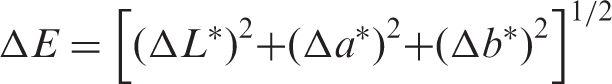Abstract
Cotton fabrics were alkali-treated with sodium hydroxide at different concentrations in the range from 1 M to 6 M in order to investigate the effect of alkali treatment on the formation of silver nanoparticles. Alkali-treated fabrics were dipped into the silver nitrate solution followed by chemical reduction, which caused in situ formation of silver nanoparticles on the surface of fabrics. As-prepared fabrics were characterized by scanning electron microscope, energy-dispersive X-ray spectroscopy, and X-ray diffraction analysis and also antimicrobial test was carried out. In addition, the color variation, antibacterial durability, and mechanical properties of the fabrics were assessed. The considerable effect of alkali pre-treatment on loading and stabilizing silver nanoparticles was demonstrated. Fabrics treated at higher sodium hydroxide concentrations have higher silver content. Silver nanoparticles are homogeneous and evenly distributed on fibers with high surface coverage. Treated fabrics exhibit strong antibacterial activity against Escherichia coli and Staphylococcus aureus, even after 10 laundering cycles. This procedure is facile, cost-effective and versatile and provides a simple route to manufacturing of cotton textiles with high silver content.
Introduction
Textiles’ surface modification with nanoscale materials is an interesting approach for the fabrication of functionalized fibers/fabrics. At present, a considerable amount of research has been carried out for immobilization of various nanoparticles on textile materials, bringing new properties [1–17]. Each of these nanomaterials is able to provide special effects. For instance, using ZnO nanostructures for UV-blocking [1–3], silver for antimicrobial [4–7], sodium montmorillonite clay for flame retardant [8], silver, silica, and polysiloxane for water repellency [9–12], TiO2 for self-cleaning [13–15], silver and ZnO for conductivity [16,17] protection/modification of textile products is reported. The results of these studies demonstrate the key role of nanotechnology in functional finishes of textiles so that using other materials achieving these special effects is not possible. Moreover, by combination of two or more nanomaterial, some new nanostructured fibrous textile composites have been constructed for advanced applications such as water purification, electrochemical capacitors, and smart fabrics [18–21].
Elemental silver and silver salts are well known as antimicrobial agents in curative and preventive health care for centuries [22]. It is well known that silver has a broad antibacterial activity while exhibiting low toxicity toward mammalian cells. Over the last decades, silver nanoparticles (AgNPs) have found applications in catalysis, optics, electronics and other areas due to their unique size-dependent optical, electrical, and magnetic properties. Currently, most of the applications of silver nanoparticles are in antibacterial/antifungal agents in biotechnology and bioengineering, water treatment, and silver-based consumer products. Moreover, in the case of textile engineering, it has been demonstrated that AgNPs have the high potential to be an excellent agent for the antimicrobial finishing of textile materials, so that various antimicrobial textiles have been fabricated by using AgNPs.
Different methods have been used for the deposition of AgNPs on fabrics. One strategy consists of the textile dip-coating with colloidal suspension of AgNPs [23–25]. Another approach consists of the textile surface modification and then coating of fabrics by using colloidal silver [4,26]. In situ generation of AgNPs within textile materials is a particularly efficient approach to such nanocomposites [27–30]. In this approach, at first Ag+ ions are adsorbed onto fiber surfaces by electrostatic or Van der Waals forces and then are reduced into the AgNPs by various reduction methods. However, most of the utilized methods suffer from insufficient deposition and also require long reaction times, and/or multiple steps, which are major drawbacks for practical applications. Consequently, finding easy and efficient methods for the uniform and high-density coating of textile substrates with AgNPs is one of the challenging issues.
Mercerization is a treatment for cotton fabric and thread with alkali hydroxides namely NaOH solution that gives fabric or yarns a lustrous appearance and strengthens them. The structure of the fiber interconverts from alpha-cellulose to a thermodynamically more favorable beta-cellulose polymorph. Mercerization has a substantial influence on physical (morphological, molecular, and supramolecular) and chemical properties of cellulose polymer fibers causing changes in crystallinity, dyeability, and reactivity [31–33]. The effect of alkali treatment on the dye sorption of lyocell fabrics has been investigated by Siroky et al. [34]. It was shown that alkali treatment has a positive effect on the adsorption of C. I. Reactive Red 120 and the greatest adsorption of dye occurred for fabrics treated with 2.53 and 3.33 mol dm−3 aqueous NaOH solutions. Goswami et al. [35] reported that NaOH pre-treatment significantly improves the color yield, exhaustion, and fixation for different types of reactive dyes. It was observed that the concentration of NaOH has an influence on the adsorption of dyes to the fabrics. Therefore, the possibility of using alkali treatment for increasing the deposition value of AgNPs to the cellulosic textiles can be very useful and has a high potential for practical applications.
