Abstract
Treating wound is challenging if there is microbial colonization. In this paper, we tried to review comparison of bacterial inhibition zone of different herbal plant bioactive loaded polymer electrospun nanofibrous wound dressings. A systematic literature search called preferred reporting items for systematic reviews and meta-analyses (PRISMA) was used to review bacterial inhibition zone of the different herbal plant bioactive loaded polymer electrospun nanofibrous mats used for wound dressing applications. The literatures were searched from PubMed, Scopus, Web of Science, and Google Scholar databases published in English from 2010 to 2021. Two hundred articles were searched out; from these, 93 articles are selected and studied their bacterial inhibition zone, among them eight have the highest bacterial inhibition zone greater than or equal to 20 mm. From the studied plant bioactive extract loaded polymer electrospun nanofibrous mat wound dressings, PVA/Tridax procumbensviz have the highest antibacterial property (42 mm and 35 mm bacterial inhibition zone to Staphylococcus aureus and Escherichia coli, respectively). The second highest antibacterial property is PVA/honey/Curcuma longa with bacterial inhibition zone of 38 mm to S. aureus and the third one is PCL/carboxyethyl chitosan/PVA/Matricaria chamomile L. with an inhibition zone of 37.5 mm to E. coli bacteria.
Keywords
Introduction
There are decisive requirements of wound dressings such as biocompatibility, porosity, nontoxicity, permeability, preventing infection from exogenous or bacterial and the likes.1,2 Wound dressings are based on textile constructions in the form of fibers, yarns, nonwoven, woven, composites and electrospun nanofibrous materials. Textile wound dressings can be traditional, modern, bioactive, tissue engineered skin substitutes, and medicated wound dressings.1,3 Amongst the above constructions, electrospun nanofibrous mats are new, advanced and alternative potential wound management materials due to numerous unique advantageous properties over others such as applicable for drug delivery materials, high porosity, excellent pore interconnectivity and large surface to area, its morphological similarity to extracellular matrices (ECM), permeability to oxygen. Nanofibrous are materials that mimics ECM with diameters in the range of nanometers produced from different polymers that can provide the natural environment for tissue formation, promote cell addition, and proliferation for wound due to their high surface to volume ratio. 4
Infection of wound is the main problem that delays the wound healing process. Wound can be infected by different pathogens such as Escherichia coli (E. coli), Salmonella typhi (S. typhi), and Staphylococcus aureus (S. aureus), among these pathogens, E. coli and S. aureus are identified as pathogens leading to severe wound infection.5–7 In wound healing process, infection prevention may be achieved by adding various antibacterial agents into dressing mats. 8 To avoid the wound infections, researchers are producing electrospun nanofibers functionalize wound dressings with antimicrobial agents such as nanoparticles and antibiotics. 1
Nanofibrous mats are novel materials as wound dressings and nowadays they are used as drug delivery apart from their biodegradability, biocompatibility, low toxicity, high porosity, excellent pore interconnectivity and large surface area. 9 Electrospinning is one technique of producing nanofibers which is simple, straightforward, cost-effective, ability to produce fibers with the high surface-area, porosity, flexibility in material selection and ability to incorporate drugs which makes attractive candidate for medical applications. There are different methods of incorporating the different synthetic nanoparticles, drugs, antibacterial agents into electrospun nanofibers such as blend electrospinning, emulsion electrospinning, co-axial electrospinning and surface immobilization. 10 Blend electrospinning is simple, one-step process and the most predominant method of incorporating bioactive molecules into nanofibers. 11
Synthetic nanoparticles such as zinc oxide (ZnO), and sliver nanoparticles have been approved as therapeutic effects since they have antibacterial effects, thus can be incorporated in to nanofibrous wound dressings. By nature, higher concentrations of ZnO nanoparticles are toxic, however, incorporating less amount of ZnO nanoparticles (0.5–1% ZnO concentrations) into different polymers can produce less toxic nanofibrous scaffolds such as sodium alginate/PVA/ZnO nanoparticles, 12 chitosan/PVA/ZnO nanoparticles. 13 Zinc oxide nanoparticles can also be combined with synthetic antibiotics to give better wound dressing properties such as PVA/carboxymethyl cellulose/ZnO nanoparticles/erythromycin (drug). 14 Silver nanoparticles have inhibitory and bactericidal effects by releasing silver ions continuously to provide protection against bacteria. Silver nanoparticles were incorporated into PVA/β-cyclodextrin, 15 PVA/carbon nanotubes, 16 PLA, 17 and PAN/polyurethane. 18
