Abstract
Constructing an easily recyclable and reusable filter is highly efficient for removing Cd, Pb2+ and organic contaminants from aqueous solution. This study investigates the optimization of Cd, Pb2+ adsorption process by thiol functionalized Agave americana fibre. Additionally, polyacrylonitrile (PAN) nanofibre membrane is used as a substrate to filter the organic contaminants. The properties of adsorbents are characterized by various techniques such as Attenuated Total Reflection- Fourier transforms infrared spectroscopy (ATR-FTIR), tensile tester and scanning electron microscopy (SEM). The filter’s performance has been investigated with three variables of thiol functionalized fibre packing density, length of the functionalized fibre and nanofibre spinning time. The fibre packing density and nanofibre spinning time are the most significant parameters that affect all responses. The impact of independent variables on responses has been critically analysed. The optimized filter is identified by regression analysis. The developed filter satisfies heavy metal ion adsorption (Cd, Pb2+) and organic contaminants.
Keywords
Introduction
Water is an essential requirement for life. In recent years, pollution in water bodies has increased because of urbanization, industrialization and population. The disposal of toxic effluents into freshwater bodies causes severe damage to humans and the ecosystem.1,2 Even the treated industrial effluents contain toxic substances such as dye, heavy metal ions, salts, solvents, etc.,3,4 Toxic heavy metal ions are inorganic pollutants and it is discharged from industries such as tanneries, mining, fertilizer, paper and pesticides. 5 The most commonly found toxic heavy metal ions are lead, chromium, mercury, arsenic and cadmium, which are not biodegradable and affects living organisms. The continuous discharge of treated wastewater into water bodies also becomes a paramount concern due to their bioaccumulation and long-time persistence.6,7
The maximum permissible standards that Environmental Protection Agency sets for lead, cadmium, chromium, mercury and arsenic in drinking water are 0.006 mg L−1, 0.01 mg L−1, 0.05 mg L−1, 0.000,003 mg L−1, 0.05 mg L−1, respectively. 8 Among the heavy metal ions, Cd and Pb2+ are carcinogenic and can chronically affect humans even at a lower concentration. At an extreme level of consumption, they can cause severe health effects, including reduced growth, organ damage, nervous system damage etc. There are many conventional heavy metal ion removal treatment techniques to remove metals but are inconvenient considering their reusability, cost-effectiveness and high waste generation.9,10 Among the techniques, adsorption is the most effective process with significant advantages such as high performance, low operation cost and flexibility in design. Various organic, inorganic and their hybrid adsorbents are widely used to remove heavy metal ions from wastewater.11,12
Porous organic adsorbent has gained more attention in wastewater treatment such as cellulose. To date, many researchers have focused on natural cellulose for heavy metal ion adsorption due to their large availability in nature. Chemical modification of porous cellulosic adsorbents can enhance efficiency of heavy metal ions from wastewater and its porous nature helps attach more functional groups to adsorbent, which can increase the adsorption sites.13,14,15 The construction of cellulose base filters would play an important role in wastewater treatment. In general, there are two adsorption methods used in filter such as continuous fixed-bed adsorption and batch adsorption. A continuous fixed-bed is more feasible under continuous flow conditions. Continuous flow helps treat large quantities of wastewater with cyclic adsorption and desorption of heavy metal ions.16,17 For instance, Wu et al. worked on thiol functionalized cellulosic biomass to adsorb Pb2+ from glucose solution. The adsorption of Pb2+ increased without loss of glucose by thiol modified cellulose biomass. 18 Daochalermwong et al. extracted the cellulose from pineapple leaves and functionalized it using ethylene diamine tetra acetic acid (EDTA) and carboxymethyl (CM) groups for removal of (Pb2+). Cellulose-EDTA achieved good efficiency of 41.2 mg g−1 for Pb2+. Whereas cellulose-CM exhibited excellent adsorption capacity of 63.4 mg/g−1 for Pb2+. The covalent bonding of cellulose-EDTA with Pb2+ is slower than the ionic bonding formation of Cellulose-CM, which decreases the adsorption of Cellulose-EDTA. 19 Yao et al. introduced aldehyde functional groups on cellulose nanofibrils to adsorb Cu2+ and Pb2+ from aqueous solutions. The authors attained a maximum adsorption capacity of 157.73 mg g−1 and 38.36 mg g−1 for lead and copper, respectively. The Langmuir isotherm model revealed a monolayer chemisorption process and the sorption was endothermic. 20 Zhang et al. introduced the functional groups of amino and carboxyl into microcrystalline cellulose. The functionalized cellulose can remove heavy metal ions through the chelating process. The Langmuir isotherm model achieved the maximum adsorption capacity of 3571.1 mg g-1 and 217.3 mg g−1 for Cd and Pb2+, which indicates that the functionalized microcrystalline cellulose acts as a promising candidate in heavy metal ion adsorption. 21 Apart from cellulosic natural fibre, nanofibre is a promising candidate in wastewater treatment due to its attractive properties such as high porosity, high surface area to volume ratio and good water permeation. 22 The polyacrylonitrile (PAN) nanofibre is porous and has nitrile groups that can act as an adsorbent for heavy metal ions. To date, a dual functional filter (heavy metal ions adsorption and organic waste removal) has not been developed with high efficiency.