In this paper, a novel method based on the surface pre-activation of cotton cellulose via alkali treatment was applied for in situ formation of AgNPs on cotton fabrics. Alkali-activated fabrics were treated with silver nitrate solution and subsequently with ascorbic acid which caused deposition of AgNPs to the fibers surface. The effect of NaOH concentration on the nanoparticle morphology, degree of functionalization, colorimetric changes, and antibacterial properties is also investigated.
Experimental
Materials
Sodium hydroxide (Caustic soda) pellets (NaOH, 99%), silver nitrate (AgNO3, 99.9% pure), and ascorbic acid (C6H8O6, extra pure crystals) were purchased from Merck, Germany. Desized, scoured, and bleached plain weave 100% cotton fabric (the wrap density of 40 yarns/cm, the weft density of 20 yarns/cm, and the fabric weight of 230 g m−2) was supplied by Yazd Baf Co., Ltd., Iran. The fabric was washed with nonionic detergent before use. Distilled water was used throughout the experiment. All experiments were performed at room temperature (25 ± 2°C).
Alkali treatment
Alkali treatment was carried out in slack condition, without tension, because the aim is to study the surface activation of cotton cellulose and its effect on adsorption and formation of AgNPs. One gram cotton fabric was dipped into the 30 mL aqueous solutions of NaOH for 5 min at room temperature. Fabrics were then removed from the bath and extensively washed with distilled water. NaOH concentration in the alkali bath was varied, using concentrations of 1–6 M.
In situ synthesis of AgNPs on fabrics
Untreated and alkali-treated fabrics (immediately after treatment without neutralizing and drying) were immersed into the 0.02 M AgNO3 aqueous solution (50 mL) and left for 30 min. Then, the fabrics were removed from the bath and rinsed completely with water for removing the loosely adsorbed Ag+ ions. AgNPs were then synthesized by immersing fabrics into the 50 mL aqueous solution of ascorbic acid (0.01 M) for 30 min. The samples were subsequently removed from the bath, rinsed thoroughly with water, and then immersed in 0.5% aqueous acetic acid at room temperature for 15 min. Samples were rinsed in cold water, then with ethanol, and dried in oven at 65°C. The as-prepared samples were labeled as Cot-0, Cot-1, Cot-2, Cot-3, Cot-4, Cot-5, and Cot-6 in increasing order of NaOH concentration used in alkali bath.
Scanning electron microscopy
Surface morphology and surface composition of samples was investigated using an AIS-2100 scanning electron microscope (SEM) equipped with energy-dispersive X-ray spectroscopy (EDS). Fabric samples were mounted on conductive carbon adhesive tabs and then were sputter-coated (SC 7620 EMITECH) with a thin layer of gold before imaging. The operation voltage was adjusted at 10 kV.
X-ray diffractometry
X-ray diffraction (XRD) patterns of samples were recorded on a Philips PW3040 X-Ray diffractometer system by monitoring the diffraction angle from 5° to 80° (2θ) using monochromatized Cu Kα (k = 1.54051 A°) radiation.
Silver content
Inductively coupled plasma-optical emission spectrometry (ICP-OES) CCD simultaneous was used on Varian Vista Pro (argon plasma, Ag 328.068-nm excitation, Ag sensitivity 0.004 mg L−1), Australia, to measure the quantity of silver concentration on fabric samples [4].
