Abstract
This review presents state-of-the-art progress in the field of polystyrene (PS)/graphene nanocomposite. Graphene is a monoatomic thick nanoallotrope of carbon. It has attracted tremendous research consideration owing to chemical functionalization aptitude and remarkable physical properties. Graphene has been used as a potential nanofiller to dramatically improve the performance of polymeric nanocomposite. PS is an important synthetic aromatic thermoplastic polymer. Graphene has been used to enhance the mechanical strength, thermal stability, electrical conductivity, and thermal conductivity of PS/graphene nanocomposite. Dispersion routes and synthetic methods of graphene and PS/graphene nanocomposite have also been reviewed. PS/graphene nanocomposites have been explored for anticorrosion, electromagnetic interference shielding, batteries, electrocatalysis, and microextraction applications. In spite of interesting developments, a lot remains to be done with regard to fundamental understanding of structure–property relationship and designing materials to operate for advanced high performance applications. This review is also concluded listing current challenges associated with processing and future perspectives of nanocomposite.
Introduction
Polystyrene (PS) is a thermoplastic polymer. It has an amorphous polymer having aromatic backbone structure leading to high performance.
1
PS has fine thermal stability and high glass transition temperature (
This review highlights the essential aspects related to PS/graphene-based materials and applications. These nanocomposite categories possess advantageous features and applications depending upon graphene functionalization, dispersion, properties, and processing techniques. Following the vital properties of nanocomposite, cutting-edge application areas of PS/graphene, including anticorrosion, EMI shielding, batteries, electrocatalysis, and microextraction, have been discussed. Toward the end, challenges and future prospects of these nanocomposites have been deliberated.
Polystyrene
PS is a multipurpose thermoplastic polymer. 11 PS has several advantageous features, such as low price, processability, optical transparency, chemical resistance, thermal stability, and mechanical performance. 12 PS is a commercialized material having applications in electronics, packaging, construction, composites, and other areas. 13 In PS-based materials, various types of nanofillers, such as nanocarbon and inorganic nanoparticles, have been used. PS-based nanocomposites have been prepared using emulsion polymerization, bulk polymerization, solution polymerization, and mechanical mixing methods. Such materials have low cost, lightweight, strength, flexibility, and environmental friendliness. 14 Thermal stability and nonflammability performance of PS have been a major concern in academia and industries. In this regard, nanoadditives or nanofillers have been used in PS. 15 PS-based nanocomposites have been studied for property improvement. 16 Research has also focused the hydrophobicity and nonbiodegradability of PS. 17 Nanofiller dispersion in PS can be enhanced by polymer modification and functionalization. 18 Polar groups can also be introduced on synthetic polymer to render hydrophilicity to PS. 19 Functional PS has also been used for water treatment processes. 20