This study aims to develop a novel proto-type filter setup to satisfy both adsorptions of heavy metal ions and the removal of organic contaminants. The Agave americana fibre is thiol functionalized and it is stuffed into the filtration tube as per the experimental design. Further, polyacrylonitrile (PAN) nanofibre is wrapped on a filtration tube. Functionalized Agave americana fibre is used to remove heavy metal ions and organic contaminants. Whereas PAN nanofibrous membrane can filter finer particles in the solution. Hence, the removal of heavy metal ions, macro and micro size organic contaminants can be achieved by a single state filtration process. The Box-Behnken experimental design has been used to optimize the filter parameters with three independent variables (X1-thiol functionalized fibre packing density, X2-length of the functionalized fibre and X3-nanofibre spinning time). The developed filtration system adsorbs Cd, Pb2+ and also filters the organic pollutants. The intermolecular interaction of the Agave americana fibres are characterized by Attenuated Total Reflection (ATR) and PAN nanofibre membrane are analyzed by Fourier transform infrared spectroscopy (FTIR). The surface morphology of the Agave americana and nanofibre membrane is characterized by scanning electron microscopy (SEM). The tensile strength of the nanofibre membrane has been investigated using Instron tensile tester. Rejection of organic contaminants are evaluated by Total Organic Contaminant analyser (TOC). The heavy metal ion adsorption capacity has been observed by inductively coupled plasma - optical emission spectrometry (ICP-OES). Fifteen filters have been constructed based on experimental design.
Materials and method
Materials
(3-mercaptopropyl) trimethoxysilane (3-MPTMS, purity 95%, M = 196.34 g/mol) has been purchased from Sigma Aldrich Ltd., India. Glacial acetic acid, Sodium hydroxide (NaOH, purity ≥96.0%) pellets, Dimethylformamide (DMF), Cadmium nitrate ((Cd(NO3)2, purity 99%), Lead nitrate ((Pb(NO3)2, purity 99%) and Ethanol have been purchased from Sisco Research Laboratories Pvt. Ltd, India, with analytical grade. Polyacrylonitrile (PAN) with MW 150,000 g/mol has been obtained from Sigma Aldrich.
Fibre Extraction
The Agave americana fibre has been collected from in and around Coimbatore, India. The long spiky collected leaves are crushed and immersed in distilled water for 1 week. Then, the adhered pulpy region is removed through washing in distilled water. Finally, the fibres are dried in sunlight for 1 week to remove moisture.
Alkali treatment
Agave americana fibre has been treated with a 5% solution of NaOH (by weight) for 3 h at 80°C. After the alkali treatment, the fibre is washed with distilled water until pH reached neutral. Then, the fibres are kept in the oven at 80°C to remove the moisture.
Functionalization of fibre
The 7 wt % of 3- MPTMS is diluted with a 95/5 vol % ethanol/water mixture. Glacial acidic acid is added to the solution until the pH reached 3. The fibre is cut into different lengths (0.5, 1 and 1.5 cm) as per the experimental plan. Then, Agave americana fibre is added to the mixture of 3-MPTMS, ethanol and water. Further, the reaction mixture is stirred for 2 h and the sample is cured at 120°C for 30 min.
PAN solution preparation
10 wt % of PAN is dissolved in DMF. The solution is stirred for 4 h at room temperature to achieve a homogeneous state.
Aqueous solution
The Cd and Pb2+ solutions of 10 mg L−1 are prepared by dissolving cadmium nitrate and lead nitrate in 1000 mL distilled water.
Filtration setup
Experimental design for three independent variables with the actual response.


