Abstract
The chemical oxidative polymerization and co-precipitation methods were employed for the preparations of polyaniline (PANI) and silver-doped zinc sulfide (ZnS) nanoparticles to be used for electromagnetic interference (EMI) shielding. PVC-based composite films were fabricated by the incorporation of Ag/ZnS and PANI-Ag/ZnS nanoparticles. These nanoparticles were first analyzed by X-ray diffraction and zetasizer for their crystal structure and particle size. Prepared nanocomposite films were then analyzed for various properties like electrical conductivity, transmission in the near-infrared region (700 nm to 2500 nm), and EMI shielding efficiency in the microwave region (0.1 GHz to 20 GHz). These parameters were characterized by DC Conductivity, NIR spectroscopy, and vector network analyzer. It was found that with the addition of the concentration of nanoparticles, both values of conductivity and shielding efficiency improved. The highest attenuation value in 0.1 to 20 GHz reached 52.5 dB in 0.1 to 20 GHz frequency for 20 wt% PANI-Ag/ZnS and < 0.5% transmission was evaluated in the NIR region.
Introduction
The development in the production of advanced telecommunication and high-density electrical instruments in recent times has led to the unwanted noise of electromagnetic radiation with the addition of tremendous heat absorption and become a thoughtful concern of the present time.1,2 Electromagnetic interferences by electromagnetic noise not only malfunctioning the electrical system but also causing the permanent manipulation of electrical circuits during the heat dissipation of thermally conductive nanomaterials when it is not properly functioning.3–5 The effect of electromagnetic interferences is causing damage to the telecommunication system, electronic systems, and military application as well as creating problems to human health.6,7 The electromagnetic interferences can be avoided by several shielding mechanisms that have been established. The general mechanism consists of absorption, reflection, as well as multiple reflections within the shielding nanomaterials to reduce the EM radiations. The overall shielding efficiency for electromagnetic interference is the result of all these three mechanisms. 8 The total shielding efficiency of 10 dB considered as the threshold efficiency as it can attenuate almost 90% of electromagnetic radiation and thought to be target value for use as a shielding material in commercial applications.9–11 The mechanism of reflection of electromagnetic interference shielding is primarily described as the charge carriers directly contact to the EM field that reflects the radiation.8,12,13 While the absorption of electromagnetic interference shielding is the interaction of the magnetic and electric dipoles with the waves. Multiple reflections is a broader term used for the reflecting radiations within the shielding materials.14–17
Polyaniline and silver have very high electrical conductivity, and being used in several applications including electromagnetic interference (EMI) shielding.18–20 Several shielding materials are using for EMI shielding such a metal, multilayered metal as well as conducting materials. Metals are considering the early choice for EMI shielding nanomaterials because of their excellent electrical conductivity. 21 Conductive polymers have various useful features for electromagnetic interference shielding applications such as absorption properties, low mass density, easy process-ability, and environmental friendly.19–21 Metal/polymer nanocomposites have gained much attention because of the firm adhesion of metal to the polymer surface. These kinds of nanocomposites are a high potential source for EMI shielding.22–24 Kumaran et al. synthesized the Ag-graphite/PVDF nanocomposites that have novel properties of conductivity of 2.7S/cm and observed the shielding efficiency of 29.1 dB at 12.4 GHz. 25 Wang et al. prepared polyaniline and graphene aerogel (GA) to study the microwave absorption properties. The measured polyaniline (PANI)/graphene aerogel (GA) exhibited high electromagnetic interference shielding—42.3 dB in 11.2 GHz frequency range with 3 mm thickness of nanomaterials. 26 Modak synthesizes electrically conducting nanocomposites for EMI shielding. Polyaniline/graphene nanocomposite was synthesized through in-situ chemical oxidative polymerization of aniline in addition to graphene. 27 The value of electromagnetic interference shielding efficiency found to be highest at 5% weight concentration of graphene that was 51–52 dB in the 2–12 GHz frequency range which was above the value of efficiency at the commercial level than the needed value of electromagnetic interference shielding efficiency (20 dB) and could be utilized as lightweight electromagnetic interference shielding nanomaterials to shield electrical equipment and machines from electromagnetic radiations.28–30
Mostly graphene, carbon nanotubes, MXene, ferrites, and related materials are being used for the EMI shielding purpose. All of these materials are in the form of nanosheets, nanorods, or nanoparticles and are very difficult to synthesis on industrial scale. On the other hand PANI, Ag/ZNS particles are cheap and easy to produce at industrial scale. So, in the present work, Ag/ZnS and PANI-Ag/ZnS nanocomposites were synthesized and compared the shielding efficiencies of these composites by different characterization techniques. Ag/ZnS was chosen as it has very high electrical conductivity. According to the EMI theory, high electrical conductivity is required for EMI shielding.31–33
Materials and methods
All the chemicals and reagents used in this study were of analytical grade and were used without further purification. Aniline (C6H5NH2 99%), APS ((NH4)2S2O8 98%), Zinc acetate dihydrate (Zn (CH3COO)2·2H2O 99%), and hydrochloric acid (HCl) were obtained from Daejung Korea. Thiourea (99%) was purchased from Merk (Germany) and silver nitrate (AgNO3 99%) was attained from Sigma-Aldrich (Germany). Distilled water was used throughout experimentation which was obtained from the local water purification system.
Synthesis of Ag/ZnS
Ag/ZnS was synthesized using the hydrothermal reaction method. 21.9 g, 7.6 g, and 17 g of zinc acetate dihydrate, thiourea, and silver nitrate, respectively, were mixed separately in 100 mL of double distilled water with continuous stirring. Molarity of all solutions was 0.1 M. After having homogeneous solutions, all three were mixed together and transferred for the hydrothermal process to an autoclave reactor made of stainless steel lined with Teflon at 180°C for 12 h. The obtained silver doped zinc sulfide precipitates were filtered and washed using distilled water and ethanol multiple times. In the end, the precipitates were dried in oven at 130°C for 16 h.
