Abstract
Metal-organic frameworks nanocomposites emerged as a novel functional material that has been reported to combine with metal nanoparticles for enhancing antibacterial activity through synergy. Herein, Ag nanoparticles and zinc-metal-organic frameworks nanocomposites are successfully in-situ synthesized on the surface of non-woven fabrics carrier via solution-immerse-growth method. The obtained product (ZA/NWF) shows superior utilization of visible light than the sample that prepared without Ag nanoparticles. Moreover, the generated reactive oxygen species by ZA/NWF is also more abundant, which indicates the effective visible light utilization of the as-obtained material. ZA/NWF shows considerable bactericidal effect against Escherichia coli with a nearly 100% bactericidal rate under visible light illumination, which is significantly higher than that of Zn2+ (70.3%) and 2-methylimidazole (71.8%). A proposed antibacterial mechanism suggests that hydroxyl radical (·OH) and superoxide radicals (·O2-) generated during the antibacterial process are conducive to the inhibition of Escherichia coli and Staphylococcus aureus. Additionally, ZA/NWF shows comparable sterilization efficiency of over 95% after the sixth antibacterial assay. Overall, a facile and convenient antibacterial material integrated structure and property is presented in this work and the composite with competitive photocatalytic antibacterial activity can be potentially applied in the large-scale antibacterial study.
Introduction
The antibacterial property of Ag nanoparticles (Ag-NPs) was recognized for decades, and this benefitted the development antibacterial materials.1,2 Ag-NPs with large surface area to volume ratio and outstanding optical property exhibit a broad spectrum of antibacterial activity towards mucedine, Gram-positive and Gram-negative bacteria via the generation of reactive oxygen species and damaging of bacterial cell walls. 3 However, the aggregation of Ag-NPs is an inevitable issue due to their high surface energy, resulting in the thermal instability and decrease in the antibacterial effect of Ag-NPs. 4 Accordingly, various strategies aiming at finding suitable substrates have been proposed, which should meet the requirements of both retaining the original antibacterial activity and keep the well disperse of Ag-NPs.
Numerous reports on the immobilization of Ag-NPs onto different carriers have concluded that nanocelluloses, 5 multi walled carbon nanotubes, aluminosilicate nanotubes, polymer substrate 6 and metal-organic frameworks (MOFs) 7 can be used as effective matrices for further exploration. Researches have been devoted to finding effective methods for the incorporation of Ag-NPs into matrices, including in-situ coating, loading of prefabricated nanoparticles, and doping into a synthetic mixture. For instance, the work in 2019 with the loading of Ag-NPs on modified multi walled carbon nanotubes used natural reductant agent to convert Ag ions to Ag-NPs and stabilize them. The primary task is to prepare S. lavandulifolia extracts as reductant agent, which is time consuming and tedious. 8 Another approach is to use colloidal method to synthesize Ag-NPs on aluminosilicate nanotubes without utilizing of reducing agents or any organic additives, and the nanotube/nanoparticle hybrid showed strong antibacterial activity. 9 Although these strategies have made considerable progress, there still many problems need to be addressed such as utilization of external reducing agent, complex modification and difficulty in recovery. 10
In recent years, non-woven fabrics (NWF) is increasingly popular for the loading of active antibacterial substances due to its high thermal stability and biostability, durability, convenience, and low cost. Li’s group prepared a dense layer of metal nanoparticles on the convenient NWF carrier, 11 which shows strong application feasibility than the powder substrates. The fabrics such as cotton, polyester (PE), polyethylene terephthalate (PET) and polypropylene (PP) is closely related to people’s lives.12–14 For example, PE NWF is widely used in biomedical applications and endowing the fabrics with antibacterial property can avoid the direct and indirect spread of microorganisms in certain circumstances. Therefore, it’s of great significance to develop antibacterial modified fabrics in both scientific and practical areas.