a) Filtration setup b) Tensile experiment procedure c) Mechanism of metal ion adsorption by thiol functionalionalized Agave americana fibre.
Specifications of filtration setup.

Box Behnken experimental design
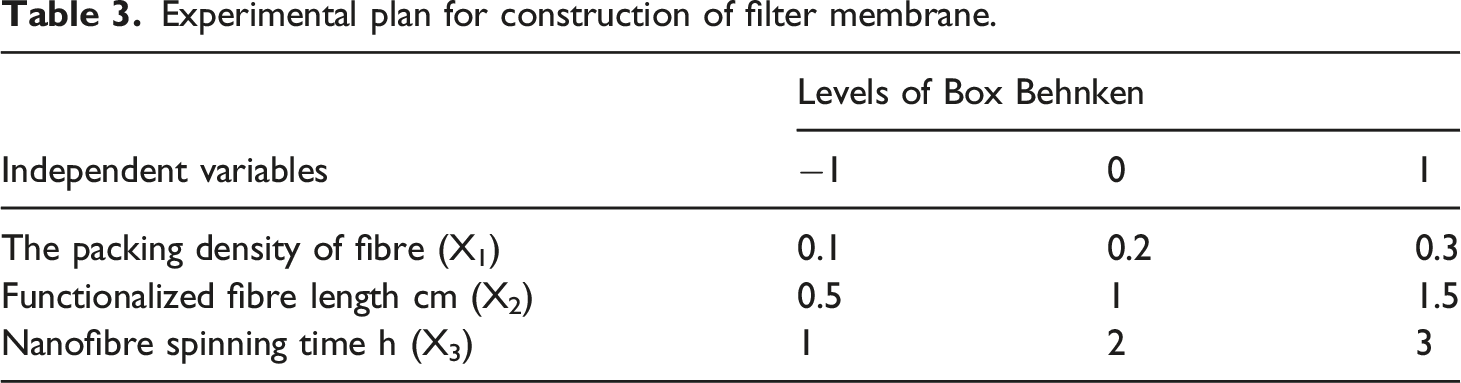
Experimental plan for construction of filter membrane.
The regression analysis correlates the relationship between independent variables and response. The polynomial regression equation is given in equation (1).
Characterization techniques
Electrospinning
Polyacrylonitrile solution is poured into a syringe that had stainless steel needle. Here, a drum collector is used to collect the nanofibre membrane. The electrospinning parameters are set at voltage of 18 kV and a feed rate of 0.75 mL/h. The rotation of the drum collector is fixed at 20 rpm.
ATR – FTIR Spectroscopy
Fourier transforms infrared spectroscopy spectra of PAN nanofibre has been recorded using Jasco FT/IR-6600 USA with a scan range from 4000 cm−1 to 400 cm−1. The PAN nanofibre membrane is cut into small pieces and KBr pelletized. Then, the sample is placed on the sample holder and IR spectrum is recorded. Attenuated Total Reflection (ATR) spectra of Agave americana fibre are examined by Bruker alpha diamond crystal Attenuated Total Reflection.
Mechanical strength
The tensile strength of the nanofibre has been analyzed using INSTRON 3369-UTM, US. The PAN nanofibre mat is fixed on a stretching fixture, and the gauge length was 20 mm with a crosshead speed of 5 mm/min shown in Figure 1(b).
Scanning electron microscopy (SEM)
The surface morphology is analysed by scanning electron microscopy (TESCAN VEGA) with a field emission gun operated at 25 kV.
Organic contaminants rejection and permeation
Humic acid iss dissolved in distilled water (0.1 g L−1). 0.001 M CaCl2 is added to the solution. The pH of the solution is adjusted to 6 to add a small amount of 0.1 M HCl or 0.1 M NaOH. The prepared standard solution has been used for rejection analysis. The filtration through each filter is carried out individually. The total organic contaminants of feed and permeate solution are estimated using a Total Organic Carbon analyzer (Shimadzu, TOC-V CPH). The organic contaminants rejection and permeation calculation are established from the literature.
25
The percentage of organic contaminants rejection is calculated using equation (2).
The permeate is collected, and the filtration time of organic contaminants is noted. The permeate flux is calculated by equation (3)
Inductively coupled plasma-optical emission spectrometry (ICP-OES)
The initial and feed concentrations of Cd and Pb2+ are analysed by inductively coupled plasma-optical emission spectroscopy (ICP-OES (Thermofisher ICAP-6000 series)). The heavy metal ion calculation is established from the literature.
