclassified as BIRADS-3 after a second-look ultrasound. Subsequently, a biopsy of the remaining seven patients did not reveal any malignancies. They all had a negative two-year follow-up. The main challenges encountered were scheduling AP-MRI screening and compliance of women.
Conclusion
In this small, single-center cohort of women with dense breasts, supplemental AP MRI screening did not identify additional cancers and was associated with false-positive findings that could lead to overdiagnosis and unnecessary biopsies. Larger, prospective, multicenter studies are needed to clarify the role of AP MRI in breast cancer screening and to determine its optimal integration into existing screening programs.
Introduction
Breast cancer is the most common cancer type and the second leading cause of cancer-related mortality in women worldwide, according to the 2020 global cancer statistics. 1 It is known that the survival time and quality of life increase with the early diagnosis of breast cancer. 2 Randomized controlled studies have shown a reduction in mortality of up to 30% when breast cancer screening is performed with mammography (MG), and it is the standard screening method.3–5 The major limitation of MG is the 2-dimensional, anatomic imaging technique that could mask breast cancer in dense breasts. Studies have shown that, within groups of the same age, there are two to six times higher risk of breast cancer in dense breasts than in non-dense breasts.6–8 Therefore, cancer screening in women with dense breasts needs further attention.
Whole-breast ultrasound (WBUS) is a supplemental screening method for women with dense breasts, with a moderate cancer detection rate (CDR) of 2.0–4.7 per 1000 screened cases.9–13 Although it increases sensitivity, WBUS is associated with high false-positive rates and short-term follow-up.10,14,15Digital breast tomosynthesis (DBT) shows higher sensitivity and specificity than mammography in dense breasts. In the ASTOUND-2 trial of women with mammography-negative dense breasts, incremental CDR was 2.83/1000 for tomosynthesis and 4.90/1000 for ultrasound. 9 Magnetic resonance imaging (MRI) is the most sensitive modality for breast cancer detection, allowing identification of cancers <5 mm due to high soft tissue resolution and multiplanar imaging.16–21 The American College of Radiology recommends MRI for women with a personal cancer history, dense breasts, or diagnosis under age 50. 22 Until March 2022, no guidelines supported MRI screening in average-risk women with dense breasts. In March 2022, EUSOBI advised supplemental MRI every 2–4 years for women aged 50–70 with extremely dense breasts. 23
While ultrasound and DBT are supplemental options, they face limitations: ultrasound has high false-positive rates,10,14 and DBT's incremental CDR remains modest. 9 Full-protocol MRI, though sensitive, is costly and time-intensive. Abbreviated-Protocol MRI (AP-MRI) offers a balance, reducing scan time/cost while preserving accuracy, warranting evaluation in resource-limited settings.
Many studies have investigated new options to decrease the high costs and scan time of complete diagnostic MRI to make it feasible. Kuhl et al introduced the abbreviated protocol (AP) for breast MRI, demonstrating equivalent diagnostic accuracy to complete breast MRI in women with dense breasts. 25 Several studies have been published showing the high diagnostic accuracy of AP breast MRI with faster performance and lower cost.26–34 AP breast MRI protocols described in the literature are broadly consistent, with minor variations primarily seen in the T2 and diffusion-weighted series.35–37 Generally, the AP consists of a T1 W precontrast, a first post-contrast T1 W sequence, a subtracted image, and maximum intensity projection (MIP). Unlike benign lesions, the technique is based on the early enhancement of breast cancers. During the initial phases of dynamic images, background parenchymal enhancement is lower, which can minimize the masking effect in dense breasts.
Although there is a national screening program in Turkey, mammographic screenings are mostly opportunistic.38,39 Bahcesehir Breast Cancer Screening Project (BMSCP) is a pilot study and an organized population-based biennial screening program that has been carried out on women aged 40–69 living in the country Bahcesehir between 2009 and 2019. 40 This screening program investigated the feasibility of a population-based organized breast cancer screening program in Turkey as a middle-income country. In this study, we aimed to investigate the feasibility of supplemental AP breast MRI screening in women with dense breasts in the BMSCP screening programme.
Materials and Methods
The study was approved by the Institutional Review Board of Istanbul University, School of Medicine (Application No.: 2007/152, Date: 24.01.2007). Informed consent was obtained from those eligible women invited to the study who accepted to participate.