MOFs have attracted great attention in the preparation of novel hybrid materials with other active composites via synergistic effect to obtain remarkable performance.15–19 Especially in the recent decades, great effort has been made to investigate the possibility developing MOFs as promising antibacterial material due to their instinct merits such as large surface area, highly porous structure, biocompatibility, and adjustable function.4,20 Zeolitic imidazolate framework-8 (ZIF-8) has been widely applied as antibacterial agent, however, the poor recyclability and limited visible light utilization has restricted its practical use. 21 Great efforts have been made to endowing MOFs with enhanced antibacterial property by introducing active groups or particles with MOFs, including in-situ doping, surface modification, and encapsulation of active species.22–24 Zinc 1, 3, 5-benzenetricarboxylate combined with antibiotic has been reported with significantly improved antibacterial efficacy due to synergy. 25 According to previous reports, the antibacterial activity of MOF can obviously improve by combining MOFs with noble metals.26,27 Therefore, zeolitic imidazolate framework-L (ZIF-L) with a slight visible-light-driven characteristics can serve as a good component for the combining with Ag-NPs for further improving the photocatalytic and antibacterial properties of the materials.
In this study, Ag-NPs and sheet-like ZIF-L were in-situ synthesized on non-woven fabrics through a simple solution-immerse-growth method for antibacterial activity investigation. Ag-NPs were coated on non-woven fabrics via a simple hydrothermal reaction (Ag-NPs/NWF), then ZIF-L further grew on the Ag-NPs/NWF fabrics by immersing in the Zn(NO3)2 and 2-methylimidazole mixture. The well dispersion of Ag-NPs and ZIF-L components on the fabric substrate, on one hand, solves the problem of aggregation and decrease of antibacterial effect, on the other hand, it improves operational flexibility and reuse rate of materials. The detailed antibacterial performance of prepared ZA/NWF was systematically investigated against Escherichia coli (E. coli) and Staphylococcus aureus (S. aureus) under visible light irradiation and the mechanism was analyzed. ZA/NWF presented here showed great potential as a photocatalytic antibacterial candidate driven by sunlight for further practical demand and it highlights the promising scope to develop the potential applications of non-woven fabric based convenient antibacterial materials in the people’s lives.
Experimental section
Materials and reagents
All chemicals in this work were used as received unless otherwise is stated. Polyester non-woven fabrics (Shenzhen Changtai Packaging Material Co., Ltd) purchased from the local supermarket were used as the carrier material. The average fiber diameter of the samples was 15 μm and basis weight 12.5 g m−2 for polyester non-woven fabrics. Silver nitrate (AgNO3), hydrochloric acid (HCl, 37%), zinc nitrate hexahydrate (Zn(NO3)2·6H2O), 2-methylimidazole (H-MeIM, C4H6N2, 98%), ethanol, N,N-dimethylformamide (DMF, C3H7NO, ≥99.5%), yeast extract, tryptone, NaCl, phosphate buffer solution (PBS, pH = 7.4), agar, 5,5-dimethyl-1-pyrrolidine-N-oxide (DMPO), dimethyl sulfoxide (DMSO), SYTO nine green fluorescent nucleic acid stain and propidium iodide (PI) were provided from Sinopharm Chemical Regent Co., Ltd. (Shanghai, China). Deionized water used in the experiment were obtained from the laboratory system.
Characterizations
The morphologies and dimensions of different samples were observed using a scanning electron microscope (SEM Hitachi S-4800) equipped with an EDS attachment. The samples were fixed on the conductive tape and treated with metal spraying before measurement. X-ray powder diffraction (XRD, Shimadzu XRD-6100) was used to examine the crystal structure of sample in the 2-theta angle ranging from 5° to 80° with a scanning rate of 5°min−1. The composition and of as-prepared samples were performed on an X-ray photoelectron spectrometer (XPS, Thermo ESCALAB 250 XI). The photoabsorption properties of the as-prepared composites were tested by UV-visible diffuse reflectance spectroscopy (DRS) on a Shimadzu UV-2450 spectrophotometer. Electron spin resonance (ESR) signals were recorded using a Magnet Tech MS400 spectrometer with 5, 5-diemthyl-1-pyrroline-N-oxide. The confocal fluorescence images of bacteria were characterized by Leica SP5 laser scanning confocal microscope (LSCM). In this work, the visible-light irradiation was obtained from a 300 W Xe arc lamp with a cut-off filter of 380 nm and the light intensity is 100 mW cm−2 (CEL-HXF300, Ceaulight, Beijing).