26
The percentage of metal ions are calculated by equation (4)
Results and Discussion
The thiol functionalization mechanism involves two main reactions, namely, hydrolysis and condensation. The silanol group is formed by hydrolysis after the removal of methoxy groups. The –OH group of silanol is attached to the hydroxyl group of cellulose by hydrogen bonding. The condensation reaction occurs at 120°C of curing temperature. At that temperature, a covalent linkage is formed between the –OH group of silanol and hydroxyl on the cellulose chain with concomitant water loss. Finally, the thiol functionalized fibre can adsorb metal ions. The adsorption mechanism shown is in Figure 1(c). In this study, the soft coordinating group of thiol can adsorb soft metal ions of Cd and Pb2+ effectively. 27
Figure 2(a) shows characteristic bands of raw Agave americana fibre, alkali-treated and thiol functionalized fibres. The absorption band at 3328 cm−1 is observed due to the stretching vibration of O-H. In alkali-treated fibre, the intensity of the peak increases due to the breaking down of the ester bond, which creates a free OH group.
28
In raw fibre, the peak at 1735 cm−1 corresponds to the stretching vibration of C = O hemicellulose. The observed peak around 1245 cm−1 is assigned to the stretching vibration of the acetyl group of lignin. After the treatment with NaOH, both the peaks disappear. The band at 1433, 1322, 1159, 1021 and 893 cm−1 are ascribed to the stretching and bending vibrations of -CH2, -CH, -OH and C-O bonds of cellulose.
29
In thiol functionalized Agave americana fibre, a new peak at a wavelength of 790 cm−1 appears, which is responsible for stretching vibration of Si-C/Si-O. A weak band is observed at 2555 cm-1, and it is related to the stretching vibration of S-H. This suggests that the thiol group has been successfully introduced onto the fibre.
30
Figure 2(b) shows FTIR spectra of PAN nanofibre. In PAN nanofibre, the peaks at 2943 and 1453 cm−1 are associated with C-H stretching and bending vibration. A band at 2240 cm−1 is related to stretching vibration of C≡N.
31
a) Attenuated total reflection spectra for Agave americana fibre b) Fourier transforms infrared spectroscopy spectra for polyacrylonitrile nanofibre.
Mechanical property of Polyacrylonitrile nanofibre.

The two-way interaction of factors towards total organic contaminants rejection is shown in Figures 3(a)–(c). The influence of thiol functionalized fibre packing density and functionalized fibre length on total organic contaminants rejection is shown in Figure 3(a). The rejection of organic contaminants increases from 30.66 to 84.02% when fibre packing density increases. The dense layer of fibre can capture the organic contaminants and increase the rejection rate. An increase in the fibre length does not affect organic contaminants rejection much. Figure 3(b) describes the effect of functionalized fibre length and nanofibre spinning time on organic contaminants rejection. It is observed that when the nanofibre spinning time increases from 1 h to 3 h, the rejection of organic contaminants also increases from 30. 66%–84.02%. According to the plot, the maximum rejection rate is obtained when the functionalized fibre has a 1 cm length and 3 h nanofibre spinning time which suggests the dense nanofibre layer can capture more contaminants. Figure 3(c) represents the effect of functionalized fibre packing density and nanofibre spinning time. The results indicate that maximum rejection has been achieved when fibre packing density and nanofibre spinning time is kept at the highest level. The reason is that the closely packed fibre, and a highly dense nanofibre layer can effectively remove contaminants due to inter and intra pores of the agave americana and PAN nanofibre. 3D surface plots depict interactions among different process variables on organic permeations and total organic contaminants rejection. (a, d) Functionalized fibre packing density, fibre length (b, e) Fibre length, nanofibre spinning time (c, f) functionalized fibre packing density, nanofibre spinning time.
Response surface 3D plots for permeate flux is shown in Figures 3(d)–(f). Figure 3(d) shows permeate flux variation in response to functionalized fibre packing density and fibre length. A low permeate flux is observed when both variables interact at the high setting. The permeate flux is dependent much on the changes in fibre packing density. The flux rate is high when the fibre packing density is low. The effect of functionalized fibre length and nanofibre spinning time on permeate flux is illustrated in Figure 3(e). An increase in the nanofibre spinning time decreases the permeate flux due to pore blockage by contaminants. The interaction of functionalized fibre packing density and nanofibre spinning time is shown in Figure 3(f). The high setting of both variables decreases the permeation. Meanwhile, the highest permeation is observed when both variables are low.