Graphene
Graphene is a two-dimensional one-atom-thick nanoallotrope of carbon.21,22 Graphene has several unique properties, such as transparency, heat and electrical conductivity, and high strength. 23 Graphene also possesses large quantum oscillations, nonlinear diamagnetism, bipolar transistor effect, and ballistic transport of charges. 24 The single-layer graphene can also be converted into other nanocarbon forms, such as fullerene and carbon nanotube (Figure 1). Several methods have been employed for graphene synthesis, including unzipping carbon nanosheet, plasma etching of graphite, liquid phase exfoliation, graphene oxide (GO) reduction, arc discharge, dry ice, shock wave, and chemical vapor deposition.25–28 In polymer matrices, graphene nanofiller renders unique electrical, mechanical, chemical, and optical properties. 29 However, GNS may have self-stacking and aggregation tendencies. To facilitate nanofiller dispersion, graphene has been surface modified to prevent agglomeration in matrices. Surface-modified graphene has been employed in polymer nanocomposite applications, such as solar cell, sensor, biosensor, supercapacitors, transistors, and drug delivery systems. 30
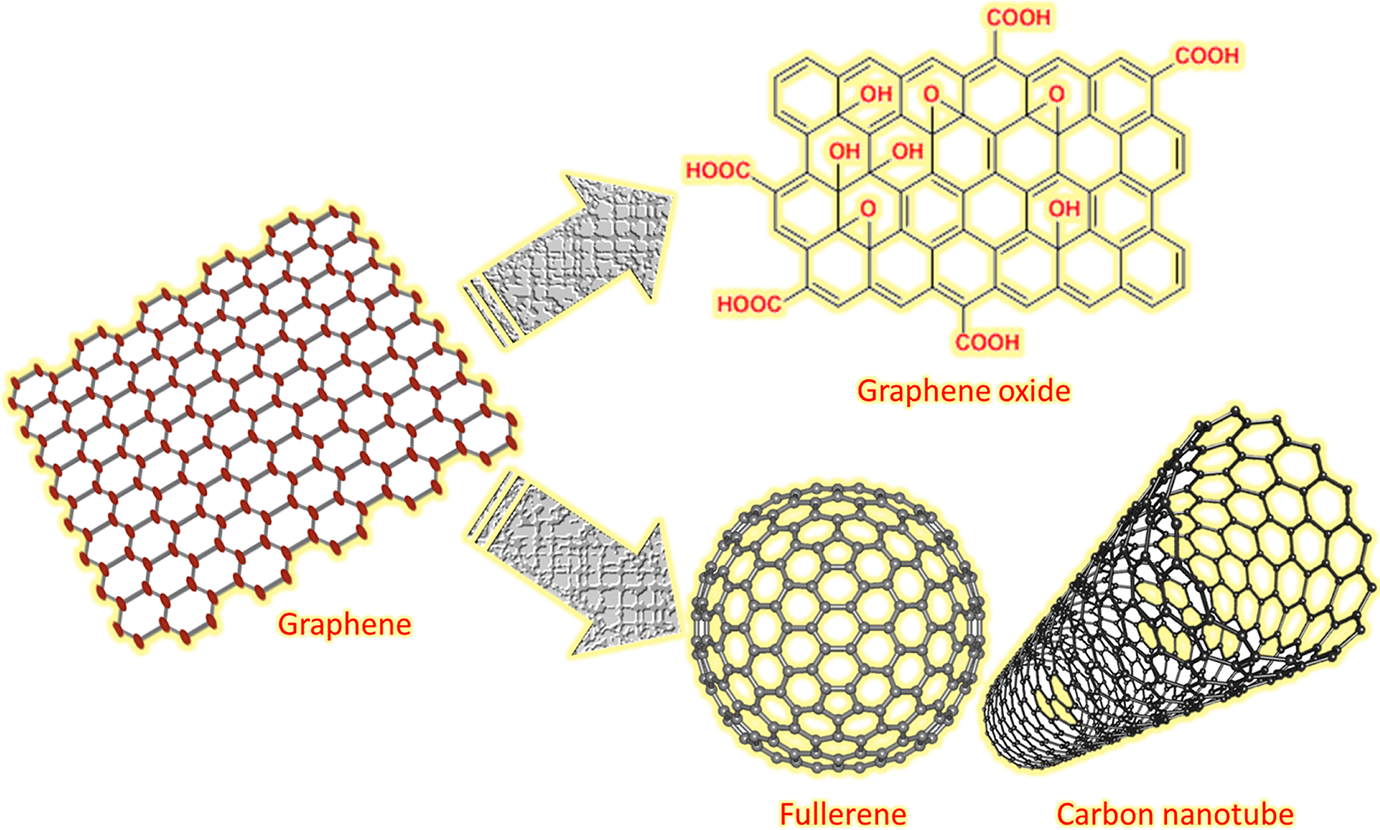
Graphene and its derivatives.
PS/graphene nanocomposite
GNSs have been used to improve the physical properties of polymers.31–33 The films of PS, graphene, and PS/graphene were found hydrophobic in nature and were much stable in water.
34
The PS/graphene nanocomposite has shown
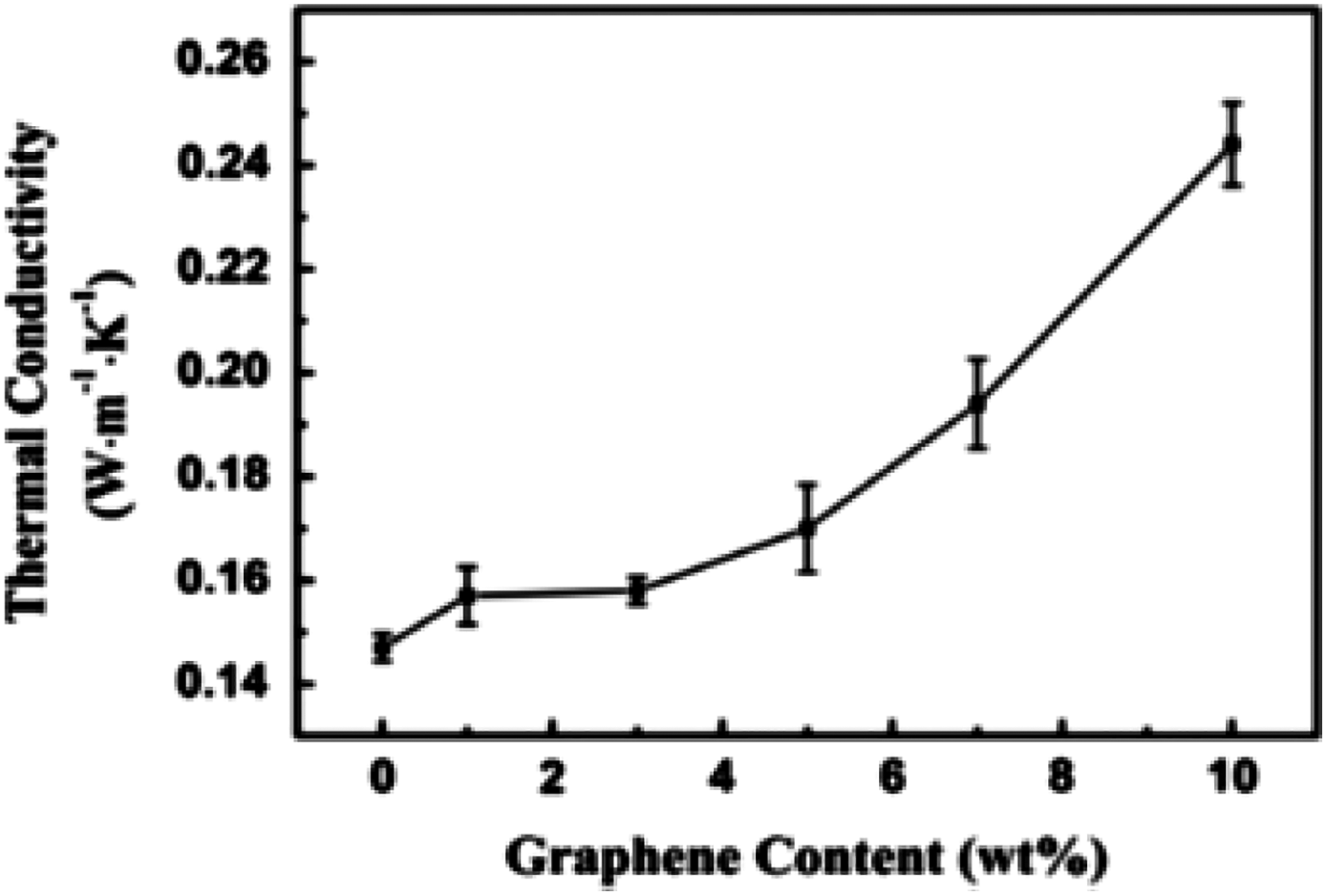
