Abstract
The integration of high conductive networks and textiles has become a favorable technical route to fulfill the objectives of wearable electronics. Herein, high stretchable and recoverable PET fabric coated with a layer of silver nanowire network by a simple and scalable polyol-method is provided. The electrothermal performance, resistance temperature-sensitivity, electromagnetic shielding performance, strain sensing, and washability of silver nanowire (AgNWs)/PET fabrics with different coating times were performed. The conductivity of the fabric coated AgNWs of 2.8 mg/cm2 is as high as 175 S/m, the EMI shielding effectiveness is 37 dB, and it gives a highly sensitive strain response to human movement (gauge factor of −6.16 under 10% strain) and an underwater oil repellent angle of 125°. The heating temperature can reach above 100°C within 27 s under an applied current of 0.10 A. In addition, an excellent linearity of the resistance temperature-sensitive behavior for AgNWs/PET fabrics is obtained, and fabric Ag-5 gives a negative temperature coefficient of resistance (TCR) of −0.05%/°C. Knitted fabric with multi-function is obtained by use of silver nanowire coating. This method provides a simple, low-cost and easy-to-scalable process for the production of electronic textiles, such as fabric heater, microwave blocker, sensor, and other technologies.
Keywords
Introduction
Smart wearable textiles have attracted widespread attention due to their flexibility, high sensitivity, stretchability, and light weight. Generally, conductive polymers (polyaniline,1,2 polypyrrole, and3,4 polythiophene5,6), carbon materials, 7 metal particles 8 and piezoelectric yarns, 9 nanowires, 10 nanotube, 11 PVDF and ZnO nanofibrous composites, 12 photovoltaic energy textile, 13 copper, 14 nonmonotonic piezoresistive fibers, 15 and other nanomaterials can all make textiles conductive. However, conductive polymer as a conductive fill gives a relatively high conductivity, 16 but poor hand feeling and air permeability. Conductive metal wires and short carbon fiber have also been used to demonstrate wearable strain sensing fabrics,17,18 but these materials give rigid properties, which are not suitable for wearing, and cannot provide satisfactory biocompatibility, flexibility, durability, and intelligent functions such as integrated smart sensing, and their low electrical conductivity and the complex preparation methods are also limit their application. 19 Carbon-based materials require a high vacuum environment or toxic chemical processes to show high electrical conductivity.20,21 Therefore, for electronic and smart textiles,22,23 the development of conductive materials with excellent electrical and thermal conductivity and combined with fibers is still an unsolved problem.
AgNWs can be used as conductive fillers to render fabric conductivity. At present, conductive fabric have been widely used in electromagnetic interference shielding, liquid crystal display, light-emitting diode, electronic packaging, radio frequency identification, bio-nano sensor, and other fields.24–26 AgNWs has attracted much attention ascribed to its high electrical and thermal conductivity, which is an important pillar in many fields. In addition, AgNWs possesses excellent flexibility. It can adapt to large deformation as the diameter of AgNWs is usually less than 100 nm, and the length is from hundreds of nanometers to tens of microns. The processing of textiles with AgNWs is simple and easy to synthesize.27,28 AgNWs will form a permeable network to give textiles excellent electrical conductivity, 29 basically does not affect the breathability, moisture permeability and wearing comfort of the fabric, and the textiles also have good antibacterial and UV protection properties. 30
Herein, we propose a simple method to prepare a multifunctional silver nanowire coating PET fabric for applications such as wearable sensing, electric heating, and electromagnetic interference shielding. The AgNWs synthesized by the polyol method are uniformly dispersed in anhydrous ethanol solution, and then the AgNWs are coated on the surface of PET fabric through the dipping-drying process. The electrical conductivity, EMI shielding effect (SE) electrothermal performance, resistance temperature-sensitivity, strain sensing, and washability will be performed. This multifunctional coating gives the PET fabric with outstanding good flexibility, stretchability, breathability, electrical conductivity, which can be used as wearable fabric heaters for temperature control, strain sensor to monitor various body movements, and EMI block materials for shield electromagnetic radiation.
Experimental Section
Materials
Polyvinyl pyrrolidone (PVP, Mw ≈ 58,000), ethylene glycol (EG, Mw = 98.08), deionized water (0.5 us/cm), copper chloride dehydrate (CuCl2·2H2O, Mw = 170.48), absolute ethanol (C2H5OH, Mw = 62.07), acetone (CH3COCH3, Mw = 58.08), and silver nitrate (AgNO3, Mw = 169.87) were purchased from Sinopharm Chemical Reagent Co., Ltd, China.