Population
Digital mammograms were collected from Bahcesehir screening programme., which is an organized, population-based, 10-year (2009-2019) mammography screening program in Turkey During the 10 years, biennially, screening was carried out in 8758 women between ages 40 and 69 living in the region who were registered to the Bahcesehir Screening Center between January 2009 and January 2019. A total of 22621 bilateral mammography was recorded in the archival system. In the last year of the screening program (between January 2018 and January 2019), mammography was performed on 3299 women. The number of women with heterogeneous dense and extremely dense breast tissue (type C and D) was 1285. Additional screening AP breast MRI was offered to women randomized in a ratio of 1 to 1. As a result, 629 women were invited to the study for AP MRI. Of those, 387 women did not undergo additional MRI screening for the following reasons: 231 didn’t respond, 145 declined to participate, 9 had claustrophobia, and 2 had contrast agent allergy. Finally, 242 agreed to an MRI screening, but images in 4 MR examinations were inadequate to evaluate following a quality assessment. Finally, 238 patients who had heterogeneous dense and extremely dense breast tissue (type C and D) according to the American College of Radiology (ACR) criteria and negative physical examination with negative mammography results during five rounds of screening were included in the study prospectively. The study flow and the number of patients included and excluded from the study are explained in detail in Figure 1.

Study Flow.
Pathology results obtained with biopsies performed after abbreviated MRI scans and normal screening mammography results within two years were accepted as the reference standard.
Mammograms
Digital mammography images were obtained using a full-field digital mammographic device (Selenia, Hologic) from the screening center. Two projections, mediolateral oblique and craniocaudal, were obtained for each woman. Two breast radiologists with more than five years of experience independently read mammograms in the screening center. In case of inconsistency between the readers, a third radiologist with more than 20 years of experience interpreted the findings for the final decision. Mammographic findings were evaluated under the guidance of the fourth edition of Breast Imaging-Reporting and Data System (BIRADS) of the ACR because Bahcesehir screening programme had already started before the last updated fifth version of BIRADS. MRI findings were evaluated under the guidance of the fifth edition of Breast Imaging-Reporting and Data System of the BIRADS. 41
If a suspicious abnormality or highly suggestive of malignancy (BI-RADS 4 or 5 cases) was detected in the imaging in the final report, the radiologists decided on whether to perform a core needle aspiration biopsy (CNB) guided by ultrasonography (14-16 gauge), or a vacuum assisted large core (11 gauge) stereotactic (VALCS) biopsy. Screening is biennial, giving a window of 24 months for an interval cancer to become symptomatic after a negative screening mammography. The prospectively collected outcome data was entered into a computer database as suggested by ACR. Data on pathologic features, any administered therapies, follow-up, and survival outcomes were gathered from our patient and pathology databases.
Magnetic Resonance Imaging
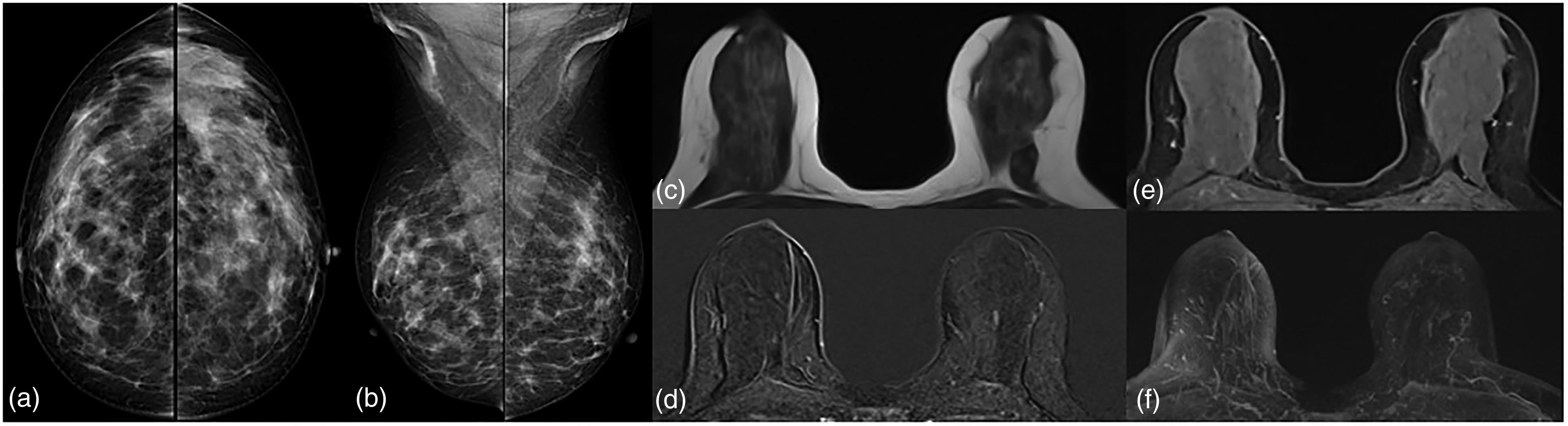