Color measurements
Color measurements were performed on a Lambda 35 UV-visible spectrophotometer using the CIE L* a* b* color space at D65/10°. The color strengths (K/S value) of the untreated and AgNPs-loaded fabrics were measured using the equation (1):
The color difference (ΔE) between the untreated and the treated cotton samples was calculated based on equation (2):
Antimicrobial activity
Antibacterial activity of the fabrics was studied using ISO 20645-2004 (agar diffusion plate method). Staphylococcus aureus (S. aureus, ATCC 25923, Gram-positive bacterium) and Escherichia coli (E. coli, ATCC 25922, Gram-negative bacterium) were used as model challenge microorganisms. In this method, two-layered agar plates were prepared. The lower agar layer consisted of 10 mL of ordinary agar whereas the upper layer consisted of 5 ± 1 mL agar inoculated with bacteria, whereby 1 mL of bacteria working solution with a concentration of 1–5 × 108 colony forming units per milliliter (CFU/mL) was added per 150 mL of agar. Perpendicular cotton specimens, about 50 × 10 mm, were uniformly pressed on the agar and incubated for 24 h at 37 ± 1°C. After incubation, the antibacterial effect of the textiles was assessed by evaluating different parameters; growth of the bacteria underneath of and the presence of at least 1 mm of inhibition zone around the specimen. Ideally, no bacterial growth should be observed on the fabric. Thus a “no growth” category represents “ideal” antibacterial efficacy; that is, no bactericide elutes into the bulk of the agar with no bacterial growth on the cotton.
Laundering durability
In order to get sufficient rubbing and reflect the true laundering durability under normal conditions, the treated cotton fabrics were washed with other loading fabrics. The samples were washed for 10 cycles (according to the ISO 105-C10:2006(C)) with 5 g L−1 soap solution, having liquor ratio of 50:1, at 50°C for 45 min. The samples were rinsed in cold distilled water and held under cold tap water for 10 min and dried at room temperature. Antibacterial test was carried out on washed samples.
Mechanical properties
Tensile strength and elongation of untreated, alkali-treated, and AgNPs-loaded alkali-treated fabrics were evaluated using a MESDAN LAB instrument according to the standard test method of ISO 5081 (strip method). The constant cross-speed of approximately 50 mm min−1 was used throughout the experiments. Measurements were performed in the warp direction of fabrics. Average values were obtained from measurements of four samples.
Results and discussion
Alkali treatment of cellulose is an important initial step in the production of many cellulose derivatives as cellulose is activated by the alkali solution for the main reactions [36]. In this research, the effect of alkali treatment on immobilization of AgNPs to cotton fabric was investigated. The alkali treatment of cotton with NaOH results in transformation of non-active cellulose to alkali-activated cellulose. The sodium salt of Cell.O−Na+ was exchanged to the silver salt of Cell.O−Ag+ (due to higher electronegativity of silver (1.93) toward sodium (0.93)) in the aqueous solution of AgNO3. In situ formation of AgNPs on fabrics was carried out via reduction of Cell.O−Ag+ by treatment with ascorbic acid solution.
Surface morphology
Figure 1 shows SEM images of modified fabrics as a function of NaOH concentration used in alkali treatment. It can be observed that a few numbers of AgNPs was formed on the surface of native cotton (Figure 1(a)). Interestingly, for the alkali-treated fabrics, the population of the deposited AgNPs is much higher and samples treated at higher concentrations of NaOH contain more particles. Particles are uniform and evenly distributed on the fabrics. For samples treated at lower NaOH concentrations, the formed particles are mono-disperse whereas fabrics treated at higher concentrations contain agglomerated particles. It is obvious that for the sample treated at 3 M NaOH, particles started to become agglomerate and the population of agglomerated particles is growing from 3 M to 6 M. The sample treated at highest NaOH concentration (6 M) is composed of a lot of agglomerated particles with the same shape and size. The SEM observations demonstrate that the formation of Cell.O−Na+ thorough alkali treatment served as effective host functional groups to introduce guest metallic ions with strong ionic interactions. The silver ions aggregate with each other by chemical reduction, resulting in AgNPs immobilized onto cellulose fibers with a high-density growth.
Scanning electron microscope (SEM) images of silver nanoparticles (AgNPs)-loaded cotton fabrics: (a) Cot-0, (b) Cot-1, (c) Cot-2, (d) Cot-3, (e) Cot-4, (f) Cot-5, and (g) Cot-6.
Surface chemical composition
EDS was used to characterize the surface chemical structures of cotton fabric before and after AgNPs deposition, as shown in Figure 2. For the untreated fabric, peaks corresponding to C and O were observed (Figure 2(a)). A strong peak of Au at ca. 2 keV was also seen. This peak corresponds to the gold coating of fabric for SEM imaging. After surface modification with AgNPs, new peak appeared at ca. 3 keV, which is attributed to silver signal (Figure 2(b)). This result together with the SEM images is strong evidence of the formation of AgNPs on the surface of cotton fabrics.