Analysis of 3D surface plots help to determine the independent variable, and it is the effect on Cd adsorption shown in Figures 4(a)–(c). The combined effects of thiol functionalized fibre packing density and functionalized fibre length are presented in Figure 4(a). It clearly shows that the adsorption efficiency of Cd increases when fibre packing density increases from 0.1 to 0.2. The reason behind this is that the highest fibre packing density has more active thiol groups on fibre, which adsorb more metal ions of Cd. The results indicate that the adsorption of Cd increases significantly while increasing the functionalized fibre length from 0.5 to 1 cm. The shorter fibres can be packed densely in filtration tubes when compared to longer fibre. At the same time, the shortest fibre can leach out though permeate water. The longest fibre is not densely packed compared to the shortest fibre, but the fibre leachate problem does not occur. Hence, 1 cm of functionalized fibre optimum for adsorption of Cd ion. Similarly, the interactive effect of functionalized fibre length and nanofibre spinning time is also assessed in Figure 4(b). Maximum adsorption occurs from 2 and 3 h PAN nanofibre spinning time due to availability of more nitrile groups. Additionally, the nitrile group of the PAN nanofibre can also adsorb metal ions.
33
Figure 4(c) depicts the surface plot of fibre packing density and nanofibre spinning time. The high adsorption efficiency of Cd has been achieved when both the fibre packing density and nanofibre spinning time are higher. 3D surface plots depict interactions among different process variables on cadmium and lead removal. (a, d) Functionalized fibre packing density, fibre length (b, e) Fibre length, nanofibre spinning time (c, f) functionalized fibre packing density, nanofibre spinning time.
Figures 4(d)–(f) shows the effect of thiol functionalized fibre packing density, functionalized fibre length and nanofibre spinning time on Pb2+ removal efficiency. Figure 4(d) represents the fibre packing density and functionalized fibre length on Pb2+ adsorption. It has been observed that with an increase in functionalized fibre packing density from 0.2 to 0.3, a slight increase in efficiency from 94 to 99.84% has been observed. Further, when the length of the functionalized fibre increases from 0.5 to 1.5, the removal efficiency decreases from 99.84 to 94%. The plot obtains maximum efficiency with a fibre packing density of 0.3 and functionalized fibre length of 0.5. The effects of functionalized fibre length and nanofibre spinning time is illustrated in Figure 4(e). With an increase in the nanofibre spinning time from 1 to 3, there is a significant increase in Pb2+ removal efficiency. The maximum adsorption of Pb2+ has been observed when nanofibre spinning time is around 3 h. Figure 4(f) shows the interactive effect of fibre packing density and nanofibre spinning time. The maximum adsorption efficiency is achieved at maximum fibre packing density and spinning time.
Looking into the previous studies, many researchers have developed adsorbents for Cd and Pb2+. The study reported by Rahaman et al. 34 revealed that jute derived cellulose composite effectively adsorbed Cd, Pb2+ ions, and maximum efficiency was achieved at 85% and 90%, respectively. Liang et al. 35 prepared thiol functionalized sepiolite for heavy metal ion adsorption. The maximum adsorption efficiency of 84.23% and 57.75% was obtained for Pb2+ and Cd ions, respectively. Mohammad et al. 36 studied alkali modified almond shells for Pb2+ and Cd ions removal. They observed that the maximum adsorption capacity was 9 mg/g for Pb2+ and 7 mg/g for Cd. A similar trend has been observed in the present study, with the adsorption capacity of Pb2+ higher than Cd, which proves that Pb2+ has more surface positive ions than Cd. Also various researchers reported both metal ion and organic contaminant rejection. Hideaki et al. 37 studied Cu, Cr, and organic contaminant rejection by thermosensitive polymers. The adsorption of Cu and Cr was observed at 85% and 90%, respectively. In contrast, organic contaminant rejection was achieved at around 80%. Another study by Ragavendra et al. 38 proved that polyetherimide membrane for Cd, Pb2+ and organic contaminants removal showed that the organic contaminants were rejected by up to 87%. The Cd ion was removed up to 42% and Pb2+ around 48%. The study showed that the adsorption of metal ions decreased. At the same time, the rejection rate of organic contaminants was higher due to pore blockage or cake formation of organic contaminants on the membrane. In the present study, thiol modification enhances metal ion adsorption, and the pores of Agave americana fibre and PAN nanofibres aid the filter medium to remove the organic contaminants as well. Hence, the developed filter achieves higher performance on metal ion and organic contaminant rejection.
