Abstract
Polylactic acid-based membranes received considerable attention due to its novel biocompatibility, renewability, and biodegradability. In this study, PLA electrospun nanofibrous membrane was prepared and 2-dimethylaminoethyl methacrylate (DMAEMA) was used as a monomer for surface grafting of polymer chains via the atom transfer radical polymerization method. Then the PLA nanofibers were quaternized by using bromoethane. The characterization of poly(DMAEMA) graft PLA nanofiber (poly(DMAEMA)-g-PLA) membranes was done by scanning electron microscopy, Fourier transform infrared spectroscopy, X-ray photoelectron spectroscopy, and energy dispersive X-ray spectroscopy. The results showed that the diameter of PLA nanofibers increased 15% as the concentration increased from 10% to 12% and then increased 23% as the concentration of PLA solution increased from 10% to 15%. But the regularity of average diameters is best achieved at 12% concentration.
Keywords
Introduction
Poly(lactic acid) or polylactide (PLA) is a biodegradable thermoplastic polyester polymer. The past researches have proved it as the potential replacement of conventional petrochemical polymers [1]. A vast field of research has been done to modify it and made it usable for different applications including biomedical fields. Its physical and mechanical properties can be deployed with its polymer architecture [2].
The PLA has the advantage to provide reactive sites to other materials and making it very favorable to use as a base material for the antimicrobial agents. Along with the reactive sites, PLA has a web-like physical structure to encapsulate the small molecules [3,4]. This encapsulation occurred due to the availability of a certain number of sites produced by the chemical active ends and physical appearance. These physical and chemical traps play a key role in enzyme immobilization [5].
Nowadays, electrospinning technology has been largely applied in the areas of battery electrode materials [6], sensors [7], tissue engineering scaffold [8], and drug carrier [9] that rely upon on manageable fiber diameter, high specific surface area, high porosity and cost-effective. With the progress of research, further materials including high polymers, inorganic fillers, and so on were used in the fabrication of nanofibers by electrospinning [1,2].
Atom transfer radical polymerization (ATRP) was selected to advocate the active sites on the surface of the nanofiber membrane to increase the amount of enzyme entrapped per unit weight [5]. It has been often used for making linear polymer chains with controlled molecular lengths; subsequently, the propagation centers do not undergo chain termination and chain transfer through polymerization [10]. Therefore, the molecular weights increase linearly with the conversion of monomers [11]. Another benefit of this technique is the use of a variety of monomers [12], for example, acrylonitrile [13], acrylate, acrylamide and many others [14].
In the present research work, the electrospun PLA nanofibrous membrane was fabricated first; subsequently, 2-dimethylaminoethyl methacrylate (DMAEMA) was selected as the monomers for surface-grafting of polymer chains onto PLA nanofibers via the ATRP method. Note that the optimal polymerization times were determined to control the length/thickness of grafted chains/brushes of poly (DMAEMA).
Materials and method
Materials
PLA (Mw = 1 × 105) was purchased from Shenzhen Esun Industry (Shanghai, China). Dichloromethane (DCM) was purchased from Alladin Chemicals and reagents (Shanghai, China) were used as solvents. H2O2 (30% solution in water) was purchased from Merch (Shanghai, China). Potassium chloride (KCl), potassium dihydrogen phosphate (KH2PO4), potassium hydrogen phosphate (K2HPO4), copper chloride (CuCl), sodium hydroxide (NaOH), sodium chloride (NaCl), hydrochloric acid (HCl), Ammonium acetate (CH3COONH4), boric acid (H3BO3), isopropyl alcohol (C3H8O), coomassie brilliant blue G250 (CBBG), chloroform (CHCl3), dimethyl sulfoxide (DMSO), N, N-dimethylformamide (DMF), diethyl aminoethyl chloride, tetrahydrofuran (THF), 2-bromoisobutyryl bromide (2-BIB), dimethylamino ethyl methacrylate (DMAEMA), acrylic acid (AA), and 1,4,8,11-tetraazacyclotetradecane were also purchased from Merck (Shanghai, China). All the chemicals and materials were used without further purifications.
Instrumentation
The morphology and properties of electrospun PLA/CTAC-MMT fibrous membranes were observed with a scanning electron microscope (SEM, SU1510, Hitachi, Japan), and energy-dispersive X-ray spectroscopic (EDS).
A Nicolet Nexus 470 FT-IR spectrometer was employed to study the formation of different functional groups on the nanofibers, and the FT-IR spectra were acquired by scanning the specimens for 32 times in the wavenumber range from 4000 to 450 cm−1 with the resolution of 4 cm−1.
X-ray photoelectron spectroscopy measurements were carried out on a Kratos XSAM800 XPS system with Kα source and a charge neutralizer.
