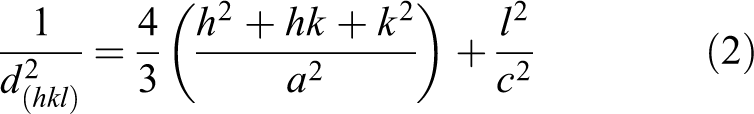Abstract
Recently, an increase in the production of intelligent nanomaterials has been reported for the application of solid surface coating. These nanomaterials provide a wide number of functionalities such as anticorrosive, antibacterial, and self-cleaning properties. Hence, titanium dioxide (TiO2) and zinc oxide (ZnO) nanoparticles were synthesized using a green chemistry approach. These nanoparticles were fully characterized by scanning electron microscopy, energy-dispersive X-ray, high-resolution transmission electron microscopy, X-ray diffraction, ultraviolet (UV)–visible spectroscopy, Brunauer–Emmett–Teller test, and nitrogen adsorption–desorption isotherm. Then, a commercial enamel-type paint was modified by using different concentrations (2, 3.5, and 5 w/v%) of nanoparticles. These nanofilled paints were then brushed onto the surface of different types of materials such as carbon steel sheets, wood sheets, and aluminum disks. Anticorrosive, self-cleaning, and antibacterial properties of the nanofilled paints were evaluated, with the aim to determine the capability for this application. According to the characterization results, TiO2 and ZnO nanoparticles exhibited similar physicochemical properties compared to those synthesized using traditional methods. The anticorrosion results revealed that nanofilled paints provide a barrier using low concentrations of nanoparticles, due to the decrease of agglomerates on the surface avoiding the presence of high porosity. In the case of self-cleaning, a proposed mechanism of degradation demonstrated that the presence of both nanoparticles in the paint provided high degradation of methylene blue due to the high surface area offered by the nanoparticles. On the other hand, antibacterial activity under UV light was observed only for ZnO nanoparticles, which may be related to the diffusion of nanoparticles into the cell membrane of the bacteria, affecting the normal function. These results showed to be promising for the modification of paints with TiO2 and ZnO nanoparticles, and the application on solid surfaces for the construction, and even in textile fields.
Introduction
The growing research focused on the synthesis and modification of materials in the nanoscale has increased their applicability in different fields such as biomedicine, electronics, optics, computing, solar thermal energy, construction, and intelligent coatings. 1 –6 Among the multifunctional properties of these nanocomposites are anticorrosive, antibacterial, self-cleaning, and eco-friendly effects, in which nanocomposites play a prominent role for surface treatments in the contribution of superior physical effects in products. 7 –11 The incorporation of nanomaterials in these products such as resins, aerosols, and paints may contribute to the reduction of additives highly toxic which are considered as health hazard. 12 –14 Anticorrosive properties are important for the fabrication of paints, in which the aim is to protect those structures exposed to chemical or electrochemical corrosion due to the pollution and marine atmosphere in large populated and coastal areas. 15,16 Through the photocatalytic activity of nanomaterials as additives, high hydrophobicity and self-cleaning ability may reduce surface contamination by degrading pollutants, in order to improve the quality and appearance of the structural surface. 17 –19 Moreover, the incorporation of antibacterial agents is being intensively applied in specific products, with the aim to avoid the growth of harmful microorganisms onto different types of surfaces, which would enhance the quality of life in developed and developing countries. The presence of nanoparticles in paints may lead to the inhibition activity of a wide number of Gram-positive and Gram-negative bacteria, which is very hard to prevent nowadays. 10,20,21
Titanium dioxide (TiO2) and zinc oxide (ZnO) nanoparticles have been extensively studied for the application as self-cleaning, anticorrosive, and antibacterial additives in construction materials, 22 especially in external surfaces, due to the improvement offered to the aesthetic quality of buildings, leading to a reduction in maintenance cost. 23 –29 Among the properties of these mesoporous photocatalytic nanomaterials are those to improve the surface characteristics of a limestone building such as adhesion efficiency, mechanical resistance, and hydrophobic behavior, as well as the degradation by solar radiation of other compounds such as methylene blue (MB) and carbon soot for self-cleaning purposes. 25,30 –33 TiO2 and ZnO production has been focused in the use of green chemistry with the aim to reduce the high energy consumption, toxic chemical, and high cost, needed for the synthesis using traditional methods. Hence, natural sources such as plants, organic, and agricultural wastes are considered as raw material environmentally friendly, in which concentrated extracts can be obtained allowing the reduction of metal precursor without using hazard chemicals. 34 –39 Different species of plants have been used to prepare leaf extracts for the synthesis of TiO2 and ZnO nanoparticles, including Phyllanthus niruri, Trigonella foenum-graecum, Psidium guajava, Sesbania grandiflora, Azadirachta indica, Typha latifolia, Averrhoa bilimbi, and Chamaecostus cuspidatus. 34,36,40 –45 Current study proposed the use of Cymbopogon citratus (commonly known as lemongrass) leaf extract as reducing of metal precursors and capping agent, due to the high content of phytochemicals such as neral, citral, geranial, pinito, allantoin flavonoids, hydrocarbons, waxy esters, sterol esters, ketones, aldehydes, terpenoids, phenols, fatty alcohols, fatty acids, among others. 46 –52 This plant is widely available in tropical and semitropical environment zones throughout Asia, Africa, and South America. 53,54 Therefore, lemongrass leaves were proposed to be part of the raw material in the synthesis of TiO2 and ZnO nanoparticles since they are easily found in the Department of Bolivar (Cartagena, Colombia) and are no part of the food chain.