Synthesis process of materials
Preparation of Ag-NPs/NWF
The synthesis of Ag-NPs coated non-woven fabrics was described as follows via a simple hydrothermal reaction.28,29 In detail, the pristine white non-woven fabrics (5 cm ×5 cm) was treated with 5 mM HCl for 2 h to remove the impurities on the surface and commercial AgNO3 was dispersed in DMF to prepare a AgNO3 solution with the concentration of 8 g L−1. Then, the activated non-woven fabrics was immersed in 100 mL of AgNO3 solution under magnetic stirring for 0.5 h to absorb Ag+. After that, the mixture solution was transferred into a Teflon reactor and heated at 80°C for 4 h. The solvent DMF was used as the reducing agent to obtain Ag-NPs. Finally, the modified non-woven fabrics was taken out and washed by deionized water and ethanol, respectively. The dried non-woven fabrics with a silver light was denoted as Ag-NPs/NWF, and the yield was 15.2–17.6%.
Preparation of ZA/NWF
For the preparation of ZA/NWF, at first, 1.6 g of H-MeIM was dispersed in 50 mL of deionized water under ultrasonic for 10 min, then the as-prepared Ag-NPs/NWF was immersed in the mixture. Subsequently, another 25 mL of deionized water with 0.75 g of Zn(NO3)2 was dropwise added into the above solution and sonicated for 5 min to form homogenous mixture. The mixture was stirred for 4 h at 40°C to in-situ construct ZIF-L on the fabric.30,31 Finally, the obtained argent ZA/NWF was washed with plenty of deionized water and ethanol and dried for use. The yield of the ZIF-L on the Ag-NPs/NWF was calculated to be 69.5%. For contrast, ZIF-L/NWF sample was also prepared in the similar way by replacing Ag-NPs/NWF with pristine non-woven fabrics. The synthesis process of Ag-NPs/NWF and ZA/NWF is shown in Figure 1. Synthesis process of Ag-NPs/NWF and ZA/NWF.
Optical property test and antibacterial assay
The UV-vis diffuse reflectance spectroscopy (DRS) spectra of ZIF-L/NWF and ZA/NWF were measured in a range of light from 380 nm to 800 nm to evaluate the light absorption behavior of the samples. Moreover, the corresponding band gap of ZIF-L/NWF and ZA/NWF were calculated by equation (1) to assess the light use efficiency.
where α represents the absorption coefficient, h, v, A and E g are Planck constant, light frequency, proportionality constant and band gap energy, respectively.32,33
The radicals trapping experiment was also performed to compare the generated reactive oxygen species (ROS) generation ability of the as-prepared ZIF-L/NWF and ZA/NWF. DMPO was used as radical trapper to form stable complexes with hydroxyl radical (OH) and superoxide radicals (O2-) (DMPO-OH and DMPO-O2-) with or without visible light illumination that provided by a halogen lamp of 300-Watt quartz.
The antibacterial property of as-prepared ZA/NWF was assessed against E. coli and S. aureus according to our previous study with slight modification. Typically, Luria-Bertani solid medium was prepared by solving 5 g of peptone, 2.5 g of yeast extract, 5 g of agar and 5 g of NaCl in 500 mL of deionized water. And the Luria-Bertani liquid medium was prepared in the same method without agar. E. coli and S. aureus were grown for 10 h in 50 mL of Luria-Bertani liquid medium at 37°C with 200 r/min rotation. Afterwards, both bacterial were diluted with PBS (pH = 7.4) to 106 cfu mL−1 (optical density at 600 nm) for the following antibacterial evaluation, respectively. Specific methods are as follows:
To intuitively evaluate the antibacterial activity of ZA/NWF, 100 μL of diluted bacteria culture fluid was evenly coated on Luria-Bertani solid medium and then the sample exposed to the environment for 2 days was placed in the center of the Luria-Bertani solid medium. The treated medium was putted 0.7 m away from visible light and illuminated by light source for 30 min, and cultured at constant temperature with 37°C. After culturing for different times (1 day, 5 days and 10 days), the medium was taken photographs to determine the antibacterial activity. As contrast, pristine NWF was also treated in the same way.
Subsequently, the diluted bacteria culture fluid was also inoculated into the Luria-Bertani liquid culture medium and the mixture was transferred to the cell culture plate to ensure the same amount of broth was present in each well of the plate. Zn(NO3)2, H-MeIM and ZA/NWF with equal amount were put in the individual well of the plate, respectively, and then the cell culture plate was treated for 30 min under a visible light source before cultured at 37°C in a temperature humidity chamber. A well without any samples was prepared as a control. After processing for 10 h, the bacterial concentration was measured with UV-vis to determine the survival rate of the bacteria. In addition, the experiment was also carried out in dark for comparison. The tests were performed in triplicate. All the instruments and materials used in the experimental test were sterilized in an autoclave for 30 min, and the experiment process was conducted in the aseptic conditions.
