Abstract
In this work, microemulsion and hydrothermal methods were combined to synthesize hydroxyapatite nanomaterials. Competitive adsorption kinetics of aqueous Pb2+ and Cu2+ on nano-hydroxyapatite surfaces were investigated by performing adsorption kinetic experiments. Results showed that the adsorption quantity of Pb2+ on nano-hydroxyapatite was higher than that of Cu2+ in the single system. However, the competitive adsorption of Pb2+ and Cu2+ occurred in the coexistence system of Pb2+ and Cu2+. The adsorption quantity of Cu2+ on nano-hydroxyapatite increased, whereas that of Pb2+ decreased. The adsorption quantities of Pb2+ and Cu2+ on nano-hydroxyapatite presented a marked linear relationship with Ca2+ release from nano-hydroxyapatite (
Introduction
Nano-hydroxyapatite (nano-HAP), a slightly alkaline substance with good biocompatibility, is in good harmony with the environment and will hardly cause secondary pollution. Since the 1990s, artificial synthesis methods for nano-HAP preparation have increased; such strategies mainly include natural combustion method, 1,2 hydrothermal method, 3 sol–gel method, 4,5 and ultrasonic method. 6 Hydrothermal method is a method that makes indissolvable or insoluble materials dissolved and recrystallized under high-temperature and high-pressure environment in a closed reactor using water solution as reaction medium. It can be used to grow various monocrystals and prepare ceramic powder and inorganic fiber– or whisker-reinforced materials with controllable size, no or few agglomeration, and highly pure and perfect crystals. However, hydrothermal method is mainly disadvantageous of high requirements on device strength and high cost.
The basic principle of sol–gel method is that active monomers are produced through hydrolysis reaction of metal alkoxides or inorganic salts. Next, polymerization and gelling of solute are implemented, followed by gel drying and roasting. Finally, inorganic nanomaterial is gained. Particles prepared by sol–gel method have good chemical homogeneity, high purity, and small size. Moreover, this method accommodates indissolvable or insoluble components and dried gel particles that have low sintering temperature. Nevertheless, this method has some shortcomings, such as poor sinterability between gel particles as well as poor sinterability and remarkable shrinkage upon drying of bulk material.
Natural combustion method is based on sol–gel method and can realize in situ oxidation under low temperature through reactions between nitrate and carboxylic acid. It can synthesize primary powder of products quickly, shortening the preparation period significantly.
Sarig and Kahana produced the supersaturated solution of HAP by fast microwave radiation heating and got about 2- to 4-μm microspheres formed by microcrystal aggregates of HAP after quick cooling. 7 It is important to note that since Sarig and Kahana accomplished the experiment in extremely dilute solution, such HAP microsphere preparation method has some limitations in industrial applications. Most nano-HAPs synthesized by these methods tend to aggregate. Thus, research on modification of nano-HAPs has been developed. Considering the characteristics of nano-HAP, such as small particle size, large specific surface area, and absence of secondary pollution in the environment, nano-HAPs have been commonly used as mineral materials to govern sewage and soil pollution. Given the strong adsorption capacity of nano-HAP for heavy metal ions in sewage, it is generally used to remove lead ions, 8 chromium (II) ions, 9 fluorine ions, and other metals in water bodies. 10,11 Consequently, heavy metal lead and copper pollution in water bodies has become one of the environmental pollution problems deserving the most concern in modern society. As heavy metal lead and copper pollution requires difficult deposition and exerts long effect duration, this challenge has drawn more profound attention than many other environmental problems. Faced with increasingly strict environmental standards in various countries, toxic substances should be urgently removed from water bodies. Liu and Peng conducted a series of experiments on deposition of Pb2+ and Cd2+ in sewage using natural phosphate ore and artificially synthesized nano-HAP, 12 and their results showed that apatite demonstrated good removal efficiency on most heavy metal ions. Corami et al. found that HAP can sustain the adsorption of copper and zinc ions in aqueous solution with adsorption rates of 97.3–98.6% and 94.2–97.7%, respectively. 13 Compared with single-metal adsorption, the adsorption capacities of Cu2+ and Zn2+ in two heavy metal systems correspondingly reduced by 13% to 76% and 10% to 63% because of competitive adsorption of the two heavy metals. However, for different ions, particularly lead ions and other heavy metal ions, the competitive adsorption mechanisms remain unclear.
The current study first adopted a combined hydrothermal microemulsion method to prepare HAP and characterize its crystal structure. In addition, this study investigated the competitive adsorption capacities of Pb2+ and Cu2+ on nano-HAP and discussed the competitive adsorption mechanisms during coexistence of Pb2+ and Cu2+. Overall, this study will provide theoretical foundation for the application of nano-HAP in water treatment and polluted soil remediation.