Synthesis of PANI
Polyaniline was synthesized by a chemical oxidative technique by polymerization of aniline monomer.29,30 4.8 g of ammonium persulfate was dissolved in 50 mL of formic acid to prepare the APS solution. Aniline monomer of 2 mL also dissolved in 50 mL of formic acid to prepare the aniline solution. Both of these solutions were kept in the ice bath to a complete reaction temperature of 5°C. APS solution dropwise was added in aniline solution with constant stirring at 0°C. A light green color appeared that indicated the start of polymerization. It took 3 h to complete the polymerization reaction. The solution containing PANI particles was centrifuge, washed with water, and dried in an oven at 80°C overnight.31,32
Synthesis of PANI-Ag/ZnS
For the synthesis of PANI-Ag/ZnS, polyaniline and Ag/ZnS are dispersed in THF separately and sonicated for 2 hours, and stirred for 5 h. Both solutions were mixed in a 1:1 ratio and sonicated and stirred for 2 h and 5 h, respectively. The obtained mixture dried in an oven at 80°C overnight. The obtained precipitate contained 50% PANI and 50% Ag/ZnS.
Fabrication of nanocomposites film
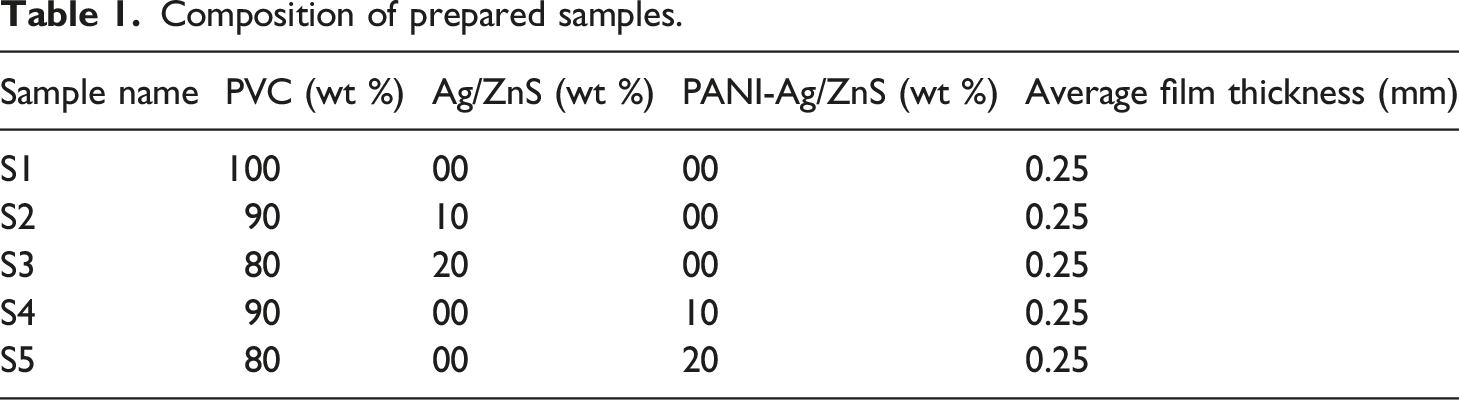
Nanocomposite films based on PVC, Ag/ZnS, and ANI Ag/ZnS were prepared using solution casting method. Required amount of nanoparticles were dispersed in 40 mL of THF. They were stirred for an hour and sonicated for 2 h. PVC was then added and stirred again for 24 h. It was cast in petridish and left to dry at ambient condition for 5 h and then dried in vacuum oven at 50 °C for 5 h (Figure 1) (Table 1). Flowchart representation of the synthesis of nanocomposite film. Composition of prepared samples.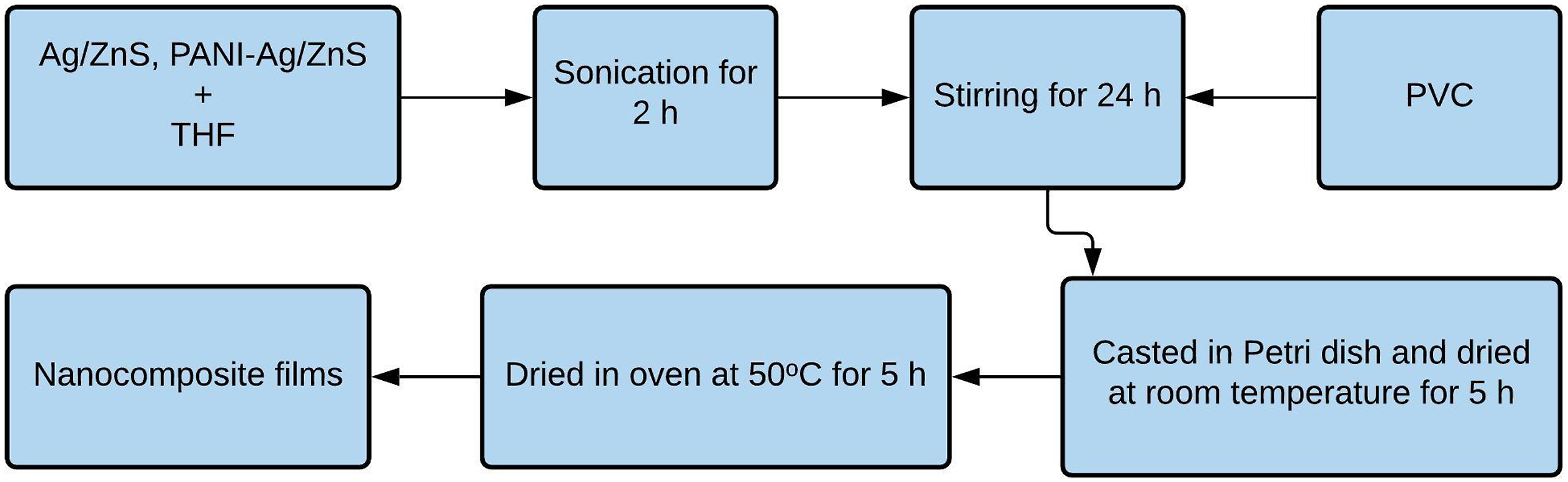
Characterizations
X-ray diffraction
D8 Discover of BRUKER was used for the X-ray analyses from 2θ 10° to 70° with the increment of 0.05° and stay time of 1 sec. The obtained graphs are shown in the X-ray diffraction section.