Methods
Electrospinning of PLA
Firstly, the three separate electrospinning solution was prepared by mixing 10%, 12%, and 15% PLA on the weight of DCM at 25°C temperature. Put these mixtures on stirring for 12 h. Secondly, these solutions were filled in syringes and placed on the syringe holder at the electrospinning machine. The applied voltage was set as 15 kV with a working distance of 15 cm from the stainless steel needle tip to the collector site (circular rotating drum covered with aluminum foil), and the flow rate was kept at 0.1 mL/h and the rotating speed of the collector cylinder was set at 100 r/min for all three solutions [10]. The whole assembly is represented in Figure 1. Then the collected PLA membranes were separately hydrolyzed by immersion into 0.05 M NaOH solution at 25°C temperature for 24 h. Then washed it with distilled water three times and dried in a vacuum oven at 45°C [10,15].
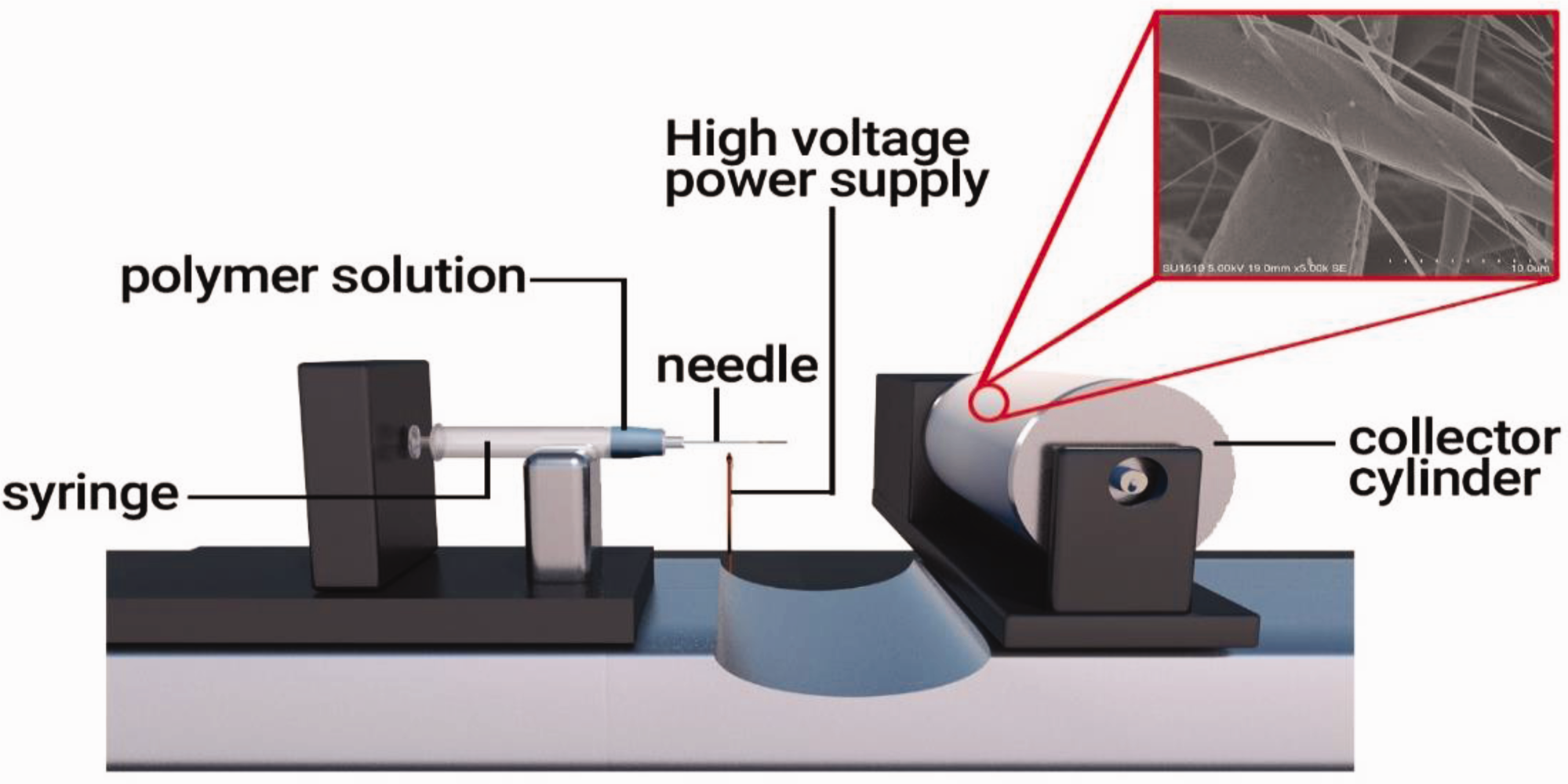
Schematic illustration of the electrospinning technique for the preparation of PLA fibrous membranes.
