Abstract
Many industries discharge oil-in-water emulsion in the waste stream, often above the permissible limit causing serious environmental hazards. Porous media such as membrane and coalescence beds are employed to treat oily wastewater. A coalescence bed filter consists of either fibrous or granular packing and is used for removing larger oil droplets less than 100 μm from secondary emulsions. Fibrous media have higher porosities and specific surface areas than coarse granular media and hence give higher oil removal efficiency. To design an efficient fibrous coalescence bed filter for treating industrial discharge, understanding the mechanism of oil separation is important. This involves the surface wettability of fibers relating to surface chemistry and roughness. Further, fiber diameter, filter bed’s height, porosity, and pore size in relation to the oil droplet size and throughput and influent oil concentration are interactive parameters that affect the efficiency of coalescence. The performance of coalescence filtration is evaluated by analyzing the oil concentrations and D50 droplet sizes in the influent and effluent. In this article, the above-mentioned subjects are comprehensively reviewed from the reported research works, which highlights the complex nature of fibrous coalescence filtration.
Introduction
One of the major concerns around the world is the contamination of water by oil. It is caused either by oil spills in the oceans or by industrial effluent mixing into the oceans and rivers. This happens when lubricants present in the industrial effluents (emulsions) contaminate land and water bodies. 1 Inland oil spills caused by leakage of oil pipes and storage facilities, human errors, unlawful dumping of industrial effluents, and natural disasters have become major sources of deteriorating land and groundwater quality. 2 Industries such as gas and oil production, transportation, petrochemical, shipping, metal cutting, aluminum, steel, food, leather, and textile discharge oily water in the waste stream.3–5 The spilled oil percolates into the subsurface and forms free products or accumulates above the water table within the capillary fringe. 2 They are water-immiscible and sometimes referred to as light non-aqueous phase liquids and are sources of pollution of groundwater. 2 Petroleum processing, metal manufacturing and machining, and food processing are the three major industries that produce oily wastewater. 6 The overall global oil emissions from the transport of petroleum hydrocarbons alone are expected to exceed up to 10 million metric tons. 7 The concentration of oily wastewater caused by industrial operations could reach up to 40,000 mg L−1. 8 For the ecologically sensitive region and aquatic culture, the amount of oil-in-water does not reach 0.1 mg L−1, according to India’s central pollution control board. The oil that has been deposited underwater is causing serious issues for fish, sea plants, corals, and other marine life. 9 As economic activities are increasing over time, oily water in inland waterways and the coastal zone has become one of the most severe issues of water pollution. Industries contributing to this pollution should take pollution control measures to separate oil from wastewater before discharging them, as mandated by governments across the world. Further, industries should explore ways to recycle and reuse water recovered from treated effluent.
Major discharging sources of oily wastewater
Food processing industries
In the food industry, oil effluents are created exclusively using lubricant solutions, water, and pure oil to achieve the finished product. There are large amounts of oily wastewater discharged by the food industry. 10 Due to slaughtering and cleaning practices, the meat processing industries discharging significant amounts of wastewater containing oil. 11 The refining of vegetable oils such as sesame, rice bran, sunflower, groundnut, and soybean creates wastewater from the vegetable oil refinery. 12
Petroleum industry
Crude oil consists of a complex blend of several chemical elements, primarily alicyclic, aromatic, and aliphatic hydrocarbons. 13 During various refinery processing phases, the petroleum industry produces a significant amount of oily sludge. Oily sludge is defined as the emulsion of heavy oil, solid particles, and water. In addition to the inadequate treatment and leakage of transport in storage tanks and pipes, the accidental release of oil–water sludge can also lead to numerous problems in the ecosystem. 14 Contamination of water due to human activity can result in major diseases such as typhoid fever, cholera, diarrhea, vomiting, skin infections, gastroenteritis, and renal failure. Furthermore, contaminated water has the potential to inflict significant harm to the environment and ecosystem. 15 Therefore, chemical industries involved in handling wastewater must have safe disposal of oil sludge and various oil treatment techniques. 16
Metal cutting industry
The metalworking industries generate different types of wastewater because of processing, storage of metal parts/products, and industrial machinery.17,18 Metalworking fluids are used in various mechanical parts for the lubrication and cooling purposes of metal components. 19 These metalworking fluids are usually categorized into two types: (a) oil-based metalworking fluids or oil–water emulsion and (b) water-based metalworking fluids. 20 After using these metalworking fluids for various uses, these oil suspension liquids create a waste stream called spent cutting oil. The spent emulsion of cutting oil is discharged from metallurgical factories in large volumes as oily wastewater. 21 In one year, nearly 2,000,000 m3 of metalworking fluids is utilized globally, and the wastewater emitted from this origin of metalworking fluids is ten times greater than the dilute metalworking fluids. 22 There are different standards for treating spent metalworking fluids due to the presence of several toxins and hazardous chemicals. 23 The operations of manufacturing and rebuilding/maintenance result in the generation of a wide variety of oils and surfactants. 6
Miscellaneous
Restaurants, domestic sewage discharges, pharmaceutical companies, and chemical plants are other important causes of oily wastewater. 24 It is known as one of the most challenging contaminants to extract because of the presence of oil and grease. 25 Municipal wastewater contains grease and oil, but not in excessive amounts since industrial discharges are regulated. The entry of oil into the system is restricted to domestic activities like cooking or cleaning, and the problem is generally managed at the source by installing grease traps. The oil and grease content of domestic wastewaters has been observed to range between 50 and 150 mg L−1. 25
Oil separation methods
Generally, oil remains in oily waters in three different forms, as shown in Figure 1: floating, dissolved, and emulsion. Oil in the free form can be removed by various processes such as settling,
26
centrifugation,
27
hydro cyclones,
28
and oil absorbing pads. Oil in the dissolved form can be separated by various processes such as vacuum evaporation,
29
distillation,
30
membrane reactors,
31
and biological treatments using microbes.
