Abstract
Magnetic polymer microspheres have attracted a great deal of attention due to their magnetic properties and good biocompatibility. In this study, novel magnetic cross-linked poly(divinylbenzene-co-acrylic acid) microspheres were successfully prepared via improved emulsion polymerization. The microstructures of nanocomposites were characterized using scanning electron microscopy, transmission electron microscope, energy-dispersive X-ray spectrometry, X-ray diffraction, and saturation magnetization measurements. It was noteworthy that the nanocomposites displayed good water dispersibility, a rapid absorption rate, a high absorption capacity, and high magnetic separability, results that are promising in terms of these nanocomposites’ future application in biomedical fields.

Introduction
In recent years, magnetic polymer microspheres have attracted a great deal of attention due to their magnetic properties and good biocompatibility.1–3 On the one hand, magnetic materials can be easily guided or separated under an external magnetic field.4,5 On the other hand, magnetic polymer microspheres possess many of the characteristics of polymers, such as polymerization and surface modification.6,7 Thus, magnetic polymer microspheres are widely used in the fields of biotechnology, isolation, catalysis, and medicine in applications such as immobilization of enzymes, immune analysis, cell separation, and magnetic hyperthermia treatment of tumors.8–11 With the development of life science, fast and effective isolations of protein as well as targeting drug delivery system based on magnetic polymer microspheres are more and more important.12–16 The magnetic polymer microspheres synthesized by traditional polymerization have many problems such as poor dispersity, low biocompatibility, and low separability.17,18 Thus, the novel magnetic hypercrosslinked polymer microspheres via improved emulsion polymerization have become the research focus by virtue of their surface, synthesis conditions, and source of monomer.19,20
In this work, a type of novel core–shell structured magnetic hypercrosslinked polymer microsphere with good dispersity was designed and prepared. First, the functionalized magnetic nanosilica were fabricated and modified by 3-(methacryloxypropyl) trimethoxy silane (KH570). Then, the magnetic cross-linked poly(divinylbenzene-co-acrylic acid) (poly(DVB-co-AA)) microspheres were obtained by improved emulsion polymerization. The magnetic polymer microspheres were characterized by scanning electron microscopy (SEM), transmission electron microscope (TEM), energy-dispersive X-ray spectrometry (EDS), X-ray diffraction (XRD), and saturation magnetization measurements. The applicability of the novel core–shell magnetic polymer microspheres was evaluated based on the water dispersibility and magnetic separation properties.
Experimental section
Materials
The Fe3O4 nanoparticles were obtained according to the co-precipitation method. 21 Tetraethoxysilane (TEOS) was obtained from 9 Ding Chemistry (China); 3-(methacryloxypropyl) trimethoxy silane (KH570) was purchased from Nanjing Xuanhao New Material Technology Co. Ltd. Divinylbenzene and acrylic acid were purchased from Shanghai Aladdin Biochemical Technology Co. Ltd.
Preparation of magnetic silica nanocomposites
In a typical procedure, magnetic Fe3O4 nanoparticles, methyl alcohol, and aqueous ammonia solution were placed in a flask. Subsequently, TEOS was added dropwise; then, the mixture was stirred at room temperature and kept for 30 min. The magnetic silica nanocomposites were obtained after the mixture was washed with deionized water.
Preparation of functionalized magnetic silica nanocomposites
In a typical procedure, the magnetic silica nanocomposites were dispersed in deionized water. After sonication, a well-dispersed solution was formed. After pH was adjusted to 3 with ethanedioic acid, 3-(methacryloxypropyl)trimethoxy silane (KH570) was added dropwise; then, the mixture was stirred at 80 °C and kept for 2 h. After the reaction was completed, the precipitate was washed with ethanol and deionized water, respectively. The functionalized magnetic silica nanocomposites were stored as an aqueous solution.
Preparation of magnetic cross-linked poly(DVB-co-AA) microspheres
The magnetic cross-linked poly(DVB-co-AA) microspheres were obtained via improved emulsion polymerization. In a typical procedure, 5.5 mL of divinylbenzene, 3 mL of acrylic acid, 50 mL of deionized water, 0.5 mg of sodium dodecyl benzene sulfonate, and functionalized magnetic silica nanocomposites were placed in a beaker. After being sonicated for 20 min, a milk-like emulsion was obtained. An amount of 50 mL of deionized water and 0.1 g of potassium persulfate (K2S2O8) were placed into a flask and flushed with nitrogen. Then, the above emulsion was added dropwise into the solution and polymerized at 80 °C for 10 h with constant stirring. After it cooled to room temperature, the magnetic cross-linked poly(DVB-co-AA) microspheres were obtained. These were subsequently washed with ethanol and deionized water, respectively.
Adsorption test
The magnetic cross-linked poly(DVB-co-AA) nanocomposites as adsorbent for protein were investigated. The magnetic poly(DVB-co-AA) microspheres (10 mg) were suspended in 10 mL of bovine serum albumin (BSA) aqueous solution (1.0 mg mL−1). The pH of solution was adjusted to 4.8 with citric acid. The sample was vibrated on a rocking table at room temperature for 0.5, 1, 3, 5, and 7 h. Subsequently, the adsorbent was separated by an external magnetic field. The contents of BSA in suspension were detected at 278 nm using an UV−vis spectrophotometer.
Characterization
The morphologies of magnetic nanosilica particles and magnetic cross-linked poly(DVB-co-AA) nanocomposites were characterized by scanning electron microscopy (Sirion 200) and transmission electron microscope (JEOL 2010). The X-ray diffraction measurements were carried out on a D/Max-rB diffractometer, and samples were measured in air with a scan range of 5°–70°. The photographic images of the magnetic cross-linked poly(DVB-co-AA) nanocomposites were obtained using a digital camera.
Results and discussion
Morphology analysis
Figure 1 shows the morphologies of magnetic nanosilica particles and magnetic cross-linked poly(DVB-co-AA) nanocomposites characterized by SEM. In this study, we synthesized a new core–shell structured magnetic polymer microsphere with binding sites on the surface. First, the magnetic nanosilica were fabricated and functionalized by introducing vinyl bonds. This was followed by copolymerization of the functional monomers to prepare magnetic cross-linked poly(DVB-co-AA) nanocomposites by improved emulsion polymerization. As shown in Figure 1(a), the magnetic nanosilica particles presented spherical morphology. Compared to the image in Figure 1(a), the core–shell structured magnetic cross-linked poly(DVB-co-AA) microspheres were observed in the SEM images with different magnifications (Figure 1(b)–(d)). The magnetic nanosilica particles were densely packed on the surface of the poly(DVB-co-AA) microspheres, which can also be clearly observed.