Modification of paints with nanomaterials has been recently investigated, as was reported by Zhou et al., in which a self-cleaning transparent coating based on fluorocarbon and semicrystalline colloidal particles of TiO2-SiO2 was prepared, achieving 96% of methyl orange discoloration using a concentration of 1.5%. 55 Truppi et al. studied the self-cleaning and photocatalytic properties of TiO2/AuNRs-SiO2 nanocomposite for application in construction materials. 56 Wang et al. developed a pseudo alloy Zn-Al-Mg-TiO2 coating by cold spraying technique on a Q235 substrate, in which the performance of Zn-Al-Mg-TiO2 coating on marine metallic equipment was studied using saltwater dynamic corrosion tests, electrochemical tests, and wear and tear tests. 57 The results reported by Wang et al. revealed that the Zn-Al-Mg-TiO2 coating provided excellent anticorrosion and wear resistance, providing a stable and long-term protection for the metallic substrate, due to the nanoparticle content acted as a physical barrier, preventing the penetration of O2 and H2O. 13,58 On the other hand, the antibacterial activity of TiO2 and ZnO nanoparticles was widely studied and reported in the scientific literature. 59 –64 Shankar et al. used three types of ZnO nanoparticles to manufacture a PLA/PBAT-ZnONP composite film, offering a good inhibition activity against Escherichia coli and Listeria monocytogenes. 65 In a similar study, Thakur et al. investigated the green synthesis of TiO2 nanoparticles and their antibacterial activity against several bacteria such as E. coli, Bacillus subtilis, Salmonella typhi, and Klebsiella pneumoniae, in which the results indicated a high inhibition. 36
Additionally, recent research has also studied the mechanical properties, thermal stability, and weather resistance for resin-based coatings and paints modified with nano- and microscale particles such as SiO2, Ag, CeO2, TiO2, and ZnO. 66,67 Hence, adhesion tests have been applied using the ASTM D3359 (cross-cut), ISO 2409 (cross-cut), and ASTM D4541-09 (pull-off) standards. The durability of the paints after irradiation can be obtained using a test widely known as weathering durability. 68,69 Results from these tests may provide details about the effect on the adhesion and durability with the incorporation of nanoparticles in paints, in which no significant impact has been evidenced in most of the cases. Nguyen et al. 70 analyzed the effect of Rutile (R-TiO2) and ZnO nanoparticles on ultraviolet (UV) shielding efficiency of a water-based acrylic coating. The adhesion and weathering durability tests were performed using a UV/condensation weathering chamber equipped with UV-B-313 fluorescent lamps. After 60 cycles of UV/condensation weathering chamber exposure, no significant changes of the color were evidenced, and the adhesion values slightly decreased from 2.5 and 2.4 N/mm2 to 2.3 and 2.1 N/mm2 for paints modified with R-TiO2 and ZnO nanoparticles, respectively.
We hereby report a green approach for the synthesis of TiO2 and ZnO nanoparticles using lemongrass extract as a capping agent for the control size of synthesized nanoparticles. We found that the phytochemical content of extract, promoted the formation of nanoparticles as was observed through the morphologic information obtained from the microscopy images. The physicochemical properties of the as-prepared nanoparticles are similar to those nanoparticles synthesized using the traditional techniques. Finally, the influence of TiO2 and ZnO nanoparticles in paints was evaluated to determine the efficiency of the anticorrosive, antibacterial, and self-cleaning.
Materials and methods
Materials
Fresh lemongrass leaves (C. citratus) were collected from Cartagena, Colombia. Titanium (IV) isopropoxide (C12H28O4Ti) solution (95%) and anhydrous zinc chloride (ZnCl2, ≥97%) were purchased from Sigma-Aldrich (Burlington, MA). All reactions were carried out using ACS Reagent chemicals. The commercial enamel paint for exteriors was supplied by the brand Kölor®, which is basically made from polyester (alkyd resin) modified with fatty acids and sometimes contains glass powder or tiny metal flake fragments instead of pigments. Varsol (100%) produced by ALGRECO® was acquired in order to increase the shine and hardness of the paint, which is mainly made from distillates of petroleum (aliphatic hydrocarbons).
Green synthesis of TiO2 and ZnO nanoparticles
The procedure for the preparation of aqueous extract of lemongrass, and then the green synthesis of TiO2 nanoparticles, was according to the methodology reported previously. 71 Lemongrass leaves were cut, washed, and dried to remove impurities, and then crushed manually. One hundred grams of biomass was added in cloth bags for further solvent extraction using 500 mL of distilled water for 6 h. The lemongrass extract thus prepared was then placed in a storage at 4°C. Afterward, 100 mL of this extract was mixed with 20 mL of the precursor C12H28O4Ti by dropwise addition in a 250 mL beaker. The solution was placed in an ultrasound processor (ultrasonic processor, WiseClean WUC-A06H, Acinterlab (Miami, Florida), 60 Hz), allowing to react for 30 min approximately. The nanoparticles were washed using distilled water and ethanol reagent grade, and then collected by centrifugation at 5000 r/min for 30 min, repeating the procedure several times to remove unreacted precursor. Calcination at 550°C for 3 h was performed with the aim to obtain the anatase phase of TiO2 nanoparticles.