32
Oil in the emulsified form can be separated by various processes such as membrane separation,
33
vacuum evaporation,
34
coagulation,
35
flocculation chemical treatment,
36
flotation,37,38 reverse osmosis/adsorption, deep bed filtration,
39
packed bed coalescence,35,40–48 electro-flotation,
36
electrocoagulation,
49
and coalescence.50,51 But certain issues have become impediments to traditional techniques. The challenges had been that the existing processes are complicated, poor in performance, with high running costs and often have poor oil recovery. In contrast, the fibrous coalescence filter has the advantage of easy device requirement, suitable operation, high processing speed, no requirement of external chemicals, high oil separation efficiency, and recovery.
52
Schematic representation of oily wastewater separation by different processes.
The membrane has pores smaller than the dispersed droplet sizes. The membrane contains pores ranging from 0.1 to 10 μm for microfiltration and 0.001–0.1 μm for ultrafiltration. However, membrane filtration is costly, involving high operating and capital costs, and hence, restricts its affordability in a manufacturing environment. Further, the membrane is prone to fouling, and hence, pre-treatment of the oily water is required before membrane filtration. 53 Finely dispersed oil–water emulsion (secondary emulsions) generated in industries with droplet sizes <100 μm cannot be effectively separated by buoyancy and gravitational forces because of low settling velocity. Porous fibrous media can be used as a coalescence filter, due to superior performance and process economies, and are commonly employed to separate secondary emulsions in droplet sizes <100 μm.
Coalescence is defined as the process by which two or more liquid droplets merge to form one larger droplet. Coalescence allows droplets to sediment more quickly in an emulsion due to an increase in their size. Besides, it may lead to a change in an emulsion’s appearance, as larger droplets usually scatter light less effectively than smaller ones. Hence, the emulsion may be less turbid and more deeply colored when coalescence happens. A coalescence filter is defined as media to separate liquid aerosols and droplets from a gas/effluent. They are specifically designed to remove submicron oil, water, and other liquid droplets from airflow/emulsion flow. Traditionally, a coalescence filter has been used to remove small amounts of water from relatively large quantities of oil. 54 Most recently, the coalescence filter has been used to extract comparatively small quantities of oil from large amounts of water by using hydrophobic filter media.54,55
When an oil–water emulsion is transported to the fibrous filter, the oil droplets come and adhere to the fiber surface before coalescence. In a laminar flow, fiber captures a droplet by direct interception as both have specific sizes. Direct interception is considered the primary mechanism for oil droplets deposited on the filter bed.56,57 Various mechanisms which facilitate the capture of dispersed droplets by a fiber is described in Figure 2. Smaller oil droplets are caught onto the fiber surface by inertial impaction, Brownian movement, or electrostatic attraction.58–60 According to Sareen et al.
60
electrostatic phenomena occur sometimes and may help in the coalescence of the dispersed phase by promoting attraction and adhesion between the droplet and the fiber surface. Schematic representation of the capture mechanisms of dispersed droplets. Adapted from Ref. 61.
Coalescence filtration through porous fibrous media
There are two basic mechanisms of coalescence using porous fibrous media to separate disperse phase from the continuous phase as shown in Figure 3: Depth filtration and surface filtration. Schematic representation of the basic mechanism of coalescence. (a) Coalescence using depth filter and (b) separation using the surface filter.
In coalescence depth filtration, the dispersed liquid droplets coming in with the continuous phase strike the fibers due to various capture mechanisms, coalesce with other droplets, grow big, and eventually drain out of the filter medium.
In coalescence surface filtration, the droplets are often larger than the pore size of the filter medium and would not pass through it. The droplets on the surface of the filter grow in size with the incoming droplets attaching to them. The filtration occurs at the inlet surface of the medium. Surface filters are very thin compared to depth filters.
In the treatment of oily wastewater, fibrous coalescence technology has gained tremendous interest for the removal of the disperse phase using depth filter bed and surface filters. In the coalescence process, using a fibrous filter, oil droplets are transferred through the filter and attached to the fiber surfaces. They agglomerate into bigger droplets on the fibers and are finally removed as oil residence time is prolonged. 64 In recent years, there have been significant advances to explore fibrous materials having oleophilic surfaces to isolate oil from water and high surface energy materials to separate water droplets from oil.64,65
The following steps are involved in the coalescence filtration process using porous fibrous media as shown in Figures 4 and 5: (1) Transport of fluid having dispersed droplets (emulsion) to the filter; (2) Collision of droplets and subsequent coalescence (collision coalescence) or agglomeration or repulsion,
66
and the transport of merged droplets to the fiber; (3) Wetting coalescence processes comprising (a) droplet attachment to the fiber surface and (b) merging and enlargement of the droplets on to the fiber (wetting coalescence); and (4) Releasing of drops from the filter.60,67–74 The collision coalescence process is outlined in Figure 4, showing emulsion breakage and the formation of large droplets. The probability of an oil droplet intercepted by the fibers would be larger than the probability of its collision and coalescence with another droplet in the flow stream.
75
Collision coalescence showing emulsion breakage processes. Adapted from Ref. 76. Wetting coalescence processes within the porous bed. Adapted from Ref. 77.
