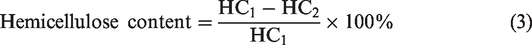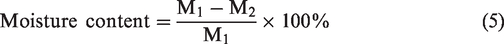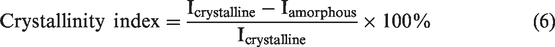Abstract
Coir fibre and pith are eco-friendly material used in the preparation of composites. Coir fibre and coir pith were treated with sodium hydroxide to study the effect of alkali for prolonged exposure on its properties. Fibre and pith were treated with different concentrations of NaOH from 5% to 30% for 24 hours at room temperature. Alkali treatment removes the impurities and the expose the crystalline cellulose and to enhance the surface properties. The alkali-treated coir fibre and pith were characterized by Scanning electron microscopy for morphology, X-ray diffraction for crystallinity index, thermogravimetry for thermal stability, Fourier-transform infrared spectroscopy for structural changes. The chemical composition was analysed for both fibre and pith. Physical properties such as bulk density, particle density and porosity were determined for pith. After alkali treatment, the properties (physical and chemical) of the treated fibre (5% to 20%) has been enhanced. For coir pith the properties has been diminished. Untreated/treated coir fibre/pith filter (CFP) were developed to study the removal of heavy metal ions from the waste water, where untreated (CFP) as a control. The removal efficiency of the developed CFP filter for heavy metal ions were determined by Inductively coupled plasma - optical emission spectrometry (ICP-OES). 5% CFP filter exhibited higher efficiency for removal of heavy metal ions.
Introduction
In waste water, heavy metal ion is a major problem and it has great threat to human beings, and animals. Several removal technologies were employed such as adsorption, chemical precipitation, membrane filtration, ion exchange, coagulation and flocculation. Adsorption is the method widely used compared to other conventional techniques [1]. Cellulosic materials were used due to its low cost, available in abundant quantity and effective removal of heavy metal ions [2]. Removal of heavy metal ions (Cu, Zn and Ni) by lignocellulosic fibres from aqueous solutions by extraction was reported [3]. Cellulose prepared from pineapple leaf fibre was modified for the removal of heavy metal Cadmium (Cd) and Lead (Pb) [4]. Flax fibres were used for the removal of heavy metals such as Cadmium, Lead and Zinc [5]. Wheat straw as a natural and modified biosorbent were used for the removal of cadmium, chromium, Copper, Lead and nickel [6–9]. Nickel was removed from the aqueous solution using sugarcane bagasse [10]. Coconut trees (Cocos nucifera) are extensively grown in tropical areas and it is used for various applications which are of commercial importance [11]. Coconut consists of thick fibrous outer exocarp called husk and inner hard endocarp or shell. Coir pith is a spongy material that binds with the fibre. Coir fibre and pith are extracted from the outer shell (husk) of the coconut while the latter is considered as an agro-waste [12–13]. Coir fibre and pith are lignocellulosic, which consists of crystalline cellulose and amorphous non-cellulosic lignin, hemicellulose, wax and pectin. Coir fibre is biodegradable, cost-effective and possesses good thermal and mechanical properties, but the coir pith is not easily degradable [13–14]. The incompatibility that exists between the hydrophilic lignocellulosic material and hydrophobic composite matrix, coir fibre and pith are not preferred as a reinforcement filler for composites. Thus, coir fibre and pith are treated chemically for surface modification to enhance the properties and to adhere to the composite matrix [13,15]. Previous works have reported the effect of alkali treatment on fibre properties by varying the concentration of alkali and soaking time. For coir pith, alkali treatment with different concentrations was not reported. Alkali-treated fibres were used in preparation of polymer composites and used for different application but not in filtration. In this work, coir fibre and pith were treated with different concentrations of sodium hydroxide for 24 hours at room temperature (30 ˚C) to study the effect of alkali treatment on its properties. Alkali treatment was performed to remove the impurities such as hemicellulose, lignin and wax and to expose the crystalline cellulose present on the surface of pith and fibre. Development of alkali-treated coir fibre-pith filter and then the heavy metal ion removal efficiency was investigated.
Experimental
Materials and method
Chemical treatment
Coir fibre and coir pith were collected from a coir processing unit (Tamil Nadu, India). Using different concentrations of sodium hydroxide (Sisco Research Laboratories, India) such as 5%, 10%, 15%, 20%, 25% and 30%, Coir fibre (CF) and Coir pith (CP) were treated for 24 hours at room temperature to study the effect of alkali treatment on its properties. Both the treatments were carried out separately.
Chemical composition for fibre and pith
Cellulose content
Coir fibre and coir pith of known weight were soaked in the mixture of 1.72% w/v sodium chloride and three drops of concentred sulphuric acid for one hour. After washing and drying 30 ml of liquid ammonia was added for neutralization. Then the fibre and pith were washed, dried and weighed. The cellulose content of treated and untreated fibre was determined by the below equation [16].
Lignin content
In the process of determination of lignin content of cellulose, 1 g of coir fibre was immersed in sulphuric acid (H2SO4) with the concentration of 7.6% and the M:L ratio of 1:300. The experiment was carried out at room temperature and then refluxed for 2 hours. The residue was taken out, washed, dried and weighed. A similar procedure was carried out for the coir pith [16].
Hemicellulose content
The fibre and pith of known weight were soaked in 5% NaOH solution for 30 minutes and then it was neutralized by HCl. Finally, the samples were dried and weighed [16].
Wax content
The wax content of fibre and pith was measured using a Soxhlet apparatus by immersing in carbon tetrachloride and refluxed for one hour. Then the fibre and pith were dried in a hot air oven and weighed [16].
Moisture content
Known conditioned (65% RH and 27 °C) quantity of coir pith and coir fibre was kept in a hot air oven for 2 hours at 105 °C and kept in the desiccator. Finally, the sample was weighed [16].
Characterisation of fibres/pith
Scanning electron microscopy
The morphology of treated/untreated coir fibre and coir pith was studied using scanning electron microscopy (Hitachi S-3400N). The treated and untreated fibre/pith were mounted on the stub and gold sputtering was done with the operating voltage of 15 kV. The cross-sectional view of the fibre was also observed using sem.
X-ray diffraction
The X-ray diffraction pattern of treated/untreated fibre/pith was obtained using Bruker D8 Advance operated at 45 kV with Cu radiation (λ = 1.5406). Samples were placed on the sample holder and measured for 2θ angle range varying from 10˚ to 80˚ at the scanning rate of 1.2˚/min. The crystallinity index of the treated/untreated fibre and pith were determined by the Segal method.
Fourier transform infrared spectroscopy
Fourier transform infrared spectroscopy was conducted by Shimadzu FT-IR spectrometer in the range of 4000-500 cm−1. FTIR analysis was performed in Attenuated total reflectance mode and the spectrum is obtained to study the functional groups of pith and fibre after alkali treatment.
Thermogravimetric analysis
TGA of coir fibre and pith were carried out by Q-50 (TA Instruments Ins, US) at a flow rate of 30 ml/min under nitrogen atmosphere at a scanning rate of 35 °C/min from room temperature to 700 ˚C. A thermogram was obtained for fibre/pith as a function of weight loss percentage against increasing temperature to examine the thermal stability and decomposition of these lignocellulosic materials.
Bulk density of coir pith
Bulk density is calculated as the ratio of the mass of coir pith to the volume of coir pith. Coir pith of known mass was taken and filled in the graduated cylinder. After filling the sample, the volume of the cylinder was noted [17].
Particle density of coir pith
The particle density of treated and untreated coir pith was calculated using a pycnometer by the below-given equation [18].
Dw = Density of water (g/cm3)
Mpcp = Mass of pycnometer and coir pith (g)
Mp = Mass of empty pycnometer (g)
Mpcpw = Mass of pycnometer, coir pith and water (g)
Mpw = Mass of pycnometer and water (g)
Porosity of coir pith
The porosity of treated and untreated coir pith was calculated based on bulk density and particle density [17].
Preparation of coir fibre/pith filter
Equal proportion 50% of Coir fibre and 50% of pith were taken for preparation of filter. Fibres were arranged vertically and pith has been placed inside the fibre where 5% chitosan solution acts as a binder for the filter. Filters were prepared with untreated and alkali-treated fibre/pith, where untreated fibre/pith will act as a control. Construction of filter is represented in Figure 1. As prepared CFP filters have a length of 10 cm and a diameter of 2.54 cm. The packing density of the CFP filter was calculated using the formula given below.