Besides, the reusability of ZA/NWF was tested for six cycles under visible light and the sterilization rate after each cycle was calculated. Then, the operations were repeated for another cycle, and the results of the six cycles were contrasted to evaluate the recycling property of the as-prepared ZA/NWF. The reusability test was performed in triplicate.
Results and discussion
Morphology analysis
The microtopography of Ag-NPs/NWF, ZIF-L/NWF and ZA/NWF were confirmed by SEM (Figures 2(a) to (d)). As shown in Figure 2(a), the pristine non-woven fabrics with a diameter about 15 μm are evenly coated with Ag-NPs, and the size of Ag-NPs is about 300–500 nm. Figure 2(b) shows the microstructure of ZIF-L/NWF, it’s clear that there are many ellipse slices of uniform size vertically grew on the surface of non-woven fabrics, indicating the construction of ZIF-L structure on the substrate. In addition, ZIF-L were designed to in-situ form on Ag-NPs/NWF for investigation. Compared with ZIF-L/NWF, the surface of Ag-NPs/NWF was also wrapped with ZIF-L and the fabrics diameter increased slightly (Figure 2(c)). From the enlarged SEM image in Figure 2(d), the morphology of both Ag-NPs and ZIF-L did not change, suggesting that the material is facile to prepare via simple water bath heating reaction. EDS element mapping results (Figure 2(e)) present a well distribution of Zn and Ag elements along the pristine non-woven fabrics composed of C, N and O elements, indicating the coexistence of Ag-NPs and ZIF-L in the composite material. SEM images of Ag-NPs/NWF (a), ZIF-L/NWF (b) and ZA/NWF (c and d). Elemental mapping images of ZA/NWF (e).
Composition analysis
The composition of as prepared material was confirmed by XRD, FT-IR and XPS characterizations. Figure 3(a) shows the XRD pattern of ZA/NWF. The characteristic peaks displayed from 2θ=5°–50° are in good agreement with the simulated ZIF-L (CCDC number:1,509,273).
34
Additionally, the peaks at 64.367° and 77.362° are well assigned to [220] and [311] crystalline Ag-NPs (JCPDS No.87–0597).
35
These observed diffraction peaks confirmed the successful preparation of ZA/NWF. Moreover, the FT-IR spectra of NWF and ZA/NWF is presented in Figure S1. Compared with pristine NWF, the FT-IR spectrum of ZA/NWF shows evident difference in the range of 700–1600 cm−1, which should be assigned to the characteristic peaks of ZIF-L.
30
XRD pattern of ZA/NWF (a); XPS spectra of ZA/NWF: survey (b), Ag 3p (c) and Zn 2p (d).
XPS analysis on the ZA/NWF sample is displayed in Figures 3(b) to (d). It’s revealed from the XPS survey spectrum of ZA/NWF (Figure 3(b)) that Zn, Ag, C, O and N elements emerged at 1022.1, 367.1, 284.7, 529.8 and 399.8 eV co-exist in the material, which is consistent with the EDS mapping results. Figure 3(c) shows the high resolution XPS spectra of Ag 3p. Two peaks can be observed at 368.4 eV and 374.0 eV that corresponds to the energies of Ag 3d5/2 and Ag 3d3/2, implying the existence form of metallic Ag (Ag0) and Ag+ species in the composite. 36 The detected Ag+ species might be due to the silver oxide formed on the pristine non-woven fabrics during the preparation process. The high-resolution Zn 2p spectra (Figure 3(d)) presented two peaks appeared at 1020.7 and 1043.8 eV. According to the previous report, Zn element existed in the following two species: Zn 2p3/2 and Zn 2p1/2, which representatively assigned to the chemical state of Zn2+. 37 Hence, the results further demonstrated the successful synthesis of ZA/NWF.
Optical property analysis
In order to investigate the photoabsorption performance of the prepared samples, the UV-vis diffuse reflectance spectra (DRS) test was carried out (Figure 4(a)). For ZIF-L/NWF, it adsorbed UV to visible light with an adsorption edge at 400 nm, suggesting that ZIF-L has slight visible-light-driven characteristics of its own. In contrast, ZA/NWF exhibited enhanced optical absorption in visible light region after the introducing of Ag species, hence enhancing the efficient utilization capacity of the solar energy of ZA/NWF. This is might ascribed to the surface plasmon resonance effect of Ag-NPs itself that can enhance the light adsorption and the electron transfer between Ag-NPs and ZIF-L.