Incorporation of antibiotics drugs into nanofibrous is the best way of transferring drugs in to the skin/wound through nanofibrous mats. Different researchers tried to incorporate a number of antibiotics into different biopolymers to produce active nanofibrous wound dressings by electrospinning to prevent wound infections like chitosan/alginate/gentamicin, 19 silica/PCL/gentamicin, 20 chitosan/PVA/Tetracycline hydrochloride, 21 chitosan/PVA/cefadroxil monohydrate, 22 chitosan/PEO/Teicoplanin, 23 modified hydrophilic sodium alginate micro particles/poly (lactic-co-glycolic acid)/ciprofloxacin, 24 sodium alginate/PVA/Moxifloxacin, ALG/polyethylene oxide (PEO)/Vancomycin, 25 sericin/PVA/Tigecycline, 26 polylactide/Doxycyclines, 27 polyvinylidene fluoride/enrofloxacin, 28 PCL/gelatin/ZnO/amoxicillin, amoxicillin, 29 and manufactured PCL/gelatin/ciprofloxacin hydrochloride. 30
However, the use or incorporation of these nanoparticles and synthetic antibiotics has a negative effect on the environment, human health, and/or bacteria resistance issues have prompted the natural antimicrobial compounds to be used. 1 Also, synthetic drugs and antibiotics are costly but developing countries are finding cost alternative therapies for wound treatment. 31 Due to these, natural antimicrobial compounds extracted from plants to nanofibrous mats are becoming the new research area with the advantage that these natural compounds have inherent medicinal properties, non-toxic or fewer side effects, environmentally sustainable, easily available, and less cost. 1
According to World Health Organization, medicinal plants would be the best source of different drugs. 80% of people in the developed countries use traditional medicines from plants. 32 There are different plats used in the treatment of wound in the form of ornaments, pure extractions, hydrogels, etc., but their physical form have disadvantages such as restrain migration of cell, and limit cell-to-cell commutation that lead to a slow healing process and increased risk of wound infection, 33 delicate shape and dry up quickly upon exposure to air. 34 In order to avoid such complications, electrospinning is one of the approaches applied to combine the advantages of utilizing plant extracts in the form of suitable products to serve as wound dressing. 33
Therefore, incorporating natural wound remedial plant extracts into electrospun nanofibrous mats showed good potential to be used in various wound applications,7,9 providing higher performance and synergistic effect for wound healing. 33 There are phytochemicals (bioactive compounds) in the traditional plants such as steroids, sugars, triterpenoids, alkaloids, reducing sugars, tannins, flavonoids, sesquiterpene lactones, and phenolic which are responsible for antibacterial properties. 9
Zhang, W., S. Ronca, and E. Mele 35 investigated antibacterial activity of selected plant components contained in electrospun fibers and their applications including in wound healing. Sukhwinder K. Bhullar and H.S. Buttar 31 reviewed about the methods of electrospinning used to fabricate nanofibers as delivery of plant-derived agents for wound healing. Sofi, H.S. et al. (2020), studied recent advancement of plant extract incorporated electrospun nanofibrous by electrospinning for tissue engineering, drug delivery and wound healing applications, focusing on fabrication methods, preparations of plant and extract methods for electrospinning. 36 Rather, A.H. et al. (2021) also studied the encapsulation of various therapeutic essential oils in polymeric nanofibers to develop functional membranes for biomedical and food-packaging applications. 37 Das, U. et al. 34 have reviewed the importance of medicinal plant bioactive molecules and bioactive molecules loaded polymeric constructs for wound healing. These constructs are plant extract/polymeric hydrogel, plant extract/polymeric membrane, plant extract/polymeric porous, plant extract/polymeric nanofibrous, plant extract/polymeric nanocomposite constructs. The constructs advantages and disadvantages are also discussed. Pilehvar-Soltanahmadi, Y. et al. 38 have also reviewed natural derived substances (plant and animal origin) loaded electrospun nanofibrous potential wound dressings.