Fabrication of AgNWs/PET knitted fabric (AgNWs/fabric)
The silver nanowire fabric synthesized by dipping-drying is shown in Figure 1. Firstly, (i) weighing 7 mg CuCl2·2H2O in a beaker containing 10 mL ethylene glycol. Then weighing 942 mg PVP and 479 mg silver nitrate into a beaker containing 30 mL EG, and dissolving it in an ultrasonic instrument at 25°C for half an hour. This process is protected from light to prevent silver nitrate from decomposing in light. (ii) Measuring 100 mL EG in a 500 mL three-necked flask with a graduated cylinder and heating it in a 160°C water bath accompanied by a serpentine condenser at reflux for 1.5 h. During the process, EG reacts with oxygen to form glyoxal at high temperature, and the reducibility of the aldehyde group reduces the Ag+ to Ag atoms. (iii) Using a pipette to measure 800 μL of the ultrasonically dissolved CuCl2·2H2O solution and adding it to the reacted ethylene glycol at a time. After 15 min, the prepared 30 mL PVP ethylene glycol solution was slowly added to the above ethylene glycol. Add the prepared 30 mL silver nitrate ethylene glycol solution to the reacted ethylene glycol. The dripping ends in 15–20 min, then the reaction is carried out at 160°C reflux for 1.5 h until the reaction is complete. Add the crystal form inducers of CuCl2 and AgNO3 to the system to produce AgCl precursors, so that AgCl is transformed into the growth seed crystal of silver wire. And the Ag atoms in the system are continuously adsorbed on the surface of the seed crystal by PVP, which results in directional growth. Then high aspect ratio silver nanowires are generated. With the gradual progress of the reaction, it can be observed that the solution changes from colorless to light yellow, and finally becomes silvery white, indicating that the silver nanowires have been successfully prepared by the polyol method. (iv) After the experiment is cooled to room temperature, adding the acetone solution of the reaction solution, transfer to centrifuge for 20 min at 8000 r/min, then pouring out the supernatant. (v) Repeating the previous step 5 cycles. (vi) Repeated centrifugation with acetone and absolute ethanol are performed for two cycles to wash away the active agent PVP molecules remaining on the surface of the silver nanowires, and finally the synthesized silver nanowires are dispersed in absolute ethanol. Schematic illustration of the preparation for AgNWs/PET fabrics and their applications on electromagnetic shielding, electrical heating and piezo-resistance strain.
Characterization of AgNWs/PET fabric
Using the two-point probe method to observe the resistance of all fabrics by a resistance tester (Keithley DMM 6500, Tektronix Co. Ltd, China) and the conductivity of AgNWs/PET fabric is the average of five tests. A commercial weft-knit polyester (PET) fabric (0.016 g/cm2, 22 wales × 19 course cm−1) was cut into squares with width by length of 6 × 6 cm2, followed by cleaning with ethanol and deionized water and then dried at 60°C in oven. A self-assembled dynamic resistance testing instrument was used to record the resistance changes of fabric samples under different strain levels, including a resistance meter (DMM 6500, Tektronix Co., Ltd, China) and an electronic universal material testing machine (EJA SERIES, Thwing–Albert Co., USA). For electromechanical testing, the sample was cut into a size of 30 mm × 30 mm, and two copper foils were connected as electrodes. All temperature distributions and thermal images of AgNWs/PET fabric were measured by an infrared thermal imager (VarioCAM® hr head, Infra Tec, FLIR system, Germany). The applied constant current is supplied by a three-way programmable DC power supply (IT6322, ITECH, USA).
SEM (ULTRA-55, JEOL, Japan) was used to characterize the morphology of the AgNWs/PET fabric with an accelerating voltage of 3 kV at room temperature, and the dynamic contact angle of the fabric coating was tested by dynamic contact angle. The instrument for characterizing electromagnetic shielding is FY800 Fabric EM Shielding Properties Tester (Wenzhou Fangyuan Instrument CO. Ltd). Using SW-24G washing fastness tester (Wenzhou, China) and YG909-II fabric head UV tester (Wenzhou, China) to evaluate the resistance after washing and UV absorption performance of fabrics. In detail, 15 g of AATCC standard detergent is added, and the entire washing process of each cycle is at 40°C and lasts for 30 min. The samples were placed in a 60°C oven to dry for 30 min after each washing cycle. Then, the resistance, UV absorption performance and weight loss were tested.
Results and discussion
Basic composition and morphology of AgNWs/PET fabric
The amount of AgNWs deposited on the knitted PET fabrics.

AgNWs can adhere to the surface of the fiber due to its own entanglement and electrostatic adsorption ability to form well adhesion ability between micro-sized fibers as prior reports.31,32 The scanning electron microscope images of Figures 2(a) to (d) shows that the silver nanowires are both adsorbed on the surface of the fibers, filled into the voids between the fibers, and bonded to each other to form a network structure, which is consistent with previous reports.