Reagents and instruments
Ultrapure water (prepared by Milli-Q ultrapure water purifier from Switzerland) was used in the experiments. Transmission electron microscope (TEM; JEM-2100 model, Japan), X-ray diffractometer (D/max2500VB2+/PC model, Japan), specific surface area instrument (Micromeritics Accusorb2100E model, Norcross, Georgia, USA), atomic adsorption spectrophotometer (HITACHI, Z-2000 model, Japan), earth pillar, high-pressure constant flow pump (P230 model, China), auto-fraction collector (BS-100A model, China), ultrasonic cleaner (KQ-3000VDE model, China), and Thermo Nicolet 380 model Fourier transform infrared (FTIR) instrument (Nicolet Corporation, Madison, Wisconsin, USA) were also employed.
Preparation of nano-HAP with combined hydrothermal microemulsion method
About 10 ml of 0.5 mol l−1 calcium nitrate aqueous solution was added into mixed oil phase consisting of 56 ml cyclohexane, 16 ml TX-100, and 16 ml
Experimental method
Characterization of nano-HAP
Transmission electron microscope: Japan JEM-2100 model high-resolution electron microscope was used to observe the particle morphology and particle sizes. Testing conditions: An accelerating voltage of 200 kV was adopted. X-ray diffractometer: D/max2500VB2+/PC model X-ray diffractometer from Japan Rigaku Corporation was used to assess the crystal structures. Testing conditions: copper (Cu)
Adsorption kinetics test
The testing device is shown in Figure 1. 14 The height of the packing column was 3.0 cm with inner diameter of 1.2 cm. Both ends of the packing column were correspondingly installed with cellulose acetate membrane and 20 μm nylon membrane. Up to 0.5 g of nano-HAP was weighed and poured into the column and then tamped. Pure water was used to prepare the following nitrate stock solutions of heavy metal ions with pH value of 5.5: 0.1 mmol l−1 single Pb2+ solution, 0.1 mmol l−1 single Cu2+ solution, and complex solution [n(Cu)/n(P) was 1] of 0.1 mmol l−1 Cu2+ and 0.1 mmol l−1 Pb2+. Simultaneously, pure water was taken as contrast. Calorstat was kept at 25°C, after which high-pressure pump was started. Afterward, the solution was pumped from the bottom up into the packing column through high-pressure constant-flow pump. The flow velocity of the stock solution, which flowed through the packing column was 1.0 ml min−1. An auto-fraction collector was used to collect outflow solution within a certain time interval. Pb2+, Cu2+, and Ca2+ concentrations in the collected solution were measured by an atomic adsorption spectrophotometer.
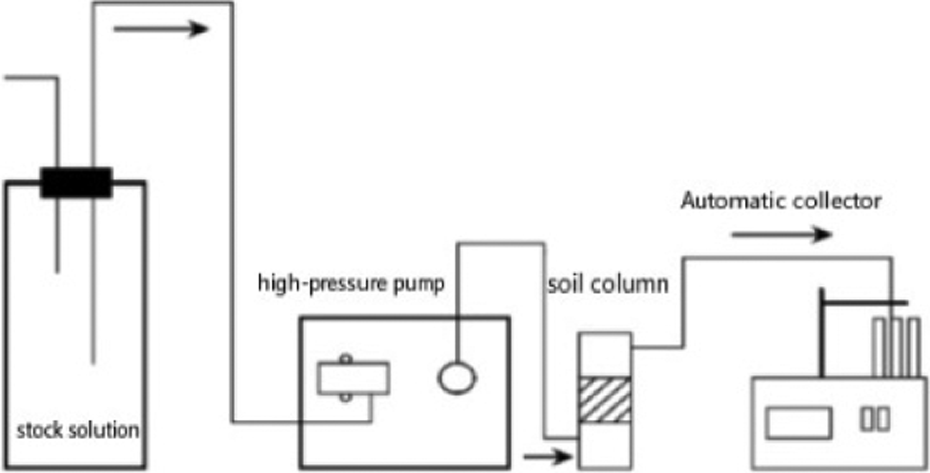
A dynamic adsorption apparatus.
Measurement of ζ electric potential of nano-HAP colloid suspension
Up to 0.50 g of nano-HAP powder was accurately weighed before and after the test (nano-HAP was removed after the reaction from the material column, washed with deionized water, and dried), and a constant volume was achieved with pure water. The solutions were then poured into a 1-L volumetric flask, which was shaken well to achieve a mixture. Subsequently, the solutions were subjected to ultrasonic dispersion for 30 min, and nano-HAP suspension was prepared. After standing for 2 days, the mixture was shaken well, and a superstratum suspension was adsorbed to measure the ζ electric potential.