A method to investigate the antibacterial property of materials in the pre-diagnostic test at laboratory level is good by using the zone of inhibition test method. This is specially very important for the final output to see whether a particular antibiotic is effective against particular bacteria, the method is very easy, relatively quick to determine the diameter of the zone of inhibition. 39
None of the research articles and review articles did study and compare the medicinal plant extract electrospun nanofibrous mat wound dressings’ antibacterial properties by using any methods of antibacterial evaluation. Therefore, in this research the antibacterial activity of different plant extract electrospun nanofibrous mats by inhibition zone is discussed, and also compared. The antibacterial mechanism of plant phytochemical coumpunds is also discussed.
Methodology
Figure 1 shows the number of original research articles of electrospun nanofibers that contains medicinal plant extracts. This review research was conducted in accordance with preferred reporting items for systematic reviews and meta-analyses (PRISMA),
40
as shown in Figure 2. PRISMA system contains general guidelines to prepare the review protocol. Number of electrospun nanofibrous wound dressing published articles containing medicinal plant extracts in the Web of Science, PubMed, Scopus, and Google Scholar database from the year 2010 till 2021. Study flow chart.

The official Web pages of the databases were used to extract information on the range of journals covered, search facilities and restrictions, and update frequency. Web of Science and Scopus are two world-leading and competing citation databases increasingly used in academic papers. 41 Based on the interdisciplinary coverage on journals, and data quality, the use of these two for research evaluation may introduce biases that favor Natural Sciences and Engineering as well as biomedical research. 42 Harzing, A.-W. and S. Alakangas (2016) also showed that the three data bases: Web of Science, Scopus and Google Scholar are consistent and stable in coverage for both citations and publications for multidiscipline including Engineering and science. 43 Another database resource used by researcher looking literature in biological science is PubMed, which is the largest number one and most well-organized, 44 and accessed for free. 45
Based on the above explanations, this paper is reviewed systematically by using databases of Web of Science, PubMed, Scopus, and Google Scholar to explore research articles. The key words used to explore the publications/articles from these data basses are medicinal plants, electrospun nanofibrous, wound dressings and bacterial inhibition zone.
In the systematic review, we searched out articles that contained or described medicinal plant loaded electrospun nanofibrous used for wound dressings published in English within the predefined period of time (2010–2021). Ninety-eight articles were identified for preliminary review from Web of Science, PubMed, and Scopus, and Google Scholar data searches. Relevant articles that constitute bacterial inhibition zone information quantitatively and qualitatively are screened and selected for study; a total of 93 research articles underwent a full-text review which have met the inclusion criteria. The excluding criteria are as follows: the first criteria is those articles that do not contains inhibition zone information qualitatively and quantitatively in their study are excluded because this review is about antibacterial property of medicinal plant extract loaded electrospun nanofibrous wound dressings by using inhibition zone method, the second criteria is those articles that could not be accessed their full text are excluded because authors cannot get any information about their antibacterial property by inhibition zone method. Figure 2 shows the flow chart illustrating selection of articles and number of articles included in the study.
There are several antimicrobial property screening and evaluating methods: both qualitative and quantitative methods. Disk diffusion and broth or agar dilution are well known and commonly used methods. For any method, when the antibacterial diffuses, it creates a radial around the disk, the susceptibility of the bacteria to the antibacterial, is called inhibition zone (no growth) around the disk. The diameter of inhibition zone can be measured in millimeter by a ruler, sliding caliper, template, or electronic instrument and image analysis.46–48 Agar diffusion is one of qualitative methods which is simple, fast, and advantageous for large number of samples test. 48 Disk diffusion method is one of the quantitative methods of antibacterial property test. In disk diffusion method, disk is impregnated with a defined concentration of an antibacterial solution; the disk is then placed in a reservoir in the agar. 47
There are different standard methods to measure antibacterial activity on textile, the most widely used are qualitative standards: AATCC147:2004, ISO20645:2004 and JISL1902:2008 Halo method and quantitative methods: AATCC100:2004, ISO20743:2007–Absorption method and JISL1902:2008–Absorption methods. 48
Discussions
Inhibition zone of medicinal plant extract loaded electrospun nanofibrous wound dressings
Some plant extract bioactive have wound healing properties and they have been used for many years; currently, there are many herbal plants extensively used as traditional wound treatment in different parts of the world. 33 These plants have different bioactive chemical compounds that can act with synergistic effects. The active compounds found in plants have lower risk of toxicity than synthetic molecules, environmental friendly and fewer side effects. 49
Bacterial inhibition zone for different medicinal plant–based electrospun wound dressings.
