Abstract
Polystyrene (PS)–poly(lactic acid) (PLA)–PS triblock copolymer is prepared with PLA and styrene (St) via atom transfer radical polymerization (ATRP). The structure and thermal properties of the copolymer are analyzed by infrared spectroscopy, gel permeation chromatography, nuclear magnetic resonance, thermogravimetry, differential scanning calorimetry and alcoholysis. The ATRP of St using bromine-terminated PLA as the initiator exhibits first-order kinetics and a low-molecular-weight distribution (M w/M n ≈ 1.5). The thermal stability of PLA is improved by copolymerization. Both the initial temperature of thermal decomposition and glass transition temperature are increased with an increasing length of PS block.
Introduction
Poly(lactic acid) (PLA) is a thermoplastic polymer that has been widely used for many medical purposes, such as suturing and bound fixture due to its excellent biodegradability by hydrolysis. Also, PLA can be thermoformed by general thermoplastic process. However, the low glass transition temperature (T g) of PLA (60°C) limits its applications. 1 T g of a polymer is an important intrinsic characteristic that influences its material properties and potential applications. Furthermore, polymers having high values of T g are attractive for industrial applications.
To improve the thermal properties and reduce the cost of production, lactic acid could be polymerized with other monomers or blended with other polymers. Attentions were focused on blending with other nanocomposites
2,3
and heat-resisting materials
4,5
to improve its thermal and mechanical properties; for example, Li and Yang used 4,4-methylene diphenyl diisocyanate to extend the chain
6
; Loo et al. devised a cross-linked structure using a cross-linking agent
7
; Mohamed et al. studied a PLA/polystyrene (PS) bioblends
8
; Shi et al. synthesized a poly(ethylene oxide) methyl ether/PS/poly(
Atom transfer radical polymerization (ATRP) has its roots in organic chemistry’s atom transfer radical addition. 11 The ATRP method has been proven effectively for a wide range of monomers (styrene (St), (meth)acrylates, acrylonitrile, etc.) either in bulk or in a variety of solvents (benzene, water, etc.). 12 The ATRP process is a transition metal-mediated living radical polymerization of vinyl monomers. However, compared with other polymerization methods, ATRP can decrease the molecular weight distribution (MWD) of a copolymer and improve the controllability of a reaction. 13,14 Consequently, ATRP has become an important method of polymer structure design. Moreover, compared with living ionic polymerization, ATRP has better controllability and can be applied to most of the monomers under mild reaction conditions. 15,16 Thus, ATRP has a considerable potential use in various industries. In particular, ATRP has been successfully employed in the synthesis of a large range of previously unknown well-defined block copolymers.
The block copolymers that have well-defined structures, such as known number-averaged molecular weight, MWD, compositions, architectures and end-group functionalities, are in high demand. In this article, a PS-PLA-PS copolymer with a controllable molecular weight was then obtained via an ATRP reaction with double bromine (Br)-terminated PLA (Br-PLA-Br) and PS. The purpose of the present study is to improve the thermal stability of PLA by copolymerizing PS with dihydroxyl-terminated PLA (HO-PLA-OH).
Experimental
Materials
The lactic acid (LA; Anhui Fengyuan Company, Anhui, China) was prepared and purified by recrystallization before use. St (Sinopharm Chemical Reagent Co. Ltd, Shanghai, China) was vacuum distilled from calcium hydride and stored under a nitrogen atmosphere at 4°C. Stannous octoate (Sn(Oct)2; Sigma, St.Louis, USA) and other reagents (Tianjin Fuchen Chemical Reagent Company, China) were used without further purification.
Preparation of polylactide
An amount of 1.0 g of purified LA, 1,4-butylene glycol (0.05%, mol%) and Sn(Oct)2 (0.1%, mol%) were added into 10 mL ampule, vacuum and backfilling with nitrogen (five cycles) were applied to remove oxygen and then sealed under vacuum. The mixture was kept to react in an oven at 130°C for 2 h; the ampule was then scraped after the reaction. All the products including glass fragments were transferred into a clean conical beaker, dissolved in chloroform and then the glass fragments were filtered out. The obtained product HO-PLA-OH was precipitated in ethanol, filtered out and dried under vacuum until it attains a constant weight.