Data analysis
The ion adsorption quantity of nano-HAP was calculated on the basis of the initial adsorbent concentration and the concentration of outflow solution.
15
–17
The adsorption quantities of Cu2+ and Pb2+ on nano-HAP were calculated using the equation
Results and analysis
Characterization of nano-HAP
Figure 2 shows the TEM graph of nano-HAP. Nano-HAP is rod shaped with an average particle size of 140 nm and a diameter of 20 nm. The X-ray diffraction (XRD) analysis results are consistent with those on HAP standard card (JCPDS 9-432). The value of
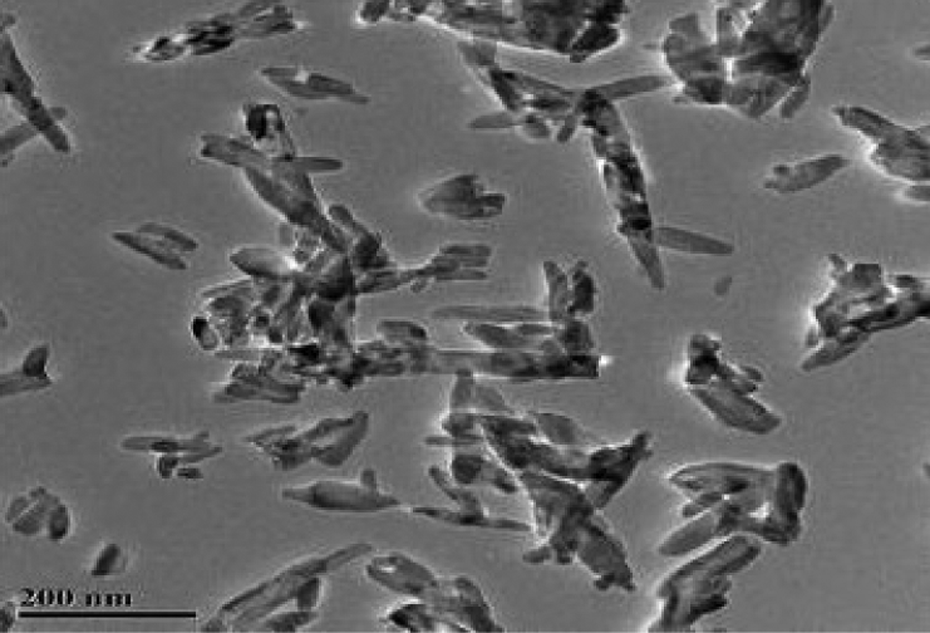
TEM imaging of nano-HAP. TEM: transmission electron microscopy.
The XRD spectrogram of the nano-HAP specimen is shown in Figure 3(a), in which the positions of all diffraction peaks of the nano-HAP specimen are consistent with those of the nano-HAP standard cards. No other impure peaks were observed, indicating that the specimen prepared in this experiment was HAP. After calculation, the particle size of HAP prepared in this experiment was 24.39 nm, and it is considered as nano-HAP.

XRD and FTIR spectrogram of the particle samples. XRD: X-ray diffraction; FTIR: Fourier transform infrared.
Figure 3(b) shows that at 3435 and 631 cm−1 –OH stretching vibration peak and bending vibration adsorption peak, the 567, 603, 959, 1030, and 1091 cm−1 adsorption peaks of
Pb2+ and Cu2+ competitive adsorption kinetics on nano-HAP
Figure 4 shows the competitive adsorption curves on nano-HAP. The following was indicated by the adsorption quantity per unit time in Figure 4(a): With progressing time, regardless whether under single or coexisting conditions, the adsorption quantities per unit time on the HAP surface of Pb2+ and Cu2+ gradually decreased and reached equilibrium. When Pb2+ and Cu2+ coexisted in a solution, competitive adsorption occurred, and the adsorption quantities and equilibrium time of Pb2+ and Cu2+ slightly changed compared with those under single-ion existing conditions. When the adsorption reached a saturation state, the sizes of adsorption quantity per unit time were satisfied as follows: Pb2+ (single) > Pb2+ (coexistence) > Cu2+ (coexistence) > Cu2+ (single).

Adsorption kinetics of Pb2+ and Cu2+ on nano-HAP. HAP: hydroxyapatite.