Scanning electron microscopy
“JEOL-instrument JSM-6490A” was used to capture micrograph of nanocomposites films. Films were dipped in liquid nitrogen and were broke to expose fresh unaltered cross-sectional area. SEM images of cross-sectional are shown in results and discussion part of this article.
UV/Vis/NIR spectroscopy
“Lambda 950, Perkin Elmer” were used to evaluate the transmission of UV/Vis/NIR waves through the nanocomposite films.
DC Conductivity
Keithley 2450 was used to measure the DC conductivity of prepared nanocomposite films. Two probs were place at a distance of 5 mm and 5 V is applied. Current was measured by a highly sensitive ammeter. By using the relations given in section, DC conductivity was calculated and the measurement setup schematic is shown in Figure 2. Schematic representation of the DC conductivity measurement setup.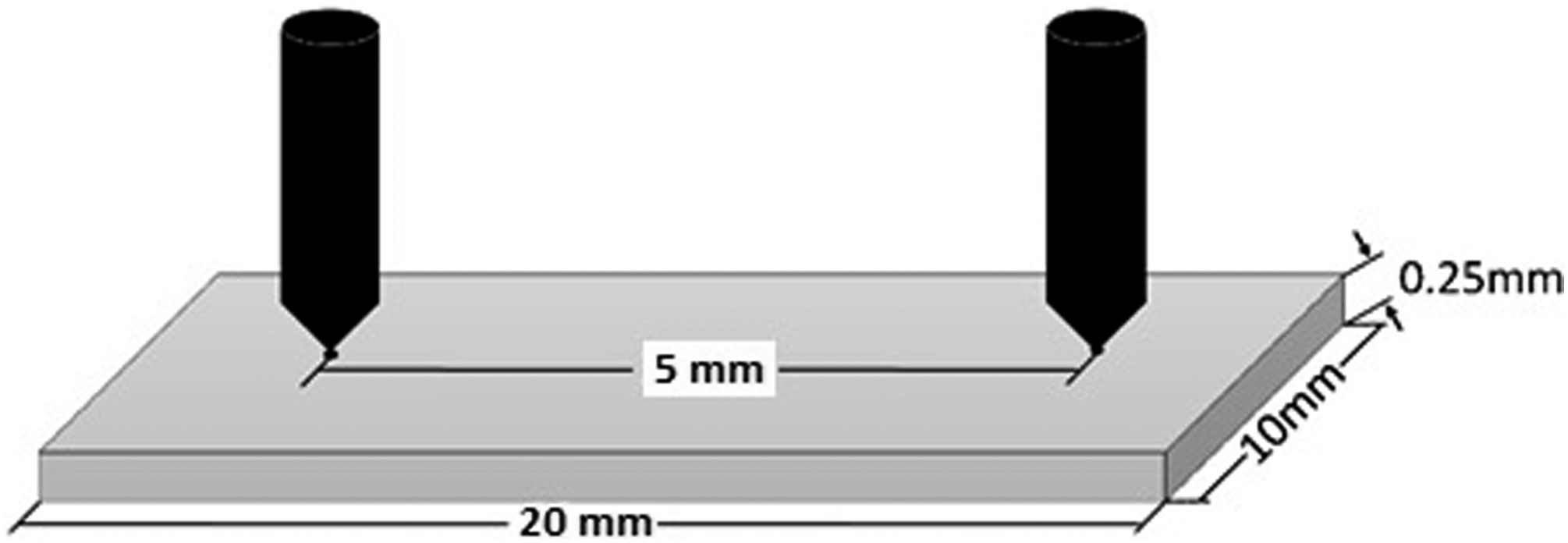
Zetasizer
Zetasizer AT is an instrument of “Malvern Pananalytics” that uses light scattering to measure the particle size from 1 nm to 1000 nm range with the accuracy of +/− 2%.