Energy-dispersive X-ray spectroscopy (EDS) spectra of the fabrics: (a) untreated and (b) (AgNPs)-loaded alkali-treated (sample Cot-6).
Silver content
Silver content of the (AgNPs)-loaded fabrics determined by ICP-OES technique.

According to these results, the concentration of NaOH solution used in alkali treatment has a significant effect on the amount of AgNPs deposited on the fabrics. It can be assumed that treating with higher NaOH concentration results formation of more cellulosate (Cell.O−Na+), which possess higher Ag+ adsorption, and therefore higher possibility to create oxygen-silver bonds. Consequently, the amount of AgNPs formed on the fabrics increased. These results clearly show the significant effect of alkali treatment on increasing the amount of AgNPs deposited on cotton fabrics and are consistent with the SEM observations.
Color changes of the fabrics
Color coordinates (CIE L* a* b*), color differences (ΔE), and color strengths (K/S) of the untreated and (AgNPs)-loaded fabrics.

The K/S value is commonly used to represent the amount of dye fixation or dye content of a given textile. Due to the coloring of the fabrics provided by AgNPs (the particles do absorb certain wavelength of light through the surface Plasmon effect and the spectrophotometer can measure the increase in absorbance due to the deposition of the particles), we can use the increase of the K/S value as a characteristic of the increased deposition of the AgNPs on the fabrics. The higher K/S value shows greater color intensity and hence better AgNPs deposition. The K/S values of the samples are presented in Table 2. The data show that all the AgNPs-loaded samples are significantly darker in color (i.e. higher in K/S values) than the untreated ones. It can be seen that the K/S value for the fabric Cot-0 is closer to the untreated fabric, because of the very low silver content, when compared with the increase in K/S value for alkali-treated fabrics. The K/S value increased with the increase of alkali concentration, which shows higher loading of AgNPs to the samples. The spectrophotometry assessment of the fabrics color is consistent with the observation on the SEM images and also the data obtained from silver content.
XRD analysis
XRD patterns for the untreated fabric and AgNPs-loaded fabric (Cot-6) are shown in Figure 3. The strong diffraction peaks at 2θ = 14.9°, 16.8°, 22.8°, and 34.7° are characteristic peaks of cellulose I crystalline form. The new peaks that appeared at 2θ = 38.1°, 44.3°, 64.5°, and 77.5° represent (1 1 1), (2 0 0), (2 2 0), and (3 1 1) planes of silver, respectively. The silver oxide phase was not detected in the deposit. The mean particle size of nanoparticles is estimated to be 28 nm by analyzing the broadening of the (111) reflection calculated by the Scherrer equation [38]:
X-ray diffraction (XRD) patterns of the fabrics: (a) untreated and (b) (AgNPs)-loaded alkali-treated (sample Cot-6).

By comparing XRD patterns shown in Figure 3(a) and (b), the characteristic peak intensity of the cellulose substrate is obviously decreasing, because of the structural changes of cellulose during alkali treatment and also the high covering degree of silver deposition.
Antimicrobial assessment
The antibacterial activity of the treated fabrics was determined using the Gram-positive bacterium S. aureus (Figure 4) and the Gram-negative bacterium E. coli (Figure 5). The antibacterial activity was assessed by evaluating different parameters; the growth of the bacteria underneath of and over the fabric and the presence of at least 1 mm of inhibition zone around the fabric (Table 3). Control samples (untreated fabric) did not show the presence of the inhibition zone for the S. aureus or E. coli strains. The growth of bacteria was also observed underneath the specimens, which demonstrate the lack of antibacterial effect. Despite the absence of inhibition zone for the untreated fabric containing AgNPs (Cot-0), the growth of bacteria was not observed on the agar medium underneath the sample, which shows its ability to inhibit the growth of bacteria. A significant inhibition zone (at least 1 mm wide) was observed in all alkali-treated samples in contact with S. aureus (Figure 4 and Table 3), indicating complete inhibition of the bacterial growth. The inhibition zone is greater for samples Cot-5 and Cot-6, due to the higher silver content. In the case of E. coli, the AgNPs-loaded fabrics did not show significant inhibition zone and only a very narrow line was observed around the samples (Figure 4 and Table 3). The agar medium underneath these fabrics was assessed, which is free of bacterium growth, demonstrating good antibacterial activity. The difference in inhibition zone for S. aureus and E. coli suggested that the AgNPs/cotton fabrics had better antimicrobial effects against S. aureus and Gram-positive bacteria than Gram-negative E. coli [39]. In summary, the tests clearly show that the presence of the AgNPs on the fabrics caused significant antibacterial effect on both Gram-positive and Gram-negative strains.