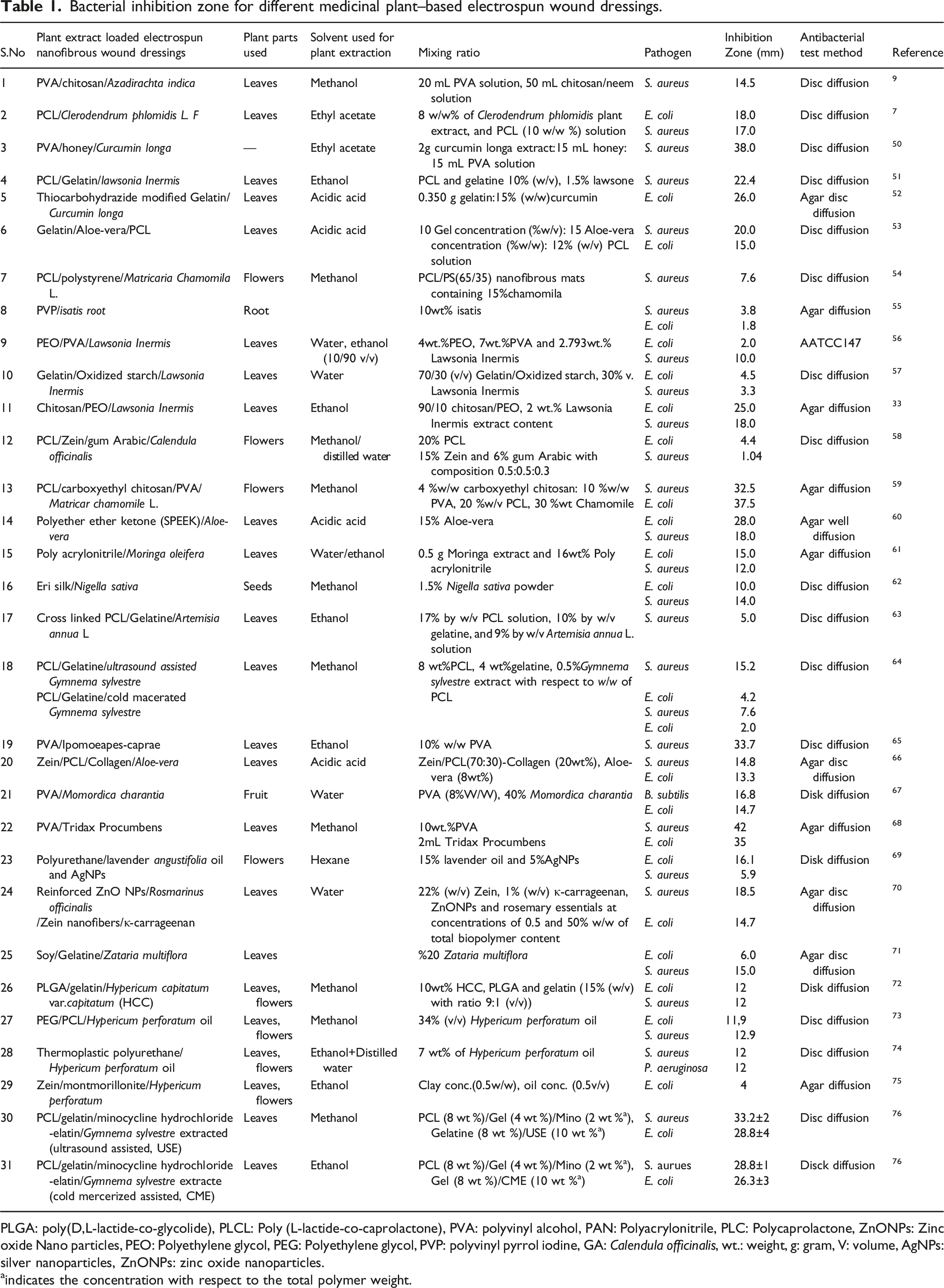
PLGA: poly(D,L-lactide-co-glycolide), PLCL: Poly (L-lactide-co-caprolactone), PVA: polyvinyl alcohol, PAN: Polyacrylonitrile, PLC: Polycaprolactone, ZnONPs: Zinc oxide Nano particles, PEO: Polyethylene glycol, PEG: Polyethylene glycol, PVP: polyvinyl pyrrol iodine, GA: Calendula officinalis, wt.: weight, g: gram, V: volume, AgNPs: silver nanoparticles, ZnONPs: zinc oxide nanoparticles.
aindicates the concentration with respect to the total polymer weight.
