Abstract
This research aimed at sonochemical synthesis and deposition of Fe3O4/ZnO nanocomposites on cotton/polyester fabric using the ultrasonic bath at 80℃. For this purpose, in situ sonosynthesis of magnetite nanoparticles on the fabric surface was carried out and followed by the generation of crystalline ZnO nanoparticles through the sonochemical approach, eliminating the need for high temperature after-treatment of the fabrics. This effect was achieved thanks to the shock waves formed by the cavitation bubbles also called microcalcination, resulting in the formation and adsorption of crystalline nanomaterials on the fabric surface. Magnetic properties with saturation magnetization of 1.5 emu/g, self-cleaning activities toward methylene blue discoloration, and excellent antibacterial and antifungal properties along with low cell toxicity were among the obtained properties. The current study is the first research indicating the potential of the sonochemical method to produce non-toxic cotton/polyester fabric with various properties including magnetic, photocatalytic, and antibacterial activities.
Introduction
Since 1994 there has been a significant development in the application of ultrasound energy to synthesize the novel nanomaterials with superior features [1]. Vast number of studies has been also carried out to functionalize the surfaces by the insertion of nanomaterials through high intensity ultrasound [2]. Parallel with this approach, several scientists worked at sonochemical synthesis and deposition of nanoparticles on various textile substrates. Metal nanoparticles such as Ag, Ni, and metal oxides including TiO2, ZnO, CuO and Fe3O4 are some of the nanomaterials sono-synthesized on different textile surfaces such as cotton, wool and polyester [3–18]. Mild preparation temperature eliminating the need for subsequent calcination step, creation of smaller nanoparticles in the absence of any dispersing agents, and strong adsorption and durability of the nanoparticles on the surface are some of the potential advantages of the chemical and physical effects of ultrasound [19]. Recently, the ability of ultrasound for sustainable surface modification of polyester fibers has been also confirmed [20]. Hydroxylation of terephthalate through water sonolysis forming hydroxyl radicals has been claimed as the driving force for the creation of functional groups on polyester surface, improving the surface wettability [20]. In addition to the sonosynthesis of several nanoparticles, nanocomposites of TiO2/Ag, ZnO/Ag, TiO2/ZnO/Ag and TiO2/Fe3O4/Ag have been in situ sono-synthesized and deposited on wool and polyester fabrics, producing textile fabrics with multifunctional properties [2,21].
Recently, magnetic nanophotocatalysts have been developed not only for enhancing the photocatalytic activities, but also with magnetic separation properties benefiting for the remediation of environmental pollutions [22]. Magnetic and optical properties of Fe3O4/ZnO core/shell nanocrystals were studied by Chiu et al. [23]. Karunakaran et al. [24] synthesized the Fe3O4-implanted ZnO nanosheets via hydrothermal method and analyzed the photocatalytic activity of the multifunctional nanosheets. Photodegradation of methylene blue as well as cell viability of Fe3O4-ZnO hybrid nanoparticles were studied for targeted photodynamic therapy by Patel et al. [25]. Liu et al. [26] proposed the effective route to synthesize Fe3O4-ZnO hybrid nanocrystals with different nanoshell thicknesses. Moreover, attention into the preparation procedure of the magnetic nanocomposites could result in higher stability of the composite structures, especially in case of iron corrosion. For instance, synthesis of ZnO nanoparticles on iron surface has been reported to produce superhydrophobic iron with anti-corrosion property [27].
In spite of vast number of studies concerning the synthesis of magnetite/zinc oxide nanocomposites, all the reported studies are mainly dealing with the core/shell structures through coating of ZnO on the magnetite by the precipitation method involving the calcination step for the crystallization of ZnO nanoparticles. Looking into literatures, in situ sonosynthesis of magnetic zinc oxide nanocomposites on textile substrates has not been reported yet. Here, we aimed at the usage of ultrasound irradiation to synthesize the Fe3O4/ZnO nanocomposites on cotton/polyester fabric. In this regard, magnetite nanoparticles were sono-synthesized on the fibers through the oxidation of Fe2+ to Fe3+ via hydroxyl radicals generated by the bubbles collapse in the ultrasonic bath [5]. This step was followed by the creation of crystalline zinc oxide nanoparticles, forming Fe3O4/ZnO nanocomposites. The two step formation of the nanocomposite involved the creation of ZnO nanoparticles on the surface of iron oxide components, developing more stability towards oxygen, reducing the chance of iron corrosion. Moreover, the physical and chemical effects of ultrasound promoted the nucleation and growth of ZnO nanoparticles at 80℃ in ultrasonic bath, eliminating the need for final heating of fabrics to convert the zinc hydroxide to zinc oxide nanoparticles. In addition, the treated samples benefited from the characteristics of both components including photocatalytic, magnetic, antibacterial and antifungal properties. Two step creation of the nanocomposite in the proposed manner also resulted in the alteration in the color of the treated sample changing from black to the light brown color. Besides, the effect of different concentration of precursors on self-cleaning and weight change of samples was statistically investigated. Detailed studies on the safety of the treated samples were also carried out through MTT cytotoxicity test. To the best of our knowledge, this is the first study on the preparation of cotton/polyester fabric sono-treated with Fe3O4/ZnO nanocomposites.