AP MRI scans were performed in 2 different radiology centers. All MRI scans were applied using a 1.5T MR scanner (Symphony, Siemens Medical Solutions), and a dedicated channel phased-array breast coil was used. Before dynamic sequences, an intravenous bolus injection of 0.2 ml/kg (standard dose) gadopentetate dimeglumine (Magnevist; Bayer Schering Pharma, Berlin, Germany) was administered, followed by 20 ml of saline. Each patient underwent AP breast MRI in 0–6 months after screening mammography. In premenopausal women, MRI examinations were scheduled according to the menstrual cycle to minimize background parenchymal enhancement. Whenever feasible, imaging was performed during the second week of the cycle (days 7-14), when BPE is typically at its lowest. This timing was maintained to reduce variability in interpretation attributable to hormonal influences on breast parenchyma. For postmenopausal women, MRI scheduling was not cycle-dependent. The AP consists of the dynamic series’ first and second post-contrast phases and axial T1W and axial T2W-fat sat (TRIM) sequences (Figure 2). Scan acquisition time was 7–10 min. On a pixel-by-pixel basis, unenhanced images were subtracted from contrast-enhanced images, producing a single subtracted post-contrast subtraction sequence. Two dedicated breast radiologists performed image quality assessment, who evaluated the images for fat saturation, artifact severity, and quality of normal anatomic structures as described before. 35 When MRI images were evaluated, normal and benign findings were concluded as BIRADS-1 and 2, the cases MRI or US follow-up was recommended after six months concluded as BIRADS-3 and the patients for whom biopsy was recommended concluded as BIRADS-4.

Composition Abbreviated Protocol. Loc, Localizer Images; FS, fat-Saturated Images; STIR, Short-tau Inversion Recovery; Post, Dynamic Postcontrast Images; sub, T1 fat-Saturated Postcontrast Subtraction Images.
Statistical Analysis
Statistical software SPSS 25 was used for analyses (Statistical Package for Social Sciences; SPSS, IBM Corp., Armonk, NY, USA) 17.0 (SPSS Inc., Chicago, IL, USA). Categorical variables were evaluated by the Pearson Chi-square test. Mann-Whitney U test was used to determine the differences between continuous variables. A p-value equal to or less than 0.05 was statistically significant. Cancer detection rate (CDR), recall rate, biopsy rate, and positive predictive value (PPV) were calculated per 1000 screenings. PPV was defined as true positives / (true + false positives).
Results
A total of 238 women with type C and D breasts were included in the study prospectively. The median age was 44 (min, 40-62), and the mean was 45.5+/- 0.34 years. The physical examination findings of all these women were evaluated as normal. Of those, 14 women were in the extremely dense (ACR type D) group, and 224 were in the heterogeneous dense (ACR type C) group. The screening mammography results were as follows: 88 women had BIRADS-1, and 150 women had BIRADS-2 imaging findings (Table-1). All patients had an MRI after MMG in a month to 6 months due to the restrictions on organizing the appointment in busy diagnostic MRI schedules.
Patient Demographics (n = 238).

*Mann Whitney U test was used. **Pearson Chi-square test was used in the analyses.
Of the MRI scan results, 201 women (84.4%) had normal and benign MRI findings and were classified as BIRADS-1 and 2. Furthermore, 25 women (10.5%) were recommended MRI or US follow-up six months later and were classified as BIRADS-3. The remaining 12 women (5.1%) were recommended for biopsy based on MRI findings and were classified as BIRADS-4. After the second look ultrasound, five women were recategorized as BIRADS-3, and ultrasound follow-up was recommended. The follow-up of these five women and 25 women with BIRADS-3 did not show any malignancy for two years.