Vector network analyzer
Rohde & Schwarz’ ZNB20 was used in this research work. The cable coaxial method was utilized to measure the EMI shielding effectiveness in 0.1–20 GHz frequency range. The prepared sample for VNA with 7 mm outer and 3 mm inner diameter were cut with highly precise laser cutter and its schematic is shown in Figure 3. Schematic representation of the synthesized sample for VNA.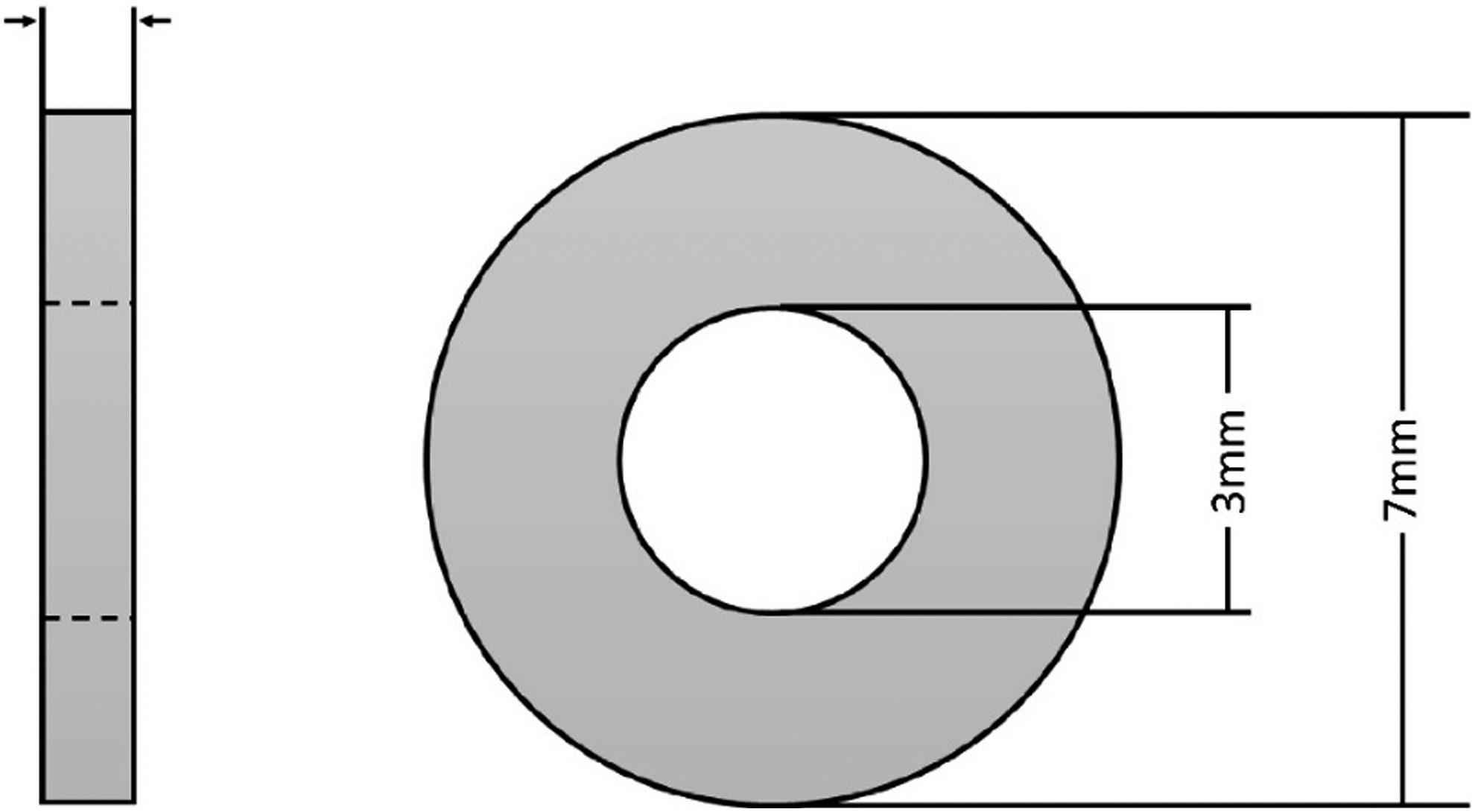
Results
X-ray diffraction
X-ray diffraction patterns of Ag/ZnS and PANI are presented in Figure 4. The sharp characteristic peaks in the XRD pattern of Ag/ZnS at 2ϴ=28.910°, 47.838°, and 56.532° are ascribed to the (111), (220), and (311) crystal planes (JCPDS: 01-080-0020).
33
In PANI composite, the weak reflection peaks at 19° and 26° are the characteristic peaks of amorphous PANI. The sharpness in XRD peaks proposed the high purity of the prepared samples.29–31 X-ray diffraction of Ag/ZnS and PANI-Ag/ZnS.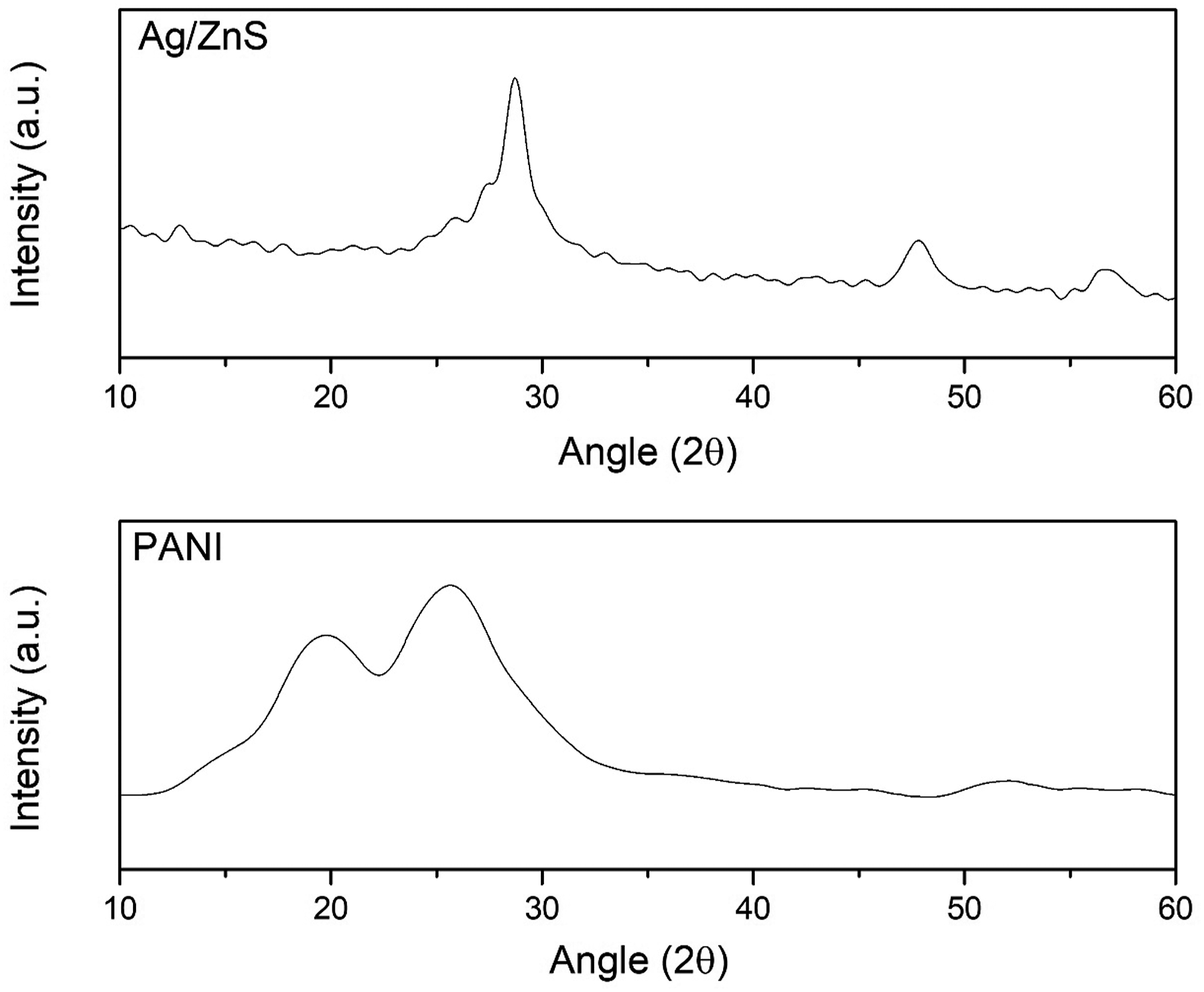
DC conductivity
The electrical conductivity test was performed by the “Two probe technique.” The value current is obtained by an extremely sensitive ammeter by applying a potential of 5V. The distance between the probs was 20 mm. A schematic is shown in Figure 2. The value of conductivity was obtained by the following relation.