Experimental
Materials and methods
The applied fabric in this study was a 30/70 cotton/polyester fabric purchased from Yazdbaft Co., Iran, with Warp: 16 open-end, Weft: 16 open-end, Warp density: 35 yarn/cm, Weft density: 24 yarn/cm, Weave: Serge 3/1 and Weight: 220 g/m2. Ferrous sulfate (FeSO4. 7H2O), zinc acetate dehydrate (Zn (CH3COO)2. 2H2O), cetyltrimethylammonium bromide (CTAB) and sodium hydroxide were supplied by Merck, Germany. Methylene Blue was obtained from Uhao Co., China.
Central composite design for weight change percentage and self-cleaning properties of the treated samples.

Characterization tests
Statistical design based on response surface methodology was used considering the concentration of iron sulfate and zinc acetate as numerical factors, while weight change percentage and self-cleaning properties of the treated samples were regarded as response. The self-cleaning property of the samples was determined by staining the fabrics with 0.05% methylene blue. Stained samples were scanned by scanner and exposed to sunlight irradiation for 18 h, after which the samples were again scanned. The color differences between the samples before and after the irradiation were evaluated by image processing in RGB color space according to equation (1), using the Matlab software

Evaluation of the weight change of the treated samples was done under the standard conditions based on equation (2)

Field emission scanning electron microscopy images were obtained using MIRA, Tescan FE-SEM for studying the morphology of the synthesized nanocomposites on the fabric surface and estimating the size of the nanoparticles. Elemental analysis of the samples was also carried out using the energy-dispersive spectroscopy (EDX). X-ray diffractogram (XRD) was performed using the XPert MPD (Philips, Holland), to confirm the creation of crystalline nanoparticles on the surface.
Antibacterial activity of the optimum sample was tested against two bacteria of 
Cell toxicity of the optimum sample was evaluated by the MTT assay [21,28,29]. Normal human dermal fibroblasts at passage 3 were grown in Dulbecco’s modified Eagle’s medium supplemented with 10% fetal bovine serum and 2 mM l-glutamine and incubated at 37℃ in a fully humidified, 5% CO2/air atmosphere. The cells were seeded in a 96-well plate at a density of 2000 cells per well and incubated for 48 h. The fabrics cut into 1 inch × 1 inch pieces were soaked in 1 and 2 mL culture medium for 24 h. The cultured medium with leaching substance (dilution of 1:2) was then serial-diluted to give dilutions of 1:4, 1:8, 1:16, 1:32, 1:64, and 1:128. The sample in each dilution was added to the cells and incubated for 24 h. The test samples were removed from the cell cultures, and the cells were re-incubated for a further 24 h in fresh medium and then tested with 3 -(4, 5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide (MTT) assay, 50 µL of MTT in phosphate-buffered saline (5 mg/mL) were added to the medium in each well, and the cells were incubated for 4 h. Medium and MTT were then aspirated from the wells, and formazan crystal was solubilized with 200 µL of dimethyl sulfoxide and 25 µL of Sorenzen’s glycine buffer, pH 10.5. Experiments were performed for five times and the obtained results were recorded as percentage of absorbance relative to untreated control cells. The cytotoxicity results were used to calculate the percentage of cell viability after incubation with the samples as shown in equation (4)