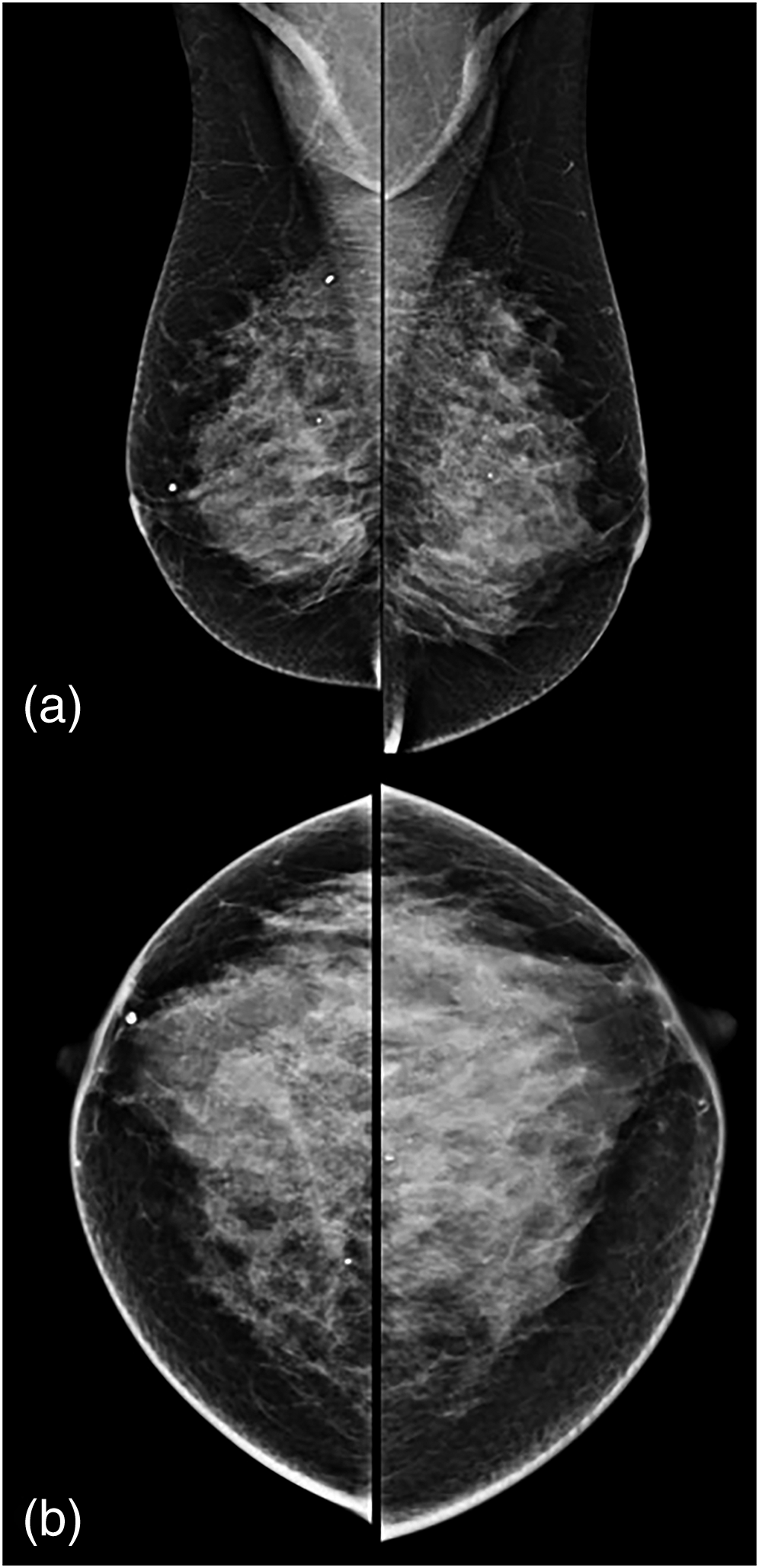
Finally, a core biopsy was performed for the remaining seven patients for whom biopsy was required according to the MRI findings. In the histopathology results, malignancy was not detected in any patient (Table 2). PPV for biopsy and CDR were 0. Recall rate was 15.5% (37/238, 156 per 1000 screenings), biopsy rate was 2.94% (7/238, 29 per 1000 screenings). However, there was only one risk lesion, an intraductal papilloma. Figure 3 shows an example of a normal case with no mammography or MRI findings. Figures 4 and 5 show a contrast-enhanced solid lesion in the lower outer quadrant of the right breast, biopsy-pathology results reported apocrine metaplasia, ductal ectasia, and stromal fibrosis. Figure 6 shows a contrast-enhanced lesion in the upper outer quadrant of the left breast, biopsy-pathology results reported fibroadenomatous changes.
52-Year-old Postmenopausal Women.
45-Year-old Postmenopausal Women.