The conductivity value of nanocomposites film is shown in Figure 5. S1 sample has a conductivity value of 2.5×10−14 which indicated the insulative nature of PVC. S2 has a conductivity value of 6.8×10−7 with 10 wt % Ag/ZnS nanoparticles. With increasing the concentration of Ag/ZnS to 20 wt %, the conductivity value increases to 3.8×10−4 in sample S3. In sample S4, 6.8× 10−6 value was obtained with 10% PANI-Ag/ZnS which is greater than S2 because it contains 50 wt % Ag/ZnS and 50 wt % PANI nanoparticles. A high conductivity value of 3.8×10−2 is obtained with a 20 wt % concentration of PANI-Ag/ZnS. The obtained results of DC conductivity clearly indicate that there is huge increase in electrical conductivity with the addition of PANI-Ag/ZnS. It can also be assumed that PANI-Ag/ZnS forms an interconnected network structure inside the polymer matrix (Scanning electron microscopy) DC Conductivity values of nanocomposites film.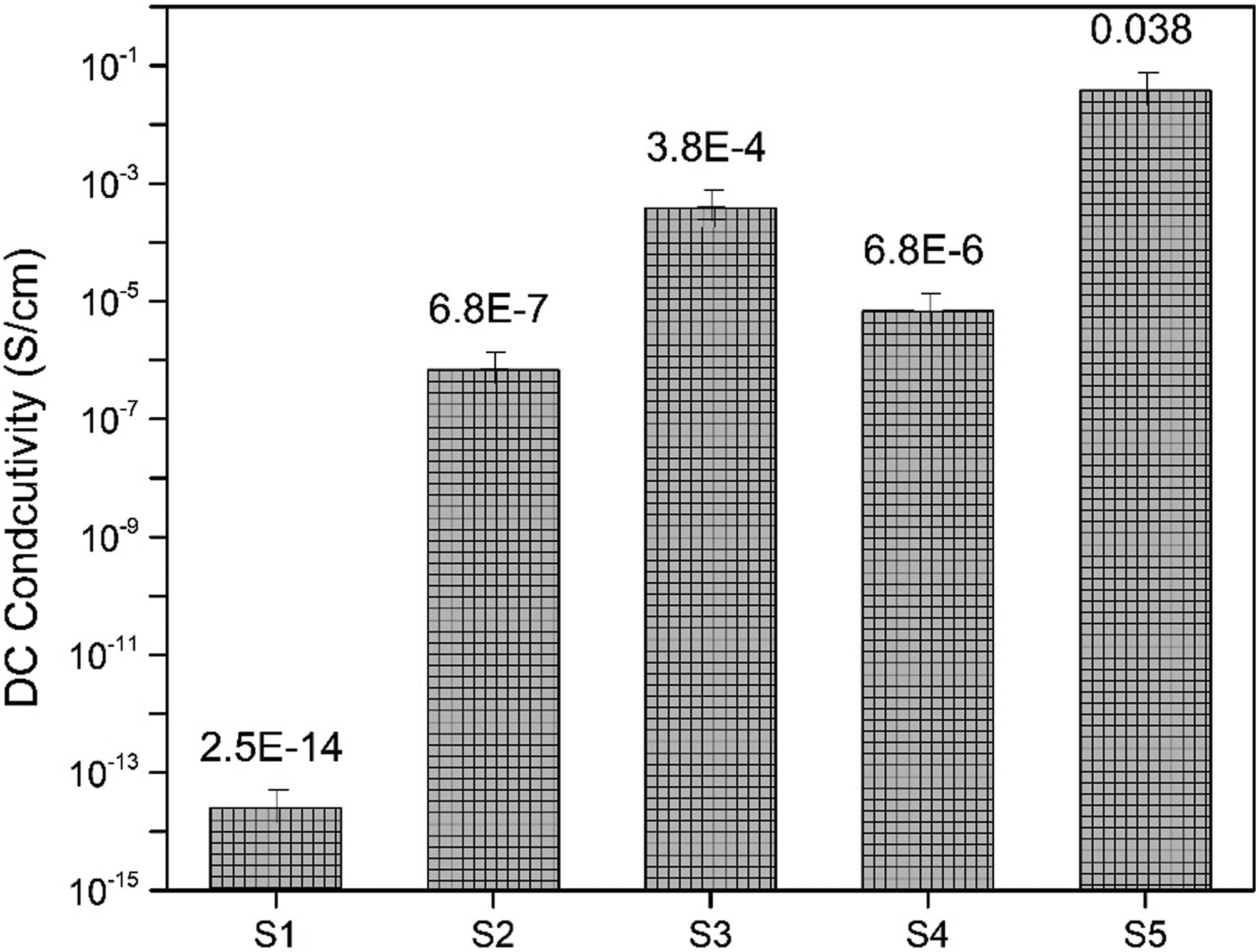
Particle size analysis