33
The hydroxyl group (-C=O) on the residual PVP molecular chain existing on the surface of AgNWs and the oxygen-containing polar groups on the surface of the fiber can form a large number of hydrogen bonds, which makes AgNWs adhere to the surface of the fiber and even cover the gaps between the PET yarns,
34
thus forming a network structure (Figure 2(c)). Figure 2(e) shows a photo of AgNWs/PET fabric connected to light-emitting diodes. Under the same voltage, the brightness of the light-emitting diode becomes brighter with the increase of AgNWs coating times, indicating that AgNWs/PET fabric has good conductivity. In addition to high electrical conductivity, this multifunctional coating fabric also demonstrate excellent underwater oil repellency and can impart self-cleaning properties to the substrate. As Figure 2(f) shown, the conductivities of coating 5, 10, and 15 times are 25 S/m, 85 S/m, and 175 S/m. The reason for the increase in conductivity is due to the superior conductivity of silver nanowires. The high aspect ratio AgNWs attached to the surface of the PET fiber to form a conductive network, which makes the PET fabric shows excellent conductivity. In addition, the underwater oil-repellent contact angles are 103°, 120°, and 125°, respectively. It can be concluded that AgNWs/PET fabric shows high conductivity, hydrophilic and oleophobic properties under water. When water and oil appear in the solution together, the fabric can be used for oil and water separation. This characteristic shows broad application prospect in the field of seawater oil pollution. SEM images of (a) pristine PET fabric, (b)-(d) AgNWs/PET fabrics with different magnification times. (e) AgNWs/PET fabrics used as a conductor to power the lamp beads under the same voltage of 3 V. The lightness corresponding to conductive performance of AgNWs/fabrics comes from different coating cycles, Ag-5, Ag-10, and Ag-15 refer to fabrics coating AgNWs five cycles, 10 cycles and 15 cycles, respectively. (f) The electrical conductivity and underwater oil-repellent contact angle verse AgNWs weight gain coated on fabrics. (g) XRD pattern of AgNWs/PET fabrics.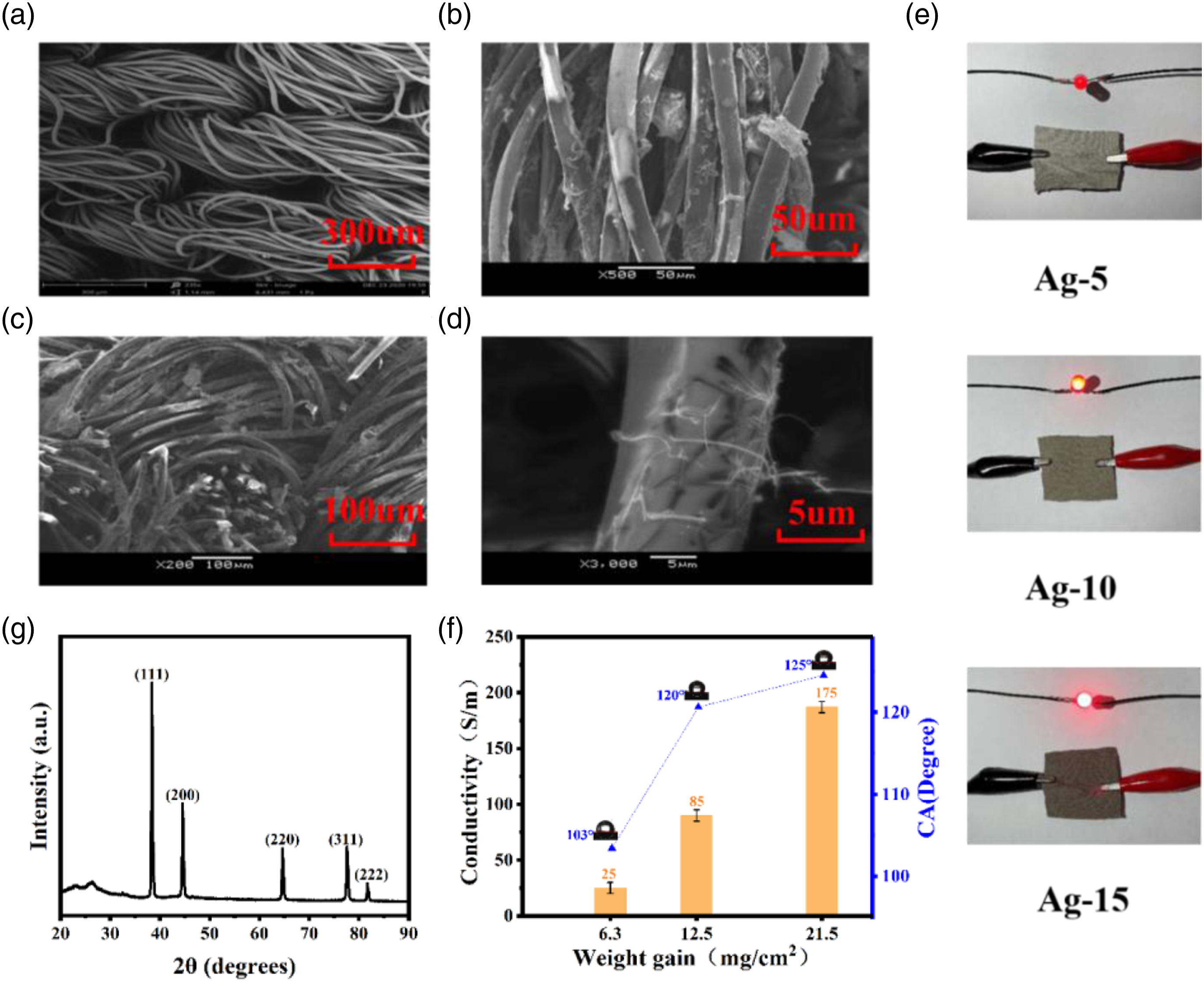
It can be seen from Figure 2(g) that the sample shows strong diffraction peaks at 2θ=38.2°, 44.38°, 64.54°, 77.5°, and 81.6°. These peaks are derived from the phase structure of silver crystals, corresponding to the (111), (200), (220), (311), and (222) crystal planes of the simple substance, respectively. Materials such as gold and silver will grow along the (111) crystal plane of the face-centered cubic system (fee) when their one-dimensional nanostructures have a high aspect ratio. It can also be seen from the Figure 2(g) that the intensity of the diffraction peak of the (111) crystal plane is very high, indicating that the crystal grows directionally along the (111) crystal plane, and its growth is higher than the growth of other crystal planes, which proves the structure of silver nanowires.
Durability of the AgNWs/PET fabrics to washing