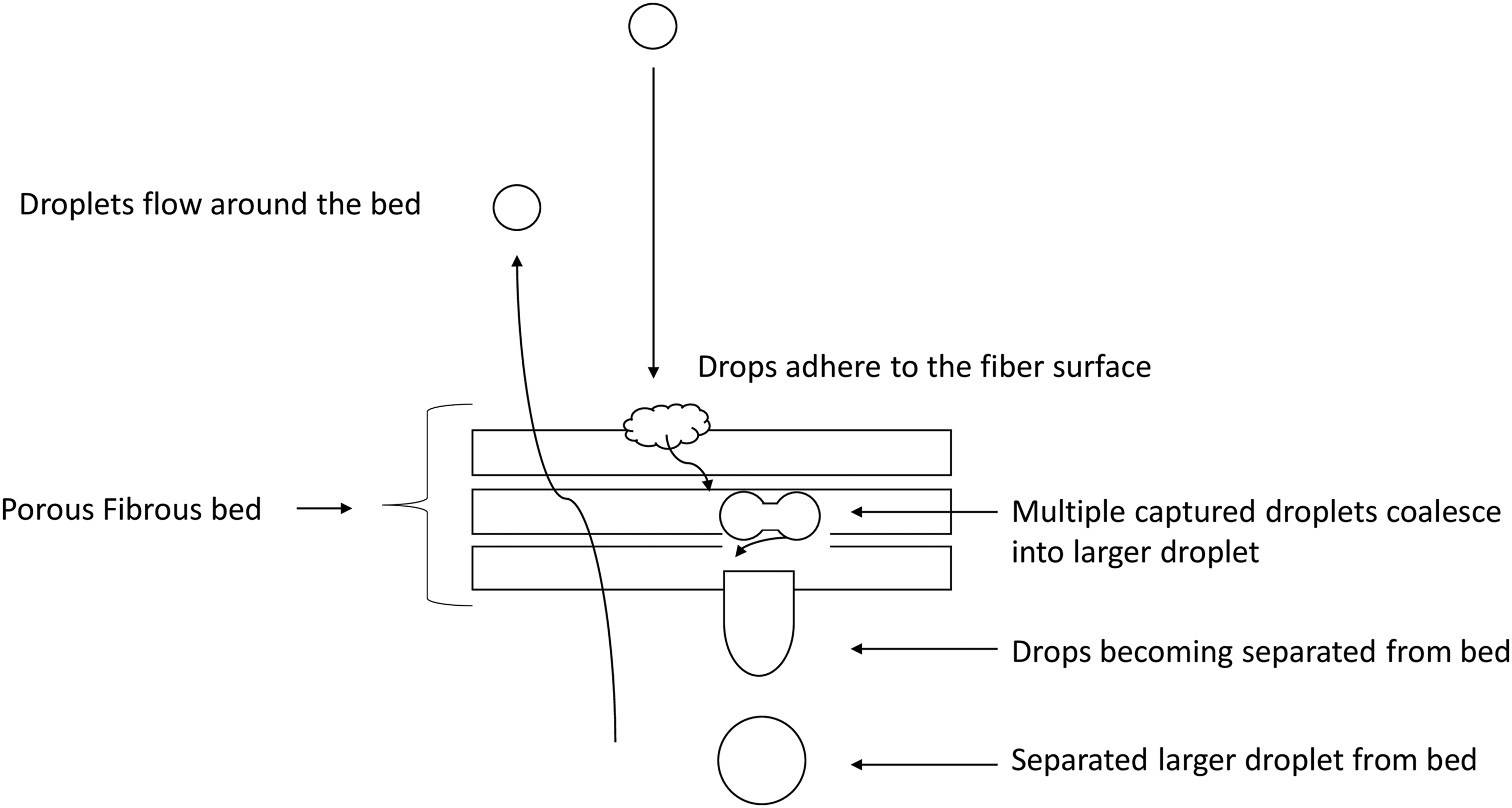
Theory and mechanisms of oil separation by porous fiber media
The probability of droplets coming into contact with the fiber and the surface wettability of fiber are significant for efficient filtration. The oil droplets must adhere and spread onto the fiber surface for coalescence to occur. This depends on the spreading coefficient of oil on the fiber surface, determined by the surface wettability of fiber. The surface energy of the fiber affects its wettability by oil. In Eqs. (1), (2), and (4), the spreading coefficient (

Young’s equation for a droplet on a smooth surface, as explained in Eq. (3) Separation mechanism for oil-in-water emulsion at (A) hydrophobic/low-energy surface and (B) hydrophilic/high-energy surface: (a) adhesion and coalescence of droplets in filters; (b) deformation of the droplet in the pore due to shear forces; and (c) droplet escape from pore and coalescence in the flow stream. Reprinted from Ref. 64 with permission.


If the pore size of the filter is very small compared to the drop size, the coalesced droplets float upstream, which can be collected as oil film. This phenomenon is called “surface filtration,” where the filter does not allow droplets to pass through the filter and hold them at the filter surface itself. 64 The retained droplets at the filter surface further coalesce into bigger droplets.
The surface energy of the fiber and the surface tension of the disperse phase (oil) determine the oil contact angle. The surface tension of oil must be lower than the surface energy of fiber. If fiber’s surface energy is very high, then a continuous phase (water) would also spread on the fiber (lower water contact angle), reducing the adhesion between the fibers and oil. As a result, the oil droplets at the fiber remain spherical and have poor contact (Figure 6(b)-(a)); hence, they are easily detached from the fiber without coalescence. Depending on the pore size to influent drop size ratio, a few or many droplets undergo deformation without coalescence on the fiber (Figure 6(a)-(c) and (b)-(b)/(c)). These droplets may coalesce in the flow stream but would be small enough to remain dispersed in the effluent. These are called “redispersed droplets.”
Factors affecting the performance of coalescence filtration by the fibrous filter
Several research findings have been reported on fibrous media such as natural and synthetic fibers, fiber diameter, surface wettability, and oil type121,122; characteristics of dispersed oil-in-water emulsion such as size distribution of oil droplets 39 and oil concentration; and flow rate 123 on the coalescence performance of fibrous filters. The parameters that affect coalescence filtration performance are (a) fiber characteristics; (b) filter medium; (c) infeed emulsion; and (d) process parameters employed during filtration.
Fiber characteristics
Fiber diameter
Fiber diameter plays a critical role in enhancing the coalescence rate.39,107,124 The specific surface area of the fiber increases with reducing fiber diameter. At constant packing density (or porosity) of fibrous filter, filter constructed with finer fibers has a greater number of fibers than the coarser fibers. Thus, a higher volume-specific area of fiber in the filter (ratio of the surface area of fiber to the volume of fibers, m−1) can be obtained with the finer fibers than with the coarser fibers. The mean hydraulic pore diameter (or capillary equivalent diameter) of the filter with finer fibers is smaller than that having coarser fibers at a constant porosity of filter media. This improves the probability of dispersed droplets being captured on the fibers by interception. The interception efficiency is proportional to the square of the mean droplet size in the influent and mean hydraulic pore diameter of the filter bed. Thus, the filter bed made of finer fibers could result in effective coalescence of the droplets, improving oil separation efficiency. In Eq. (5), the capillary equivalent diameter (
The hydraulic pore diameter 
Fiber surface wettability
Fiber surface wettability is one of the most important parameters of filter medium for an efficient coalescence to remove oil from oil-in-water emulsion. Both the surface chemistry (surface energy) and the roughness of fibers affect the surface wettability. Fibers having higher and lower surface energies are “hydrophilic” and “hydrophobic,” respectively, as shown in Figure 7. By having a different combination of fiber surface chemistry and fiber roughness, superwetting and antiwetting can be obtained. Water droplet on fiber surface with contact angles: (a) hydrophilic (superwetting); (b) intermediate state and (c) hydrophobic (antiwetting). Adapted from Ref. 103.