For the ultrasound-assisted green synthesis of ZnO nanoparticles, a procedure reported by Mohammadi et al., 72 with minor modification was performed. Twenty grams of ZnCl2 were dissolved in aqueous lemongrass extract (400 mL) with a ratio of 80:320 v/v under magnetic stirring at room temperature for 20 min. In order to adjust the pH = 12 of the solution, 15 mL of sodium hydroxide solution (5 M) was slowly added at 500 r/min. Subsequently, the suspension was placed in an ultrasonic processor (WiseClean WUC-A06H, 60 Hz) for 15 min, then followed by centrifugation at 4000 r/min for 10 min. The resulting precipitate was collected and washed four times with distilled water and twice with ethanol, and finally calcined at 500°C for 5 h.
Characterization
The morphology of TiO2 and ZnO nanoparticles was studied by using a high-resolution transmission electron microscope (HRTEM) FEI Tecnai F20 Super Twin TMP (Thermo Fisher Scientific, Hillsboro, Oregon. Additionally, an elemental analysis was performed using energy-dispersive X-ray (EDX) spectroscopy. Solutions of 10 µg/mL of TiO2 and ZnO nanoparticles in absolute ethanol were prepared by using an ultrasound bath, with the aim to obtain a good dispersion of nanoparticles in the medium. Then, a micropipette was used to place a drop of nanoparticle solution onto a carbon-coated grid and let dry for 5 min. Finally, the grid was placed in the holder, which was then introduced inside the microscope. Scanning electron microscopy (SEM) images were acquired using a Phenom XL tabletop (Thermo Fisher Scientific, Hillsboro, Oregon) with an operating voltage of 15 kV. X-ray diffraction (XRD) of the samples was performed in standard reflection (2θ: 5–60°) using an X-ray diffractometer XPert PANalytical Empyrean Series II—Alpha1, Model 2012 (Malvern Panalytical, Malvern, UK) equipped with copper anode (copper K alpha 1 (CuKα1), λ = 1.5406 Å) and operated at 40 kV and 40 mA. On the other hand, the specific surface area was measured by nitrogen (N2) adsorption–desorption analysis at −196°C using a Micromeritics (Norcross, GA) ASAP 2020 surface area and porosity analyzer. Prior to N2 adsorption–desorption analysis, the samples were degassed at 300°C for 2 h in a vacuum. Finally, ultraviolet–visible diffuse reflectance (UV-vis-DRS) was carried out in a spectrophotometer Thermo Scientific (Madison, WI) Evolution-600, with BaSO4 as a reference and using a scanning speed of 240 nm/min.
Modification of enamel paint with TiO2 and ZnO nanoparticles
Different concentrations (2, 3.5, and 5 w/v%) of the as-synthesized TiO2 and ZnO nanoparticles were used to modify a commercial enamel-type paint, according to the procedure reported by Amorim et al. 18 Initially, the nanoparticles were added into 1 mL of Varsol and then dispersed by placing the solution in an ultrasound bath for 2 min. Then, 3.5 mL of enamel paint was added into the previous solution and stirred for 1 min using a metal stirring rod. The effect of the nanoadditive-modified paints on the anticorrosion, self-cleaning, and antibacterial properties was evaluated by brushing the paints on the surface of rectangular carbon steel sheets (10 × 15 × 0.3 cm3), rectangular wooden sheets (5 × 7.5 × 0.5 cm3), and thin aluminum disks with a thickness and radius of 0.1 mm and 1 cm, respectively. The samples were then dried completely for 72 h at room temperature prior to the evaluation of the as-mentioned paint properties.
Anticorrosion, self-cleaning, and antibacterial evaluation
Anticorrosion test was performed in a salt fog chamber (Renault (Strongsville, Ohio D17 1058J) using the ASTM B117:16 for 200 h. The dimensions of the samples were adjusted to 10 × 15 × 0.3 cm3 (width, height, and thickness) and were then painted on both sides using an insulating tape for coating the edge.
The self-cleaning capacity of the samples coated with nanofilled paints was evaluated through the degradation of MB organic dye stain, as a function of the accumulated solar radiation (0, 3000, and 4000 J/m2). This test was measured using an HD 2102.2 Photoradiometer (Delta OHM, Padova, Italy) equipped with an LP 471 UVA probe (315–400 nm) (Delta OHM, Padova, Italy). Accordingly, an MB with a concentration of 20 mg/L was prepared, and 0.1 mL (a drop) was then placed in the middle of the wood samples coated with nanofilled paint, and finally, the samples were exposed to solar radiation. Samples were photographed in order to appreciate visually the decolorization (Padova, Italy) degree, and then compared using a sample coated with unmodified paint (target). To quantify the decolorization percentage, four solutions (5, 10, 15, and 20 mg/L) of MB were prepared, a drop of each solution was deposited on a rectangular wooden sheet coated with paint without nanomaterials and dried for 72 h at room temperature and darkness. Finally, the cyan composition of each spot was determined using the eyedropper tool of Adobe Illustrator CS6 software.