Development of CFP filter.
Volumetric flow rate
The volumetric flow rate was calculated for both untreated and treated Coir fibre/pith filter by the formula,
Where,
Q= volumetric flow rate (ml/min)
V= volume of the liquid (ml)t= time (min):
Inductively coupled plasma – optical emission spectrometry
Inductively coupled plasma - optical emission spectrometry (Perkin Elmer Optima 5300DV) was used to study the concentration of heavy metal ions. Aqueous solution of mixed heavy metals such as Copper (Cu), Zinc (Zn), Chromium (Cr), Lead (Pb), Arsenic (As), Ferric (Fe) and Aluminium (Al) with the concentration of 100 ppm was prepared. As prepared heavy metal ion solution was passed through the coir fibre/pith filter and the permeate was analysed by ICP-OES for removal of heavy metal ions. Removal efficiency was calculated by
Cf – Concentration of feed (ppm)
Cp – Concentration of permeate (ppm)
Results and discussion
Chemical composition and other properties of coir fibre and coir pith
The chemical composition of coir fibre and coir pith is shown in Tables 1 and 2. Hemicellulose and lignin that surrounds and protects the crystalline structure of cellulose. When coir fibre and pith are treated with sodium hydroxide, the hydroxyl group of hemicellulose is broken, reacts with water molecules and discharged out. By alkali treatment the crystalline nature of cellulose is not hydrolysed, the fibre-O-Na chain was formed by cellulose molecules [19]. The cellulose content of treated fibres and pith was increased from 44.12% to 55.40% and 29.98% to 45.02% respectively. On mild alkali treatment, the crystalline cellulose will be exposed. As the concentration and immersion time of alkali increased, Cellulose I (crystalline nature) have been converted to Cellulose II (amorphous). Hence on increasing the concentration of alkali, the cellulose content also increased. Hemicellulose was completely removed for treated coir pith and fibre except for 5% treated fibre which implies the partial removal. Wax on the surface of fibre and pith was removed completely by the alkali treatment [20]. The moisture content of treated pith/fibre was increased compared to untreated fibre/pith because of the removal of hemicellulose and lignin.
Chemical composition of coir fibre.