Preparation of Br-PLA-Br initiator
Initially, 10 g of HO-PLA-OH (M n = 5490; using gel permeation chromatography (GPC)) was dissolved in the reaction bulb with 40 mL of methylene dichloride (DCM). The bulb was then kept in an ice bath, and the pyridine and α-bromopropionyl bromide (dissolved in DCM, HO-PLA-OH : pyridine : α-bromo-propionyl bromide = 1:5:10 mol) were added dropwise and stirred for 2 h at 0°C and 48 h at room temperature. The resultant pyridinium was filtered out and the filtrate was collected and precipitated with methanol to obtain Br-PLLA-Br. Drying under vacuum until constant weight was reached.
Preparation of PS-PLA-PS copolymer
Br-PLA-Br, cuprous chloride, 2,2-bipyridyl (bpy) and St (1:1:4:200 mol, respectively) were added into a 10-mL ampule, vacuum and backfilling with nitrogen (five cycles) were applied to remove oxygen and then sealed under vacuum. The polymerization was carried out at 110°C in an oven. Then the ampule was scraped, the product and glass fragments were transferred into a clean conical beaker. DCM was added to dissolve the product and then the glass fragments were filtered out. The crude product was precipitated and washed with ethanol until the wash solution became colorless, and then the product was collected and dried under vacuum until constantly weighing product PS-PLA-PS is obtained.
Characterization
Fourier-transform infrared (FT-IR) spectra of these samples were recorded from KBr-pellets (1 wt% of polymer) on a Nicolet 380 FT-IR spectrometer in the domain ranging from 4000 to 650 cm−1 at 4 cm−1 resolution (1738 data points).
Proton-nuclear magnetic resonance ( 1 H NMR) was recorded on a Bruker AVANCE spectrometer at 400 MHz. 1 H NMR chemical shifts in cadmium chloride were reported downfield from 0.00 ppm using Tetramethoxysilane (TMS) as an internal reference.
GPC was conducted using a Dionex P680 equipped with PLgel (Polymer Laboratories Inc., [Amherst, USA]; 5 µm, 300 × 7.5) and a differential refractometer (Shodex RI 101, Kanagawa, Japan). The mobile phase was tetrahydrofuran (THF) at a flow rate of 1 mL min−1 at 35°C. Polymer concentration was 1 mg mL−1 with a 100-μL injection volume. A series of narrow MWD polystyrene standards were employed to generate a calibration curve. Average molecular masses were reported as PS equivalent.
Thermogravimetric analysis (TGA) measurements were carried out in a Perkin-Elmer Pyris I DSC (Massachusetts, USA). All the samples were heated from 25 to 550°C at a heating rate of 20°C min−1.
Differential Scanning Calorimetry (DSC) is carried out in a Perkin Elmer Pyris I DSC. Samples are heated at 20°C min−1 from 0 to 150°C; fast cooled from 150 to 0°C and scanned at a heating rate of 20°C min−1 from 0 to 150°C.
Alcoholysis of the copolymer
The PLA and copolymer (0.5 g) were dissolved in THF (10 mL) and then alcoholic NaOH (5 mL, 6 wt%) was added and stirred at 37°C. The M n of samples after alcoholysis were measured by GPC.
Results and discussion
Characterization of PS-PLA-PS
Figure 1 shows the infrared (IR) spectra of the PS, PLA and its copolymers. The FT-IR spectrum of the copolymers shows a typical PLA characteristic peak and a band corresponding to the –C=O stretching vibration band of PLA 17 in 1750 cm−1 (peak A in the Figure 1). The peaks of benzene skeleton vibration around 1600 cm−1 (peak A in the Figure 1) belong to PS. 18 These results indicate that the copolymer contain the characteristic groups of PLA and PS.