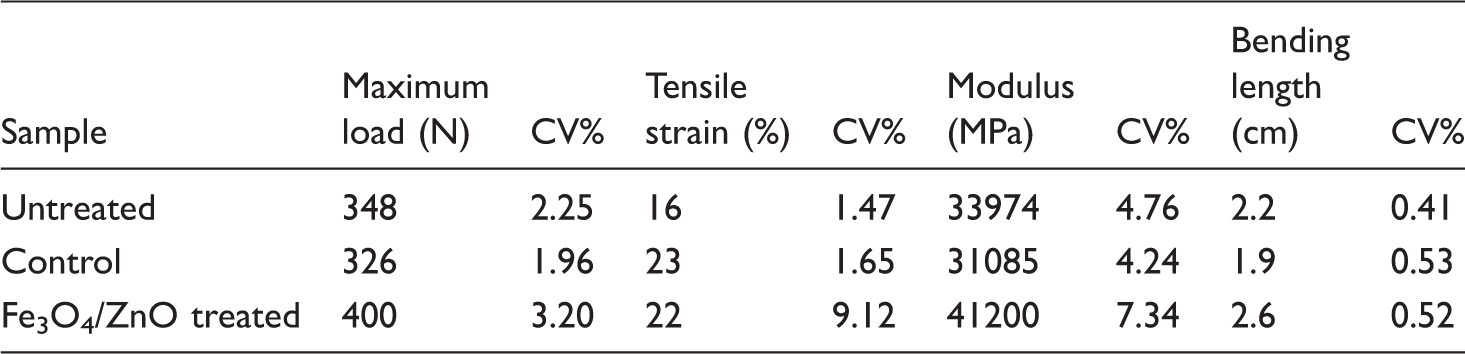
Magnetization of optimum sample was quantified by a vibrating-sample magnetometer (VSM, Iran) at room temperature. The tensile strength of the treated sample was measured using an Instron instrument (USA) with gauge length of 7 cm and extension rate of 70 mm/min for three replicates, and the average value was recorded. Bending length (cm) of the sample in warp direction was also measured by Shirley fabric stiffness tester (USA).
Results and discussion
Sonosynthesis of Fe3O4/ZnO nanocomposites on cotton/polyester fabric: Mechanism
Sonosynthesis of Fe3O4/ZnO nanocomposites on the surface of cotton/polyester fabric was carried out through the two-step procedure, briefly shown in Figure 1. Different parts of the fabric were differently affected under the applied synthesis method. Under the alkaline condition of the reaction bath, polyester fibers underwent the alkaline hydrolysis, forming hydroxyl and carboxylate functional groups increasing the chance of surface reactivity, from which the nucleation and growth of magnetite nanoparticles were promoted [30]. Part of iron (II) ions underwent oxidation to iron (III) ions through water sonolysis under the ultrasound irradiation. Hydroxylation of iron species was done and followed by the formation of magnetite seeds and the growth of nanoparticles was facilitated due to the ultrasonic energy and the related phenomenon [5]. In addition, based on the findings of our previous study, hydroxylation of terephthalate can be possibly promoted by •OH radicals formed under water sonolysis, forming the functional groups on the polyester surface improving the surface reactivity toward the nanoparticles adsorption. Detailed discussions on the possible ultrasonic phenomenon resulting in the polyester surface modification have been provided in our previous paper [20].
Schematic illustration of the synthesis of Fe3O4/ZnO nanocomposites on cotton/polyester fabric.
On the other hand, cellulosic part of the fabric was charged negatively (Cell-O−) under the alkaline condition, providing the sites for adsorption of iron (II) ions and subsequent reactions to form magnetite nanoparticles. Thus, in addition to the prominent role of NaOH in the synthesis of Fe3O4 nanoparticles, it has strong effect on the enhanced reactivity of the fiber surface for nanoparticles adsorption. Besides, fast projection of nanomaterials to the fibers by the induced microjets due to the sonochemical reactions is beneficial for the strong deposition of the nanoparticles on the surface [20].
Based on the isoelectric point of magnetite nanoparticles [31], under the alkaline condition of the synthesis of zinc oxide nanoparticles, the surface of Fe3O4 is more negative, providing the sites for adsorption of zinc (II) ions, from which the nucleation and growth of the crystalline ZnO nanoparticles were promoted, with no need for subsequent fabric heating. This effect which is famous as in situ microcalcination has been related to the ultrasound shock waves formed by the cavitation bubbles [20]. The proposed method has been speculated based on the FESEM pictures, indicating almost no individual ZnO nanoparticles on the fiber surface as shown in Figure 2.
FESEM images (a) untreated (2500 × magnification) and (b–d) fabric treated with Fe3O4/ZnO nanocomposites (6000 × 25,000 × and 75,000 × magnifications), (e) EDX spectrum and (f) mapping image. XRD patterns of untreated and fabric treated with Fe3O4/ZnO nanocomposites .