Abbreviated MR Images of the Previous Figure. Pre-Contrast non-fat-sat T1A Image

46-year-old Postmenopausal Women. T2W-fat sat
Pathological Results of Biopsy Verified Lesions (n = 7).

The follow-up mammography results of all women included in the study were evaluated after a 2-year follow-up, and no evidence of malignancy was found within two years.
No significant differences could be found between patients undergoing a breast biopsy according to the MRI findings and patients without a biopsy regarding demographic features, including menopausal status, family history, and use of hormone replacement therapy, as shown in Table 1. Patients with a lower BMI (<25) were more likely to have a mammogram with a higher density (ACR D: BMI; < 25: 9.3% vs BMI; ≥ 25: 1.8%; p = .015).
Dıscussıon
This study showed no additional cancer detection through supplemental AP MRI screening for women with average risk and dense breasts. During a 2-year follow-up period, no evidence of malignancy was observed. Nevertheless, several studies have reported a significantly higher additional cancer detection rate (CDR), ranging from 11.8 to 27.4 per 1000 screenings with supplemental MRI screening.20,42,43,44 These studies featured different inclusion criteria, encompassing both high-risk and average-risk women and various breast density types, including C and D-type breasts and D-type breasts exclusively. However, a common finding across all these studies is the elevated additional CDR compared to mammography.
Several factors may explain the unexpected result of our study. First, the sample size in our study was relatively small. Nonetheless, in a study by Chen, Shuang-Qing et al, 43 which included 478 female participants with dense breast tissue and negative mammography results, imaged with AP MRI. Fifteen cases of breast cancer were successfully screened using the AP method. Similarly, Kuhl et al 25 found a CDR of 18.2 per 1000 in 606 exams supplementary to mammography screening. Both studies closely resemble our cohort of 238 women. This suggests that with our sample size, at least two cancers should have been detected, as according to the literature, 4.3 to 6.5 additional cancers per screening should be expected for such a cohort of 238 women. However, it's important to note the very limited number of participants in our study. Considering the acceptance rate of AP MRI screening among the invited women, stood at 38.5%, with 37.8% of them completing high-quality AP MRI screening. This relatively low number of study inclusions may have introduced a bias to our results.
Second, the women in our study had undergone five rounds of mammography screening over ten years. It is well-established that after successive rounds of screening, interval cancers tend to decrease. The CDR among the women who underwent MRI was 16.5 per 1000 screenings in Bakker et al's study. 44 The second screening round of their study showed a CDR of 5.8 per 1000 screening examinations compared with 16.5 per 1000 in the first round. 45 So, Bakker et al’ results strongly support the decrease of CDR after successive screening rounds. However, in the ACRIN 6666 study, 20 it was demonstrated that after three consecutive rounds of mammography screening supplemented with US screening, they could still detect 14.7 additional cancers per 1000 screenings with MRI. Conversely, we have not detected any cancer with supplementary MRI screening.
Third, our interval cancer rate was notably lower (2.03 per 1000 screening). 40 In the Dense trial, the interval cancer rate was 5, subsequently decreasing to 0.8 per 1000 screenings after MRI screening. 44 However, our interval cancer rate was notably lower than the Dense trial. This difference may have diminished the significant impact of the supplementary AP-MRI screening results.
Despite high CDR, long scan acquisition, and interpretation time, high cost limits breast MRI for screening in all women-risk populations. Therefore, the 2007 American Cancer Society (ACS) has recommended additional breast MRI screening for women at high risk. This group includes women with genetics-based increased risk and their untested first-degree relatives, those with a calculated lifetime risk of 20% or greater, and those with a history of chest or mantle radiation therapy at a young age. 22 A more personalized approach should be favored over offering MRI screening to every woman with risk. A recent study by Lamb LR et al utilizing a mammography-based AI risk assessment tool demonstrated a higher CDR in women evaluated by this AI-based tool compared to traditional risk models. 46 Furthermore, this study highlights that the risk assessment is contingent on the fibroglandular structure observed in mammography rather than the ACR density categories or conventional risk scores. Although many guides do not recommend MRI screening for average-risk women,47–49 EUSOBI has published a new recommendation that recommends supplemental full protocol MRI screening to women with extremely dense breasts from age 50–70 and at least every four years, preferably every 2 or 3 years in March 2022. 24 ACR guideline supports that MRI screening can be beneficial for dense breasts despite the accumulation of sufficient data.20,21,49 However, a study conducted by Geuzinge HA et al 50 calibrated with the findings from the Dense trial, 44 demonstrated the cost-effectiveness of MRI screening at 4-year intervals, with a cost of €15,620 per Quality-Adjusted Life Year (QALY). This assessment utilized a threshold of €22,000 per QALY gained. Consequently, a three or 2-year interval for MRI screening was deemed not cost-effective. Although our study did not specifically assess the cost-effectiveness of MRI screening, countries with limited resources need to be mindful of the limitations associated with MRI screening and carefully evaluate its cost-effectiveness.