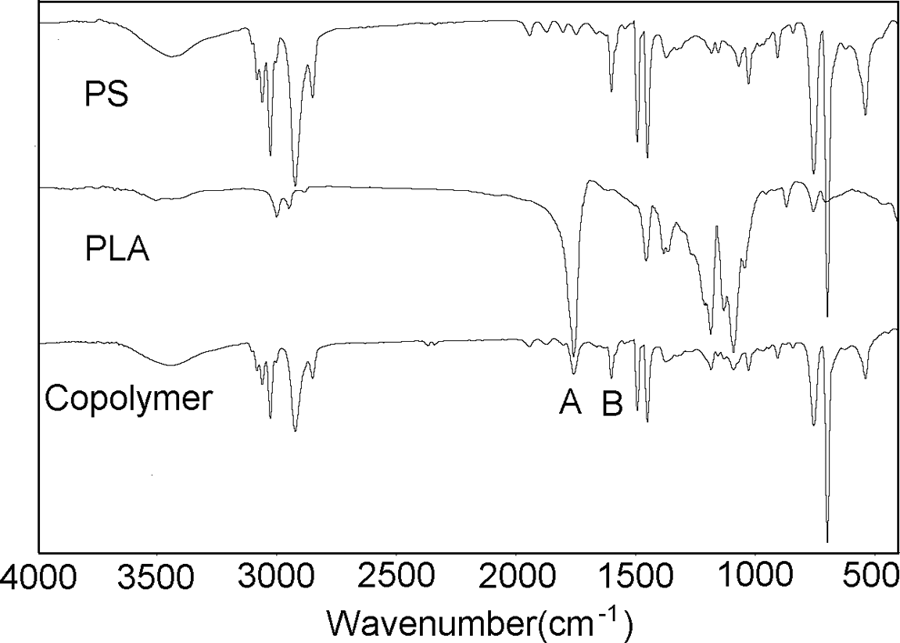
FT-IR spectra of PS, PLA and copolymer. FT-IR: Fourier-transform infrared; PLA: poly(lactic acid); PS: polystyrene.
As shown in Figure 2, the 1 H NMR spectra of PS-PLA-PS copolymer, δ of 1.60 and 5.18 ppm correspond to –CH3 (a) and –CH (b) of PLA, 19 respectively. The signal at δ of 4.17 ppm is assigned to –CH2 (c) of butanediol, whereas –CH2 (e), –CH (f) and benzene ring (g) backbone protons of PS resonate at δ of 1.87, 1.46 and 6.40–7.20 ppm, respectively. 19,20 These results verify the structure of the triblock copolymer PS-PLA-PS.
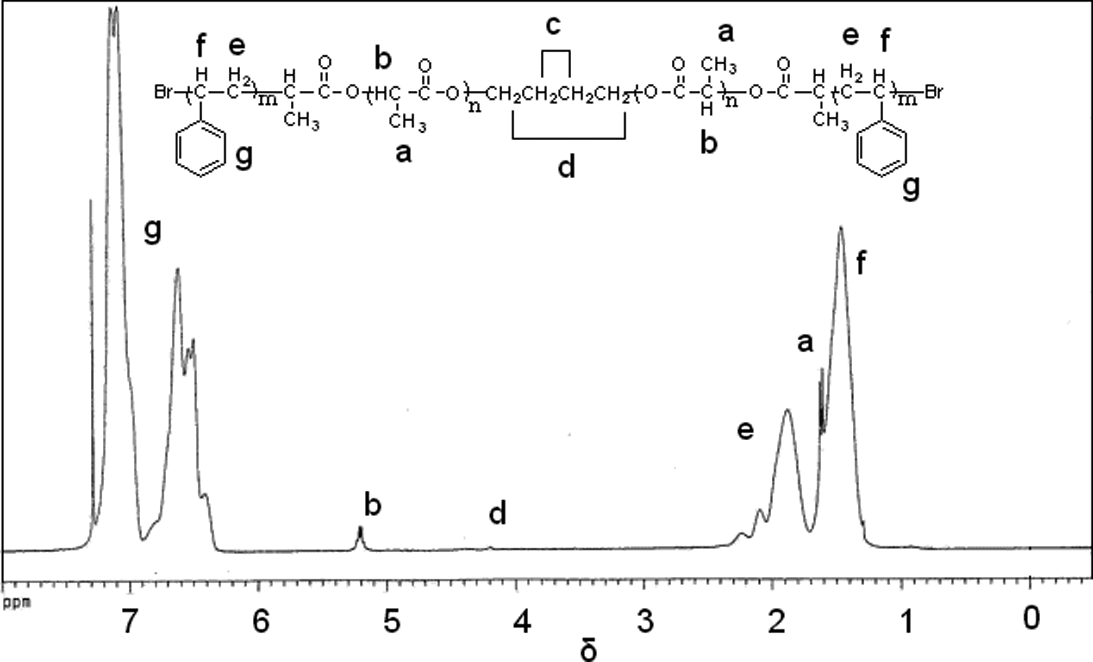
1H NMR spectra of copolymer. 1H NMR: proton-nuclear magnetic resonance.