Morphological and EDX analyses
FESEM images of the sample treated with Fe3O4/ZnO nanocomposites indicated the presence of nanoparticles with different morphological properties (Figure 2(b)) compared with untreated sample (Figure 2(a)). Semi-hexagonal-shaped nanosheets and cobblestone-like nanoparticles with an average size of 56 and 20 nm were detected at higher magnification (Figure 2(c) and (d)). Moreover, the EDX spectrum shown in Figure 2(e) confirmed the presence of Zn and Fe, in addition to C and O. Mapping images shown in Figure 2(f), also shows the uniform distribution of nanoparticles on the fiber surface.
XRD analysis
Successful synthesis and deposition of crystalline Fe3O4/ZnO nanocomposites on the fiber surface were confirmed by the X-ray diffraction analysis (XRD) (Figure 3). In comparison with the XRD spectrum of untreated cotton/polyester fabric, the presence of peaks at 2θ angles of 31.85, 34.53, 36.36, 47.62, 56.65, 62.93, and 66.41° is related to zinc oxide structure [9], while the peaks at 2θ angles of 30.1, 35, 43, 56.9 and 62.5° are referred to magnetite structure [22]. Furthermore, the crystalline size of the nanocomposites was estimated based on Scherrer’s equation around 39 nm.
Weight change percentage
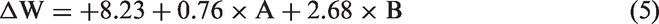
Analysis of variance for weight change percentage of the treated fabrics.

Based on the magnitude and sign of the coefficients of the variables, both zinc acetate and iron sulfate concentrations have positive linear effect on the weight change indicating weight gain (Figure 4(a)). In the other words, more nanoparticles were formed in higher zinc and iron salt concentrations. However, the model coefficient of iron sulfate (0.76) was smaller than the coefficient of zinc acetate (2.68) which represents the importance of zinc content rather than iron in weight change, within the applied concentrations. As part of NaOH was consumed to form Fe3O4 and ZnO nanoparticles, the extent of polyester alkaline hydrolysis was lower in case of treated samples compared to control fabric.
3D response surface images for the effect of zinc acetate and iron sulfate on (a) weight change percentage and (b) self-cleaning properties of the treated fabrics.
Self-cleaning properties

Analysis of variance for self-cleaning properties of the treated fabrics.
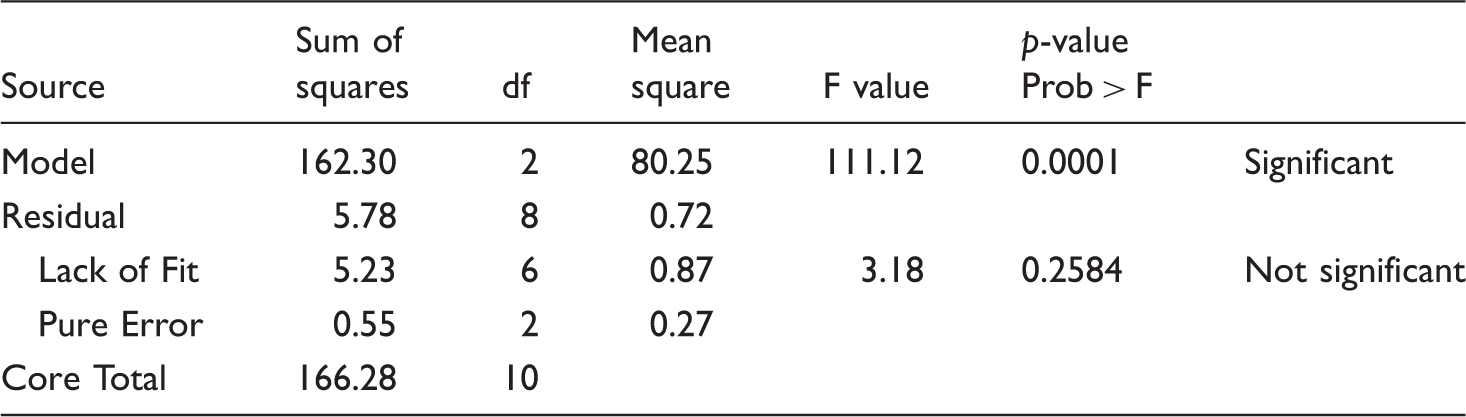
Finally, the software suggested the optimized model factors of both iron sulfate and zinc acetate concentrations at 3 (% W/V), regarding the maximum self-cleaning (greater ΔRGB) and weight alteration.