Chemical composition of coir pith.

Surface morphology
The scanning electron micrographs of the treated/untreated fibre and pith were represented in Figures 2 and 3. The surface morphology of pith and fibre was studied to find the impact of alkali treatment. Wax and other impurities present on the surface of untreated fibre/pith is responsible for the uniform and smoother surface. The fibre/pith on treatment with sodium hydroxide exposed the rough surface on the removal of impurities [21]. The pores present on the surface of coir fibre has been exposed in treated fibre. The cross-sectional view of fibre depicts the multi-lumen structure of the fibre gets damaged after alkali treatment due to the removal of lignin.

SEM images of untreated and treated coir fibre (a) untreated, (b) 5% NaOH, (c) 10% NaOH, (d) 15% NaOH, (e) 20% NaOH, (f) 25% NaOH, (g) 30% NaOH.

SEM images of untreated and treated coir pith (a) Untreated, (b) 5% NaOH, (c) 10% NaOH, (d) 15% NaOH, (e) 20% NaOH, (f) 25% NaOH, (g) 30% NaOH.
Effect of alkali treatment on the crystallinity of coir fibre and pith
XRD patterns of treated/untreated fibre and pith were represented in Figures 4 and 5 respectively. Crystallinity Index of fibre and pith was analyzed using the two peaks which reflects the crystalline phase at 2θ = 21°-22° and amorphous phase at 2θ = 17°-18°. The crystallinity index of fibre increased for the alkali-treated concentration 5% to 20% and decreased for 25% and 30% compared to untreated fibre. Initially, the alkali solution removes the amorphous hemicellulose, wax, pectin and exposure of crystalline cellulose which lies between the crystallite domains [22–23]. As the impurities were removed the crystalline cellulose will be exposed which results in increase in crystallinity index of alkali-treated fibre (5% to 20%), compared to untreated. Further increase in concentration, results in swelling of the fibre, which leads to cleavage of the cellulose chain. Thus, the cleavage of the cellulose chain causes the disordered structure of cellulose which results in a decrease of crystallinity index for alkali-treated fibre (25% and 30%). Fibre can withstand prolonged exposure to alkali treatment but the pith is not capable. Thus, there is a decrease of crystallinity index of treated coir pith 5% to 30% compared to untreated. Table 3 shows the crystallinity index of fibre and pith.