Thermal conductivity of PS composites with varied graphene contents. PS: Polystyrene. 39
Various thermomechanical data of PS/graphene nanocomposite obtained from DMA study. 38
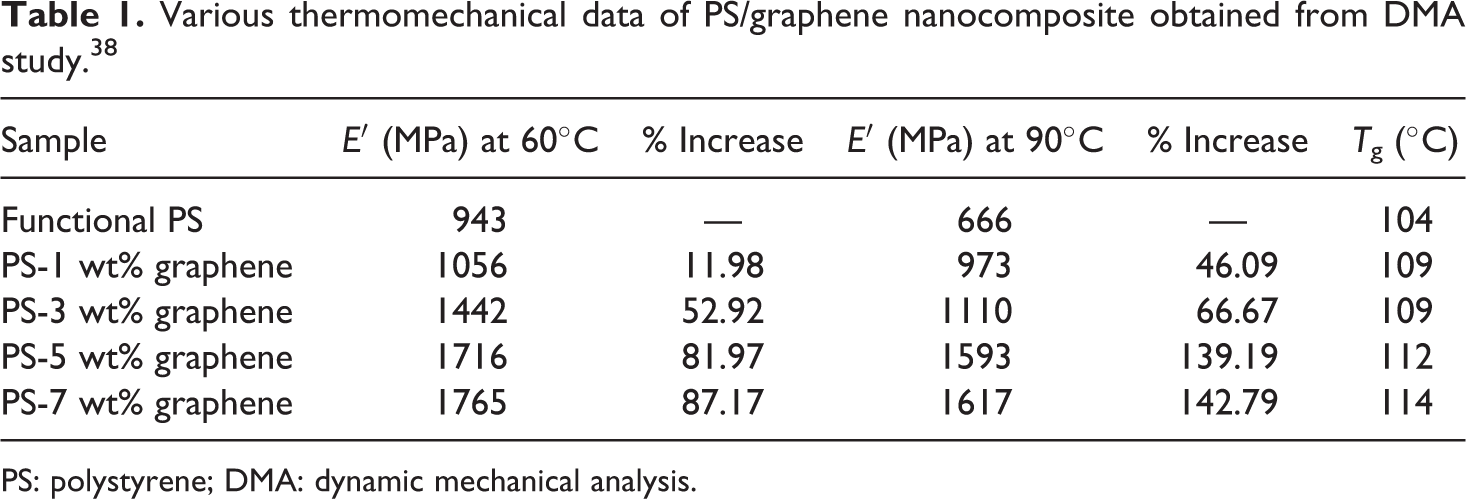
PS: polystyrene; DMA: dynamic mechanical analysis.
Hu et al.
40
prepared PS/GNS nanocomposite using in situ emulsion polymerization. Figure 3 shows thermogravimetric analysis curves of the PS and PS/GNS nanocomposites. PS degrades in the range of 270–420°C. The nanocomposite showed enhanced degradation from 350°C to 450°C. The results indicated robust interaction between the PS matrix and the GNS nanofiller. Figure 4 shows differential scanning calorimetry (DSC) plots for PS and PS/GNS. The pristine PS had low
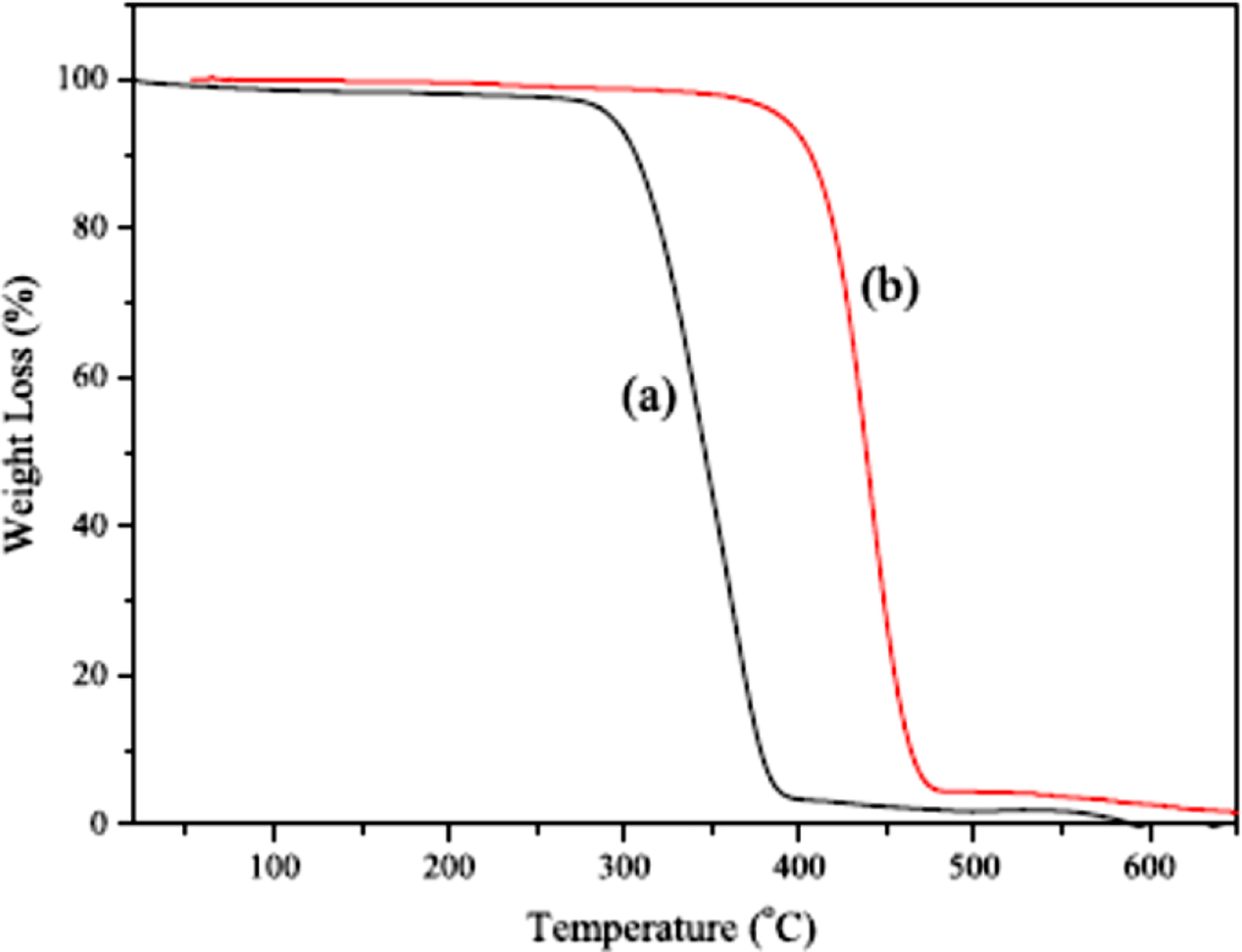
TGA curves of (a) PS and (b) PS/GNS nanocomposites. 40
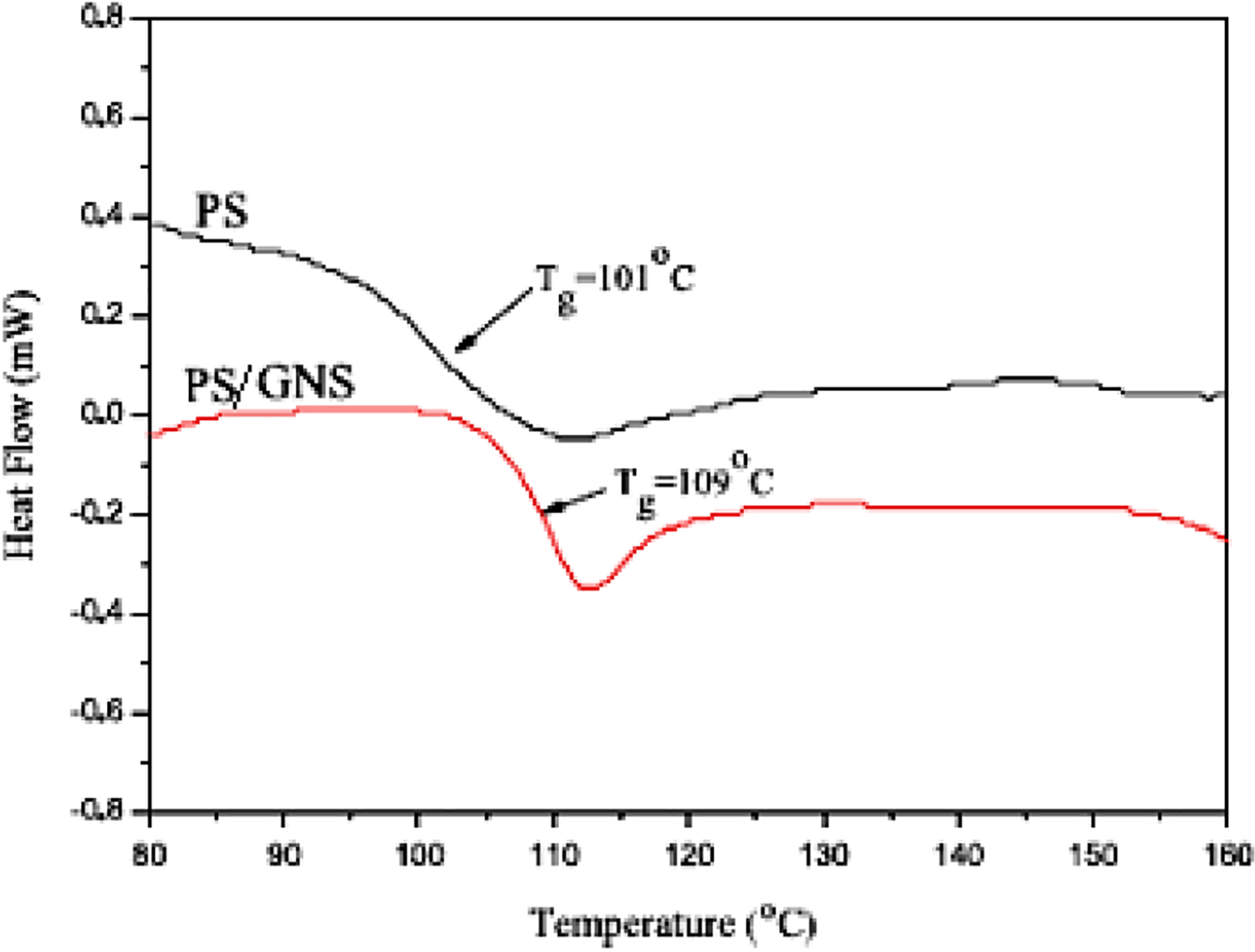
DSC curves for PS and PS/GNS. 40
It was found that the nanosheets hindered the polymer chain movement. Thus, PS-based nanocomposites have been prepared using versatile and environmentally friendly methods. The nanocomposites have been studied for remarkable mechanical properties, high electrical conductivity,
Application areas
Anticorrosion performance
Graphene may form an impermeable diffusion barrier and cause physical isolation between metal and corroding media.41,42 Bolotin et al.
43
performed electrochemical studies on graphene monolayer and multilayer graphene films. The anticorrosion efficiency of graphene was found five times higher than organic coatings. The barrier performance of graphene-coated nickel and copper has been studied.