Surface chemistry and roughness
Introducing roughness on fiber surfaces can alter their surface wettability differently by oil and water. According to Wenzel’s wetting theory,
106
the apparent contact angle on a rough surface is related to the static contact angle on the smooth surface, as in Eq. (7)
In the case of Cassie mode, a rough surface having dual surfaces/chemical heterogeneity having superhydrophilic and superhydrophobic surfaces in the proportions 
In this case, the contact angle can still be increased while the surface becomes rough.
127
Feng and Jiang
127
discussed a threshold value 
If Young’s contact angle is lower than
By combining a range of surface roughness, and surface chemistry and chemical homogeneity or heterogeneity (dual or multi-surfaces), it is possible to create superwetting (superoleophilicity or superhydrophilicity) and antiwetting (superhydrophobicity or superoleophobicity). The transformation of wetting states is illustrated in Figure 8. Transfer between four basic wetting states Superwetting/Amphiphilicity (Superhydrophilicity and Superoleophilicity) and Antiwetting/Superamphiphobicity (Superhydrophobicity and Superoleophobicity) properties, and double arrow indicates the coexistence of two properties (heterogeneity/dual scaled); reversible arrow indicates switchable properties. Adapted from Ref. 127.
Superhydrophilicity and superoleophilicity can coexist on a solid substrate having very high surface free energy and micro-/nanostructures, and the film will exhibit superamphiphilicity (Figure 8, right). Superhydrophobicity and superoleophobicity can coexist on a rough surface with a very low surface free energy, and the film will demonstrate superamphiphobicity (Figure 8, left).
When the hetrogenous surface exhibits superoleophilicity-superhydrophobicity or superhrdrophilicity-superoleophobicity, oil from water or water from oil respectively can be separated efficiently. When stimuli-responsive materials with appropriate surface geometric structures are exposed to changes in the environment, their surface wettability can be reversibly switched between superhydrophobicity and superhydrophilicity or between superoleophobicity and superoleophilicity. These are called “smart switches,” and they have the potential for constructing future generation smart devices.
When nanoscale roughness is introduced on a homogeneous (especially single polymer manmade fibers) and high-energy surface (hydrophilic), where the static contact angle is <90°, the surface becomes more wettable with water (toward superhydrophilicity). In contrast, on a low-energy surface (hydrophobic), it promotes the dewetting of water (superhydrophobicity). 128 From this, it can be inferred that hydrophobic-oleophilic nanoparticle coating on low-energy fiber surfaces alters both the surface wettability of oil and water such that it increases the water contact angle and reduces the oil contact angle, thus, promoting preferential wetting by oil than by water. This phenomenon has been reported by Bansal et al. 64 According to them, surface roughness and limited pore size (pore size is smaller than the droplet size) synergistically promote the capture of oil droplets and coalescence at the fiber surfaces (surface filtration) for the fibers with low surface energy.
Bansal et al. 64 investigated disperse phase removal from isooctane-deionized water using fibrous filters in down-flow mode. They applied nanoparticles on woven polyester, non-woven cellulose, meltblown PBT, and spunbond polyester having different porosity and pore size to prepare filter media with different surface wettability. It is reported that the mean dispersed droplet size in the effluent increases with increased fiber surface energy at a constant pore size. The low surface energy filter is easy to wet with oil but not with water (Figure 6(a)-(a)). The adhesive forces between the oil and fiber surfaces are much higher than between the water and fiber surfaces for low-energy surfaces. Hence, the dispersed oil droplets displace water quickly from the fiber surface and spread out. In the case of high surface energy materials (hydrophilic), there are low adhesive forces between oil and fiber, and oil droplets cannot easily displace the water from the fiber surfaces. Oil droplets on hydrophilic surfaces in water show a high contact angle with substantially spherical shapes on fiber surfaces (Figure 6(b)-(a)). Due to weak adhesion or droplet deformation by hydrodynamic forces, these spherical oil droplets can quickly move via high surface energy filters (Figure 6(b)-(b)). Therefore, for the same pore size in the filter, larger dispersed droplets move via high surface energy fibers than via low surface energy fibers. Hence, wettability affects the size of droplets in the effluent significantly.74,96,112,129 The oil separation efficiency decreases with an increased surface energy of fibers.
Though the surfaces can be made rough to fabricate superhydrophobicity or superhydrophilicity, the strength and durability may often worsen due to the chemical treatments. This might hinder their utility in industrial applications. Ramkumar et al. 130 investigated smart poly (vinylidene fluoride) nanofiber webs customized with rGO/TiO2 composite nanoparticles (combination of hydrophilic and hydrophobic nanoparticles on the hydrophobic surface) for application in oil/water separation. The incorporation of rGO/TiO2 nanoparticles resulted in outstanding oil–water separation efficiency of up to 98.5%. When rGO/TiO2 concentrations increased from 0% to 20%, the water contact angle of nanofiber decreased from 130° to 110°. The rGO/TiO2 hybrids have enhanced both the specific surface area and also formed dense pore structures, which gave superior controlled permeability.