Synthesis of PS-PLA-PS
The ATRP of St initiated by Br-PLA-Br was carried out at 110°C under vacuum. Since Matyjaszewski et al. 21 found that at least a 1:1 stoichiometry of ligand-to-copper(I) chloride was needed to obtain polymers with M w/M n < 1.5, but no rate data as a function of the stoichiometry were provided so the kinetically optimum ratio was not found. A 4:1 stoichiometry of ligand-to-copper(I) chloride was used in this ATRP.
As shown in Figure 3, on the early and middle stages of reaction (<36 h), a straight semi-logarithmic kinetic plot of ln([M]0/[M]) versus time indicates the first-order kinetics of this reaction; hence, the concentration of growing radicals is constant. This result together with the linear evolution of molecular weight with conversion (Figure 4) indicates that the polymerization rate was first order with respect to monomer concentration and M n of the polymer increased linearly with conversion. Thus, the process is indeed controlled. But at the end of the reaction, conversion increased slowly with the time, due to high M n and high viscosity, which is not good for polymerization.
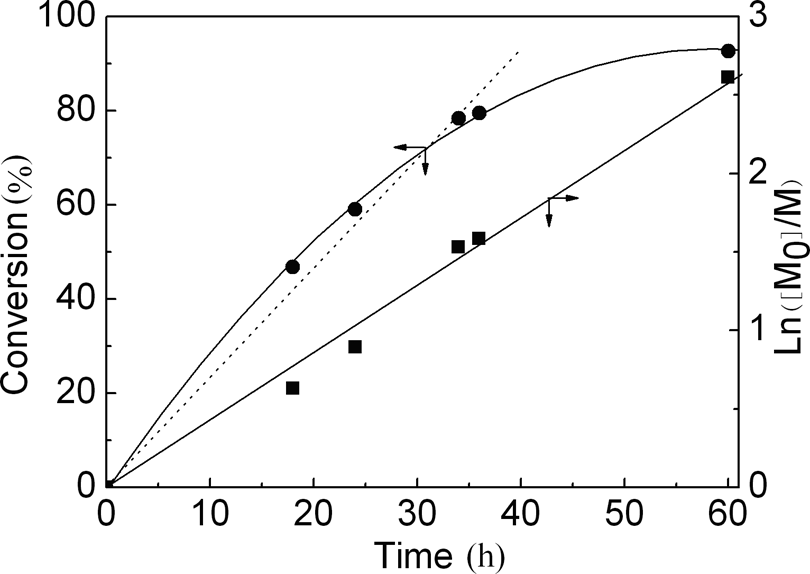
Plots of conversion and ln([St]0/[St]) against polymerization time.