44
The corrosion rate of metal was decreased with modified graphene coatings. Several studies have reported the use of conducting polymers such as polyaniline (PANI) for corrosion protective coatings.45,46 However, PANI has a limitation of poor solubility restricting its processability in anticorrosion applications.
47
Moreover, conjugated PANI may induce oxide layer formation on steel surface, whereas nonconjugated PS did not form oxide barrier.
48
The
EMI shielding performance
EMI pollution has become a serious environmental issue due to excessive use of electronic equipment, such as computer, cell phones, electronic devices, and radar system. 51 In this regard, polymer nanocomposites have been researched for better EMI shielding performance and high conductivity.52,53 Low loading level of graphene may cause high electrical conductivity and EMI shielding of polymeric nanocomposites. Thermally exfoliated and reduced graphene oxide (rGO) have been used for improving electrical and EMI shielding properties of nanocomposite.54,55 Additionally, surface modification of GNS has been used for improving the EMI shielding effectiveness.56,57 Consequently, magnetic nanoparticles have also been used for enhancement of microwave penetration and absorption by polymer/graphene nanocomposite. 58 Yan et al. 59 prepared PS/graphene nanocomposite with a specific shielding effectiveness of 64.4 dB cm 3 g−1. EMI shielding materials had lightweight and low thickness of 2.5 mm. Chen et al. 60 prepared PS-based nanocomposite with thermally reduced graphene oxide (TGO)-modified Fe3O4 nanoparticle (Fe3O4@TGO) and Fe3O4@rGO. The material has been studied for electrical conductivity and EMI shielding effectiveness. Figure 5 shows electrical conductivities of PS/Fe3O4@rGO and PS/TGO/Fe3O4 nanocomposite. The PS/Fe3O4@rGO nanocomposite had shown increased conductivity relative to PS/rGO nanocomposite at the same nanofiller content. The further higher conductivity was shown by PS/TGO/Fe3O4 nanocomposite. Figure 6(a) and (b) shows disturbing effect of Fe3O4 nanoparticle on GNS. The EMI shielding effectiveness of PS/TGO/Fe3O4 nanocomposite with 2.24 wt% loading was >30 dB in the frequency range of 9.8–12 GHz. The PS/TGO/Fe3O4 nanocomposite exhibited improved EMI shielding effectiveness than PS/TGO nanocomposite. Thus, EMI shielding performance of PS/graphene nanocomposite has found to be improved using metal nanoparticles. Interconnection of GNS and metal nanoparticle with PS matrix may result in fine microwave absorption results.