From the listed mats in Table 1, PVA/Tridax procumbensviz electrospun nanofibrous mat has the highest antibacterial property with inhibition zone of 42 mm to S. aureus and 35 mm to E. coli. A number of chemical constituents were reported from the plant T. procumbensviz such as alkaloids, flavonoids, carotenoids, ß-sitisterol,n-hexane, fumaric acid, luteolin, quercitin, oxoester, lauricacid, myristic, palmitic, arachidic, and linoleicacidandtanninetc. The methanol extract of T. procumbensviz alone has antibacterial property to E. coli and S. aureus of inhibition diameter 12 mm and 24 mm, respectively. 77 Ganesan, P. Pradeepa, P. (2017), incorporated aqueous T. procumbensviz leaves extract into PVA to develop PVA/T. procumbensviz nanofibrous mat and the antibacterial property was investigated against both S. aureus and E. coli bacteria to be 42 mm and 35 mm bacterial inhibition zone, respectively. 68 This indicates that incorporation of herbal bioactive into polymers by electrospinning increases the antibacterial property of the herbal bioactive extract.
PVA/honey/Curcuma longa mat also have the second highest with bacterial inhibition zone of 38 mm to S. aureus. The reason may be due to honey has also explicit antibacterial properties to S. aureus, 78 pure honey alone exhibited a maximum bacterial inhibition zone of 17 mm to S. aureus and 16 mm to E. coli which is considered to be moderate antibacterial activity. 79 Therefore, the combination of two antibacterial materials, that is, Curcuma longa extract and honey will bring the mat to have the highest inhibition zone to S. aureus. Curcuma longa extract was also loaded into Thiocarbohydrazide modified gelatin nanofibers for wound healing applications with inhibition zone of 26 mm to E. coli.
The third highest bacterial inhibition zone is found for PCL/carboxyethyl chitosan/PVA/Matricaria chamomila electrospun nanofibrous mat (37.5 mm to E. coli bacteria). Here, carboxyethyl chitosan also has antibacterial property with inhibition diameter of 25 mm to S. aureus, and 30 mm in case of E. coli. 80 Carboxyethyl chitosan/PVA (1:1) had higher inhibitory activity to E. coli with inhibition diameter of 38 mm. As a result, combining carboxyethyl chitosan/PVA with medicinal plant extract chamomile resulted good antibacterial property for the fibrous mat. The therapeutic activity of chamomile is due to the presence of bio-actives such as phenolic and flavonoids apigenin, quercetin, patuletin, luteolin, and their glucosides. 54 Matricaria Chamomila as antibacterial agent was incorporated into nanofibrous PCL/polystyrene blend mats but found to be low inhibitory zone of 7.6 mm to S. aureus. 54 This shows that the plant extract loaded nanofibers increased the mat inhibition zone when there is another material with antibacterial activity.
In the contrary, Calendula officinalis loaded PCL/Zein/gum Arabic electrospun nanofibrous has the lowest bacterial inhibition zone which is 1.04 mm to S. aureus. Calendula officinalis have major bioactive compounds such as carotenes, triterpenoids, terpenoids, steroids, tocopherols, quinones, phenolic compounds, carbohydrates, essential oils, minerals and fatty acids. 81 PVP/isatis root also has lower bacterial inhibition zone to E. coli with 1.8 mm. This indicates that not only the antibacterial plant extract bioactive but also the polymers used and other bioactive polymers combinations are very crucial to have antibacterial property for the nanofibrous wound dressing.
Aloe-vera has been used for traditional medicine for many years and incorporated into nanofibrous mat as an antibacterial agent. Ghorbani M. Nezhad-Mokhtari P., and R. S. (2020) 66 found that Aloe Vera loaded Zein/PCL/Collagen electrospun nanofibrous mat exhibited 13 mm and 9 mm bacterial inhibition zone to S. aureus and E. coli bacteria, respectively. To increase inhibition zone of Zein/PCL/Collagen/Aloe-vera mat, ZnO (1wt%) was incorporated to nanofibers which increase to 19.23±1.35 mm against S. aureus and 15.38±1.12 mm to E. Coli, due to antibacterial activity of ZnO. Aloe-vera (15%) was also loaded into polyether either ketone resulting an anti-bacterial activity to E. coli and S. aureus with 28 mm and 18 mm zone of inhibition, respectively. 60 Co-electrospun PCL/Gelatin/Aloe-vera nanofibrous mat was developed by Baghersad, S. et al. (2018) 53 with an inhibition zone of 20 mm and 15 mm to S. aureus, and E. coli, respectively.