Yang et al. 24 reported that polyester substrate deposited with PVDF nanofibers showed very high oil removal efficiency up to 99.5%. In an oil/water/solid three-phase system, researchers produced a unique superamphiphilic PVDF membrane with a multiscale surface structure that exhibited ultralow adhesive superoleophobicity underwater and low sticky superhydrophobicity under oil. Due to its switchable transport property, the membrane may be used to separate diverse oil-in-water and water-in-oil emulsions with droplet size larger than 20 nm. It has excellent separation efficiency, permeability, recyclability, and antifouling properties. 131
Zhang et al. 132 prepared a superhydrophobic and superlipophilic PLA/γ-Fe2O3 dual scaled nanocomposite fibrous membrane having micropores between fibers and nanopores on fibers by uniformly spreading γ-Fe2O3 nanoparticles over fibers to increase the roughness of the surface. The composite membrane had high mechanical properties in addition to being superhydrophobic and superlipophilic. The membrane showed a very high adsorption capacity of 268.6 g/g for motor oil. Shin and Chase 68 found improved droplet capture and coalescence efficiency for treating oil–water emulsion with nanofibers incorporated microfiber glass-based filter media.
Barroso-Solares et al. 133 incorporated iron nanoparticles on oleophilic and hydrophobic PMMA, poly (methyl methacrylate) nanocomposite fibrous mats. The incorporation of nanoparticles increased the hydrophobicity, showing high oil absorption capacity when the fiber mats were placed in the water-oil emulsion. Ren et al. 134 investigated magnetic nanocomposite fibrous mats. They prepared PIL poly (ionic liquid) grafted polypropylene non-woven by surface grafting of IL monomers and photocrosslinking. By anion exchange of the PIL anion from the hydrophilic bromide (Br-) to the hydrophobic bis (trifluoromethyl sulfonyl) imide (TFSI-), the nanocomposite non-wovens displayed a good and switchable separation of oil/water. By washing with an acid solution, the PIL grafted PP fibrous filters can easily be regenerated.
Hu et al. 106 investigated smooth and rough non-woven filter mats made of a mixture of glass wool, glass fibers, and cellulose fibers. They used these mats for studying coalescence filtration of oil from oil–water emulsions. The fiber mats having rough surfaces showed higher oil separation efficiency than the smoother ones. They reported that an optimum oil contact angle is required in order to balance the oil droplet capture by fibers and releasing them for effective oil removal. A lower oil contact angle only favors the capture and not the release, and the large oil film formed on the fibers in the filter exploded, caused frequent oil breakthroughs. In the case of surface filtration, an intermediate wettability allows the oil droplets to displace the water sufficient enough at the fiber-liquid interface and also allows easy release of the coalesced oil for better separation. Wang and Song 135 also made a similar observation that the optimum oil spreading angle should be kept at 70°–80° so that the absorbed oil drops on the fabric surface aggregate together rather than spreading wildly. In deep bed filtration using kapok fibers, a very high oil (diesel) removal efficiency of 100% was observed with oil contact angle of 13°. 136 This indicates that fibers used as coalescence bed filters must have a lower oil contact angle.
Natural fibers and materials
Conventionally, synthetic fibers like nylon, PP, and polyester are utilized as filter media in deep bed filtration. Due to their non-biodegradability, they cause serious environmental problems when the used filters are disposed off. Non-wovens based on biodegradable or agricultural materials such as cotton and wool were explored as filter bed materials. Some research works are reported on the suitability of natural vegetable materials, including fibers with hydrophobic and oleophilic characteristics for the separation of oil–water emulsion. Raw cotton fibers are hydrophobic as they have waxes on their surfaces; hence, they float on water. Their surface energy is above that of oils, and thus, they are highly wettable by oils. Sareen et al. 60 performed a unique experiment to investigate the performance of a tightly packed cotton fiber filter bed assisted by glass, polypropylene, and polytetrafluoroethylene fibers. They observed that of all fibers, cotton fibers gave greater coalescence. The dispersed phase (oil) had a zero-contact angle on cotton fiber. Cotton fibers did not exhibit preferential wetting due to many depressions and obstructions on the fiber surface exhibiting surface roughness. Deschamps et al. 137 observed that a cotton filter bed could be used to get effluent almost oil-free recovered oil from oily water. Das et al. 138 synthesized fluorinated silyl functionalized zirconia using the sol–gel technique and coated them on cotton fabrics by immersion technique to create superhydrophobic surfaces. The coated fabrics had a remarkably high separation efficiency of 98.8% in separating oil from oil/water emulsion.
Riberio et al. 139 reported that Salvinia, a hydrophobic aquatic plant performed very well in separating oil from the oil–water emulsion. The large surface area, hydrophobicity, expandability due to hair-like projection of the surfaces are the reasons attributed to this observation. Varghese and Cleveland 39 observed that a kenaf fiber filter could remove about 70–95% of oil from surfactant-stabilized oil-in-water emulsions. Pasila 140 observed that reed canary grass, hemp, and flax fibers could remove oil from oily water. Milkweed, sericin derived from silk cocoon, bast fibers, wool, cotton, PP, and PU were also used for oil separation.125,141–143 The oil efficiency of kapok, cattail fiber, wood chip, coconut husk, rice husk, bagasse, and polyester fiber have been investigated by Khan et al. 144 Among them, the filter beds constructed of hydrophobic material had higher oil removal, whereas hydrophilic materials could achieve only 30% removal. The higher oil removal efficiency of the hydrophobic materials can be attributed to their water-repellent waxy surfaces.
A study on kapok fiber as the filter medium in coalescence deep bed filtration for separating diesel and hydraulic oil (a high viscous oil) from oil–water emulsion found an incredibly high oil separation efficiency close to 100%. 59 The contact angles of diesel and water with kapok were 13° and 117°, respectively. 136 Kapok fibers are hollow, permitting a high specific area, and this property could be utilized to construct the porous bed.
Wang et al. 145 applied silica nano nanoparticles onto the NaClO2 treated kapok fibers to alter their wettability. The modified fibers have outstanding hydrophobicity and gave the highest oil absorption. The oil was easily absorbed onto the surface of modified fibers, leading to a high oil sorption capacity. 146 The untreated kapok fibers themselves are hydrophobic and oleophilic, as reported earlier. According to the present authors, the nanoscale surface roughness on the modified fibers might have diminished the surface wettability of water more than enhancing the oil wettability. This differential wetting might still be useful to enhance oil absorption. The above observations are related to the removal of floating oil. To what extent the modified kapok fibers do improve the oil removal from the oil–water emulsions must be investigated since the unmodified kapok fibers themselves gave very high oil removal efficiency, as reported by Huang and Lim. 59 Further, the surface modification using nanoparticles is an additional process with time and cost constraints.