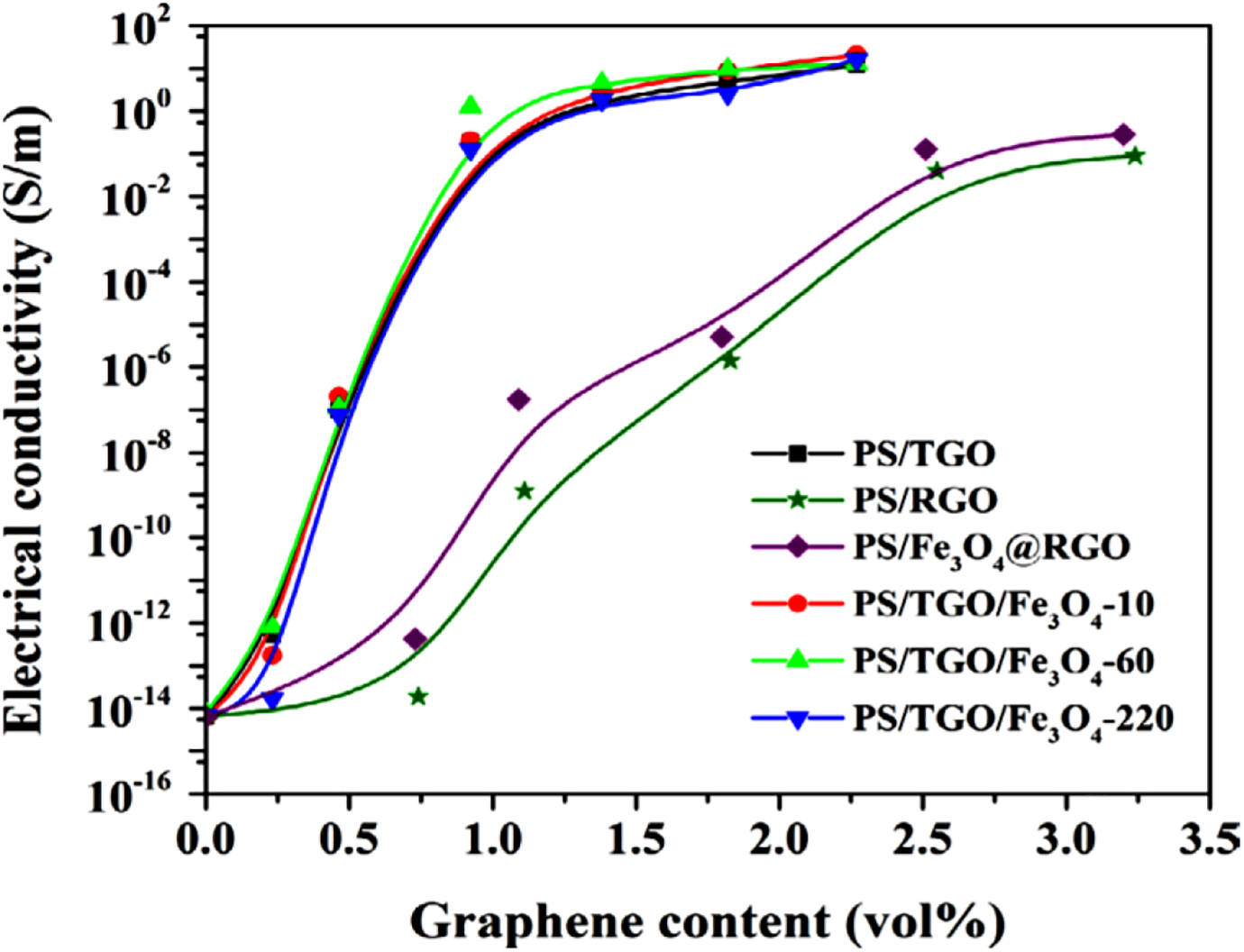
Plots of electrical conductivity versus graphene content for PS nanocomposites. 60

TEM images of (a) Fe3O4@RGO hybrid and (b) scanning electron microscope images of PS/Fe3O4@rGO. 60
Batteries
Electrochemical capacitors, rechargeable batteries, and energy storage devices with high power density, energy density, long cycle life, and fast charge–discharge process have gained recent research interest.61,62 The carbon nanomaterials have been used for high-performance energy storage devices and lithium (Li)-ion devices.63–65 Sodium (Na) ion battery electrodes must have high conductivity, mechanical strength, specific surface area, and chemical stability.66,67 The van der Waals interactions in GNS may result in low surface area and electrochemical properties. 68 Chen et al. 69 developed nanostructured PS/PANI/rGO hybrid. The material was used to prepare the electrode of Na-ion battery. The PS/PANI/rGO electrode had a specific capacitance of 180 F g−1 at a current density of 0.5 A g−1. In Na-ion battery, PS/PANI/rGO anode delivered high capacity of 155 mA hg−1 over 150 charge–discharge cycles at a constant current of 100 mA g−1. The battery had 74% capacitance retention after 5000 cycles. Liu et al. 70 prepared PS/GO nanocomposite as anode for Li-ion battery. The 3-D graphene frameworks (GFs) and nitrogen-doped 3-D graphene frameworks (N-3D GFs) were synthesized using two-step method (Figure 7). The PS nanospheres and GO nanosheets formed core–shell structure through electrostatic attraction. The core–shell structure of PS@GO nanocomposite was converted into N-3D GFs through thermal treatment with ammonia. Figure 8 shows that below 1 V, N-3D GFs had a large capacity than that of 3-D GF electrode. It was found that the nitrogen doping offered active sites for lithium intercalation. Figure 9(a) to (c) shows Nyquist plots of electrodes with two semicircles