Azadirachta indica, commonly called neem, a known medicinal plant exhibiting a wide spectrum of antimalarial activities, has different bioactive compounds such as asteroids, sugars, triterpinoids, alkaloids, reducing sugars, tannins, flavonoids, sesquiterpenelactones, and phenolic compounds. 9 It was incorporated into PVA/chitosan enhancing the formation of inhibition zone (14.5 mm) to S. aureus. 9 Clerodendrum phlomidis (with anti-bacterial activity) incorporated PCL nanofibers was investigated its antibacterial property which inhibit 18 mm and 17 mm diameter to E. coli and S. aureus, respectively. 7
Lawsonia inermis leave extract possess pharmacological ingredients and has antibacterial property that is used for wound healing application. 33 The responsible chemical constitutes for biological activities includes Lawsone (2-hydroxynaphthoquinone), mucilage, mannite, Gallic acid and tannic. 82 Even though the mechanism was not known exactly, Abulyazid, I. E.M.E. Mahdy, and R.M. Ahmed (2013) studied the possible mechanism due to presence of free hydroxyls in main chemical components that have the capability to combine with the carbohydrates and proteins in the bacterial cell wall that may have attached to enzyme sites rendering them inactive. 83 They showed the antibacterial activity of Lawsonia inermis leaves extract against E. coli. 83 Adeli-Sardou, M. et al. (2019) incorporated Lawsonia inermis extract into polycaprolactone-gelatin polymers in core-shell architecture by electrospinning and investigated its antibacterial to be found 22.43± 0.072 mm diameter inhibition zone to S. aureus. 51 Antibacterial electrospun PVA/PEO nanofibers Lawsonia Inermis leaf extracts loaded have also 2 mm and 10 mm inhibition zone diameters to E. coli and S. aureus, respectively. 56 Lawsonia Inermis extract also loaded into gelatin-oxidized starch nano-fibrous which has antibacterial property (inhibition zone of 4.45 mm and 3.34 mm to E. coli and S. aureus, respectively). 57 Yousefi, I. et al. (2017) also produced PEO-chitosan loaded with Lawsonia inermis that shows inhibition zone of 25 mm 18 mm to E. coli and S. aureus, respectively. 33
Momordica charantia contains phytochemicals such as proteins, polysaccharides, flavonoids, triterpenes, saponins, ascorbic acid and steroids with many biological activities including antibacterial activity. 84 With this property Mirbehbahani, F.S. et al. (2020), incorporated this plant extract into PVA to develop PVA/M. charantia nanofibrous mat resulting 14.5 mm inhibition zone to E. coli 67
Fayemi, O.E. et al. (2018), investigated that Poly acrylonitrile/Moringa oleifera electrospun nanofibrous wound mat has antibacterial activity due to incorporation of Moringa extract. 61 The ethanoic and ethyl acetate extract of Moringa stenopetala proved to have an inhibitory zone in the range of 25.7 mm–31.7 mm and 17 mm–26.8 mm. 85
Artemisia annua L. has constitutes such as monoterpenes, sesquiterpenes, diterpenes, sterols and triterpenes, aliphatic hydrocarbons, alcohols, aldehydes and acids, aromatic alcohols, ketones and acids, phenylpropanoids, and flavonoids that gives antibacterial effect. Its incorporated into PCL/Gelatine to produce nanofibrous mat PCL/Gelatine/A. annua L. with inhibition zone of 15.2 mm to S. aureus and 4.2 to E. coli. 64
The therapeutic properties of Nigella sativa seed extract is due to the presence of major bioactive component called thymoquinone, 86 and approved as antimicrobial property to E. coli and S. aureus, 87 which have been widely used in the treatment of different diseases and Muthumanickkam, A. et al. (2020) 62 incorporated this seed powder into Eri silk nanofibrous mat by electrospinning. They found that the seed powder loaded nanofibrous mat showed inhibition diameter of 10.0 mm and 14.0 mm to E. coli and S. aureus, respectively.