It is evident from the above discussions that the surface wettability of fiber plays a substantial role in the initial attachment of oil droplets and spreading them onto the fiber surface. The fiber surface should be oleophilic and hydrophobic so that the work of adhesion between the oil and the fibers must be higher compared to the water-fiber interface. In other words, the fiber must be wettable by oil and not by water. Hence, water can flow continuously and rapidly through the pore channels of the fibrous medium without hindrance. This also improves the throughput of the filtration process. The fiber wettability is a function of fiber surface chemistry and surface roughness. The surface chemistry is an inherent property, and the fibers’ surface wettability can be modified by incorporating different scales of roughness, viz., micro, nano, or both, or chemical heterogeneity on the surfaces.
Characteristics of the filter medium
Porosity, pore size, and porous structure of bed
Porosity provides information on the total pore volume of the filter and is defined as the non-solid volume ratio (voids) to the total volume of the filter.147,148 A closely packed fibrous bed would have a smaller capillary equivalent diameter and the mean hydraulic pore diameter as discussed earlier (refer Eqs (5) and (6)). This would increase the droplet interception and coalescence efficiency. Hu et al. 106 investigated non-woven fiber mats reinforced with a binder. Fiber mats bonded with resin had lower porosity and average pore size, with uneven pore size distribution exhibiting pore blockage compared to the unbonded ones. In addition, resin-bonded mats showed lower oil separation efficiency due to the smaller fiber surface area for capturing the oil droplets. This causes high flow resistance and subsequently low separation efficiency.
In laminar flow, the Hagen–Poiseuille is applied as in Eq. (10)
Mechanisms of filtration of oil from oil-in-water emulsion concerning surface energy of fibers and the ratio of the size of pore size to droplet size.

Bed height
In the coalescence process, bed height plays a significant role.17,102,151–154 Zhou et al.
155
studied the bed height on effluent oil concentrations. Having a shorter bed might result in lesser interaction between fiber and oil droplets and insufficient drops coalescence. Li and Gu
107
investigated bed heights (
Sareen et al. 60 working on cotton fiber beds backed by fibers such as polypropylene, polytetrafluoroethylene, and glass found improved coalescence when bed the height was increased from 0.25 to 1.25 inches. However, they observed redispersion of the coalesced droplets above a certain bed height. Radmila et al. 156 also found similar observations. The longer beds behaved as two beds in succession if the fluid flow velocity is above the critical velocity. Above the critical velocity of the fluid through the bed, the oil concentration in effluent increased initially, reached a maximum, and decreased with increasing bed height. Beds of 7 and 15 cm gave almost the same oil removal efficiency, whereas the bed with 10 cm height gave low efficiency. As the oil droplets enlarged by coalescence pass through the remainder of the bed, they are redispersed into smaller droplets and may coalesce again. This is analogous to the operation of two beds constructed sequentially. High effluent concentration for 10 cm bed may be attributed to redispersion of drops without coalescence, whereas the low concentration in the effluent obtained with 15 cm bed is due to both redispersion and repeated coalescence. Bed height is observed to have a major effect on the coalescence performance, and a suitable bed height must be selected to maximize the filtration performance. However, the optimum bed height very much depends on the bed porosity and the desired flow rate, and it must be selected based on trials.
Emulsion (oil-in-water) parameters
Oil concentration
Oil concentrations in the oil–water emulsions play an important role in coalescence filtration. At higher concentrations, there are more droplets in the flow stream; thus, the ability of droplets to collide on the fiber or colliding among themselves increases, which may increase coalescence efficiency. 59 This is too simple to state, ignoring the complex nature of coalescence. Li et al. 107 observed that the poor coalescence efficiency at higher oil concentrations might be due to the coalescence occurring in the bath rather than on the fiber surface. Radmila et al. 156 have observed that increased oil concentration in the influent increases the effluent concentration. At higher flow rates, the effect is more pronounced because of fluid velocities are above the critical velocity. At low flow rates, the separation efficiency is independent of the concentration of oil in the influent. A high oil concentration in the influent substantially reduces the oil separation efficiency at a higher working velocity. In the case of an emulsion having high oil concentration, a larger amount of oil is held in the pores. This reduces the hydraulic conductivity/permeability of the filter, encountering high interstitial velocity in the pores, consequently more redispersion of droplets into smaller sizes. These findings show that the filter bed must be operated at low throughput if the influent has high oil concentration.
Oil droplet size distribution
There are several approaches for measuring the size and distribution of oil droplets, including optical counting methods, laser diffraction methods, and dynamic light scattering methods. Among them, the dynamic light scattering is mostly used to determine the size distribution of oil droplets in oily wastewater.
157
A typical droplet size distribution in oily wastewater measured using dynamic light scattering is shown in Figure 9. The D50 droplet diameter (discussed elsewhere) in the oily wastewater in this case is 10 μm. Droplet size distribution in oily wastewater.
Industrial effluent streams often have droplet of sizes <10 μm with narrow size distribution.143,158 The stirring time plays an important role in obtaining the required size and distribution of the oil droplets than the stirring rate.26,99,159,160 If the stirring time is low, that is, <2 min, it leads to the larger size of bimodal distribution, and if it is >10 min, it leads to the narrow distribution of droplet size ranging between 1 and 10 μm in diameter. As the oil droplet size reduces, the probability of it getting captured by the fibers reduces; hence, a reduction in interception efficiency and oil separation efficiency is expected. Smaller droplets have high collision frequency and are rigid. 161 Therefore, they don’t spread quickly on the fibers reducing the separation efficiency. 158 Oil droplets smaller than 10 μm are almost spherical in shape and make point contact with fiber rather than on multiple fibers. The oil droplets larger than 10 μm deform easily under hydrodynamic forces to allowing high contact areas with several fibers.