For electronic products made of textiles, durability is a concern. As twists and other mechanical movements will affect the use of the fabric be in use. For the study of the durability of AgNWs/fabric, the fabric was cut into 3 × 3 cm2 square, and the mass, conductivity, contact angle, and UV absorption of the fabrics before and after washing were recorded to analyze its washing durability. As shown in Figure 3(a), the weight of these three AgNWs/PET fabrics decreased by 5.7%, 10.2%, and 16.5% after two, four, and six wash cycles, respectively, which results from some AgNWs stick to the fabric only by electrostatic adsorption and will fall off while suffering washing exercise. The peeling of AgNWs will lead to a decrease in conductivity as Figure 3(b) shown. The conductivity of Ag-15 for unwashed, washed two cycles, washed four cycles, and washed six cycles are of 175 S/m, 93 S/m, 13 S/m, and 0.17 S/m individually. And the conductivity result of each fabric is the average of five measurements. As the washing time increases, the reduction rate of conductivity for fabric weight becomes smaller and smaller. This may be ascribed to the residual AgNWs are supported on PET fibers through hydrogen bonds and intermolecular forces, their forces are great to ensure not easy to fall off the surface of the fibers. The relationship between the conductivity of AgNWs/PET fabric and the number of nano-silver coatings is described. (a) PET fabric weight loss under different washing cycles. (b) Plots of conductivity of the AgNWs/PET fabric under different washing cycles. (c) UV absorption of untreated polyester fabric, Ag-15 fabric, Ag-15 fabric after washing for 6 times. Unless noted, the samples in this text refer to unwashed fabrics. (d) The σ/σo of AgNWs/PET fabric soaking in different solutions for 24 h, σo refers to the conductivity of AgNWs/PET fabric, σ denotes to the conductivity of AgNWs/PET fabric after 24 h of soaking in the solvent.
The ultraviolet-visible absorption spectra of the original PET fabric, AgNWs/PET fabric, and AgNWs/PET fabric washed six times are shown in Figure 3(c), and the ultraviolet-visible spectrum is in the wavelength range of 200–1100 nm. The original PET fabric has an absorption peak at 300 nm and consistent with previous reports. 35 The fabric of Ag-15 gives an absorbance peak at 400 nm and this peak is the surface plasmon resonance (SPR) of spherical AgNWs,36,37 this result is consistent with the previously discovered nano-silver colloid.
The conductivity changes of nano silver fabric immersed in different solutions such as ethanol, water, deionized (DI) water, acetone, formic acid, and glycol for 24 h were studied. As shown in Figure 5(d), the conductivity of AgNWs/PET fabrics deteriorated 11%–29% after being immersed in six kinds of solvents for 24 h. The AgNWs/PET fabrics show better conductivity stability in water, deionized water, and ethanol, and the residual value in DI water and ethanol is as high as 80%. This structural performance can be attributed to the effect of the interface interaction between AgNWs and PET fibers, which should be addressed for the study on coated functional fabric.
EMI shielding performance of AgNWs/PET fabrics
EMI shielding effectiveness of competitive coated fabrics.