XRD pattern of untreated/treated fibre.

XRD pattern of untreated/treated pith.
Crystallinity index of fibre and pith.

Effect of alkali treatment on structural change in fibre/pith
Fourier Transform Infrared Spectroscopy graphs of pith and fibre represented in Figures 6 and 7. In fibre and pith, the peak around 3300 cm−1 and 2900 cm−1was assigned to OH stretching and C-H stretching of cellulose respectively. The broadening of the peak at 3354 cm−1 was observed for alkali-treated fibre compared to untreated due to the changes in intra and intermolecular hydrogen bonding of cellulose. The peak observed at 1241 cm−1 and 1737 cm−1 indicates the presence of C-O stretching and C = O stretching in the acetyl group of hemicellulose respectively. These peaks were absent for the treated fibres except for 5% alkali concentration which indicates the removal of hemicellulose. For 5% alkali-treated fibre partial removal of hemicellulose was noticed [24]. The peak at 1029 cm−1 was due to the presence of (C-O-C) ether linkages in lignin. As the fibres are exposed to alkali treatment at higher concentration and longer duration, the peak intensity was reduced when compared to untreated which specifies the breakage of the ether linkage. The peak at 1423 cm−1 and 876 cm−1 indicates the CH2 scissoring motion and C-O-C stretching at β-

FTIR spectrum of untreated/treated fibre.

FTIR spectrum of untreated/treated pith.
In untreated coir pith, the peak observed at 1728 cm−1 assigned for C = O stretching of hemicellulose disappeared for treated pith after the removal of hemicellulose. The intensity of peak at 3323 cm−1 corresponds to intramolecular hydrogen bonding increased for treated pith compared to the untreated due to demethylation. This implies that the cellulose in treated coir pith started to lose its crystalline nature and has been converted into amorphous cellulose. Demethylation of lignin takes place in treated pith as the concentration of NaOH increases, hence there is an increase in peak intensity at 1030 cm−1. Slowly, the lignin started to degrade in treated pith which indicates the partial removal of lignin, hence there is a change in intensity of peak at 1610 cm−1 [27].
Effect of alkali treatment on thermal properties of fibre/pith
The Thermogram (TG) and Derivative Thermogram (DTG) of treated/untreated fibre and pith were depicted in Figures 8 and 9. Three stages of thermal degradation take place for fibre and pith. The first stage of decomposition occurs below 100˚C due to evaporation of moisture and other impurities. The second stage of decomposition is due to hemicellulose degradation below 300 ˚C because it was easily hydrolyzed due to the amorphous structure. The final stage of degradation is by cellulose which starts above 300 °C which is responsible for the thermal stability of fibre and pith [19]. Lignin degradation takes place slowly from 290 °C to 600 °C. In DTG, double and single degradation peaks were observed for untreated fibre/pith and treated fibre/pith respectively. The first peak observed at 290˚C for untreated fibre is due to the degradation of hemicellulose and the second peak at 370˚C corresponds to degradation of cellulose [28]. The single peak around 350-370˚C in treated fibres corresponds to the decomposition of cellulose. The thermal stability of treated fibre decreased after alkali treatment compared to untreated because of the degradation of cellulose. For untreated pith, the first step and second step degradation were observed at 320˚C and 460 ˚C corresponds to the decomposition of cellulose and lignin respectively [29]. For treated coir pith, the thermal stability was decreased drastically as the single-step degradation was observed between 310-340 ˚C. The Cellulose I present in untreated pith maintained the thermal stability of the pith and in treated coir pith the Cellulose I has been converted to amorphous cellulose (Cellulose II). The amorphous cellulose degrades soon and hence there is a decrease in thermal stability of treated pith. The residue content of the treated/untreated coir fibre (26.05% to 32.25%) and pith (9.48% to 42.38%) was increased when the samples are treated at 680 °C. Derivative thermogram temperature and residue at 680 ˚C of fibre and pith were represented in Table 4.

TG/DTG curve of untreated and treated fibre.

TG/DTG curve of untreated and treated pith.
Derivative thermogram temperature and residue at 680˚C of fibre and pith.