Smaller oil droplets are subjected to high buoyancy forces due to low settling or rising or terminal velocity. They take more time to reach the fiber surfaces. In-depth filtration processes, a highly effective coalescing medium facilitates the formation of larger droplets. The larger droplets have less buoyancy, reach the fiber surfaces quickly, and have more time to grow.
35
The rising velocity (
The resident times for larger oil droplets in oil separation are longer as they reach the filter surface much earlier than the smaller droplets do. This favors high oil separation efficiency if the dispersed droplets are larger in the influent. It can be concluded that the larger droplets favor high oil separation efficiency.
Viscosity and pH
Oil having high viscosity has lower hydraulic conductivity due to the formation of thick film layers on the walls of the flow channels.102,163–166 This implies that filtration must be performed at low flow rates while separating the high viscous oil from emulsions.
Varghese et al. reported 39 that with the increase in pH, oil droplets become negatively charged. But generally, textile fibers used are negatively charged in nature. Hence the adhesion of oil droplets goes down as the pH increases. Deshiikan et al. 167 reported that pH is more important for controlling oil coalescence droplets. They also concluded that coalescence time increased with the pH of emulsion.
Process parameters
Coalescer configurations
In an industrial setup, both horizontal and vertical coalescers are used to treat oily emulsions. It is beyond the scope of this article to review all the setups as many are complicated in design. However, these are discussed briefly. The coalescer configuration is based on the flow orientation employed during filtration. They are (a) horizontal, (b) vertical downward, and (c) vertical upward, which are shown in Figures 10, 11, and 12, respectively. The mostly used coalescer configuration in the industry is horizontal followed by vertical upward. In down-flow mode, influent is injected into the filter medium from the top. In up-flow mode, influent is injected into the filter through the bottom and the oil film formed within the structure breaks and moves up to the surface of the water column. In up-flow mode, air entrapment in the filter column is lesser compared to down-flow mode. In both the up-flow and down-flow modes, more filter medium could come in contact with the oil droplets, unlike horizontal mode.
102
Horizontal flow mode. Adapted from Ref. 168. Vertical downward flow mode. Adapted from Ref. 168. Vertical upward flow mode. Adapted from Ref. 168.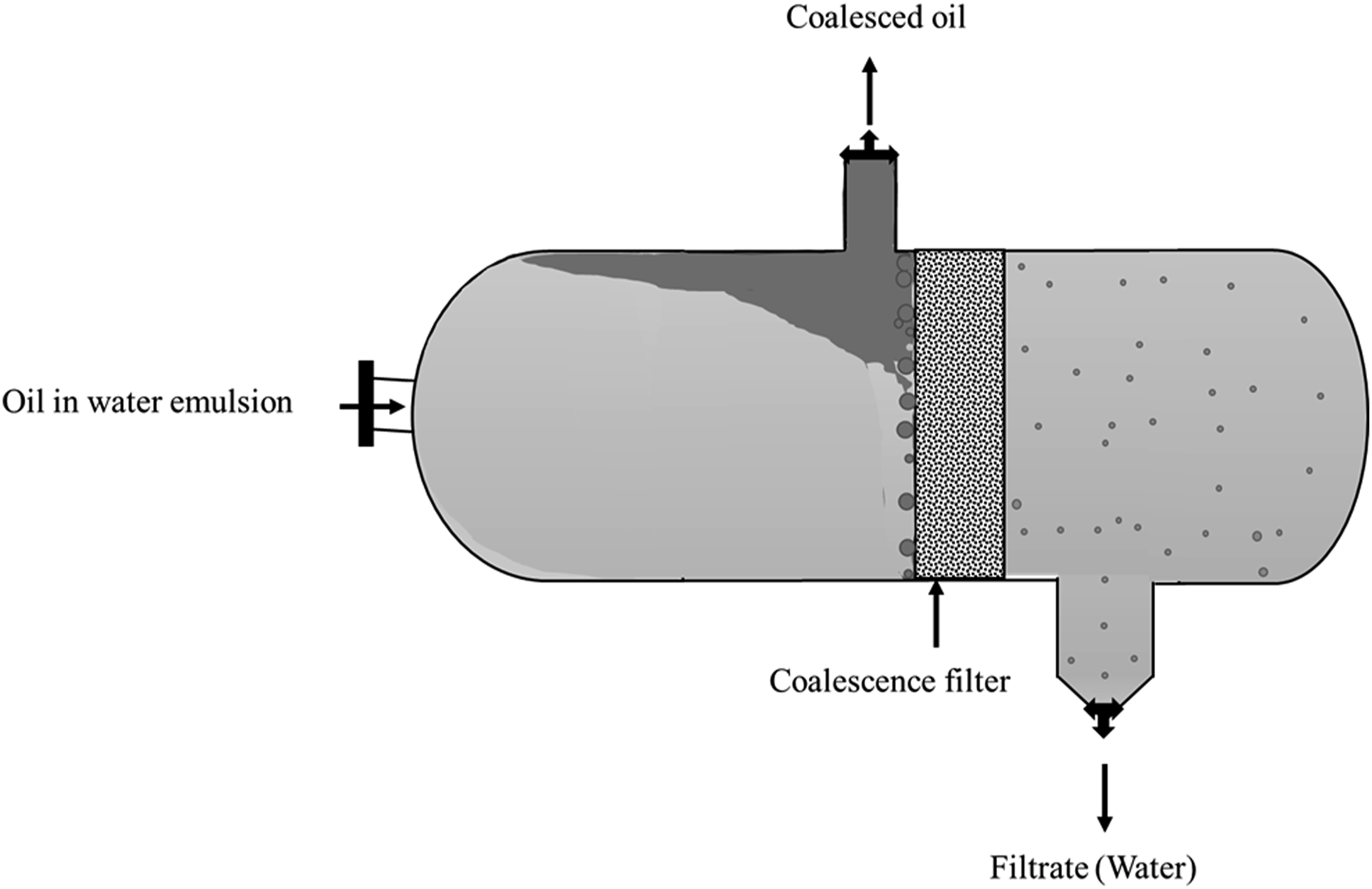


The flow path of the emulsion and the structure of the fibrous bed coalescer have a significant effect on coalescence performance. Sokolovic et al. 102 investigated coalescing geometry on steady-state bed coalescence. They demonstrated that all the vertical fluid flow modes had poor separation efficiency. Coalescer with vertical flow modes has extremely low working velocity. Horizontal flow coalescer is more efficient at high fluid velocities than vertical ones at cruising velocities, though there is no noticeable difference. The up-flow process is the least effective at high fluid rates. 141 Radmila et al. 151 investigated the critical fluid velocities in PU filter beds having different porosity under different flow modes. The critical fluid velocities are higher for horizontal flow mode followed by down-flow and up-flow, and maximum flow rates that can be employed would follow in that order.