SEM images of (a) magnetic nanosilica particles and (b–d) the magnetic cross-linked poly(DVB-co-AA) nanocomposites at different magnifications.
To investigate the morphologies of magnetic nanosilica particles and magnetic cross-linked poly(DVB-co-AA) nanocomposites, the transmission electron microscope has also been used (Figure 2). As shown in Figure 2(a), the spherical Fe3O4@SiO2 nanoparticles (with an average diameter of ~10 nm) were well observed. The core–shell structured magnetic polymer microspheres with poly(DVB-co-AA) as cores and Fe3O4@SiO2 as shell were further confirmed in Figure 2(b), which were consistent with the results of SEM.

TEM images of (a) magnetic nanosilica particles and (b) the magnetic cross-linked poly(DVB-co-AA) nanocomposites.
Compared to the general structure of magnetic polymer microspheres with magnetic nanoparticles as cores and polymer as shell reported in the literature, the novel core–shell structured magnetic polymer microspheres with poly(DVB-co-AA) as cores and Fe3O4@SiO2 as shell were successfully prepared via improved emulsion polymerization in this paper. The core–shell structured magnetic polymer microspheres were further confirmed using EDS (Figure 3). In the EDS spectrum, the presence of Si elements, Fe elements, O elements, and C elements was evidence of thin magnetic nanosilica shell formation on the surface of poly(DVB-co-AA) microspheres, which further demonstrated the chemical composition of the nanocomposites.

EDS element analysis of the magnetic cross-linked poly(DVB-co-AA) nanocomposites.
XRD result analysis
The crystallinity of the prepared nanocomposites was identified using XRD. The XRD patterns of magnetic Fe3O4 nanoparticles and of the magnetic cross-linked poly(DVB-co-AA) nanocomposites are depicted in Figure 4. As shown in Figure 4(a), the peaks at 30.1°, 35.6°, 43.3°, 57.2°, and 62.7° are characteristics of Fe3O4. 22 The broad peaks at 20°–30° are amorphous SiO2 and polymer diffraction peaks in Figure 4(b). These peaks also suggest that the crystal structure of the ferrite was unchanged by the polymerization process.

XRD diffraction patterns of (a) magnetic Fe3O4 nanoparticles and (b) the magnetic cross-linked poly(DVB-co-AA) nanocomposites.
Dispersibility of the magnetic cross-linked poly(DVB-co-AA) nanocomposites
The water dispersibility of the magnetic cross-linked poly(DVB-co-AA) nanocomposites was investigated. Figure 5(a) shows that the nanocomposites could be dispersed in water very well. With the aid of carboxyl groups, the magnetic cross-linked poly(DVB-co-AA) nanocomposites displayed good water dispersibility. The loading of carboxyl groups on the nanocomposites was found to be 1.2 mmol g−1 by acid–base titrations. Figure 5(b) shows the magnetic separation process of the magnetic cross-linked poly(DVB-co-AA) nanocomposites from aqueous solution using a permanent magnet. The result indicated that the nanocomposites can be rapidly separated from aqueous solution within a few minutes.