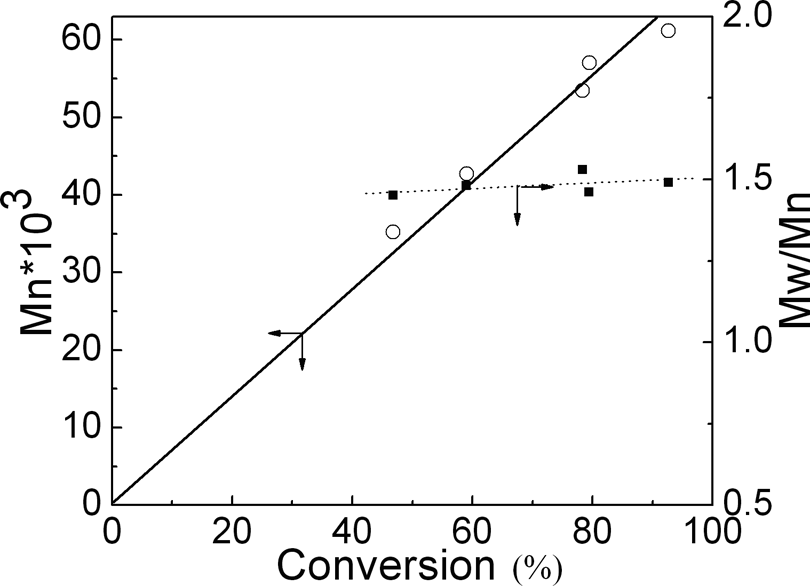
Dependence on conversion of the molecular weight and M w/M n.
MWD increased after the polymerization, although remaining lower (M w/M n ≈ 1.5) even at high conversion according to the random polymerization of St (Mw/M n ≈ 2.0). Overall, the polymerization followed the characteristics of ATRP, indicating that it is a ‘living’ polymerization.
Alcoholysis of the copolymer
Hydrolysis of PLA proceeds mainly via a surface erosion mechanism in the alkaline solution (pH 12), 22 whereas in phosphate buffer and acid solution, it proceeds homogeneously along the film cross-section mainly via a bulk erosion mechanism. 23 Consequently, PLA is unstable under alkaline condition, easy for hydrolysis and chain scission, 24 but PS is quite stable under alkaline and acid condition. 25 After PLA and its copolymer PS-PLLA-PS alcoholysis for 3 days, complete hydrolysis takes place. This is due to the absence of -C=O stretching vibration band of PLA around 1750 cm-1 by IR spectrum, and the absence of GPC peak.
According to the mechanism of ATRP, the copolymer should be PS-PLA-PS triblock structure. When the copolymer is dissolved in THF and mixed with alcoholic NaOH, the chain of PLA will hydrolyze to scission and the PS chain could be obtained without hydrolysis. The M n of PS chain was measured by GPC to confirm the structure of triblock polymer.
Alcoholysis of the copolymer.
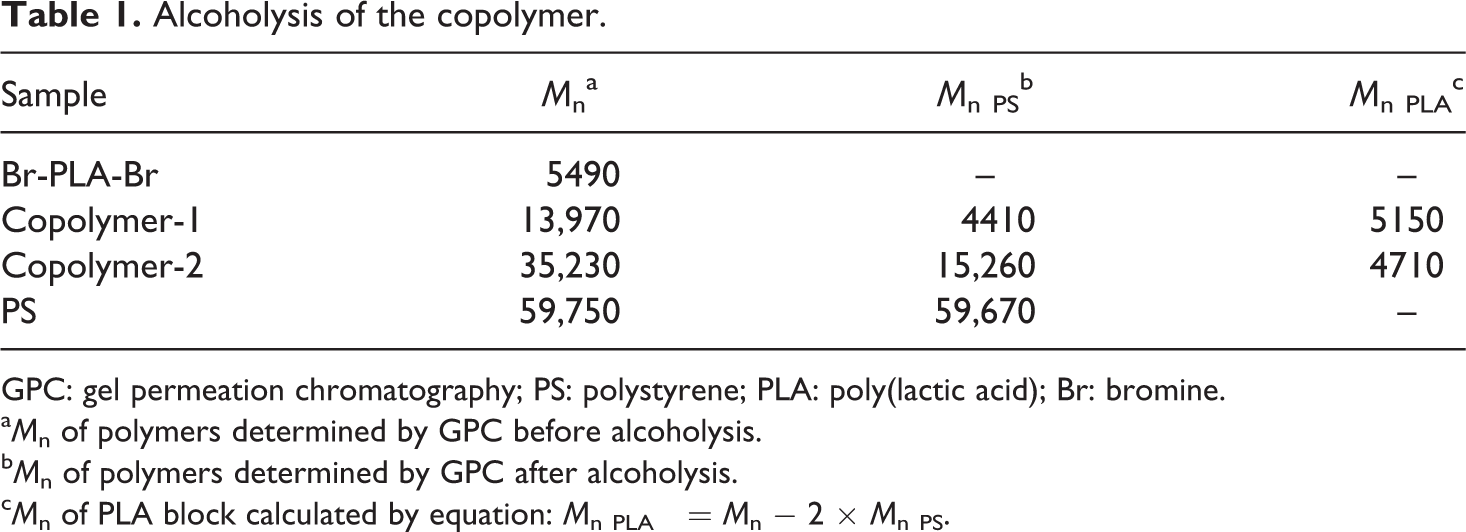
GPC: gel permeation chromatography; PS: polystyrene; PLA: poly(lactic acid); Br: bromine.