Flow rate
Flow rate104,156 is a crucial factor in coalescence filtration. At a lower flow rate, the longer residence time is available for the oil within the filter and fluid encounters lesser hydrodynamic forces. This facilitates droplet growth on the fibers and improved coalescence. With the increase in flow rate, the separation efficiency reduces due to a reduction in droplet interception efficiency by the filter. 1 But according to Angel et al., 17 with the increase in flow rate, the pressure drop increases and the filter bed gets compressed, reducing the pore size, which increases the separation efficiency. However, it should be noted that high flow rates coupled with low permeability of filter bed would increase the flow velocities above the critical velocity. The oil droplets, before coalescing to sufficient size, are broken up into small drops by the hydrodynamic forces, thus resulting in reduced droplets in the effluent and low separation efficiency.
Pressure drop
A high flow rate employed using a closely packed bed eventually leads to a high-pressure drop across the filter. The flow velocity in the pores would be high that can release the oil droplets before being sufficiently coalesced. This reduces the oil separation efficiency. The pressure drop increases initially and reaches a steady state in a highly efficient filter. 169 In the case of membrane filter, the fouling of the filter over time increases the pressure drop. The high-pressure drop is a deterrent to increasing the throughput of the process and hence the economy of the process of oil separation itself.
Material characterization
The selection of fibers is the most important for designing an efficient coalescence filter. The fibers must be hydrophobic and oleophobic. The surface wettability of fibers can be evaluated by measuring the contact angles with water and oil. Goniometer and Tensiometer could be used to measure the contact angles but are time consuming and often not very accurate. A simple method to evaluate the surface wettability fibers is to pack them inside a perforated tube with defined porosity and the measuring the oil absorption capacity. The tube can be dipped in oil bath for few minutes, taken out and allow the loosely held oil on the tube surface to drain out. The ratio of the weight of oil to dry fiber weight gives the absorption capacity. Fibers having higher oil absorption capacity would certainly be an effective material as a filter medium. Research findings on kapok fibers indicate that they have high oil absorption capacity and also give high oil removal efficiency when used as a coalescer filter bed. According to Wang and Song, 135 materials having high oil absorption capacity should be the ideal coalescence materials.
Performance evaluation of coalescence filtration
Separation efficiency
Separation efficiency (
Various techniques can estimate the oil present in the effluent and filter.
Analysis of droplets redispersion
A high oil separation efficiency indicates high filtration performance. The droplets that are coalesced to sufficiently large sizes can attain separation by settling in the filtrate. These droplets are the only ones accounted for the separation efficiency. However, many redispersed droplets, which are small, emerge out of the filter bed and get carried through the settling section by the continuous phase before they could settle out.
26
They are (1) droplets coalesced on the fiber surface that is not sufficiently large enough to attain separation (Figure 6(b)-(b)); (2) very small droplets
D50 droplet size ratio
The droplet size ratio (
Smaller the ratio, especially <1, signifies better filter performance. This indicates that only smaller droplets in the influent are uncaptured and escape through the filter. A low droplet ratio in the effluent signifies greater phase separation efficiency, and a greater ratio indicates poor filtration performance.
Permeability of filter bed and critical fluid velocity
Permeability of fibrous filter medium is also an important parameter for assessing the ability of the filter medium in oil/water separation and filtration.151,171 It varies with the packing density or porosity of the filter. According to Darcy’s law, intrinsic permeability (
The superficial/working/fluid velocity or the hydraulic conductivity is the ratio of flow rate and filter bed cross-sectional area (
Conclusions
Fibrous coalescence technology using a deep bed filter is simple in operation, cheaper, and gives excellent performance in removing oil-in-water emulsion if proper materials are selected in constructing the filter bed. The surface wettability of fibers is the most important aspect of coalescence bed filtration, and it can be modified by incorporating surface roughness. The presence of nanoscale roughness on low surface energy fibers promotes oil wetting and filtration performance. Fibers having hydrophobic-oleophilic characteristics are highly suitable for deep bed filtration. Low surface energy materials are preferred if the filter bed has a larger pore size compared to the mean droplet size in the influent. The surface energy of fibers is not important if the pore size of the filter is smaller than the influent droplet size. The porosity, pore size, and fiber bed height affect its permeability, determining critical fluid velocity and oil separation efficiency. These factors must be considered while designing a coalescer bed. The filtration performance with smaller droplets is low because they are difficult to be intercepted and have a low contact area on fiber surfaces. Oil separation efficiency is independent of the influent oil concentration at low throughput. In contrast, at high throughput, high oil concentration in the influent reduces the oil separation efficiency due to saturation in the pores and if the fiber bed’s hydraulic conductivity is low.
Further research is required on kapok, milkweed, and poplar fibers as deep bed filer materials as they are oleophilic-hydrophobic. Efficient filter beds with natural hollow fibers can be constructed with high porosity that saves the amount of fibers and also high throughputs can be achieved during filtration favoring further cost reduction. Future research must focus on developing and investigating deep bed filters using biodegradable fibers as the filter material which can be used as biofuels after usage. Simple, cost-effective approaches are also required to chemically modify the natural fibers to enhance their potential in filtering oil from oily wastewater.
It is important to design fibers with hydrophobic properties for fibrous coalescer beds and oleophilic characteristics. The throughput of the filtration must be selected based on the porosity of the filter, oil concentration in the emulsion, and bed height.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