Our study represents a feasibility assessment to explore the potential integration of AP breast MRI into a screening program. Despite numerous studies in the literature demonstrating higher CDR,42,43 our findings did not align with these previous reports. While AP breast MRI holds promise as a screening modality, several operational considerations must be addressed to facilitate its adoption into clinical practice. For instance, it is widely accepted that reducing image acquisition and interpretation times can enhance availability and cost-effectiveness. However, the reality in real-life scenarios often diverges from these expectations, particularly within government hospital systems, where cost reduction may not be readily achievable. One of the challenges we encountered during the study was incorporating screening AP-MRI into a schedule already filled with diagnostic MRI appointments. This proved to be a daily operational challenge. Additionally, while image acquisition and interpretation times may be shorter, there remains a debate about the actual overall workflow efficiency.
A study by Borthakur et al 51 investigated various aspects of workflow, including imaging setup, patient preparation, and MRI room turnover, revealing that the flow rate for an abbreviated MRI examination was lower than anticipated. Therefore, radiologists and clinicians should be aware of these practical challenges when considering the implementation of AP breast MRI into clinical practice. The challenges in clinical practice extend beyond longer-than-expected total procedure times. For example, incorporating screening AP MRI into an appointment schedule filled with diagnostic MRI appointments posed practical difficulties. Furthermore, it has been observed that AP MRI can increase the rate of unnecessary biopsies, leading to increased costs. This represents one of the primary disadvantages of incorporating screening MRI. However, the EUSOBI guideline recommends screening for women with extremely dense breasts from age 50–70 and at least every four years, which seems to offer potential advantages, particularly considering the findings from the Dense Trial. 44 Nevertheless, in contemplating the integration of AP MRI into a screening program, one must bear in mind the possible challenges given above.
Our study has some limitations. First, the number of participants was low to make an optimal comparison with MRI and mammography. Second, the number of women who did not respond was very high, possibly due to being called up after five screening rounds.
This study represents a real-life scenario involving the implementation of supplemental breast screening for women with dense breasts. The study revealed no additional cancer detection among the 238 women with dense breasts. However, this result came at the cost of a 15.5% recall rate, a 5% biopsy recommendation, and a 3% biopsy rate. Furthermore, the study encountered several challenges, including scheduling appointments within a busy MRI schedule and managing various aspects of workflow. It is crucial to consider these limitations when considering the implementation of MRI in large-scale screening programs. Meticulous criteria should be applied when selecting women for MRI screening to ensure optimal outcomes.
Conclusion
In this small, single-center cohort of women with dense breasts, supplemental AP MRI screening did not identify additional cancers and was associated with false-positive findings that could lead to overdiagnosis and unnecessary biopsies. Logistical challenges, including timely scheduling, may further limit feasibility in broader screening settings. These findings should be interpreted within the context of the limited sample size and setting, and are not intended to generalize to all populations. Larger, prospective, multicenter studies are needed to clarify the role of AP MRI in breast cancer screening and to determine its optimal integration into existing screening programs.
Footnotes
Acknowledgments
The authors thank the Bahçeşehir Breast Screening Facility, including, Nursen Has, Deniz Doğan Ozkan, Semra Yasar, and Ahu Ozdemir in particular, for providing the screening data used in this study. We are grateful to all Turkish women who participated in this prospective trial.
Statement of Ethics
All procedures performed in the present study involving human participants were by the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. The authors declare that the subjects have given their written informed consent and that the Institute's Ethical Committee of Istanbul University has approved the study protocol. (Application No.: 2007/152, Date: 24.01.2007/01).
Authors’ Contributions
The study was designed by EA and NC. IK, NC, and EA performed the initial search, literature organization, analyses, and manuscript writing. EA, MD, IK, OC, NC, and RY provided data acquisition. AS, NO, SOG, BO, VO, and AK made critical comments and typesetting corrections on the final version. All authors have read and revised the manuscript critically.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project is supported by Roche Pharmaceutical Company.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
Data can be shared if requested.