Bulk density/particle density/porosity
As the concentration of sodium hydroxide increases, bulk density (0.092-0.200 g/cm3) increased due to the removal of wax, fat and other impurities present in pith [13]. Bulk density is inversely proportional to porosity. Thus, the increase in bulk density implies low porosity. The untreated pith is more porous than the treated pith as the porosity decreased from 76% to 10%. Particle density also decreased from 0.388 to 0.123 g/cm3. Porosity and particle density are inversely proportional to particle size. When the coir pith was treated with alkali its particle size has been increased, as the impurities were removed from the surface. Thus, there is a decrease in particle density and porosity. Table 5 represents the bulk density, particle density and porosity.
Properties of coir pith.

Properties of pith and fibre with respect to heavy metal ion removal
The properties of pith and fibre influence the heavy metal ion absorption rate. It was reported that 5% alkali treatment and six hours are the optimal condition to sustain the properties [19]. The effect of alkali treatment on properties and structural degradation was analysed by increasing the concentration of NaOH from 5% to 30%. By alkali treatment, hemicellulose, lignin and impurities were removed and the internal surface of the fibre was explored. Hemicellulose and lignin were responsible for hydrophilicity of the fibre and pith. After alkali treatment the hydrophilicity of treated fibre and pith decreased, which implies that the treated fibre/pith has lower moisture absorption [30]. Sodium hydroxide reacts with cellulose to break the hydrogen bonds and explore the hydroxyl group on the surface. This hydroxyl group present on the surface of cellulose plays an important role for adsorption of heavy metal ions.
Volumetric flow rate
The developed CFP filters were represented in Figure 10. The volumetric flow rate of the CFP filter is tabulated in Table 6. The volumetric flow rate was measured to check the flow of water through the filter. Volumetric flow rate decreased for treated CFP filter when compared to untreated. Porosity of the all constructed sample (coir fibre/pith) was maintained as 40% and packing density as 0.6122 (g/cm3). After alkali treatment, due to increase in crystallinity and by maintaining the same porosity and packing density of 0.6122 (g/cm3), the flow through the filter was decreased.

CFP filters treated with different concentrations of NaOH.
Volumetric flow rate.

Inductively coupled plasma - optical emission spectrometry (ICP-OES)
After passing heavy metal ion solution through the CFP filter, the collected permeate was analysed through ICP-OES and removal efficiency were given in Table 7 and Figure 11. Untreated CFP filter acts as a control for treated filters. The 5% CFP filter absorbed higher heavy metal ions compared to untreated and other treated filters. Alkali treated CFP filter has better efficiency compared to untreated CFP filter. Thus, Alkali-treatment was removed the impurities and exposed the crystalline cellulose. The exposed crystalline structure with higher number of OH groups acts as an adsorbent for removal of heavy metal ions. Crystalline cellulose has greater resistance compared to amorphous cellulose against degradation. In general, the OH groups are interconnected by bonds with hemicellulose and lignin. On alkali treatment, these hydroxyl group gets activated after the removal of impurities and acts as an adsorption sites for heavy metal ion. Based on the crystallinity index, 5% CFP filter which has higher crystalline nature compared to other alkali treated fibres and thus higher removal efficiency was achieved. As the concentration of alkali increases, the crystalline cellulose was restructured by disintegration of molecular chains as amorphous cellulose. Amorphous cellulose is more porous than crystalline cellulose which allows to penetrate the heavy metal ion and thus there is a decrease in removal efficiency for other alkali treated fibres as the concentration of alkali increased. The decreased porosity of coir pith was another reason which increased the metal ion removal for treated CFP filter because untreated coir pith has higher porosity compared to treated coir pith. Porosity of coir pith implies that the heavy metal ions are filtered based on their size. Thus, the CFP filter acts as a filter as well as an adsorbent. When comparing all samples the lowest heavy metal ion removal of the treated CFP filter is higher than the untreated filter.
Heavy metal ion removal efficiency by CFP filter.