Statistical analysis
Analysis of variance for the response.

Figure 5(a) represents the removal efficiency of Cd and Pb2+ metal ions listed in Table 1. The percentage of Pb2+ adsorption does not differ for all experiments. All constructed filters attain more than 94% of Pb2+ adsorption because of high affinity towards Pb2+. However, more visible efficiency changes are observed in Cd adsorption due to fewer available binding sites. Similar findings have been observed in Rashi Gusain et al.
31
The maximum adsorption efficiency of both metals is achieved by Exp 9 and has the maximum fibre packing density and the lowest functionalized fibre length. The highest packing density of functionalized fibre has more thiol groups and can adsorb maximum metal ions. Meanwhile, the PAN nanofibre is also involved in metal ion adsorption through the nitrile group.
40
Hence, increasing the nanofibre spinning time can influence metal ion adsorption. The minimum adsorption efficiency is observed in Exp 2. The lowest packing density with a longer fibre length decreases the adsorption efficiency. According to Pearson’s soft acid/base theory, the soft acid coordinating group of thiol can effectively bind with soft acid metal ions of Cd and Pb2+.
41
a) Efficiency of Cd and Pb2+ metal ion. b) Performance of filter on organic contaminants rejection and permeation.
Organic contaminants rejection and permeation are shown in Figure 5(b), given in Table 1. From the results, it has been observed that when the organic contaminants rejection increases, the rate of permeation decreases.
25
An increase in the fibre packing density and nanofibre spinning time increase the rejection rate. The functionalized Agave americana fibre and nanofibre pores are blocked at the highest rejection of organic contaminants. The minimum fibre packing density and nanofibre spinning time offer low resistance to organic contaminants transport across the fibre resulting in lower organic contaminants rejection and higher flux. The organic contaminants rejection percentage is above 80 in Exp 10 and Exp 12, with maximum packing density and nanofibre spinning time. Figure 6 shows the before and after treatment of organic contaminants permeation. The images show that contaminants block the pores of nanofibre and functionalized fibre after achieving the maximum rejection rate. The filter can effectively removes the organic contaminants also. Condition of the filter before and after organic contaminants rejection.
Figure 7 shows the scanning electron microscopy images of raw fibre and nanofibre. The neat Agave americana fibre surface is relatively smooth (Figure 7(a)) after the treatment with 5% NaOH (Figure 7(b)). Groves and holes can be observed after the functionalization, and the interconnected porous morphology can be seen in Figure 7(c).
42
Figure 7(d) shows the well-defined smooth, uniform and clear surface with beadless structure. The diameters of nanofibre range from 50 to 200 nm. Scanning electron microscopy images of a) Agave americana fibre b) alkali-treated c) thiol functionalized fibre d) Polyacrylonitrile nanofibre (3 h spinning time).
Conclusion
This work has explained the detailed probability of response surface methodology for evaluating the interactive effect of three process variables on various responses such as organic contaminants rejection, permeate flux, adsorption of Cd and Pb2+. The developed filter shows that each response highly influences three independent variables. Maximum fibre packing density and maximum spinning time led to higher efficiency on removal of Cd, Pb2+ and organic contaminants. An increase in organic contaminants rejection causes low permeate flux due to pore blockage of contaminants on PAN nanofibre. The analysis of variance shows a high coefficient regression, i.e., R2 = 0.9, which confirm that the obtained value fits well with the model. Furthermore, the p-value is found to be <0.05, and it is proved that the variables have crucial control over all the responses. The results reveal that the developed filter shows a significant capacity to remove Cd, Pb2+ and organic contaminants. The Box-Behnken experimental design could effectively predict the response with maximum accuracy. The regression analysis optimize the influential factors such as fibre packing density, length of the functionalized fibre and nanofibre spinning time. The optimized parameters are fibre packing density of 0.3, fibre length of 1 cm and nanofibre spinning time of 3 h. The metal ions-Cd and Pb2+ removal efficiency at these optimum conditions are 87.19% and 99.23%, respectively.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the DST-PURSE (MH No. 7.1.3.69 DST- PURSE phase 2 program- 4. Con) and UGC_SAP (MH No. 6.2.137 UGC- SAP- textile technology- consu).