Polymerization
Initiation
The electrospun PLA nanofibrous membrane was firstly immersed in 20 mL THF for 10 min. After that, it was placed in a mixture of 63 µL 10 mM 2-BIB, 70 µL 10 mM TEA, and 50 mL THF at 35°C for 3 h. It was then thoroughly rinsed with THF and deionized water [15].
Propagation and termination
A mixture was prepared by adding 20.24 mL DMAEMA, 300 µL HMTETA, and 39.76 mL 2-propanol and deoxygenated by using freeze-pump-thaw cycling technique. It was stirred in the oxygen-free glovebox for 2 h and then 50 mg CuCl was added. Then pre-initiated PLA nanofibrous membrane was placed into the reaction bottle at 40°C for 12 h. Then, the reaction was quenched by adding ethanol and the resulting product was washed with DI water thrice. This polymerization is indicated in Figure 2.

Schematic showing the grafting of poly(DMAEMA), poly(AA), on the surface of an electrospun PLA nanofibrous membrane via the ATRP method.
Quaternization of bacterial PLA copolymer
Absolute ethanol 30 mL and 12 g bromoethane were added into a flask along with PLA-g-poly(DMAEMA) and placed at 60°C for 24 h. Then it was washed with ethanol, tetrahydrofuran and distilled water separately and then freeze-dried under vacuum until constant mass.
Characterization of nanofiber membrane
Surface morphologies of the PLA and PLA poly(DMAEMA) membranes were examined by a Hitachi S4800 SEM. Before the SEM examination, the specimens were sputter-coated with gold to avoid charge accumulation, and the average nanofiber diameter of each sample was obtained through measuring 100 randomly selected fibers using the software of Image Pro-Plus 3.0.
A Nicolet Nexus 470 FTIR spectrometer was employed to study the formation of different functional groups on the nanofibers. FTIR spectra were acquired by scanning the specimens for 32 times in the wavenumber range from 4000 to 450 cm−1 with the resolution of 4 cm−1.
The morphological structure and chemical composition of electrospun PLA and PLA-poly (DMAEMA) were examined by X-ray photoelectron spectroscopy (XPS, Escalab 250Xi, UK) and energy-dispersive X-ray spectroscopic (EDS).
Results and discussion
Electrospinning is a facile technique for the preparation of continuous fibers with diameters from the micrometer to the nanometer scale, though the preparation of required nanofibers with smooth morphology is dependent on many aspects such as solution viscosity, the used solvent, solution conductivity, surface tension, and the applied electric field intensity, etc. Hence, how the electro spinnable solution is prepared is a critical point. At low viscosities, surface tension is the significant influence on fiber morphology and under a certain polymer concentration drops will form instead of fibers.
Fiber morphology
Figure 3 shows the SEM images of three different solutions of PLA in PLA-g-poly(DMAEMA). In Figure 3(a), the diameters are not even, few fibers are thick and few are much thinner, and in Figure 3(b), all the fibers seem to have a uniform diameter and are very regular in shapes in contrast with Figure 3(c) where the diameter is the highest but irregularity is at peak. This thickness of fibers in these figures could be explained as the concentration of PLA in the solution decreases, the fiber diameter decreases and vice versa. But as we increase the percentage to 15%, the uniformity decreases and irregularity increases in the fiber diameters.

Effect of polymer concentration on nanofiber morphology – scanning electron micrographs with 10% PLA (a), 12% PLA (b), and 15% PLA (c).
The fiber diameter distribution histogram is shown in Figure 4. Figure 4(a) shows the average diameter of 1.3 µm and we can also see that the distribution is not even, few fibers are very thick and most of them are very thin. In Figure 4(b), the diameter distribution is even and most of the fiber has a nearly average diameter of 1.5 µm which is consistent with the data given in Figure 3(b). In Figure 4(c), the cluster lies at the highest level of the graph which means the average diameter is 1.61 µm and is highest among the three solutions but the regularity is not present which is also consistent with Figure 3(c) [16,17].

Fibre-diameter distribution of PLA fibres at 10% PLA (a), 12% PLA (b), and 15% PLA (c) concentration.