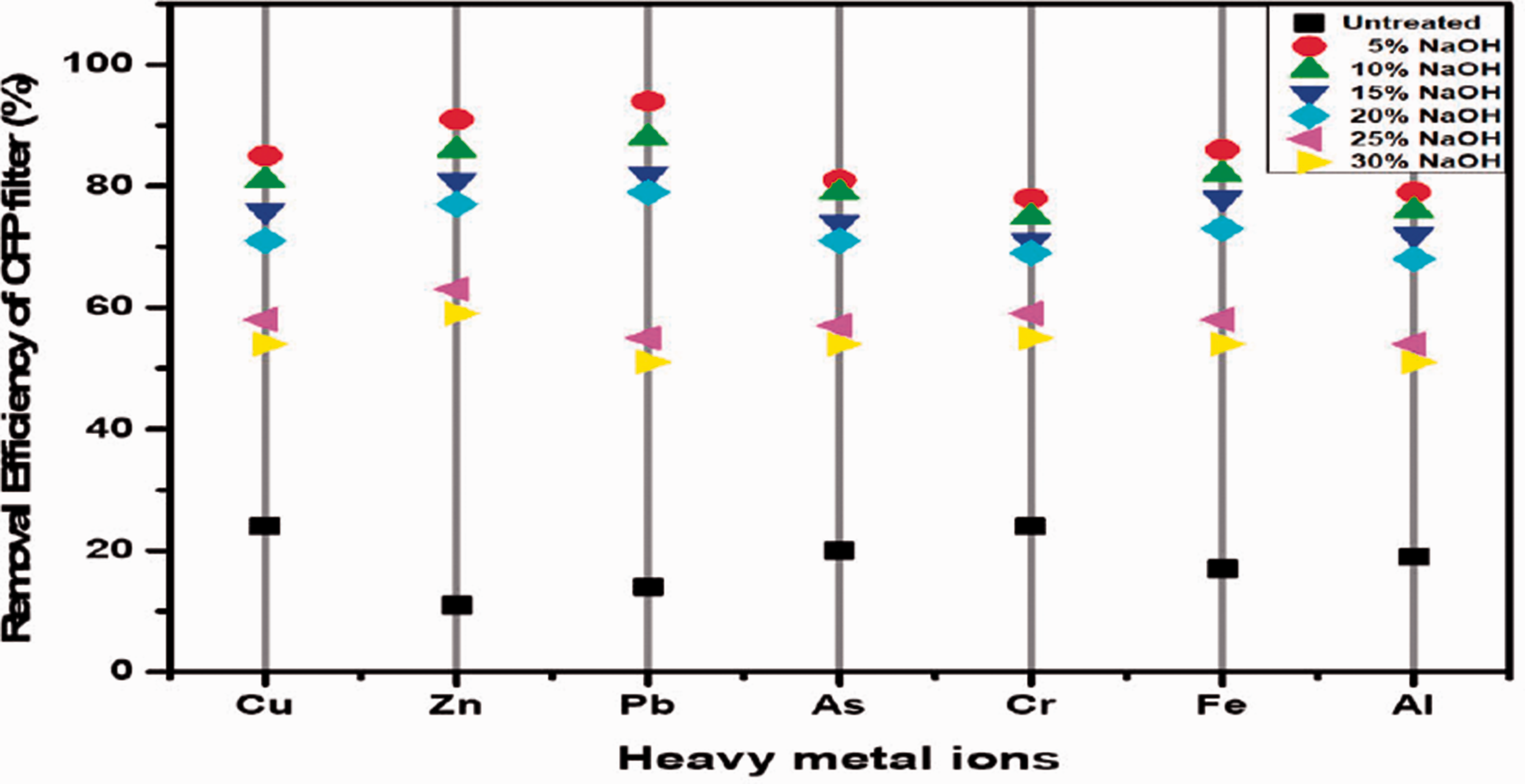
Heavy metal ion removal efficiency by CFP filter.
Conclusion
The morphology of alkali-treated fibre and pith were changed significantly by alkali treatment by the removal of hemicellulose, wax and a partial amount of lignin from the surface. Hemicellulose removal and exposure of crystalline cellulose were confirmed by FTIR and XRD. The thermal stability has been decreased slightly for fibre and drastically for coir pith, as the concentration were higher. 5% to 20% treated fibre maintained the properties (physical, and chemical) though it has been exposed to longer duration. The CFP filter was developed with treated/untreated fibre and pith, 5% alkali-treated filter shows higher removal efficiency compared to other treated filters. Further studies will focus on the varying proportion of fibre and pith for the preparation of filter and increasing the concentration of the heavy metal ion and adsorption mechanism of chitosan and CFP filter.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by University Grants Commission (UGC), New Delhi, India.