At the preliminary stage of adsorption reaction, many adsorption sites were activated on the nano-HAP surface, and Pb2+ and Cu2+ can be simultaneously adsorbed onto the nano-HAP surface. With continuous inflow of the adsorption solution, the activated adsorption sites on the nano-HAP surface gradually decreased. Consequently, competitive adsorption among the ions intensified, and the ions with more rapid adsorption rates would more easily undergo adsorption reaction and would be adsorbed in large quantities. 18 Hence, at a later stage of adsorption reaction, the adsorption quantity of Cu2+ per unit time was higher than that of Pb2+.
Figure 4(b) shows that with increasing equilibrium time, the accumulated adsorption quantities of Pb2+ and Cu2+ on nano-HAP gradually increased, and the size rule of accumulated adsorption quantity was consistent with that of the adsorption quantity per unit time. Existing studies show that the adsorption process of Pb2+ on HAP mainly focused on dissolution and precipitation, that is, Pb2+ and Ca2+ conducted ion exchange and generated HAP precipitation involving heavy metals. The adsorption process of Cu2+ involves surface complex adsorption on the adsorption sites of the HAP surface. Considering the difference between the two adsorption processes, influences exerted by them during competitive adsorption also varied. When Pb2+ and Cu2+ coexisted, the accumulated adsorption quantity of Pb2+ was obviously lower than when only Pb2+ was present. This finding indicates that the existence of Cu2+ competes against the adsorption sites of Pb2+ on HAP, which prioritizes generation of stable Pb2+ HAP when coexisting with Pb2+. 19 –21 In the reaction of HAP removing Pb2+, the Pb2+ removal rate of HAP decreased with increasing initial Pb2+ concentration. In the experiment, as the stock solution continuously entered the material column, the quantity of Pb2+, which can react with HAP in the material column, continuously increased, whereas the Pb2+ removal rate of HAP decelerated with increasing quantity of Pb2+. Moreover, the existence of Cu2+ competed against the adsorption sites of Pb2+ on HAP, thereby resulting in lower accumulated adsorption quantity of Pb2+ compared with sole Pb2+. By contrast, the accumulated adsorption quantity of Cu2+ was obviously higher than when it solely existed.
Figure 5 shows the kinetic outflow curves of Pb2+ and Cu2+ on nano-HAP. With gradually increasing metal ion adsorption quantity, Ca2+, which was exchangeable in nano-HAP, was replaced, and the outflow quantity of Ca2+ gradually increased. The accumulated outflow quantity rules of Pb2+ and Cu2+ in Figure 5(a) are consistent with the accumulated adsorption quantity rules of Pb2+ and Cu2+, that is, greater accumulated outflow quantity indicates less accumulated adsorption quantity. The sizes of accumulated outflow quantities were satisfied in the following order: Cu2+ (single) > Cu2+(interaction) > Pb2+ (interaction) > Pb2+ (single). As shown in Figure 5(b), when single Pb2+ existed, the accumulated release quantity of Ca2+ was obviously higher than when single Cu2+ existed, and the dissolving-out quantity of Ca2+ when Cu2+ existed was approximate to that when pure water went beyond the column. This finding indicated that the adsorption process of Cu2+ on nano-HAP mainly focused on statics and physical adsorption and did not cause a large dissolving-out quantity of Ca2+. However, when Pb2+ existed, the accumulated release quantity of Ca2+ obviously increased, and this result indicated that Pb2+ conducted ion exchange with Ca2+ in HAP, thereby causing a large quantity of Ca2+ released. The Ca2+ outflow quantity when Pb2+ and Cu2+ coexisted was less than that when Pb2+ solely existed. This finding indicated that the existence of Cu2+ influenced Pb2+ adsorption on nano-HAP and reduced the quantity of Ca2+ exchanged during competitive adsorption. The accumulated quantities of Pb2+ and Cu2+ and the accumulated release quantity of Ca2+ on nano-HAP were fitted, and a good linear equation was obtained (Table 1).

Effluent concentration of Pb2+ and Cu2+ on nano-HAP. HAP: hydroxyapatite.
Liner relationships between Pb2+ and Cu2+ adsorption quantities on nano-HAP and Ca2+ release quantities from nano-HAP.

HAP: hydroxyapatite.
ζ electric potential variation on nano-HAP surface before and after the experiment
After nano-HAP adsorbed Pb2+ and Cu2+, the ζ electric potential on the surface experienced a certain change (Table 2). In the pure water contrast test, as a large quantity of Ca2+ flowed with the outflow solution, the positive charge in the nano-HAP surface-diffused double layer decreased. Thus, the ζ electric negative potential increased compared with that of the original nano-HAP. During single Cu2+ adsorption, the nano-HAP surface mainly experienced static adsorption, and this adsorption did not change the quantity of superficial positive charges within the diffused double layer. As such, the ζ electric potential was relatively small.