2
UV-visible diffuse reflectance spectra in the visible light range (a) and plots of (Ahv)2 vs. photon energy (hv) of ZIF-L/NWF and ZA/NWF (b).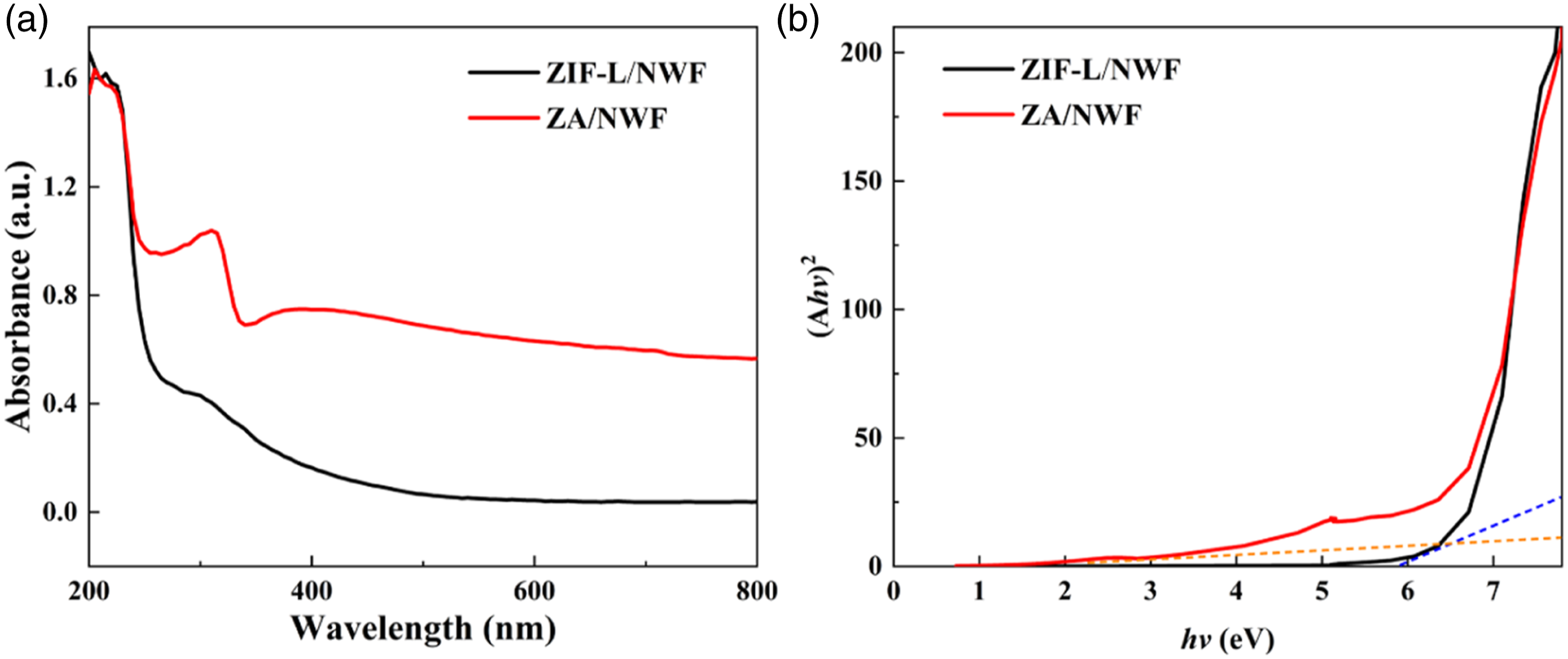
Moreover, the band gap energies of the as-prepared samples were calculated according to the equation (1). The plot of ZIF-L/NWF and ZA/NWF are displayed in Figure 4(b). It’s estimated that the band gap energies of ZIF-L/NWF and ZA/NWF are 5.82 eV and 2.24 eV, respectively. The obvious decrease of the band gap energy for ZA/NWF composite should be ascribed to the combination effect of Ag-NPs and ZIF-L, which improves the light use efficiency and separation of photogenerated electron-hole pairs. 38 It’s well known that Ag-NPs can serve as electron acceptors to capture photogenerated electrons produced by photoexcitation, averting the rapid recombination of photogenerated electrons and holes.39,40 This supports the narrower band gap of ZA/NWF. Therefore, it can be drawn that the ZA/NWF sample possesses significant utilization of visible light.