Photographic images of (a) the magnetic cross-linked poly(DVB-co-AA) nanocomposites dispersed in water and (b) magnetic separation using permanent magnet.
Saturation magnetization analysis
The saturation magnetization of the magnetic materials was measured by a MPMS3 SQUID-VSM magnetometer (Quantum Design, America). Figure 6 presents the hysteresis curves of magnetic nanosilica and magnetic cross-linked poly(DVB-co-AA) nanocomposites. As shown in the figure, the saturation magnetization (Ms = 0.37 emu g−1) of magnetic nanosilica is significantly higher than that of the magnetic poly(DVB-co-AA) nanocomposites (Ms = 0.029 emu g−1). This indicates that the saturation magnetization decreases with the increase in non-magnetic materials, which results in a weakening in magnetism.23,24 In fact, the magnetic cross-linked poly(DVB-co-AA) nanocomposites exhibit extremely low coercivity (Hc = 31 Oe), which could be observed by further amplifying the hysteresis loop (inset in Figure 6). However, the magnetic cross-linked poly(DVB-co-AA) nanocomposites can be readily separated from the aqueous solution by an external magnetic field, which meets the requirement of the experiments.

Hysteresis lines of (a) magnetic nanosilica and (b) the magnetic cross-linked poly(DVB-co-AA) nanocomposites.
Adsorption properties of the magnetic cross-linked poly(DVB-co-AA) microspheres
Use of the magnetic cross-linked poly(DVB-co-AA) nanocomposites as an efficient adsorbent for protein was investigated. BSA was chosen as a model protein. Figure 7 shows the curve of the BSA’s adsorption of polymer microspheres. It can be seen from the diagram that 98% of the BSA could be adsorbed by the magnetic poly(DVB-co-AA) microspheres within 5 h at room temperature. The adsorption capacity of the magnetic cross-linked poly(DVB-co-AA) microspheres to BSA is 0.98 mg mg−1, which is significantly greater than that reported in the literature.25–28
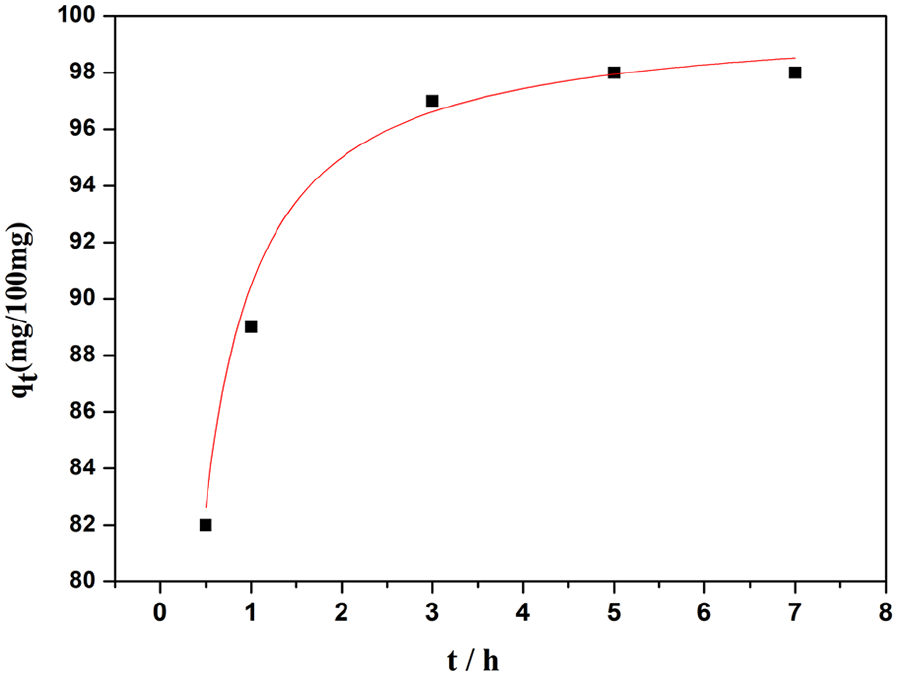
Curve of BSA adsorption on magnetic cross-linked poly(DVB-co-AA) nanocomposites.
Conclusion
In summary, magnetic cross-linked poly(DVB-co-AA) microspheres were successfully prepared via improved emulsion polymerization. The core–shell structured magnetic polymer microspheres with poly(DVB-co-AA) as cores and Fe3O4@SiO2 as shell were confirmed by TEM and SEM. The chemical composition and crystal structure of the nanocomposites were also identified separately by EDS and XRD. The magnetic property measurement system (MPMS) results showed that the saturated magnetization of the magnetic polymer nanocomposites was 0.029 emu g−1. Furthermore, it was noteworthy that 98% of the BSA could be adsorbed by the magnetic poly(DVB-co-AA) microspheres within 5 h at room temperature. The magnetic cross-linked poly(DVB-co-AA) nanocomposites displayed good water dispersibility, rapid absorption rate, high absorption capacity, and high magnetic separability, which demonstrate their potential application in biomedical fields.
Footnotes
Author contributions
Liyuan Zhang: conceptualization, methodology, writing, project administration, and funding acquisition; Xuewen Bai: investigation; Kai Zou: investigation, provision of access to various apparatuses and instruments; Linlin Zhu: writing, and funding acquisition.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Foundation of Anhui Province’s Higher Education of China (KJ2021ZD0140, KJ2019A0847) and the University Synergy Innovation Program of Anhui Program of Anhui Province (GXXT-2023-096).