ζ-Potentials change of nano-HAP colloids after adsorption kinetics.

HAP: hydroxyapatite.
During single Pb2+ adsorption, the electronegativity of the ζ electric potential on the nano-HAP surface obviously decreased. This finding indicated that a large quantity of Pb2+ diffused onto the internal surface of the nano-HAP surface-diffused double layer. Consequently, the positive charges increased, whereas the negative electric potential of the whole nano-HAP decreased. When Pb2+ and Cu2+ coexisted, the ζ electric potential on the nano-HAP surface increased compared with the original nano-HAP electronegativity, but the potential was quite approximate to that of single Cu2+ disposition. When Pb2+ and Cu2+ coexisted, Cu2+ affected the entry of Pb2+ into the internal surface of HAP surface-diffused double layer. Thus, a slight variation of ζ electric potential occurred.
Data equation fitting to adsorption kinetics of Pb2+ and Cu2+ on nano-HAP
The adsorption kinetic processes of Pb2+ and Cu2+ on the nano-HAP surface under single-ion environment and compound-ion environment were correspondingly fitted. The first-order kinetic equation, second-order kinetic equation, Elovich equation, parabolic diffusion equation, double-constant equation, and Lennard–Jones (LJ) equation
22
were then adopted (Table 3). The kinetic models of adsorption reaction of Pb2+ and Cu2+ on the nano-HAP surface under single-ion environment and compound-ion environment varied. Generally, the fitting degrees of models were ranked as follows: second-order kinetic equation > double-constant equation > parabolic diffusion equation > LJ equation > Elovich equation > first-order kinetic equation. In particular, second-order kinetic equation and double-constant equation can better fit the adsorption process of Pb2+ and Cu2+ on the nano-HAP surface, and the correlation coefficient satisfied
Adsorption kinetic equations of Pb2+ and Cu2+ on the nano-HAP.

HAP: hydroxyapatite; LJ: Lennard–Jones.
According to the kinetic characteristics of the models, if the reaction conformed to second-order kinetic equation, then this reaction process exhibited kinetic characteristics of the chemical reaction with the reaction order being 2. If the reaction conformed to a double-constant model, this reaction was a complicated kinetic process, and the reaction process exhibited adsorption characteristics featured as follows: reaction rate presented power-function exponential-order increasing tendency. Hence, the adsorption kinetics of Pb2+ and Cu2+ on the nano-HAP surface in single-ion and compound-ion environments represented a complicated adsorption process, in which both chemical reaction and physical adsorption occupied a certain proportion.
Conclusions
When single Pb2+ or single Cu2+ existed, the accumulated adsorption quantity of Pb2+ on the nano-HAP surface was higher than that of Cu2+. When Pb2+ and Cu2+ coexisted, both ions would experience competitive adsorption on the nano-HAP surface, and the accumulated adsorption quantity of Cu2+ increased as the accumulated adsorption quantity of Pb2+ decreased.
The adsorption mechanisms of Pb2+ and Cu2+ on nano-HAP showed an obvious difference. The adsorption process of Pb2+ was chemical adsorption, that is, Pb2+ and Cu2+ conducted ion exchange and generated HAP precipitation containing heavy metals. By contrast, the immobilization process of Cu2+ was static with physical adsorption occurring on the HAP surface by implementing surface complexation with special sites on the HAP surface. Different adsorption mechanisms influenced the competitive adsorption behavior on nano-HAP when Pb2+ and Cu2+ coexisted.
The first-order kinetic equation, second-order kinetic equation, parabolic equation, Elovich equation, double-constant equation, and LJ equation were all used to conduct the fitting of adsorption kinetic processes of Pb2+ and Cu2+ on nano-HAP. Among these equations, second-order kinetic and double-constant equations presented better fitting results, thereby indicating that the adsorption reaction kinetics of Pb2+ and Cu2+ on the nano-HAP surface represented a complicated adsorption process in which both chemical reaction and physical adsorption occupied a certain proportion.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by the National Natural Science Foundation of China (40871005) and funded by the Scientific Research Foundation of the Changchun University of Science and Technology. This project was also supported by the “Simulation and regulation of nitrogen and phosphorus transfer in small drainage basins on the periphery of urban areas” research program, which is funded by the 13th Five-Year Scientific Research Program of the Jilin Province Department of Education.