PDA = poly (dopamine), GF = glass fabrics, PP = Polypropylene, AFP-3 = AgNWs/Fe3O4/PDM, MXene = two-dimensional (2D) transition metal carbide or nitride, CPFCs = conductive polymer fabric composite, Ti3C2Tx = Titanium carbide, CFFs = carbon fiber fabrics.
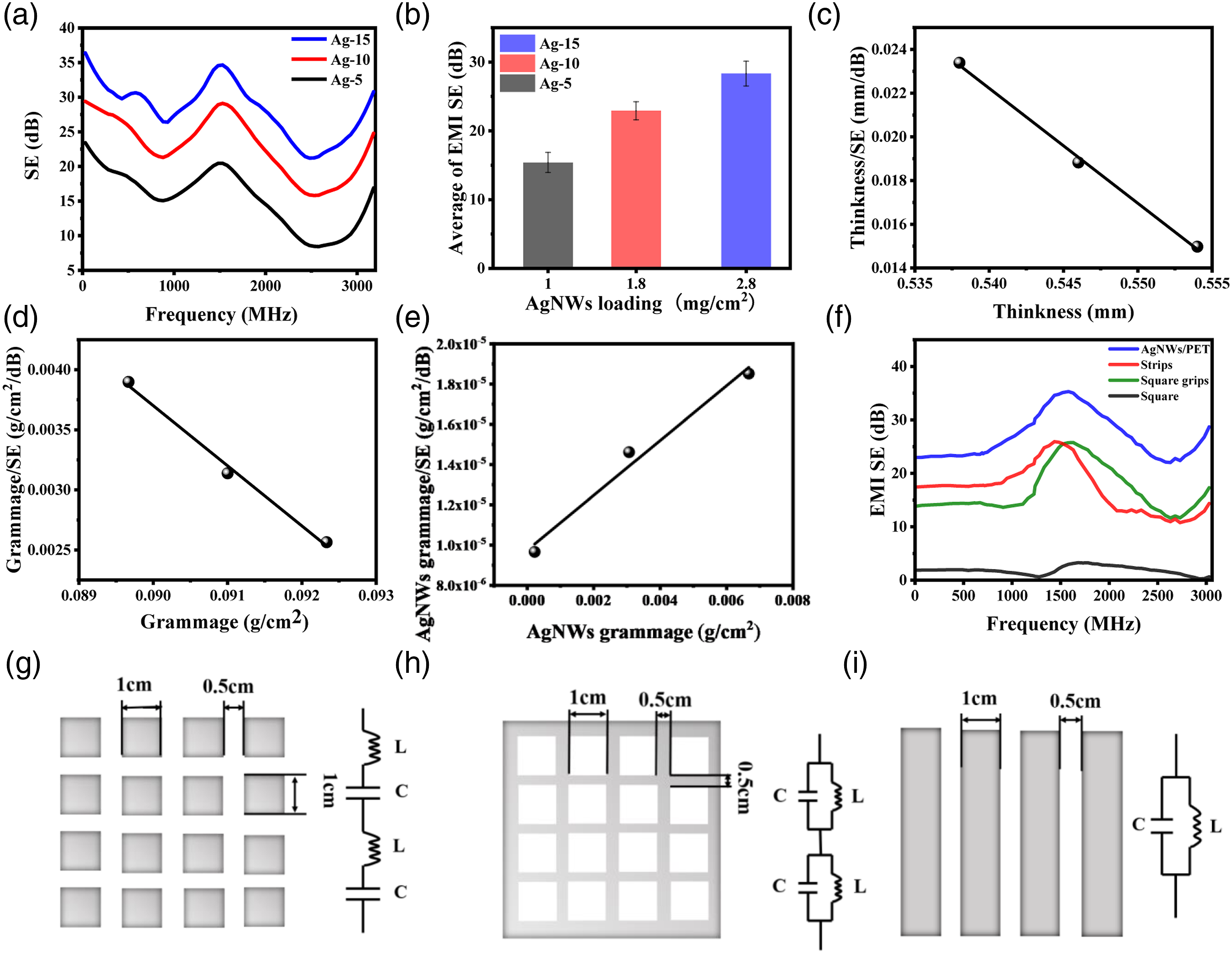
(a) The EMI SE of the coated fabric under frequency of 0.03–3.3 GHz. (b) Average EMI SE for AgNWs/PET fabric with different AgNWs loading. Plots of (c) Thickness/SE versus Thickness and (d) Grammage/SE versus Grammage and (e) Grammage/SE versus Grammage (data from Ag-15 in Table 1). Grammage refers to the square cm g weight. The curves are linear fits. AgNWs/PET fabric with unit periodic structure of (g) square, (h) square grid and (i) strip. The equivalent circuit diagram corresponding to the unit period structure is the right. (f) The electromagnetic shielding results of periodic structure of (g) square slice, (h) square grid and (i) strip.