The particle size of Ag/ZnS and PANI particles was determined by “ZetaSizer AT” using a 0.5 wt% solution of nanoparticles in water. The obtained particle size distributions are shown in Figure 6. The average particle size of Ag/ZnS and PANI is found out to be 113 nm and 178 nm respectively. Particle sizes of (a) Ag/ZnS and (b) PANI.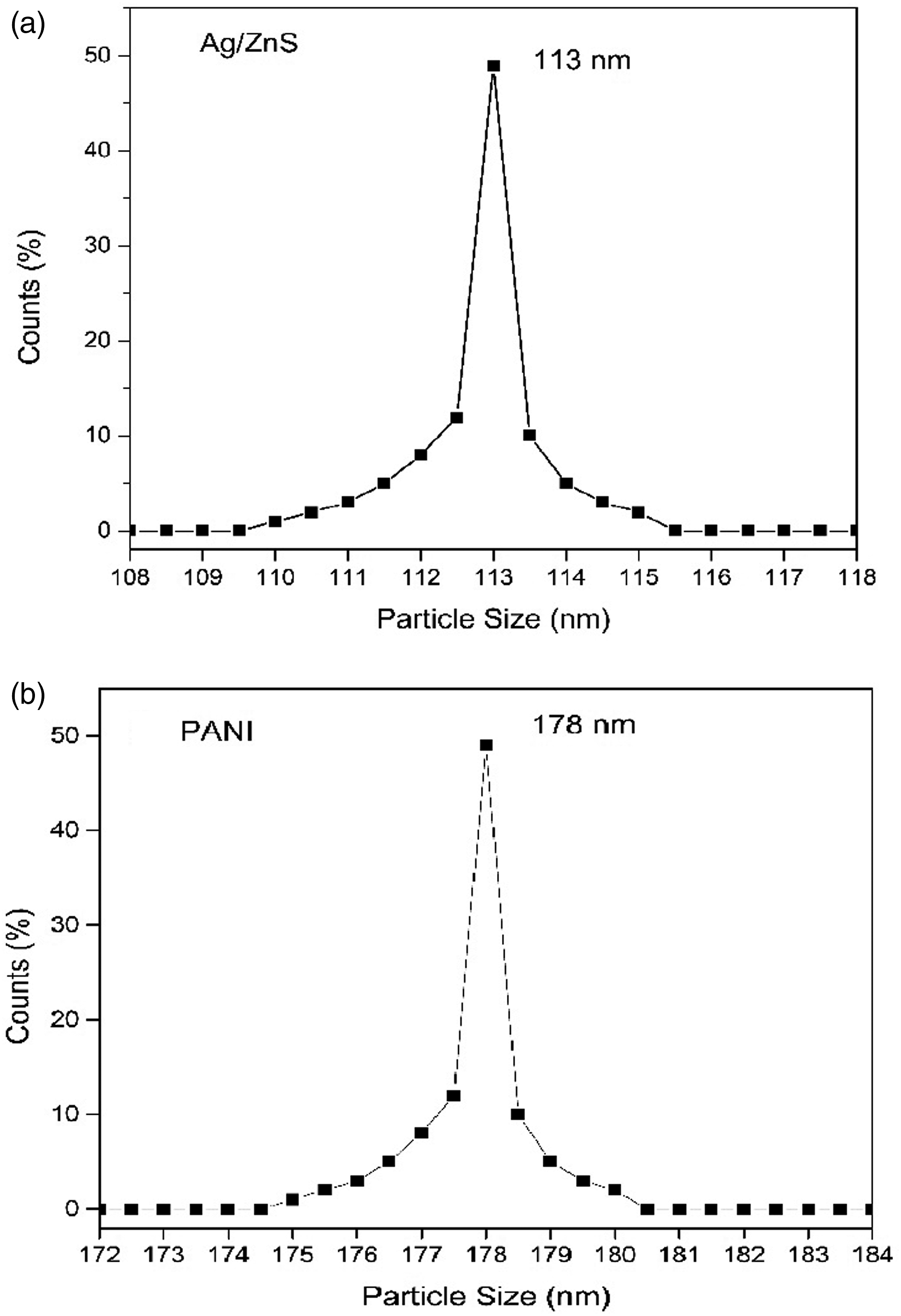
Scanning electron microscopy
The microstructure of sample S5 was observed by SEM and images are shown in Figure 7. Nanocomposite film was dipped in the liquid nitrogen and then broke to get a fresh and unaltered cross-sectional area to observe the dispersion and fabrication of interconnected network structure of nanoparticles in the polymer matrix. It is clear from the SEM images that PVC has no prominent features in the background and nanoparticles are dispersed uniformly in the PVC matrix. In Figure 7, large and bright particles of about 200 nm are PANI as discussed earlier in the Particle size analysis section and small darker particles of about 120 nm are Ag/ZnS. In Figure 7(b), it is evident that both particles are dispersed uniformly in the whole matrix and form an interconnected network structure. SEM images of S5 at (a) high resolution and (b) low resolution.