On the other hand, the antibacterial test was carried out by using aluminum disks coated with nanofilled paints for inhibition of E. coli. Fourteen grams of nutritive agar (OKOID, CM0003) were weighed following a typical formula: 1 mg/L of “Lab-Lemco” powder, 2 g/L of yeast extract, 5 g/L of peptone, 5 g/L of sodium chloride, and 15 g/L of agar. These nutrients were dissolved using a base volume of 500 mL of distilled water, which was then stirred at 500 r/min and heated up until boiling temperature was reached. The solution was cooled down to decrease the temperature up to 30°C, and the pH was then measured and adjusted to 7.35. The nutrient agar was sterilized at 121°C and 2 atm or 75 min (15 sterilizing and 60 drying). Aluminum disks of 1 cm of diameter were cut in order to perform to test by duplicate. The disks were then coated by using unmodified paints and nanofilled paints with TiO2 and ZnO nanoparticles. The antibacterial activity test against E. coli was carried out in sterile petri dishes, in which 20 mL of sterile culture medium was poured and left to solidify for 1 h. Finally, the disks were placed into the petri dishes and incubated at 37°C for 24 h.
Results and discussion
Characterization
The morphology of the as-prepared TiO2 and ZnO nanoparticles was studied by SEM, as shown in Figure 1(a) and (b), respectively. For both types of nanoparticles, the same agglomeration behavior can be easily observed using a magnification of ×500 at 100 µm, which is a typical result for the synthesis using a green approach.

SEM images of (a) TiO2 and (b) ZnO nanoparticles. SEM: scanning electron microscopy; TiO2: titanium dioxide; ZnO: zinc oxide.
HRTEM images of TiO2 nanoparticles are shown in Figure 2(a) to (d). Figure 2(a) shows an agglomerated system of nanoparticles with a similar shape of a sphere, obtaining an average size particle of 12.34 ± 1.52 nm. From Figure 2(b) the lattice spacing can be clearly evidenced, which represents the interplanar space (101) of the anatase phase with a value of 0.353 nm. In addition, rings corresponding to planes (101), (004), (200), and (105) were identified in Figure 2(c), revealing the polycrystalline character of TiO2 nanoparticles synthesized using green ultrasound-assisted chemistry. The purity of the sample was also confirmed using the EDX spectrum, the results of which are shown in Figure 2(d) and indicate the presence of Ti atoms.

HRTEM images of the (a) as-prepared TiO2 nanoparticles through green ultrasound-assisted chemistry, (b) lattice spacing, (c) SAED pattern, and (d) EDX spectrum. HRTEM: high-resolution transmission electron microscopy; TiO2: titanium dioxide; EDX: energy-dispersive X-ray.
These results are in agreement with those reported by Liang et al., 26 in which TiO2 nanoparticles were synthesized through the sol-gel method using acetylacetone as the hydrolysis control agent, and with a calcination temperature of 500°C. Equation (1) was used to identify the planes of TiO2 nanoparticles corresponding to each ring obtained in the selected area electron diffraction (SAED) pattern.
where h, k, and l are the Miller indices, a and c the lattice parameters, in which
ZnO nanoparticles were also studied by HRTEM, as shown in Figure 3(a) to (d). Figure 3(a) reveals that the green ultrasound-assisted chemistry method generates semispherical and rod-shaped nanoparticles with average particle sizes of 137.41 ± 15.80 and 54.81 ± 12.57 nm, respectively. Additionally, a wide size distribution was observed and is similar to that reported by Begum et al., 75 in which ZnO nanoparticles were synthesized using Eryngium foetidum L. The lattice spacing showed in Figure 3(b) was determined for the Wurtzite phase (100) with a value of 0.29 nm. Figure 3(c) indicates the ring corresponding to the plane (102). Finally, high purity of Zn atoms contained in the sample was confirmed in the EDX spectrum as shown in Figure 3(d). Equation (2) was used to identify the planes of ZnO nanoparticles in the SAED pattern, which presented a hexagonal structure according to the values of a = 3.2506 and c = 5.2075. 76

HRTEM images of the (a) as-prepared ZnO nanoparticles through green ultrasound-assisted chemistry, (b) lattice spacing, (c) SAED pattern and (d) EDX spectrum. HRTEM: high-resolution transmission electron microscopy; ZnO: zinc oxide; EDX: energy-dispersive X-ray.
The structural information of the as-synthesized nanoparticles was performed through XRD. Figure 4 shows patterns for TiO2 and ZnO nanoparticles prepared through the green ultrasound-assisted chemistry. The tetragonal structure of the anatase phase of TiO2 nanoparticles is attributed to the temperature and calcination time (550°C during 3 h, respectively) used during the green synthesis. Hence, the diffraction pattern revealed different peaks located at 25.37°, 37.12°, 37.90°, 48.22°, 54.12°, 55.21°, and 62.91°, corresponding to the Miller index (hkl) of (101), (103), (004), (200), (105), (211), and (204) planes, respectively. According to the pattern of the ZnO nanoparticles, several peaks can be observed between 5° and 60° corresponding to the characteristic planes of the hexagonal structure (Wurtzite), which are represented by the angles at 31.71°, 34.31°, 36.31°, 47.51°, 56.51°, 62.81°, and 66.31°. These results are in agreement with the Joint Committee on Powder Diffraction Standards (No. 79-2205). The average crystal sizes were determined using the Debye–Scherrer formula (equation (3)): 77

XRD patterns of TiO2 and ZnO nanoparticles synthesized by green ultrasound-assisted chemistry using aqueous lemongrass extract. XRD: X-ray diffraction; TiO2: titanium dioxide; ZnO: zinc oxide.