Antibacterial activities of the fabrics: (a) control, (b) Cot-0, (c) Cot-1, (d) Cot-2, (e) Cot-3, (f) Cot-4, (g) Cot-5, and (h) Cot-6 placed on the agar plate inoculated with Staphylococcus aureus. Antibacterial activities of the fabrics: (a) control, (b) Cot-0, (c) Cot-1, (d) Cot-2, (e) Cot-3, (f) Cot-4, (g) Cot-5, and (h) Cot-6 placed on the agar plate inoculated with Escherichia coli. Tested response of the bacteria strains to the untreated (control) and (AgNPs)-loaded fabrics, before and after 10 cycles washing.
a
The + indicates the growth underneath of the fabric; ++ indicates the no growth underneath of the fabric; +++ indicates the presence of a slight (less than 1 mm) inhibition ring around the textile; and ++++ indicates the presence of a significant (at least 1 mm) inhibition ring around the textile.


In order to determine the laundering durability of the AgNPs-loaded fabrics, washing was carried out according to ISO 105-C02:1989(E) standard method. The treated fabrics were subjected to repeated launderings (10 cycles) and were evaluated by antibacterial activity test. The obtained results are given in Table 3. Compared with the results of antibacterial activity before laundering, only for the sample Cot-0, antibacterial activity is reduced. This may be due to the low silver content/low stability of AgNPs and their removal from the fabric surface, during laundering treatment. In the case of alkali-treated fabrics, it is clear that the antibacterial activity did not change, which demonstrate high durability of the fabrics against laundering.
Mechanical properties
The textile performance of the modified cotton samples were evaluated in terms of its strength properties. The results of the tensile strength and elongation are shown graphically in Figures 6 and 7, respectively. The modified samples exhibited lower tensile strength when compared with the untreated ones (Cot-0). Within the limits of experimental accuracy, the tensile strengths of the alkali-treated fabrics were essentially constant for the range of NaOH concentration used (Figure 6) and comparatively small differences in tensile strengths of the fabrics were observed. However, continued shrinkage of the fabric with increased NaOH concentration was indicated by the rise in elongation (Figure 7). As the NaOH concentration increased up to 6 M, elongation of the fabrics increased up to 40%. The AgNPs-loaded alkali-treated fabrics were similar to alkali-treated fabrics in their effects.
Tensile strengths (N) of the cotton fabrics. Elongation (%) of the cotton fabrics.

Conclusions
The novel strategy for in situ deposition of AgNPs on cotton fabrics was reported by pre-activation of cotton cellulose via alkali treatment. The obtained results demonstrated that higher electronegativity of silver to sodium caused adsorption of silver ions to sodium cellulosate, which after chemical reduction lead to simultaneous synthesis and deposition of AgNPs on cotton fabrics. Alkali-treated fabrics have much higher silver content than those untreated fabrics, so that the sample treated at 6 M NaOH is completely covered with AgNPs. By increasing the concentration of NaOH, used in alkali treatment, silver content increased from 0.35 g kg−1 and reached to 17.32 g kg−1. The modified fabrics exhibited high antibacterial effect against S. aureus and E. coli as indicated by the inhibition zone and the absence of growth underneath and on the top of the fabrics. The methodology developed in this work to generate AgNPs functionalized cotton fabrics presents several advantages over those reported in literature and it is expected that this simple method can be applied for the functionalization of cellulosic textiles with other nanoparticles.
Footnotes
Funding
This research project has been financially supported by the Research Council of the Yazd Branch, Islamic Azad University.
Acknowledgment
We gratefully acknowledge Payame Noor University of Yazd, Sciences and Nanotechnology Research, for providing the facilities for testing antibacterial activities.