Figures 4(c) to (e) shows the linear relationship of thickness/SE versus thickness and gram weight of AgNWs fabric/SE versus gram weight of AgNWs fabric and gram weight of AgNWs/SE versus gram weight of AgNWs. The main mechanism is that the prepared silver nanowires are coated on the PET fabric in a small amount (not exceeding the skin depth), the shielding of electromagnetic waves has not reached saturation, and the line trend of downhill is opposite to our previous research,46,47 but the Figure 4(e) gives the same variation trend. All in all, the variation trend for gram weight/SE versus gram of functional materials coated fabric and pure functional materials are quite distinct. Which could be a selective reference to verify data.
The fabric structure determines the frequency selection characteristics of electromagnetic shielding. To further explore the influence of fabric structure on electromagnetic shielding characteristics, the EMI SE of AgNWs/PET fabric under different unit periodic structures of square, square grid and strip were performed as the Figure 4(g–i) shown. When a plane wave is incident on a periodic structure, a certain number of floquet harmonic plane waves will be excited. 48 Therefore, the equivalent circuit can be used to describe the propagation of plane waves. The equivalent circuits correspond to Figure 4(g) square slice, Figure 4 (h) square grid, and Figure 4(i) strip unit periodic structure, there are two types of equivalent circuits: inductance and capacitance in series and inductance and capacitance in parallel.
As shown in Figure 4(f), the square fabric did not give an obvious peak representing the frequency selection characteristics in the test range of 0.03–3 GHz, and showed transmission characteristics. This is because when electromagnetic waves are incident on the surface of the square fabric, the electric field parallel to the fabric generates an oscillating force on the electrons. An induced current is formed on the surface. Here, the electromagnetic wave that causes the electrons to oscillate is the electromagnetic wave absorbed by the square fabric. At a certain frequency, the incident electromagnetic wave energy is all converted into energy that makes electrons oscillate.
At this time, the transmission coefficient of the checkered fabric is zero. However, the square grid and strip give the superior peak SE values of 26 dB were given for the woven fabric at 1430 MHz and 2330 MHz, respectively. It indicated a wide range of electrons to move, so that the electrons absorb most of the energy and form an induced current in the gaps of the fabric. 49 When the incident frequency of electromagnetic waves increases, the moving range of electrons gradually decreases, and the currents formed along the gaps continue to increase. As the electrons absorb a large amount of incident wave energy, they are also radiating energy outward resulting in the loss in the form of reflection. To sum up, AgNWs/PET fabrics with periodic structures can achieve some selective electromagnetic shielding characteristic involving simple tailoring.
Piezo-resistance behaviors for Human motion detection
Figure 5(b) shows schematic illustration of electromechanical test system. The reciprocating motion Ag-15 were performed in a fabric-extension testing machine at a setting rate based on the test requirement. Meanwhile, the resistance variation ΔR was recorded in real time by a resistance tester DMM 6500. Usually, the strain of the fabric is sensed by resistance variation ΔR/R0 responsing to the stretching and releasing of the fabric. And the gauge factor (GF, GF = ((ΔR/R0)/ε) is used to characterize the sensitivity of the sensing performance.44,50,51 Figure 5(c) and (d) show that Ag-15 presents a negative resistance response (ΔR/R0) under tensile strain, the resistance of the fabric changes inversely with the strain increases, and the resistance decreases monotonously. The sensing fabric provides an outstanding deformation capacity and a wide reversible strain sensing range greater than 400% without breaking. As the suspended figure in Figure 5(c) shown, the overall GF value of Ag-15 goes down to −0.45 firstly, which may come from the contact slip adjustment of the fiber loops. The GF value of Ag-15 goes up to −0.20, which may be resulted from the deformation and elongation of fiber loops. In the strain range of 0–435%, the corresponding GF of Ag-15 is of −0.1 ∼ −0.5. As the straight tangents in Figure 5(c) shown, the real-time GFs of Ag-15 between the strain of 0150%, 150%–300%, and 300% 435% are −0.45, −0.19, and −0.04, respectively. This result indicates that the sensitivity of sensor decreases gradually with the increment of strain. The AgNWs/PET fabric strain sensor is used to detect human motions. (a) An overview of fabric sensor mounted on different sensing locations. (b) Schematic diagram of the electromechanical test system. (c) The relationship between the relative resistance change of AgNWs/PET fabric (ΔR/R0) and the applied tensile strain. (d) Relative resistance change (ΔR/R0) under cyclic tensile strains of 10%, 20% and 30%. (e) Bending of the thumb. (f) Bending of the wrist. (g) Fisting of the palm. (h) Swallowing. (i) Elbow movement. (j) Knee bending.