where
The absorbance UV-vis spectrum of the prepared nanoparticles is shown in Figure 5, indicating the typical curve for the interaction of Ti-O and Zn-O structures. The optical band gap energy (E g) was calculated according to the Kabelka–Munk function by using the following equation:
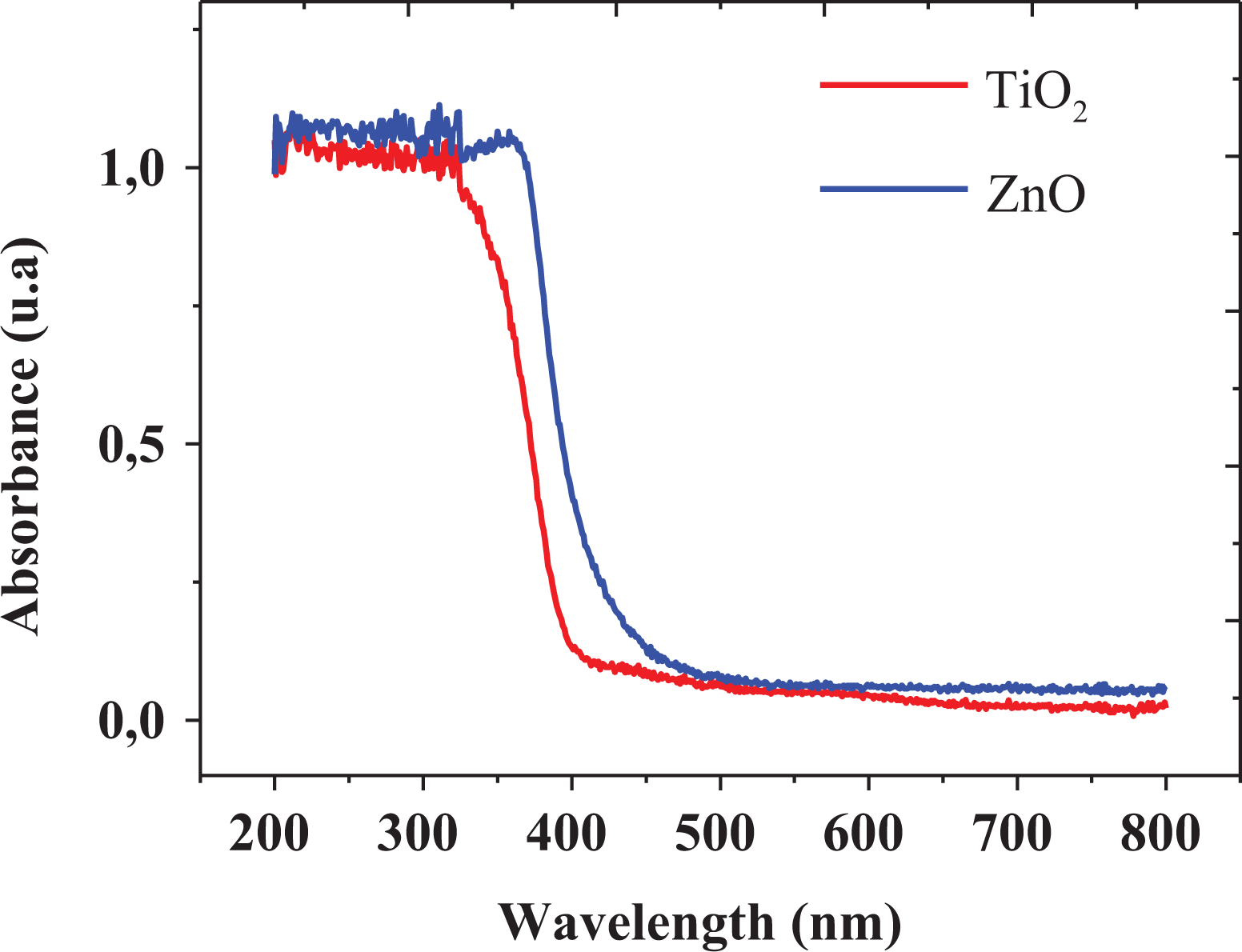
UV-vis spectra of the as-synthesized TiO2 and ZnO nanoparticles. UV-vis: ultraviolet–visible; TiO2: titanium dioxide; ZnO: zinc oxide.
where hν is the photon energy, α is the absorption coefficient, and A is an energy-dependent constant known as the band tailing parameter. Another constant is n, which is known as the power factor of the transition mode of the materials. Tauc plot was obtained from this function, assuming an indirect transition between the edge of the valence band and conduction for anatase TiO2, and direct for the case of ZnO nanoparticles. 78,79 The calculated values of n according to direct, indirect, direct forbidden, and indirect forbidden were 0.5, 2, 1.5, and 3, respectively. Accordingly, the (αhν)1/n versus hν graph is shown in Figure 6, in which the band gap energy values were 3.14 and 3.16 for TiO2 and ZnO nanoparticles, respectively. These results are in agreement with those reported in literature, suggesting that the nanoparticles offer excellent photocatalytic activity. 80,81

Optical band gap energy estimation for (a) TiO2 and (b) ZnO nanoparticles. TiO2: titanium dioxide; ZnO: zinc oxide.
Table 1 summarized the surface area of the TiO2 and ZnO nanoparticles analyzed by using a multipoint Brunauer–Emmett–Teller (BET). The surface area of TiO2 nanoparticles provided the largest value due to the smaller particle size compared to ZnO. These values were compared with those reported in literature for the same type of nanoparticles synthesized through a green chemistry approach. Furthermore, a single point adsorption total pore volume (
BET surface area analysis of TiO2 and ZnO nanoparticles.