a M n of polymers determined by GPC before alcoholysis.
b M n of polymers determined by GPC after alcoholysis.
c M n of PLA block calculated by equation: M n PLA = M n − 2 × M n PS.
Thermal properties of the copolymer
The DSC results of the copolymers with different lengths of PS chain are shown in Table 2. The T g of the copolymer has been increased after copolymerization with PS. It rises from approximately 50.3 to 62.3°C with the increased PS-block content from 0 to 5580. The T g of the copolymer never reaches the T g of PS homopolymer (105°C) due to the effect of PLA block. In addition, when the chain length of each PS block reaches 8820, T g drops to 60.9°C and two Tg s are observed, which shows phase disengagement due to incompatible PLA and PS blocks. ▵Cp 1 decreases and ▵Cp 2 increases with the growing chain length of PS, indicating a greater influence of PS on the copolymer.
DSC data of PLA, PS and their copolymers.
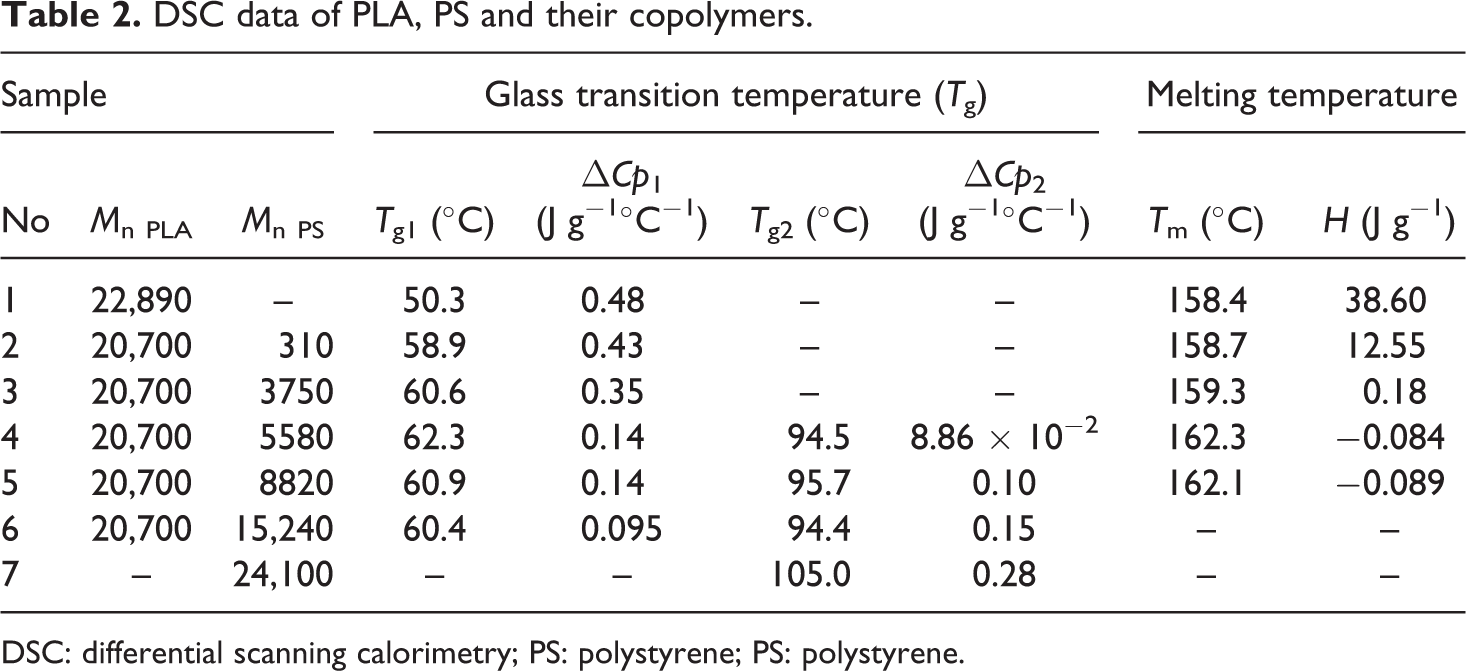
DSC: differential scanning calorimetry; PS: polystyrene; PS: polystyrene.