The piezoresistive performance of Ag-15 under cyclic tensile strains of 10%, 20%, and 30% is shown in Figure 5(d). The relative resistance change of ΔR/R0 increases monotonously with increment of tensile strain in the range of 0–30%. In actual use, the fabric tensile deformation is generally within 30%. The resistance response varies steadily in each cycle under different tensile strains, and the repeatability is strong, which indicates that the fabric has good stability and sensitivity. In summary, Ag-15 has the ideal integration of high sensitivity and wide sensing range, which broadens its application range, and is especially suitable for detecting a full range of human movement.
To explore the potential applications of as-prepared coated fabrics as wearable devices, the fabrics were attached to different parts of the human body for the real-time monitoring of human movement and detecting of human physiological signal, such as muscle movements, pulse, respiration, and facial expressions. As Figure 5(e) shown, fixed the Ag-15 sensor on the finger, when the finger joints are bent to a certain angle, the relative resistance change corresponding to the bending of the finger joints is displayed, the real-time response shows stability. In detail, the bending movement of the finger can be accurately monitored by recording changes in the resistance the coated fabric. When the finger was bent to a certain angle and the finger is stable, the resistance variations of the fabric sensor first rise rapidly, and then the resistance hold steady. As the bending angle increases, the resistance of the coated fabric further increases, forming a step signal, indicating that the coated fabric responds quickly and has high sensitivity. It is worth noting that when the fingers are suddenly bent, the coated fabric shows a small overshoot resistance, which is attributed to the viscoelastic properties of the fabric. It is of great significance to use a single strain fabric sensor to monitor the complex deformation characteristics of the human body. Herein, we use coated fabric to detect the motions of the wrist and fist (Figure 5 (f) and (g)). The amplitude and shape of the resistance plots change with the bending angle of the wrist, which shows a favorable corresponding respond, indicating our fabric sensor can easily detect motions of different positions of the human body.
It is worth noting that repeated clenched fists can be monitored by recording the resistance of the coated fabric (Figure 5(g)). The peak characteristics of the resistance curve should be combined with the influence of the anisotropic electromechanical properties of the coated fabric. By recording the resistance change of the fabric, it is convenient to monitor the elbow movement (Figure 5(i)), knee bending (Figure 5(j)), flexion of the wrist joint and shows outstanding stability. The amplitude and frequency of the resistance curve can, respectively, characterize the motion state and rate of the monitored object. Furthermore, the fabric sensor was also put on the throat (Figure 5(h)) to recognize the sound production. These results show the ability of the coated fabric as a wearable device for detection of human movement.
Electric heating behavior and resistance temperature-sensitive behavior
The AgNWs/PET fabrics can serve as a wearable heater for personal thermal management resulting from electric heating and resistance temperature-sensitivity based on their conductivity and flexibility. When a current was applied on the AgNWs/PET fabrics, the Joule heating will generate and causes a corresponding change in resistance simultaneously, and the possible mechanism is collision between the moving electrons and the phonons. Figure 6(b) shows the plots of voltage and current of AgNWs/PET fabrics, which complies with Ohm’s law and gives a low resistance of 5 Ω. As shown in Figure 6(c), even at low currents, the fabrics give a good Joule heating effect. The temperature rises rapidly in the initial stage, then slowly rises over time, and finally reaches equilibrium within 40 s. It can be observed that when the input current is only 0.04 A, the equilibrium temperature is as high as 53°C, and low current of 0.10 A, the equilibrium temperature can reach 106°C. The upper left corner is the electrothermal image of PET fabric and AgNWs PET fabric at ambient temperature. The difference is not large due to the samples at the same room temperature. (a) Schematic diagram of the measurement setting of electric heating behavior and resistance temperature-sensitive behavior for AgNWs/PET fabric electrothermal heater. (b) I-V plots of Ag-15. (c) Plots of temperature verses time under different currents of 0.02 A–0.10 A for Ag-5 fabrics, the upper left corner shows the electrothermal image of PET fabric and AgNWs PET fabric at ambient temperature. The real-time temperature verses time for (d) Ag-15 fabrics, (e) Ag-10 fabrics, and (f) Ag-5 fabrics at different applied currents. (g) Relative resistance changes and real-time temperature versus time curves of Ag-5 under 0.10 A. (h) Relative resistance variations versus steady-state temperature curves of Ag-5 under different currents. The conductive mechanism of the AgNWs/PET sensor is inserted in the left corner. (i) Relative resistance variations of Ag-5 versus temperature heated by the oven.