BET: Brunauer–Emmett–Teller; TiO2: titanium dioxide; ZnO: zinc oxide.

Nitrogen adsorption–desorption isotherms of the as-prepared TiO2 and ZnO nanoparticles. TiO2: titanium dioxide; ZnO: zinc oxide.

Calculation of the (a) cumulative pore volume and (b) incremental pore volume for the as-prepared TiO2 and ZnO nanoparticles. TiO2: titanium dioxide; ZnO: zinc oxide.
Corrosion, self-cleaning, and antibacterial testing
Corrosion tests were performed in a salt fog chamber using the ASTM B117:16. Figure 9 shows the photographic record of the carbon steel plates coated with nanofilled paints after 200 h of exposure to accelerated corrosion in a salt solution. The ASTM standard categorizes the degree of oxidation (G) with a scale between 0 and 10 based on the visible percentage of the corroded surface, in which 0% is the highest value with a percentage of oxidized surface greater than 50%, and 10% is the lowest value with a percentage equal to or less than 0.01%. According to the results, the oxidation of the sample’s surface is in agreement with the values obtained from the oxidation degree test reported in Figure 10. Hence, the optimum concentration of nanoparticles for the modification of enamel-type paints corresponded to 2 w/v%, due to the fact that samples exhibited a significantly low level of corrosion compared to those samples using higher concentrations. Therefore, increasing the concentration of nanoparticles in the paints may produce major oxidation of the substrate, which may be related to the fact that agglomerates avoid good dispersion of nanoparticles in the solvent (Varsol) as can be observed in the SEM images of the nanofilled paints in Figure 11, leading to the formation of porosity in the paint coating, and consequently the appearance of corrosion points.

Photographic record of the test saline fog camera after 200 h of exposure, using coated surface with unmodified paint (target) and paints modified with 2%, 3.5%, and 5% of TiO2 and ZnO nanoparticles. TiO2: titanium dioxide; ZnO: zinc oxide.
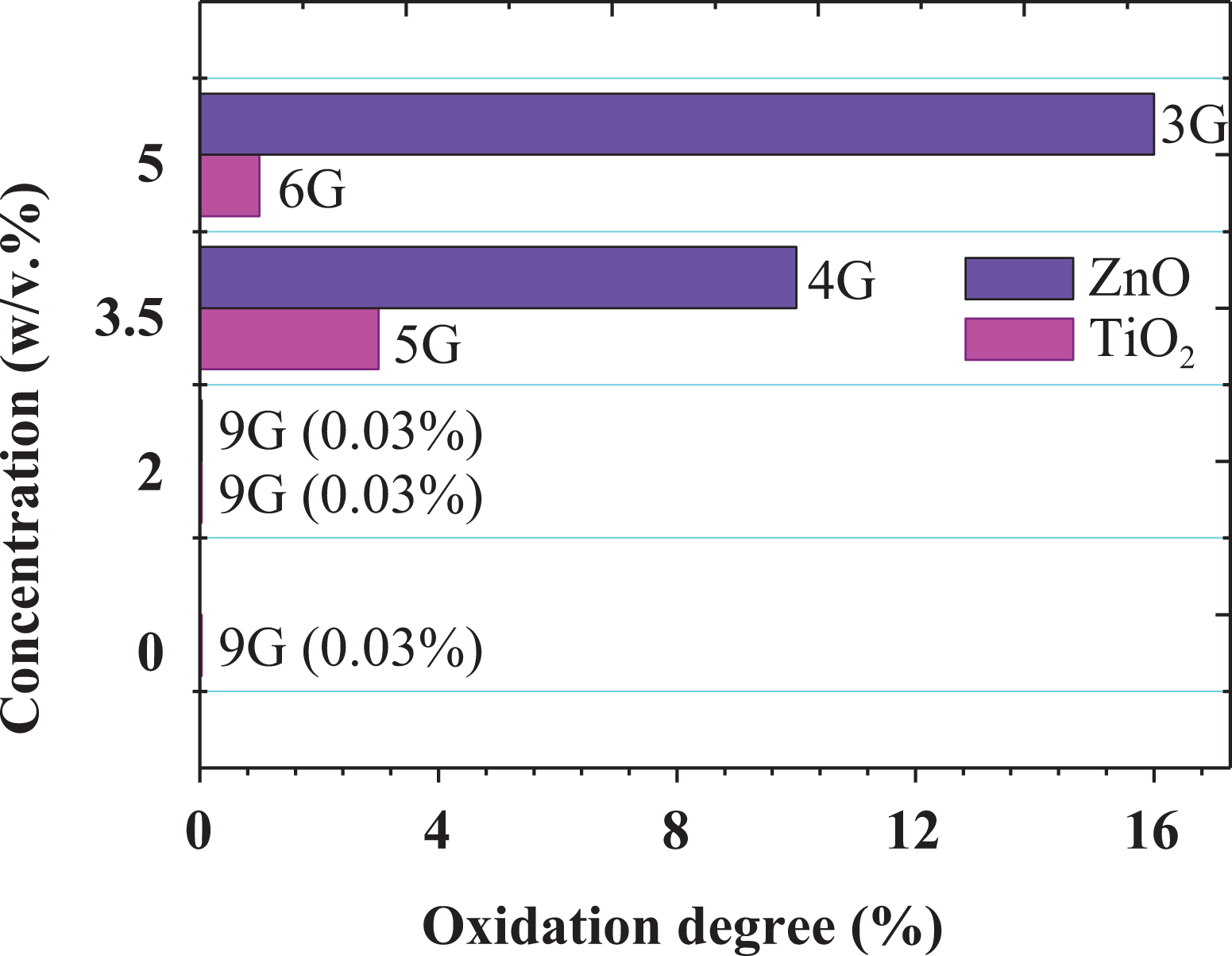
Oxidation degree presented by the samples coated with nanofilled paints by using the ASTM B117:16.