Fourier transform infrared spectroscopy
To further establish the chemical identity of the prepared nanofibers, powder X-ray diffraction (FTIR) was employed as shown in Figure 5. The spectrum acquired from the PLA-poly(DMAEMA) membrane had a band centered at 1749 cm−1; this band could be attributed to the stretching motion of carbonyl groups in DMAEMA, indicating that poly(DMAEMA) chains/brushes were generated/grafted on the surface of PLA nanofibers [18]. The needle-like peaks at 1737 and 1165 cm−1 are allocated to the carbonyl stretching C = O and stretching vibration of C–O in PLA chains. A sharp triplet of peaks at 1110, 1072, and 1025 cm−1 was linked to C–O stretching vibrations. The peaks located at 2994 and 1751 cm−1 of PLA were assigned to the stretching vibration of –CH2 and vibration of –C = O bonds, respectively. The characteristic peaks of PLA/poly-g-(DMAEMA) can be noticed from Figure 5 at 2800 and 2950 cm−1 which are due to the C–H stretching [19]. The peak for C = O bending is observed at 1751 cm−1, and the peak for C–O bending is at 1184 cm−1 [16].

FT-IR analysis graphs for electrospun PLA and PLA-g-poly(DMAEMA).
After polymerization of DMAEMA on the surface of electrospun PLA, the ratio of ethyl bending (1450 cm−1) to methyl bending (1400 cm−1) peak areas increases, and an intense increase of the absorption peak is observed at 1750 cm−1 (Figure 5 black color) conforming to the stretching vibration of an ester carbonyl group in the poly(DMAEMA).
X-ray photoelectron spectroscopy
Furthermore, XPS of the PLA/poly (DMAEMA) membrane and PLA nanofibers is shown in Figure 6, with the inset showing that the magnified images at the binding energy range from 80 to 60 eV. The survey spectrum showed the presence of C, O and Br elements (Figure 6(a)), where the large peaks observed at 533 and 282 eV correspond to O1s and C1s, respectively. The high-resolution spectrum of Br 3d located at 68 eV confirms the binding of the Br-initiator to the PLA membrane.

(a) XPS spectrum of high-resolution C/O/Br on PLA/poly(DMAEMA)) (this XPS is about PLA-Br initiator, proved the success of initiation on the surface of PLA) modified and (b) C/O on electrospun.
Energy dispersive X-ray spectroscopy analysis
For chemical characteristics, the elemental composition of electrospun PLA, and PLA-poly(DMAEMA) was determined by energy-dispersive X-ray (EDX) spectroscopy. The EDX mapping results are shown in Figure 7, where bluish, purple and black colors correspond to carbon, oxygen and Br(poly(DMAEMA), respectively. Analysis confirmed the presence of carbon and oxygen as the majority of the elements for PLA (Figure 7) [20–22]. Figure 7(a) and (b) shows the observation of O, and peaks on the EDx spectrum and the relative concentration of the above elements were about 65.46 and 34.54 wt.%, respectively.

SEM-EDX elemental mapping images of electrospun PLA. (a) C&O mapping, and SEM-EDX elemental mapping images of PLA/poly(DMAEMA) (this EDX is about PLA-Br initiator, proved the success of initiation on the surface of PLA) and (b) modified nanofibers (C&O&Br mapping).
Figure 7(b) reveals that the SEM–energy-dispersive X-ray spectroscopy (EDx) mapping images contained C, O and Br elements on the surface of PLA-Br initiator, thus indicating their uniform distribution in the PLA membrane. Additionally, Figure 7(b) shows the observation of O, C and Br peaks on the EDS spectrum, and the relative concentration of the above elements was about 60.47, 34.42 and 3.11 wt.%, respectively (Figure 7(b)).
Conclusion
Three PLA-based nanofibers were electro-spun at 18 kV and 12 mL/h rate in concentrations of 10%, 12%, and 15% separately. Poly-2-dimethylaminoethyl methacrylate (DMAEMA) of 20.24 mL was used as the monomer for surface grafting of polymer chains by ATRP polymerization technique at 40°C for 12 h. The obtained PLA nanofibers were quantized at 60°C for 24 h by a mixture of ethanol and bromoethane. The successful grafting was determined with the FTIR spectroscopy, XPS, and EDX. The surface morphology of the poly(DMAEMA) graft PLA nanofiber and simple PLA nanofibers were examined with SEM and found that the 12% concentration of PLA nanofibers solution is the best among all three as-received regular and average-sized diameter fibers at this concentration. The optimal ATRP reaction time for poly (DMAEMA) was determined at 40 min, 8 h, and 22 h. Surface modification of poly(DMAEMA) was also proved and characterized. The results also demonstrated that the diameter of microfibers was affected by varying the concentrations of PLA. In the future, researchers can make this poly(DMAEMA-g-PLA) nanofibers antimicrobial and antifungal by using different techniques like reaction with methylene blue and it can be used for different application areas.
Footnotes
Acknowledgements
This research is grateful to the nanocomposite materials with training of young academic scholars, whereas instruments support of Key Laboratory of Eco Textiles, Jiangnan University,Wuxi, Jiangsu, China.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by National first-class discipline program of Light Industry Technology and Engineering (LITE2018-21).