Magnetic properties
Magnetic properties of the optimum sample were measured by a vibrating sample magnetometer at room temperature and the induced magnetization (M) related to the applied magnetic field (H) is shown in Figure 5. For comparison, the magnetization properties of the sample only treated with magnetite nanoparticles have been also included. By increasing the applied field from 0 to 8000 Oe, a sharp increase in magnetization was observed with saturation magnetization of 3.4 and 1.5 emu/g, for Fe3O4 and Fe3O4/ZnO-treated samples, respectively. This clearly shows the decreased magnetic properties of the sample treated with the nanocomposite due to the in situ synthesis of ZnO on magnetite nanoparticles. However, the optimum sample still shows appropriate magnetic properties, confirming attraction towards a magnet.
Room-temperature magnetization curves of cotton/polyester fabrics treated with Fe3O4 and Fe3O4/ZnO nanocomposites.
The coloring effect of the applied treatment on the fabric was evaluated by the reflectance spectra of the samples. L* (lightness), a* (redness–greenness) and b* (yellowness–blueness) color values of the treated sample were obtained using Gretag Macbeth 7000 spectrophotometer. As shown in Figure 5, during the treatment, the color of the sample only treated with magnetite nanoparticles was changed to light brown due to the synthesis of zinc oxide nanoparticles on the surface of iron oxide. The L*, a*, b* colorimetric values of the sample treated with Fe3O4 were 24, 5, 2, which changed into 48, 5, 12 for Fe3O4/ZnO-treated fabric, indicating the significant color difference of ΔE = 26.
Mechanical properties
Mechanical properties of untreated, control and treated samples.
Antibacterial and antifungal properties
According to the previous studies of our own group [30], magnetite nanoparticles are capable of killing the Antibacterial and antifungal efficiencies of the control and optimum treated sample against (a) 
MTT assay
Based on several previous literatures in which MTT tests have been done, under exposure to toxic agents, the first visible change is observed in cell shape rather than any morphological changes in metabolism or proliferation [33]. Thus, the cytotoxicity test was performed to evaluate the harmful effects of the nanocomposite on dermal fibroblast through monitoring any changes in optical microscopy after three days. Moreover, the percentage of cell viability of samples comparing with control was quantitatively evaluated. As shown in Figure 7, treated fabric possessed normal cultured human dermal fibroblast with similar morphology to control, indicating minimum harmful toxic effect. This was also confirmed by the average cell viability of over 80%, indicating almost non-toxicity.
Cell viability of (a) control and (b) fabric treated with Fe3O4/ZnO nanocomposites.
Thus, comparing with control, although some cells underwent morphological changes, most of them are viable retaining their initial spindle shape. Therefore, no considerable adverse effect on human skin was found for the treated fabric. No significant morphological change can be observed in the cell lines comparing with the control sample.
Washing durability
The stability of nanocomposite adsorbed on the fabric was tested by washing the optimum sample with 1 g/L nonionic detergent, rotated under a certain temperature (60−70℃) and time (five washing cycles). No significant change has been observed in the saturation magnetization, antibacterial efficiency, and self-cleaning ability of the washed sample indicating the washing durability. The appropriate durability of the synthesized nanocomposite on the treated fabric was confirmed by the negligible change occurred in the reported saturation magnetization. In addition, the washed sample showed appropriate degradation of methylene blue stain. Thus, no significant change in self-cleaning activity was observed even after washing. Also, the washed sample was effective in killing both Gram-positive and Gram-negative bacteria (100% bacteria reduction) entirely.
Conclusion
For the first time in this study, a non-toxic cotton/polyester fabric with superior self-cleaning, antibacterial, antifungal and enhanced mechanical properties has been developed through sonochemical synthesis of Fe3O4/ZnO nanocomposites using the ultrasonic bath. The preparation condition based on the iron sulfate and zinc acetate as precursors was statistically optimized, as 3% W/V of both salts resulted in the maximum efficiencies of the treated samples. Radical formation and micro-convection by cavitation bubble had strong influence on the surface modification of polyester part of the fabric, enhancing the adsorption of the nanoparticles on the surface. In situ microcalcination was also beneficial for eliminating the need for final heating of the fabric. Thus, the crystallization of zinc oxide occurred under the shock waves was formed by the cavitation bubbles making the sonosynthesis and sonofabrication possible at 80℃. In addition, the synthesis of ZnO nanoparticles on magnetite changed the color of the Fe3O4/ZnO-treated sample from black to light brown. We believe that due to the formation of zinc oxide on magnetite, the possible detrimental effects of oxygen on iron oxide causing corrosion will be hindered, which can be further analyzed in the future studies.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