SEM images of the nanofilled paints using a concentration of nanoparticles of 3.5 w/v% for (a) TiO2 and (b) ZnO nanoparticles. TiO2: titanium dioxide; ZnO: zinc oxide.
The photographic record of the self-cleaning tests performed on wood sheets coated with nanofilled paints and stained with MB is shown in Figure 9. The reported images correspond to the concentrations of 2 and 3.5 w/v%, in which a high degradation of MB can be clearly observed, as the accumulated radiation and the concentration of nanoparticles increase, allowing the adsorption and subsequent degradation of the dye. A calibration curve was plotted based on the cyan percentage, in order to corroborate and quantify the data collected in the MB decolorization test as shown in Figure 12. In Figure 13, the photodegradation of MB can be clearly evidenced for all samples, including the target (paint without nanoparticles) with a low photocatalytic activity. The presence of TiO2 and ZnO nanoparticles in the paint considerably improved the photocatalytic degradation of MB, even using 2 w/v% as the lowest concentration tested. Although TiO2 nanoparticles showed higher degradation of MB, ZnO nanoparticles also offered this self-cleaning property with values slightly less than those obtained for TiO2. These results are related to the surface area, as reported in the BET analysis in Table 1, ZnO presented a surface area four times less indicating that an adsorption process may occur at a lower level compared to TiO2. This degradation behavior is in agreement with the results reported by Guo et al., 23 in which TiO2 (P-25) nanoparticles were used to modify white paints for potential application in compacting architectural mortars (SCAM), showing a high self-cleaning capability toward the degradation of rhodamine B under visible light irradiation.

Photographic record of self-cleaning test carried out on wood sheets coated with unmodified paint (target) and paints modified using TiO2 and ZnO nanoparticles (2 and 3.5 w/v%) under solar radiation. TiO2: titanium dioxide; ZnO: zinc oxide.

MB decolorization using unmodified paint (target) and modified with different concentrations of TiO2 and ZnO nanoparticles. MB: methylene blue; TiO2: titanium dioxide; ZnO: zinc oxide.
The proposed mechanism for self-cleaning properties of coatings with nanofilled paints using simulated spot with MB is shown in Figure 14. When samples are exposed to solar radiation, processes such as drying, adsorption, and photodegradation may occur simultaneously. Adsorbed water and oxygen play an important role in self-cleaning properties, working as precursors for the generation of reactive oxygen species by interacting with photogenerated electron/hole pairs (
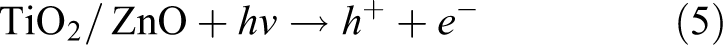








Proposed self-cleaning mechanism of the paints modified with TiO2 or ZnO nanoparticles using simulated spot with MB. MB: methylene blue; TiO2: titanium dioxide; ZnO: zinc oxide.
Antibacterial activity was performed in aluminum disks coated with nanofilled paints against E. coli activity, as is shown in Figure 15(a) to (d) and Figure 16 (a) to (d). TiO2 nanoparticles showed no inhibition of bacterial growth for all concentrations, while the incorporation of ZnO nanoparticles generates a protection barrier against the microorganism. Although some inhibition activity could be observed, no significant radius of the inhibition halos was observed when using ZnO nanoparticles, which may be attributed to the low capability to diffuse from the paint into the culture medium, avoiding contact with the cells. Velmurugan et al. 90 have reported the mechanism of antibacterial action of nanoparticles against several human pathogens. The proposed mechanism behind the inhibition of bacteria using nanoparticles can be explained as follows: (1) the nanoparticles may anchor with the bacterial cell wall and then penetrate causing structural changes in the cell membrane, such as the permeability leading the death of the cell; (2) the formation of free radicals by the nanoparticles is considered other mechanism that leads a damage of the cells; (3) the appearance of porosity in the cell membrane which can slowly lead to death cell; and (4) the inactivation of the cells through electrostatic interaction of thiol groups with vital enzymes.