at high frequency and a slope line at low frequency. The two overlapped semicircles were due to the migration of Li ions at high-frequency range (Rf) and interfacial charge transfer impedance (Rct), whereas the slope line corresponds to diffusion of lithium ions into bulk electrode (Zw). The resulting PS/graphene anode with nitrogen doping had advantages of high specific surface area and electrical conductivity. The anode had a high specific capacity of 1094 mA hg−1 after 100 cycles. According to the literature, the PS-linked GFs have exhibited superior ion storage performance and high rate capability. Nitrogen doping of electrode had also led to improved battery performance.
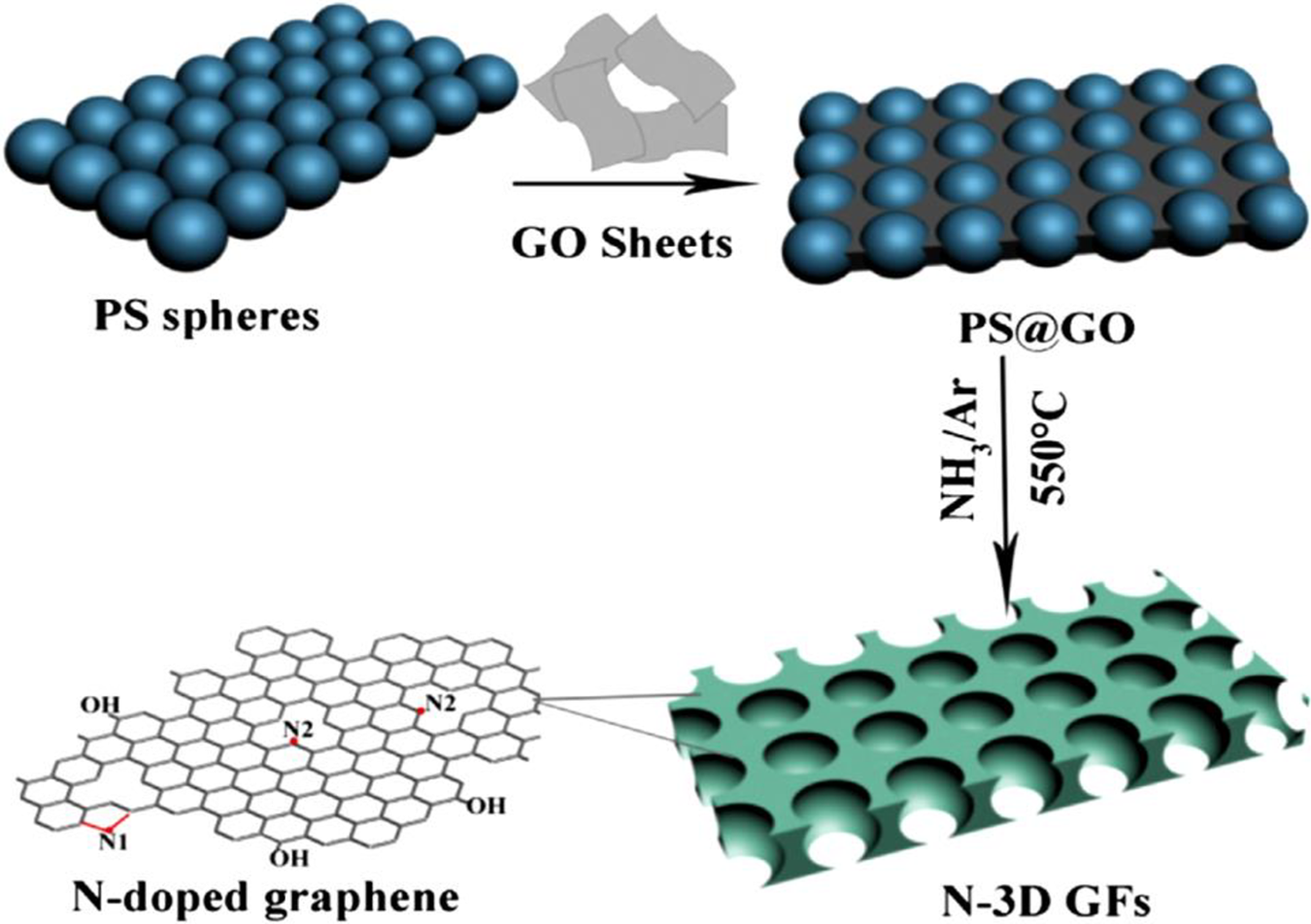
Synthesis process for N-3D GFs. 70
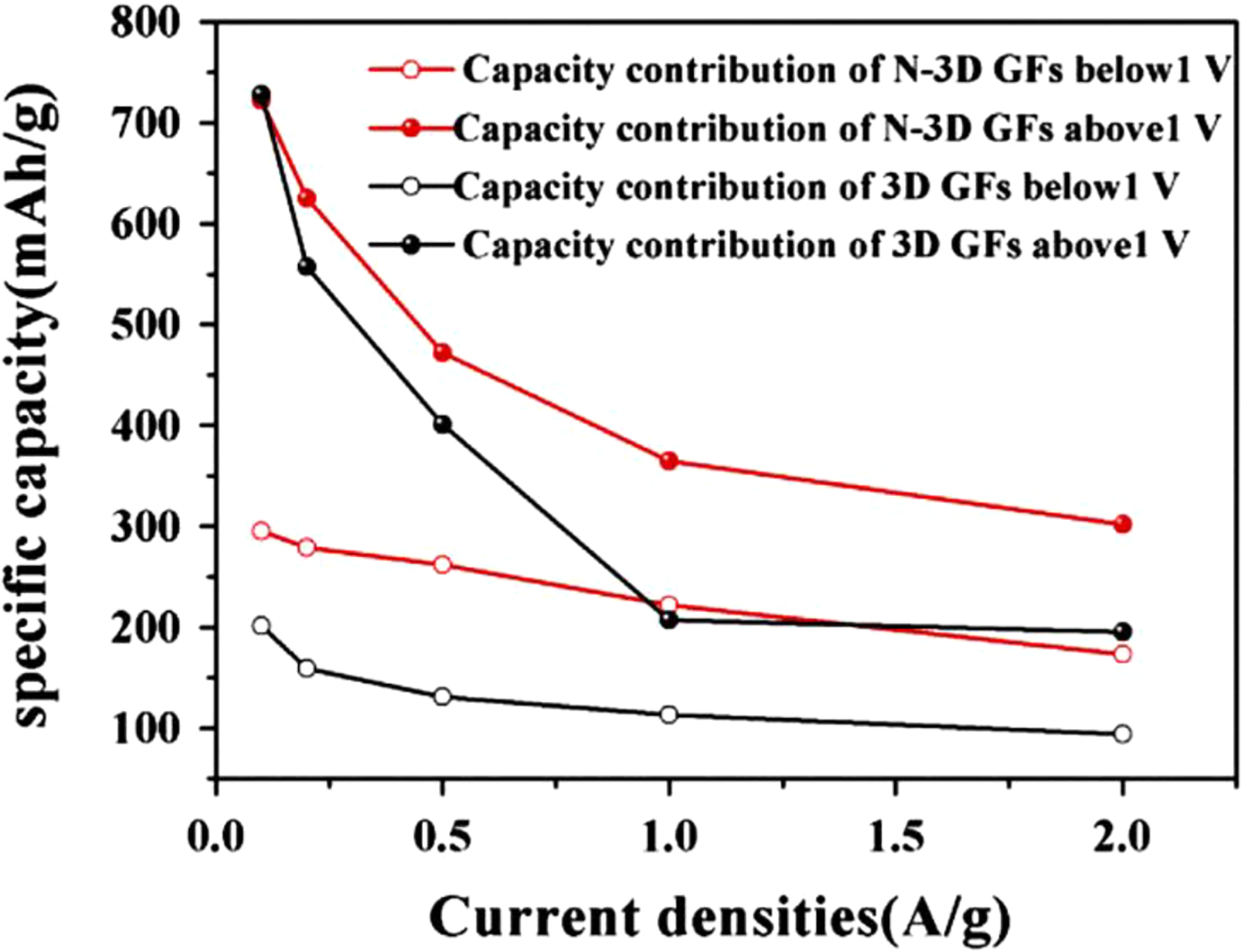
Capacity contribution of N-3D GFs and 3D GFs electrode. 70
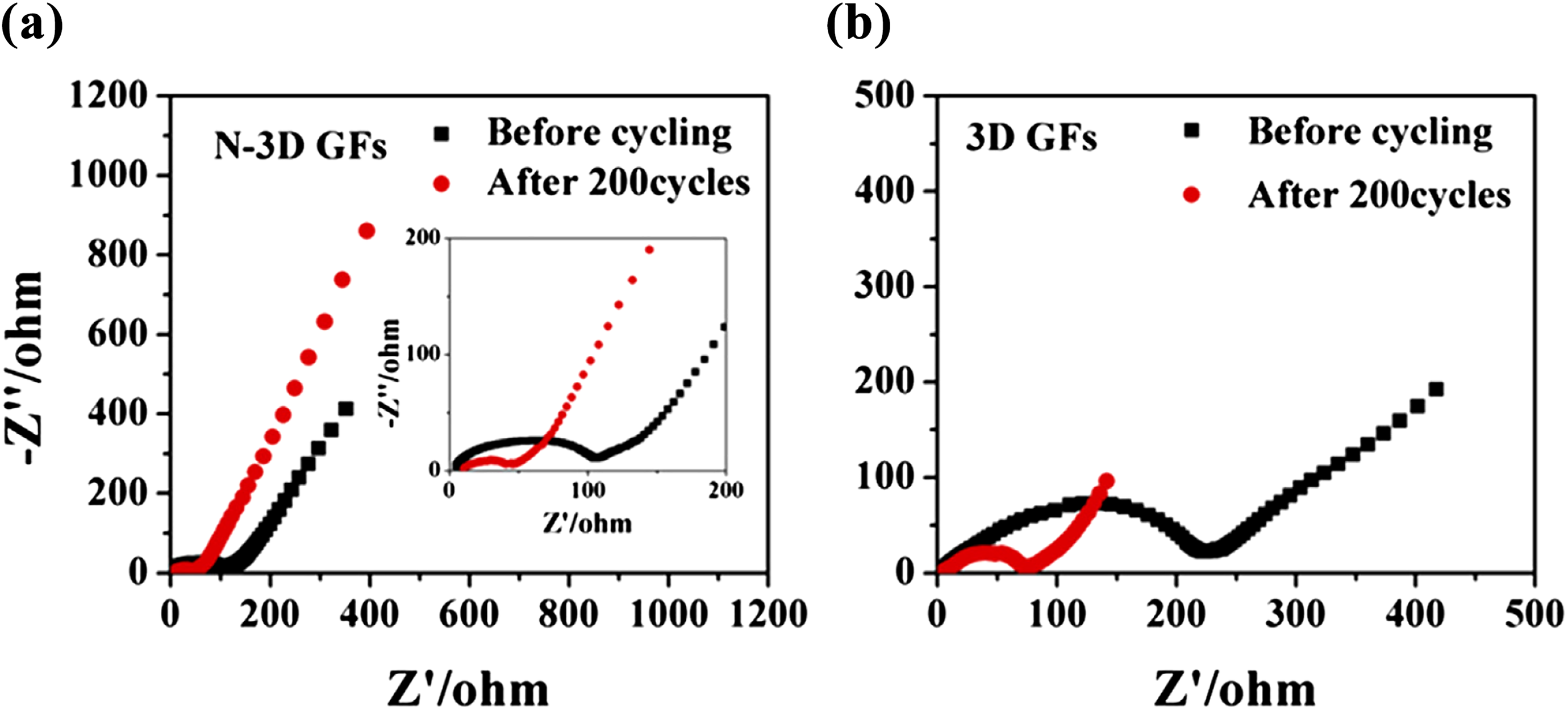
Nyquist plots of (a) N-3D GFs and (b) 3D GFs electrode obtained by applying a sine wave with amplitude of 10 mV at the frequency range of 100 kHz–10 mHz. 70
Electrocatalysis
PS/graphene nanocomposites have shown fine electrical conductivity and electrocatalytic activity.71–73 For electrocatalytic activity, PS microspheres were used. The PS microspheres with an average size of 280 nm were obtained using cationic initiator 2,2′-azobis-(2-methylpropionamidine)dihydrochloride.
74
Then, GNSs with negatively charged groups were coated on PS microspheres. During coating process, low-speed dropwise addition may lead to electrostatic interactions between PS and graphene. Li et al.
75
developed PS/graphene core–shell microspheres. The nanocomposite was highly sensitive for dopamine detection owing to electrostatic interaction and
Microextraction
Solid-phase microextraction (SPME) method has been extensively used for extraction of various organic compounds.
76
The reliability and accuracy of this method depends on extraction, concentration, and isolation of analytes.
77
However, the extraction capacity may be limited owing to low surface area to volume ratio and high thickness of coating material. Research has focused the simplicity, time efficiency, thickness, and surface area of SPME systems. He et al.
78