Electromagnetic interference shielding in NIR region
The NIR shielding efficiency of the synthesized nanocomposites was analyzed in the 750–2500 nm range of wavelength in the NIR region. IR spectroscopic technique was used to observe the shielding by obtaining the transmittance waves through the shielding materials. From Figure 8, it is indicated that S1 showed 80% transmittance that confirms the transparent nature of PVC for NIR waves which was confirmed from previous literature.
15
The S2 and S4 have transmission of 17% and 10%, respectively. These results indicated that with increasing the concentration of Ag-ZnS the transmission decreased. By the addition of conductive polyaniline nanoparticles in nanocomposites of S3 and S5, the transmission becomes 2% and <0.5%, respectively. Polyaniline as conductive nanoparticles can reflect the waves that decrease the transmission of waves within the shielding material. Transmission of waves in IR region.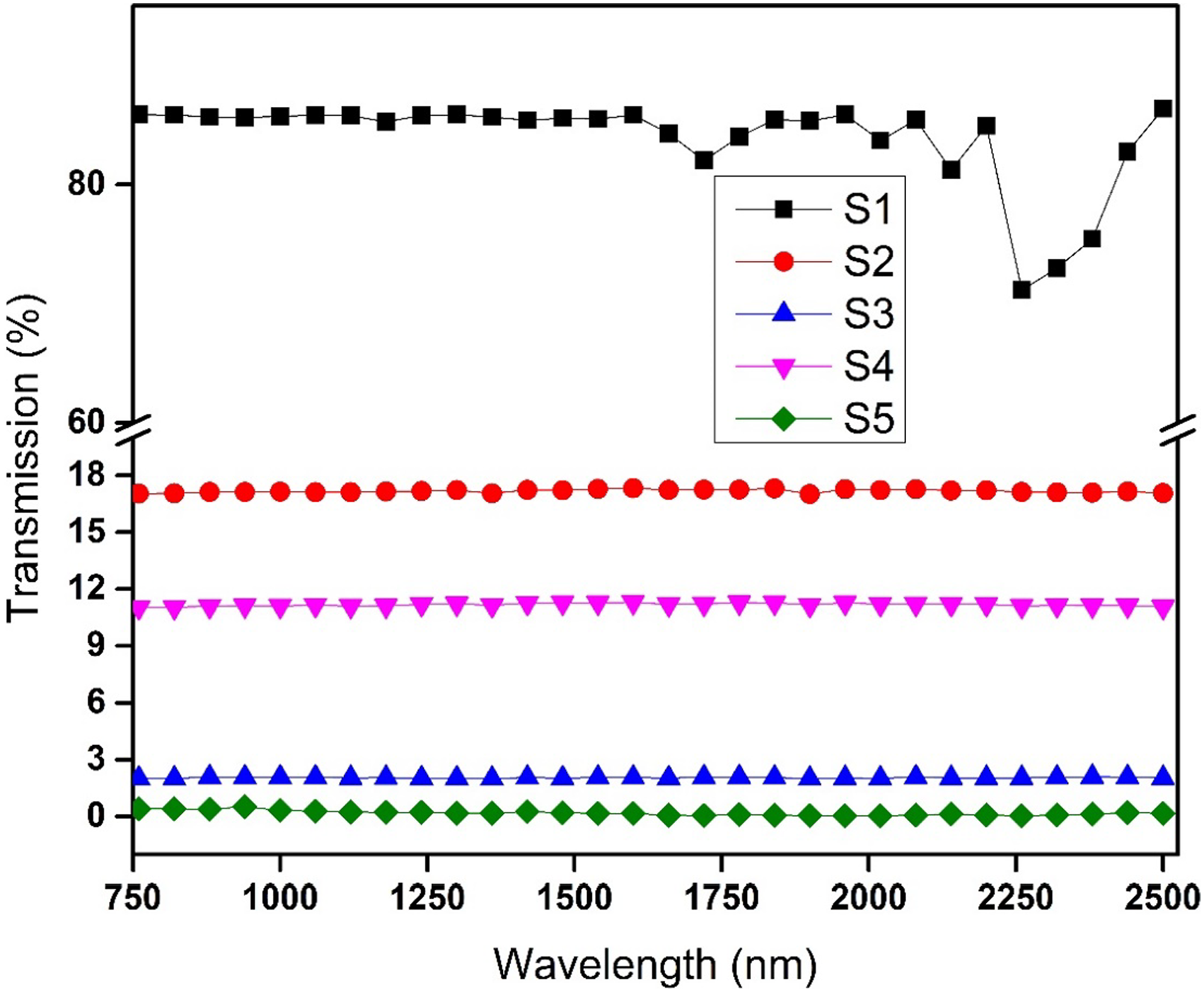
Electromagnetic interference shielding in the microwave region
Electromagnetic interference shielding is considered as the blockage of incident electromagnetic radiations and represented as the logarithm ratio of incident power to outgoing power, mathematically
34
as



The vector network analyzer by scattering the scattering parameters is used to investigate the nanocomposites for electromagnetic interference shielding effectiveness. The VNA can process constraints for electromagnetic radiation as permittivity and permeability. The scattering (S)-parameters are related to reflection and transmission coefficient as (a) SER, (b) SEA, and (c) SET for Ag-ZnS and Ag-ZnS/PANI nanocomposites.