To study the thermal stability of PS-PLA-PS, copolymers with different molecular weights were examined by TGA. At 238°C, the pyrolysis of PLA caused it to undergo a continuous heat weight loss (Figure 5). Following copolymerization, the TGA curve of copolymer shows two-stage weight loss corresponding to the pyrolysis of PLA and PS block. As a result, the initial decomposition temperature of copolymer is increased from 238 to 333°C, so the thermal properties of the copolymer are better than that of HO-PLA-OH. Notably, even a short PS block (M n = 4240 g mol−1) causes an increased initial decomposition temperature of nearly 80°C (Table 3). Intramolecular transesterification is generally accepted as the dominant thermal degradation pathway for PLA, 26 if hydroxyl chain end of PLA is capped, then the intramolecular transesterification will be inaccessible and PLA remains intact until the onset of alternate degradation pathways at much higher temperatures. 27 Therefore, changing the PS block length has no significant influence on the initial decomposition temperature and weight retention rate.
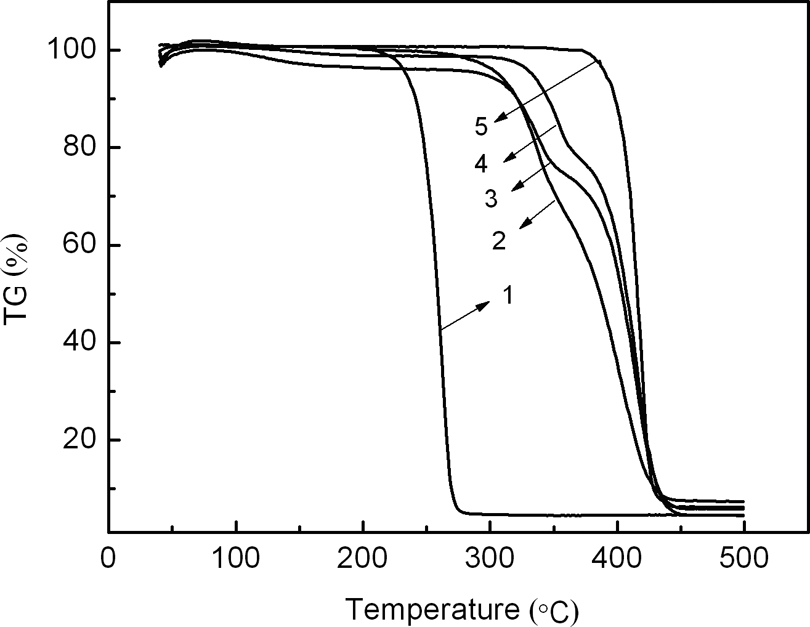
TGA curves of PLA, copolymer and PS. TGA: thermogravimetric analysis; PLA: poly(lactic acid); PS: polystyrene.
Data of the TGA curves of PLA and its copolymers.