prepared PS and carbon nanotube-based electrospun nanocomposite thin film for microextraction. The nanocomposite was used as an adsorbent for benzo[a]pyrene and 1-hydroxypyrene. Graphene is a nanomaterial with ultrahigh specific surface area and excellent mechanical, electrical, thermal, and optical properties.
79
Graphene has been found attractive for SPME application.80,81 The nanosized graphene has been used as a sorbent.
82
The aggregation of graphene may lead to decrease in accessible active surface. Huang et al.
35
prepared PS/graphene nanocomposite nanofiber for thin-film microextraction (TFME). The microextraction high-performance liquid chromatography was developed for determination of six aldehydes in human breath condensates. The limits of quantification of six aldehydes were about 13.8–64.6 nmol L−1.The recoveries were in the range of 79.8–105.6% with relative standard deviation of 16.3% (
Future and summary
The future of PS and graphene-based nanomaterials lies in numerous developments in graphene fabrication, nanofiller modification, dispersion, and processing strategies. As reviewed in this article, excellent conductivity of graphene-based nanocomposite can be used for producing corrosion protection and EMI shielding materials. The aromatic structure of PS and 2D nature of graphene render them ideal high-performance materials. In view of electronic device applications, there have been great demand for batteries and high-performance energy storage devices. Ion battery electrodes must be improved in design for high surface area, conductivity, specific capacity, and energy density. Selectivity of PS-/graphene-based SPME systems must be researched for better phase extraction and sensitivity. The
PS- and graphene-based nanocomposites have been appraised in this article. Preparation methods and physical properties of PS/graphene nanocomposite have been reviewed. The main focus of this review was on the applications of PS/graphene nanocomposite in anticorrosion, EMI shielding, batteries, electrocatalysis, and microextraction applications. However, several challenges need to be overcome for the successful use of PS/graphene nanocomposite in technical relevance and future progress in this field.