ESR characterization for detecting OH and O2- generated with or without visible light illumination was conducted using DMPO as radical trapper, of which OH was detected in aqueous solution and O2- was tested in the solvent of DMSO. Figure 5 showed the signal peaks of DMPO-·OH and DMPO-·O2- in ZIF-L/NWF and ZA/NWF under the illumination of visible light or in dark, respectively. For OH, four typical signal peaks could be observed for ZIF-L/NWF and ZA/NWF. And the signal peaks were significantly increased when the samples were treated under visible light rather than in dark. For O2-, there is also a distinct difference in the ESR spectra with or without visible light environment. DMPO-·O2- signals were almost not detected in dark, while the peaks of ZIF-L/NWF and ZA/NWF were evidently higher upon visible light illumination. The truth elucidate that more ROS could be generated under the visible-light irradiation. Furthermore, the generation amount of OH and O2- can be deduced from the peak intensity.
41
It’s obvious that the intensity of both DMPO-·OH and DMPO-·O2- signals of ZA/NWF is apparently higher than that of ZIF-L/NWF, symbolizing the superior photocatalytic performance of ZA/NWF. ESR spectra of DMPO-·OH adducts and DMPO-·O2- adducts over ZIF-L/NWF and ZA/NWF under the illumination of visible light or in dark.
Antibacterial activity and reusability of samples
The tests on the antibacterial activity of ZA/NWF were carried out using E. coli as the standard organism. Firstly, as shown in Figure 6(a), Luria-Bertani medium liquid culture medium containing E. coli culture fluid with equal amount was added into each well of the cell culture plate, and Zn2+, H-MeIM and ZA/NWF with the same weight was added, respectively. Then the antibacterial efficiency of Zn2+, H-MeIM and ZA/NWF were measured under both visible light condition and dark condition. Meanwhile, a blank control group without the addition of any samples was set to determine the effect of visible light on the bactericidal activity. It can be intuitively seen that all the Luria-Bertani medium liquid culture medium of experimental groups in dark become turbid, of which the turbidity degree is extensively higher than that under visible light. The group of ZA/NWF with visible light illumination shows a perfectly clear liquid. This implies the acceptable antibacterial activity of ZA/NWF. The antibacterial activity of Zn2+, H-MeIM and ZA/NWF against E. coli (a). The statistics results of bacteria viability after incubation with Zn2+, H-MeIM and ZA/NWF (b).
In order to further evaluate the bacterial sterilizing rates of Zn2+, H-MeIM and ZA/NWF, the bacterial concentrations in each well were measured by UV-vis. 21 The final bacteria viability was calculated according to the absorbance value with the absorbance value of the blank group in dark defining as 100% bacterial survival rate. As displayed in Figure 6(b), the bacteria viability of blank group (only E. coli) shows little difference whether in dark or there was visible light, indicating the effect of visible light on the antibacterial activity can be negligible. In contrast, after the samples treated under light source for 30 min, Zn2+ and H-MeIM showed antibacterial properties to a certain degree. The bacteria viability values of both Zn2+ and H-MeIM declined, which are also similar with or without visible light, respectively. The corresponding value decreased to 70.3% (visible light) and 77.4% (in dark) for Zn2+ against E. coli and 71.8% (visible light) and 73.4% (in dark) for H-MeIM against E. coli. Nevertheless, ZA/NWF sample showed considerable bactericidal effect with visible light illumination, the bacteria viability value is only 2.03%, suggesting nearly 100% bactericidal rate. Without light irradiation, the bacteria viability decreased to 57.7%, the result shows that ZA/NWF in dark possesses more powerful antibacterial activity than Zn2+ and H-MeIM. This efficient antibacterial activity may be due to the co-existence of Ag-NPs and ZIF-L, which can effectively inhibit the recombination of holes and electrons, thus further enhancing the photocatalytic antibacterial ability of the material.
Blank non-woven fabric and ZA/NWF were placed in the Luria-Bertani medium solid medium under irradiation for 1 day, 5 days and 10 days to visually assess the antibacterial activity, respectively (Figure S2). It’s easy to see that the bacterial colonies on the blank sample expanded as the cultured time increased, indicating no antibacterial activity. Whereas, ZA/NWF showed no bacterial colonies until the 10 days, which is distinctly different from the blank control group. Therefore, the results also verify the comparable antibacterial ability of ZA/NWF.