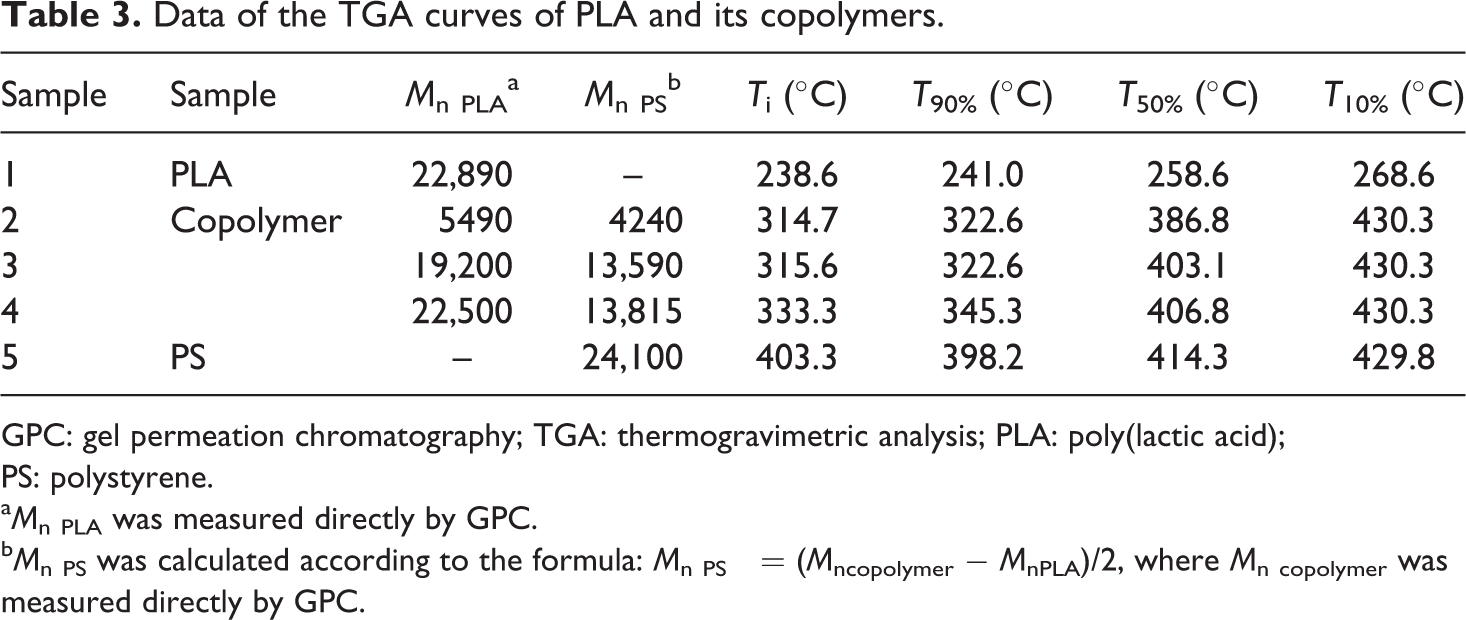
GPC: gel permeation chromatography; TGA: thermogravimetric analysis; PLA: poly(lactic acid); PS: polystyrene.
a M n PLA was measured directly by GPC.
b M n PS was calculated according to the formula: M n PS = (M ncopolymer − M nPLA)/2, where M n copolymer was measured directly by GPC.
Conclusion
In this study, a PS-PLA-PS triblock copolymer was synthesized by a Br-PLA-Br initiator with St via ATRP. The copolymerization was a first-order kinetics process indicating that the reaction had controllability and low MWD. This is a typical feature of radical polymerization via ATRP. The thermal properties of PS-PLA-PS investigated by DSC and TGA show that the initial decomposition temperature increases from 240 to 315°C, and the T g increases from 50 to 62°C. These results indicate that the thermal properties of PS-PLA-PS have been improved compared with PLA. In addition, the results of alcoholysis confirm the triblock structure of PS-PLA-PS.
Footnotes
Funding
This work was supported by the High Technology Research and Development Program of China (863 Program, 2007AA02Z200 and 2007AA06A402).