Gymnema sylvestre is also a medicinal plant traditionally used for diabetic patients to lower the blood sugar level and has wound healing properties.64,76 developed G. sylvestre extract containing PCL/gelatine nanofibers and investigated their antibacterial and biocompatibility properties. The PCL/gelatine nanofibers mats have showed no inhibition zone but nanofibrous mat comprising G. sylvestre extract by ultrasound assisted displayed a clear zone of inhibition of 15.5 mm and 4.2 mm to S. aureus and E. coli, respectively, and cold macerated assisted G. sylvestre extract loaded nanofibers (7.6 ± 2.2 mm to S. aureus and 1.9 ± 0.2 mm to E. coli). Ramalingam R, Dhand C, Mayandi V, Leung CM, Ezhilarasu H, Karuppannan SK, et al. 76 also fabricated a core–shell nanofibrous dressings containing G. sylvestre extracts using coaxial electrospinning, PCL/gelatin/minocycline hydrochloride as a shell and gelatin/G. sylvestre extracted by ultrasound-assisted and cold macerated extracts as core. The core-shell (combination of plant extract and minocycline hydrochloride) gives the high antibacterial property with ultra sound assisted extraction of 33.2 mm to S. aureus and 26.3 mm to E. coli. The cold mercerization extraction also gives inhibition zone of 28.8 mm and 26.3 mm to S. aureus and E. coli, respectively.
Hypericum perforatum and Hypericum capitatum var. capitatum are plants in the species Hypericum (Hypericaceace) that contain active agents that were used as traditional medicine for wound healing. The oil from these plant extracts shows antimicrobial activity on numerous Gram-positive and Gram-negative bacteria, but, the layer of the oil layer inhibits transfer of gas transfer.72,73 To solve this problem, Eğri, Ö. and N. Erdemi 73 incorporated H. perforatum oil into PEG and PCL polymers and produced as two layers membrane. The upper layer: PCL electrospun nanofibers for membrane integrity and mechanical strength, and bottom layer: PEG/H. perforatum oil and PCL polymer solutions by electro spraying and electrospinning to be in contact with the wound. Akşit, N.N. et al. 72 also developed PLGA/gelatin electrospun containing H. capitatum var capitatum extract. The in vitro H. capitatum var. capitatum release was 80% during the first 24 h and the antibacterial property (10wt% H. capitatum var. capitatum) against S. aureus and E. coli was 12 mm inhibition zone for both bacteria. But, Avci, H. and H. Gergeroglu 74 faced problem of high viscous solution and lack of sufficient entanglement that leads to unsteady electrospinning process when 10 wt% H. perforatum was incorporated into thermoplastic polyurethane polymer solutions for electrospinning. But, incorporation of 7% H. perforatum into thermoplastic polyurethane polymer solutions produce a good nanofiber with inhibition zone of 12 mm against S. aureus and P. aeruginosa. Gunes S, Tamburaci S, Tihminlioglu F. 75 also incorporated H. perforatum into Zein/montmorillonite to produce bilayer nanocomposite resulting 4 mm inhibition zone with 0.5 v/v oil against E. coli.
The activities and properties of electrospun hydrogels are faster than the conventional hydrogels. Development of electrospun polymer hydrogels containing plant extract loaded materials is a new research area. PVA hydrogel electrospun incorporated with Ipomoeapes-caprae leaf extract as wound dressing materials for infected wound was developed with inhibition zone of 33.7 mm, whereas the conventional PVA loaded with Ipomoeapes-caprae leaf extract one is 31.3 mm to S. aureus. 65 This may be due to release of plant active compound from the PVA electrospun hydrogel faster than that of conventional hydrogel dressing. 88 The plant has phytochemical compounds such as alkaloids, glycosides, saponins, flavonoids, tannins, and terpenoids with biological properties including antimicrobial activity. 89
There are some limitations in this study, such as only four databases (PubMed, Scopus, Web of Science and Google Scholar) are included, and papers published only in English are included in the study.