To further study the electric heating performance and influence of the number of coatings on the electric heating performance of coated fabric, the real-time temperature of the fabrics with different coating cycles under different currents are analyzed (Figures 6(d) to (f)). All results show similar trends, and the temperature graph shows three states of heating, stabilization and cooling. When the current is 0.02 A, the temperature of Ag-15 rapidly rises to a stable temperature of 34°C. When the current is increased to 0.1 A, Ag-15 gradually heats to 85°C, which proves the excellent electrothermal performance of AgNWs/PET fabric. Comparing the results of Ag-5, Ag-10, and Ag-15 fabrics under the same current, the lower stable temperature of Ag-5 is due to its lower resistance. The stable temperature of the Ag-10 heater at 0.10 A is 95°C, and the stable temperature of the Ag-5 heater at 0.10 A is 106°C, indicating these AgNWs/PET fabrics shows excellent Joule heating efficiency than that of most of previously reported fabric heaters.52–54 According to the equation P = I2R (where P is power, I is the applied current, and R is the resistance of heaters). Therefore, for electric heating applications, the less AgNWs coatings within a certain range, the better.
In order to study the effect of temperature on the resistance of AgNWs/PET fabrics, the change of fabric resistance with temperature changes was also monitored. Figure 6(g) shows the real-time temperature change and relative resistance change versus time curve of Ag-5 at 0.10 A. When a current of 0.10 A was applied, the resistance goes down about 7%, which is opposite to the increasing real-time temperature. After unloading, the resistance recovers. To further explore the relationship between the resistance and the steady-state temperature of the AgNWs/PET fabric under applied current load, the relative resistance changes of the Ag-5 heater under the applied current of 0.02 A–0.10 A were provided in Figure 6(h). The resistance of the fabric increases monotonically with the increase of the current and showing an excellent linearity. And the steady-state temperature is responding to the applied current. The negative temperature coefficient of resistance (TCR) for fabric Ag-5 is of −0.05%/°C.
In addition, to check the resistance variation is mainly come from temperature rather than the polarization caused by applied current, the relative resistance variations of Ag-5 versus temperature heated by the oven was provided as Figure 6(h) shown, which shows the same resistance temperature-sensitive behavior as the sample heated by current and gives a similar TCR of −0.06%/°C.
Therefore, the above results confirm that these fabric heaters present stable and good electrothermal properties and thermal efficiency. In particular, the remarkable linearity endows the fabric heater both temperature sensitivity based the resistance variation and temperature controlling based on the applied current.
A simulation of emotion recognition system based on AgNWs/PET fabric combined with galvanic skin response signal (GSR) was established. Changes in human emotions can cause changes in the human endocrine system and affect the sympathetic nervous system of the human body. Therefore, the skin conductance response signal contains a large amount of human sympathetic information. Since the GSR signal is related to the expansion and contraction of human blood and the secretion of sweat glands, there must be individual differences between different testers. The experimental equipment is shown in Figure 7(a), which uses finger electrodes made of AgNWs/PET fabric. The signal in Figure 7 is the GSR signal collected by placing this electrode on the two fingers of the test subject. Figure 7(b)–(f) measure the skin level of the experimenter under the GSR electrical signal when they are calm, nervous, happy, sad, and fear. It can be seen that their skin level is higher when they panic. When AgNWs/PET fabric is added to the finger electrode, the GSR skin signal of the tester is more prominent than when the fabric is not applied, as shown in Figure 7(d) and (e). It can be seen that when the tester is happy, the GSR level value is rising, and when the tester is sad, the GSR level value is falling. The reason is that the AgNWs/PET fabric has the characteristics of low resistance and high sensitivity. It is in good contact with the skin and acts as a wire in it to make the contact signal between the finger electrode and the finger more sensitive. After the GSR grade value was added to our fabric, a good classification and recognition effect was achieved, which verified the feasibility of establishing an emotion recognition model. These results prove that AgNWs/PET fabric can be used as a sensing material for GSR signals in emotion recognition systems. (a) GSR signal acquisition connection diagram. Test the GSR signal in five emotional states (b) In a calm state. (c) Under stress. (d) In a happy state. (e) In a state of grief. (f) In a state of fear.
Conclusion
In summary, we use a simple solution impregnation method to prepare multifunctional AgNW/fabrics. Ag-15 provides high sensitivity (GF is −6.16 when strain is 10%), wide sensing range (stress strain up to 435%), and good electromagnetic shielding (37 dB), Joule heating (with a current of 0.10 A in 30 s, the temperature can reach more than 100°C), strain sensing and UV protection. In addition, AgNWs/PET fabric can be heated to above 100°C with a low current of 0.10 A in less than 30 s. The excellent linearity of resistance temperature-sensitive behavior gives this fabric a temperature sensing and management function. Moreover, AgNWs/PET fabrics is highly sensitive to human movement. This work provides selective results and methods for technologies such as fabric heaters, microwave blockers, human motion, and temperature sensors.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the National Natural Science Foundation of China (NSFC 51803185), Public Welfare Project of Zhejiang Province (LGF21E030005), China Postdoctoral Science Foundation (2020M681917), Shandong Provincial Natural Science Foundation (ZR2020QF115), Postdoctoral Foundation of Zhejiang Sci-tech University Tongxiang Research Institute (TYY202013).