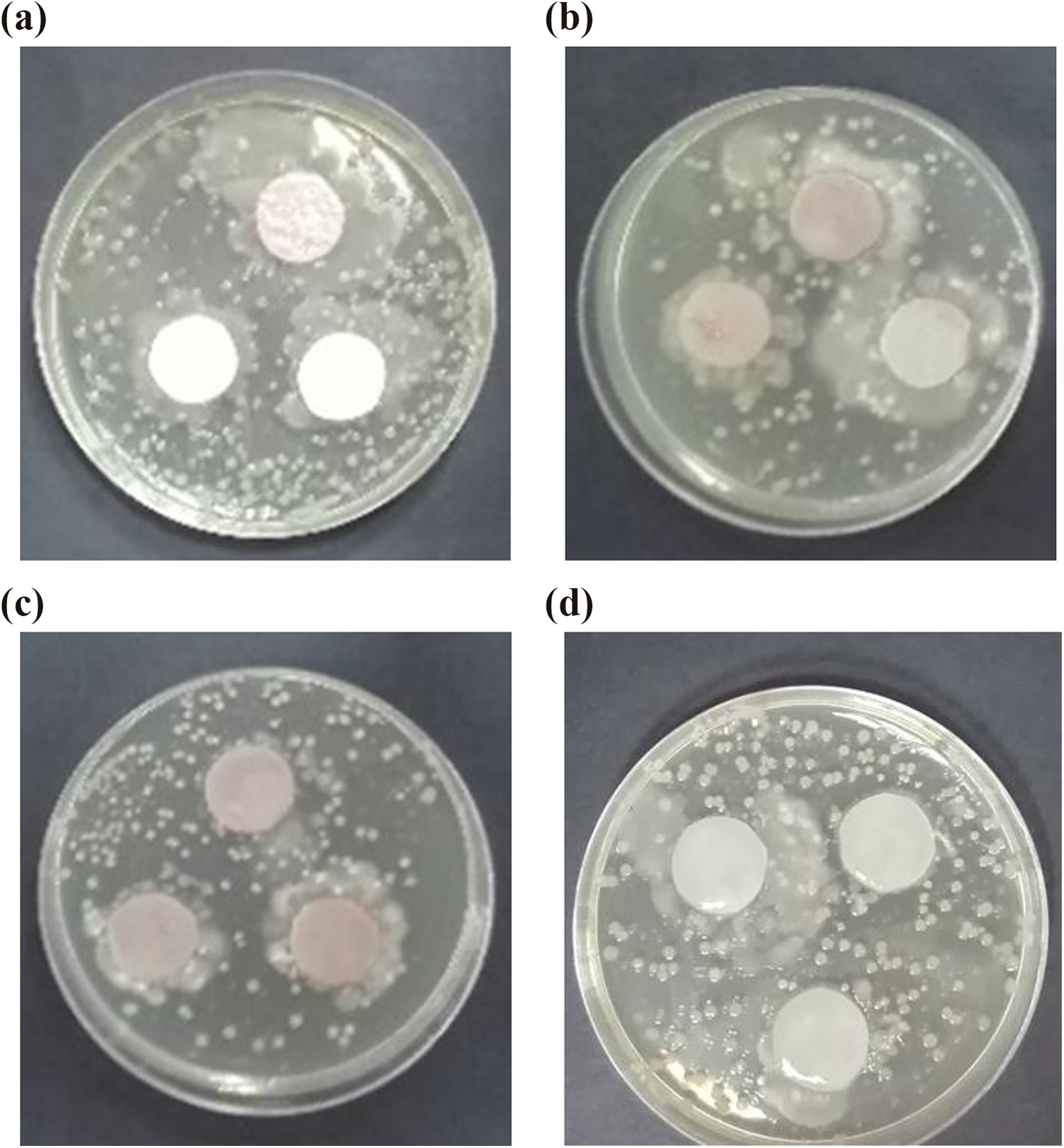
Antibacterial activity of circular disks coated with paint modified using TiO2 nanoparticles at (a) 2, (b) 3.5, and (c) 5 w/v%; compared with the (d) target (coating with paint without nanoadditives). TiO2: titanium dioxide.

Antibacterial activity of circular discs coated with paint modified using ZnO nanoparticles at (a) 2, (b) 3.5, and (c) 5 w/v%; compared with the (d) target (coating with paint without nanoadditives). ZnO: zinc oxide.
Conclusions
The green ultrasound-assisted chemistry methodology using lemongrass extract allowed to synthesize TiO2 and ZnO nanoparticles with excellent structural, morphological, optical, and photocatalytic properties observed from the characterization results, in which phytochemicals worked as capping and reducing agent, respectively. According to the results in the corrosion test, concentrations of nanoparticles in paints higher than 2 w/v% provide an accelerated oxidation of the carbon steel, due to the occurrence of agglomeration which generates more porosity on the surface. Although using less concentration of nanoparticles for the modification of paints may offer positive economic and environmental impacts, a surface modification of TiO2 and ZnO nanoparticles would possibly improve the dispersion in solvent, allowing to reduce the appearance of agglomerates in paints, and thus, the formation of porosity on the sample’s surface. We found that the same behavior of efficiency was observed in the photocatalytic test, in which the samples painted with a nanoparticle concentration of 2 w/v% were found to degrade MB, indicating this nanocomposite as promising for self-cleaning in the construction field. Although TiO2 nanoparticles are agglomerated, the photocatalytic activity was slightly higher than that for ZnO nanoparticles, which is strongly related to the small particle size, and thus, the surface area. We also found that no antibacterial inhibition for the samples using disks with the paint containing TiO2 nanoparticles. However, the results were the opposite for the samples coated with paint containing ZnO nanoparticles, which was possible to observe an increase in the antibacterial inhibition, indicating the diffusion and further interaction of some nanoparticles with the bacteria cell. These findings determined that the modification with a low concentration of TiO2 and ZnO nanoparticles offers an enhancement in the physicochemical properties of paints, allowing to be applied in the construction field as a promising nanocomposite.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The author received financial support for the research and publication of this article from Universidad de Cartagena, according to the Strengthening Plan 2018 (Act of Agreement Number 062-2018).