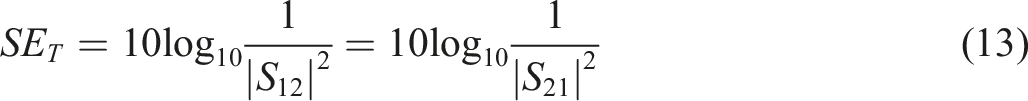
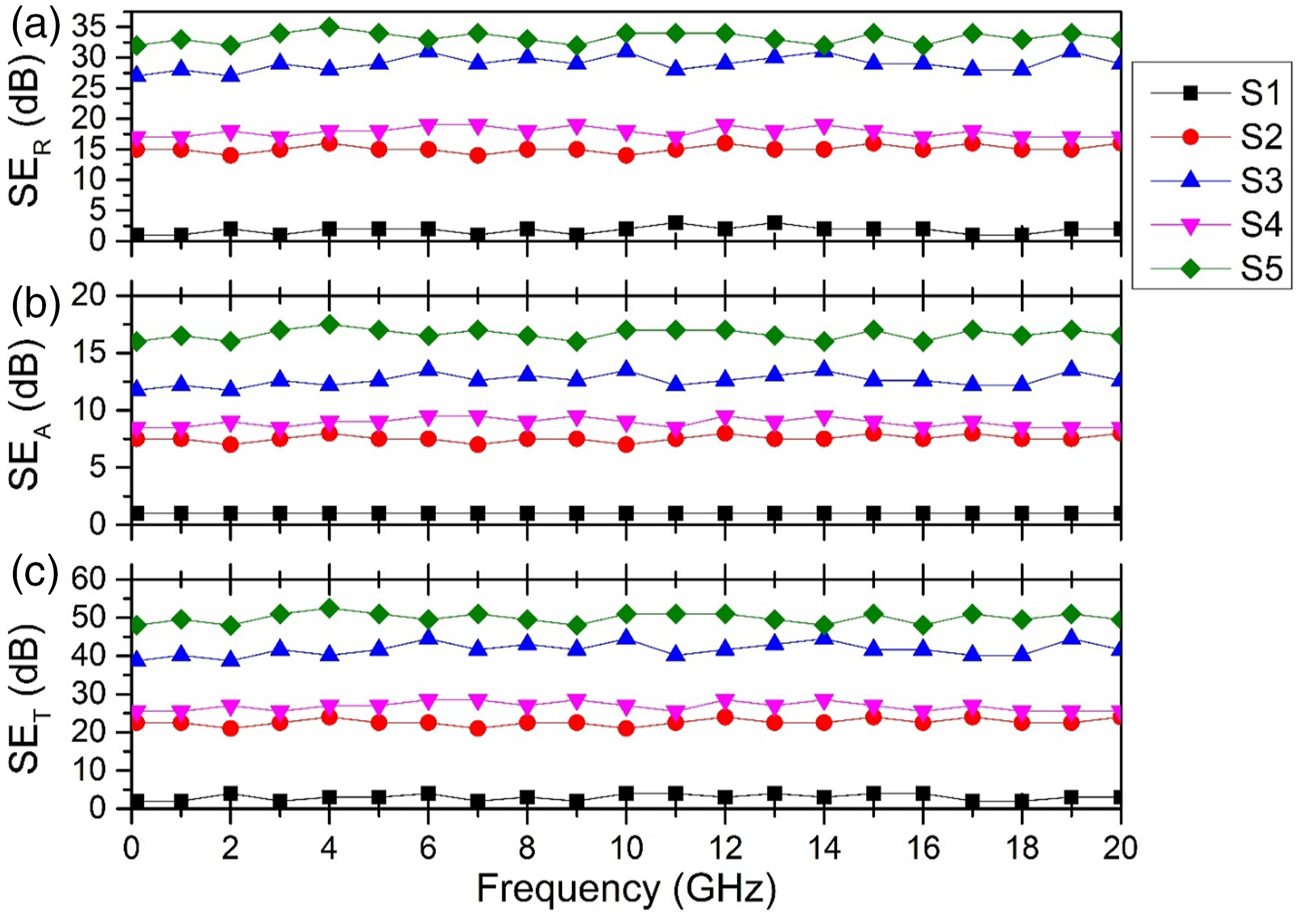
The S1 sample showed almost no attenuation of EM waves because it comprises PVC only that is insulating in nature. The samples S2 and S3 represented increased values of SER and SEA as Ag/ZnS reflected the EM waves due to mobile electrons. The particles that were present inside the nanocomposite gave a high SEA value but the presence of particles on the surface showed a high SER value. The EM radiation reflected from the Ag/ZnS interface and reflecting wave again interacts with another particle before exiting the nanocomposite and so on. In S4 and S5 samples, 10 wt% PANI-Ag/ZnS content showed increased SER and SEA values than S2, with increasing the concentration of PANI-Ag/ZnS to 20 wt% due to the electrically conductive structure formation inside the PVC matrix. The S5 sample showed the highest value of SER due to the presence of PVC/PANI interface on the surface of the composite and a high value of SEA due to PVC/PANI interface presence within the composite. The total SE depends on the absorption and reflection mechanism. The maximum attenuation value obtained in the 0.1–20 GHz region was 52.5 dB for the S5 sample. It is obvious from SEM and DC conductivity values that PANI-Ag/ZnS particles formed an interconnected network structure inside the polymer matrix. This structure becomes more mature and dense with the increasing concentration and in turn increased the electrical conductivity of the composite film. According to EMI theory, higher the electrical conductivity, higher the reflection of electromagnetic waves.
Conclusions
The nanocomposites with the addition of Ag/ZnS and PANI-Ag/ZnS nanoparticles synthesized and characterized by the XRD, NIR spectroscopy, DC conductivity, SEM, and VNA. The evaluated results indicated that the nanocomposite film of 20% PANI-Ag/ZnS was an outstanding nanomaterial for EMI shielding application. These nanocomposites obtained the conductivity value of 0.038 S/cm while the obtained shielding efficiency value for electromagnetic interference was 53 dB in the frequency range of 0.1–20 GHz. The combination of metal/polymer nanocomposites could be useful for effective shielding at commercial as well as a military application.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