Furthermore, the bacteria viability of E. coli and S. aureus were also measured by Live/Dead assay conducted on a laser scanning confocal microscope (LSCM). A Live/Dead backlight bacterial viability kit that containing both SYTO nine green fluorescent nucleic acid stain and propidium iodide was used for sample staining. Figures 7(a) and (c) display the blank group with only E. coli and S. aureus, respectively, and E. coli and S. aureus that incubated on ZA/NWF are shown in Figures 7(b) and (d), respectively. Living cells under florescence microscope present in green and the converse is red. It’s observed almost no dead E. coli and S. aureus presents in the blank group. On the contrary, the LSCM micrographs revealed increasing number of dead bacteria could be observed when cultured with ZA/NWF. The phenomenon is in consistent with the result of above antibacterial assay, implying ZA/NWF exhibits effective antibacterial property. Confocal fluorescent micrographs of pristine E. coli (a), S. aureus (c) and E. coli (b), S. aureus (d) incubated on ZA/NWF under visible light irradiation for 30 min. The sterilization rate of ZA/NWF in six cycles (e).
The reusability of the prepared ZA/NWF was tested for six cycles under visible light and the sterilization rate was converted according to the mentioned method in Figure 6(b). As can be seen in Figure 7(e), the sample showed satisfied sterilization effect on E. coli during the experiment cycles. After the sixth antibacterial assay, the sterilization efficiency of ZA/NWF still maintains over 95%, suggesting that ZA/NWF could be used for several times with sufficient activity. The superior reusability of ZA/NWF can significantly reduce cost and favor its promising application in future large-scale antimicrobial field.
Proposed antibacterial mechanism of ZA/NWF
It has been reported that the effective combination of MOF and noble metal can produce more bactericidal ROS under visible light irradiation, hence improving the antibacterial activity, which has been regarded as a new kind of photocatalytic antibacterial material. In this study, the bacteria initially surround and get adhere on ZA/NWF material, the functional composite of Ag-NPs and ZIF-L thus come into effective contact with the bacteria and the sheet-like structure of ZIF-L and irregular Ag-NPs could form physical interaction with them, leading to the deformation and apoptosis of bacteria. Under irradiation, the excited electrons in the valence band of ZIF-L leapt into the conduction band, resulting in the holes (h+) leaving behind in the valence band. Subsequently, photogenerated electrons escaped from ZIF-L to Ag-NPs, causing the photoexcitation of molecular oxygen in aqueous solution into O2- and oxidizing reaction of H2O to OH (Figure 8). This process can effectively facilitate the separation of excited charge carriers, thus accelarating the generation of O2- and OH.42–44 Besides, the slow release of silver ions could also affect membrane, DNA, and proteins. In general, the co-existance of ZIF-L and Ag-NPs on ZA/NWF expedites the separation of photogenerated electrons and holes, ensuring the large quantity of ROS for antibacterial procedure, which is the necessary substances for the inhibition of bacteria. Illustration of antibacterial mechanism of ZA/NWF.
Conclusions
In summary, ZA/NWF was in-situ synthesized with well dispersion through a simple solution-immerse-growth strategy. The effective combination of Ag-NPs and ZIF-L onto fabrics greatly avoids the aggregation issue of metal nanoparticles and significantly enhances the antibacterial activity when compared with ZIF-L/NWF sample. The optical test results demonstrate that the improved antibacterial mechanism should be ascribed to the high use efficiency of visible light and abundant generation of ROS. The bactericidal rate against E. coli is nearly 100% after treating under light source for 30 min, confirming the acceptable potential application of ZA/NWF. In addition, the sterilization efficiency of ZA/NWF still maintains over 95% after the sixth antibacterial assay. Therefore, the antibacterial material with simple process and competitive performance in this study has a promising foreground in the deep research on developing visible-light induced antibacterial agents.
Supplemental material
Supplemental material - In situ deposition of Ag/Zn-MOFs on the surface of non-woven fabrics for effective antibacterial activity
Supplemental material for In situ deposition of Ag/Zn-MOFs on the surface of non-woven fabrics for effective antibacterial activity by Yao Zhu, Kaili Mao, Jian Rong, Yunhua Zheng, Tao Zhang, Dongya Yang and Fengxian Qiu in Journal of Industrial Textiles
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.