Mechanism of antibacterial properties of plant extract
There are mainly two mechanisms of antibacterial activity: by interfering chemically with the synthesis or function of vital components of bacteria, and circumventing the conventional mechanisms of antibacterial resistance. Some antibacterial may target to protein biosynthesis and cell membrane and others may alter bacterial outer membrane permeability and finally destabilize outer membrane of bacteria, inhibit DNA replication, cell-wall biosynthesis. 49
Most traditional plants phytochemicals such as alkaloids, phenolic, terpene and the likes have antibacterial properties used for wound treatment contains. The antibacterial mechanism of these phytochemicals is discussed as follows: Alkaloids are nitrogenous-based organic compounds that exhibit properties similar to alkalis. The nitrogen atom is responsible to the antimicrobial activity of alkaloids. The mechanism is through efflux pump inhibition, inhibition of cell division, bacteria protein and DNA synthesis, destruction of bacterial membrane and adenosine triphosphate synthase inhibition mechanisms.49,90 Phenols are a large group of secondary plant metabolites, further classified into four according to the number of phenolic groups and the structural elements as: 1 flavonoid, further split into flavones, isoflavones, flavonols, flavanones, chalcones, and catechins; 2 stilbenes with resveratrol as the main compound; 3 lignans and 4 phenolic acids such as hydroxylbenzoic acid and hydroxylcinnamic acid. The antibacterial activity of phenolic compounds work based on the mechanism of rigidifying bacterial membrane, efflux pump inhibitor, inhibition of dihydrofolate reductase, and by damage to the cytoplasmic membrane of the bacteria.49,91 Plant phytochemical terpene are chemically, hydrocarbons with a 5-carbon isoprene, 92 works based on disturbing the cell membrane of the bacteria and inhibiting the transformation of cellular yeast form to the filamentous form.
As shown in Figure 3, there are different targets for the antibacterial agents such as bacterial protein biosynthesis, bacterial cell-wall biosynthesis, bacterial cell membrane destruction, bacterial DNA replication and repair, and inhibition of a metabolic pathway. Proven targets for antibacterial agents.
49

Comparisons with known antibiotics
Inhibition zone of common antibiotics.

Conclusion
There are plentiful natural plant sources that can produce bioactive compounds. These bioactive compounds from plants are easily available, inexpensive, easy to isolate or to extract, environmentally friendly, compatible and harmless to human cells. Many medicinal plant extract bioactive compounds have antibacterial properties used for wound treatment. These plant extracts can be incorporated with wound dressing textile materials by means of electrospinning that gives good bacterial inhibition. Even though, the effect varies according to the different parts of the plant used and the type of extraction, among the medicinal plant extract loaded electrospun nanofibrous wound dressings eight have the highest bacterial inhibition zone greater than or equal to 20 mm. PVA/T. procumbensviz, PVA/honey/C. longa, PCL/carboxyethyl chitosan/PVA/Chamomile, PVA/Ipomoeapes-caprae, Polyether ether ketone/Aloe-vera, Curcuma Longa/gelatin, Chitosan/PEO/Lawsonia Inermis, PCL/Gelatin/lawsone, Gelatin/Aloe-vera/PCL have inhibition zone diameter greater than 20 mm. PVA/T. procumbensviz electrospun nanofibrous (42 mm and 35 mm bacterial inhibition zone to S. aureus and E. coli, respectively) has the highest antibacterial property of all. PVA/honey/C. longa extract electrospun nanofibrous has the second highest antibacterial property (bacterial inhibition zone of 38 mm to S. aureus), and the third one is PCL/carboxyethyl chitosan/PVA/Chamomile with inhibition zone of 37.5 mm against E. coli. We also realized that combination of medicinal plant extract and other antibacterial materials will increase the synergetic effect of the wound materials. Therefore, we can conclude that antibacterial plant extract together with other bioactive polymers combination will increase bacterial inhibition zone for the nanofibrous wound dressings.
Plant phytochemicals incorporated nanofibrous mats are the encouraging wound treatment strategy. However, in the future, there is a need to determine the pure compounds responsible for antibacterial activity in the case of complex mixtures such as in the extracted and essential oils. Another thing that needs more focus is the controlling mechanisms of releasing these bioactive compounds from the electrospun nanofibrous. In the near future, we hope a number of plants will be investigated and can be incorporated into nanofibers as a potential use for wound healing and management.
Footnotes
Acknowledgments
The authors acknowledge College of Textiles, Donghua university, and Bahir Dar University, for allowing to do this paper.
Author contributions
Biruk Fentahun Adamu: Conceptualization, methodology, investigation, writing the original draft, formal analysis, investigation
Gao Jing: Conceptualization, article structure construction, reviewing, editing
Shaojie Tan: Collecting materials and editing
Esubalew Kasaw Gebeyehu: Collecting materials and editing.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
