Abstract
An all-fabric solid-state flexible supercapacitor has been fabricated using three types of commercial woven fabrics made of carbon fiber, activated carbon fiber, and polyester fiber, respectively. The activated carbon fiber fabric is viscose-based twill through carbonization and activation, followed by a deposition of CeO2 or ZnO nanoparticles through a hydrothermal process. The resultant fabric supercapacitor displays a high specific capacitance of 13.24 mF cm−2 at a scan rate of 0.2 mV s−1, an excellent capacitance retention of 87.6% at 5000 charge/discharge cycles, and a high energy density of 4.6 × 10−7 Wh cm−2 at a power density of 3.31 × 10−6 W cm−2. The supercapacitor is capable of bending in any angles without losing its performance, revealing an excellent flexibility. The facile, cost-effective fabrication process and excellent electrochemical performance allow this all-fabric solid-state flexible supercapacitor to be potentially used in new generation of wearable and self-powered electronic devices.
Introduction
Flexible solid-state supercapacitors have attracted an increasing interest for their potential applications in wearable electronic devices, hybrid electric vehicles and portable electronic devices such as mobile phones, e-readers, tablets, laptops, etc. [1]. Compared with conventional supercapacitors, flexible electrochemical supercapacitors possess several important advantages, such as flexibility, lightweight, and high safety. Being bendable, twistable, rollable, and foldable, they can take advantage of the space that may be inaccessible to the conventional supercapacitors [2]. For the purpose of fabricating high-performance flexible supercapacitors, it is very important to seek new types of flexible capacitive materials with high capacitance and to develop novel fabrication approaches.
Flexible capacitive materials
Recently, many carbon nanomaterials including carbon particles [3, 4], carbon nanotubes (CNT) [5, 6], carbon nanofibers [7, 8], and graphene [9, 10] have been utilized as electrochemical active materials and fabricated into flexible electrodes. To enhance the capacitance of flexible supercapacitors, pseudo-capacitive materials such as transition or lanthanide metal oxides such as MnO2 [11], RuO2 [12], Co3O4 [13], CeO2 [14, 15], or ZnO [16, 17], and conducting polymers such as polyaniline (PANI) [18], polypyrrole (PPy) [19], or poly(3,4-ethylenedioxythiophene) (PEDOT) [20], have been fabricated into the electrodes, usually as composites with carbon materials. For example, flexible supercapacitors fabricated by PPy/graphene oxide (GO)/ZnO nanocomposites exhibit a specific capacitance of 94.6 F g−1 at a current density of 1 A g−1. The application of pseudo-capacitive materials PPy and ZnO renders a high energy density of 10.65 Wh kg−1 and power density of 258.26 W kg−1 at 1 A g−1 [17].
Fabrication methods of flexible supercapacitors
Fabrication methods play an important role in the performance of flexible supercapacitors. A main approach for fabricating flexible electrodes is preparing a free-standing film [21–25] or fiber [26–29] from electrochemically active materials, which can be fabricated into thin flexible supercapacitors. For instance, a free-standing and flexible MoS2/GO hybrid film prepared via vacuum filtration presents a high specific capacitance of 380 F cm−3 at 10 mV s−1 [21]. A symmetric supercapacitor fabricated by N/O-enriched carbon cloth delivers a high energy density of 9.4 mW h cm−3 [22]. Flexible, free-standing, and porous carbon films in the absence of conducting additives and binders have been developed by activation of reduced graphene-oxide films with KOH. Flexible supercapacitors fabricated using these films as electrodes reveal a very high power density of about 500 kW kg−1 with a reasonably high energy density of 26 Wh kg−1 [24]. Besides the planar type electrochemical device, fiber-shaped supercapacitors fabricated by freestanding CNT fibers [26, 27] or graphene fibers [28, 29] also render excellent capacitive performance.
Another attractive approach for fabricating flexible electrodes is the deposition of carbon nanomaterials, metal oxides, and conductive polymers onto a flexible support including plastics, papers, and textile fibers [30–32]. Polyethylene terephthalate (PET) film is a relatively common use of a substrate due to its flexibility and excellent mechanical properties. The reduced GO and CNT composites [33], CNT/PANI nanocomposite [34], nanostructured a-MnO2 [35] have been deposited on PET substrate to obtain flexible electrodes. Cellulose printing papers have also been used as a substrate for their porous structure, rough surface properties, and low price [2, 36, 37]. For being able to be knitted into textiles, a number of fiber-shaped supercapacitors have been fabricated by utilizing carbon fiber, cotton thread [38], nanocellulose fibers [39, 40], and TiO2 fibers [41] as substrates. However, these fabrication approaches involve either the high cost of the capacitive materials or the elaborate process, not favorable for mass production [42, 43]. Therefore, it is essential for future research with an aim at increasing electrochemical performance as well as lowering fabrication cost of the supercapacitors.
State of current work
In this paper, we present a carbon-based all-fabric flexible supercapacitor based on an activated carbon fiber fabric, a PET fabric, and a carbon fiber fabric that are used as electrodes, separators, and current collectors, respectively. The commercial viscose fiber woven fabrics are firstly carbonized and activated into capacitive fabrics with high porosity. Then CeO2 or ZnO nanoparticles, which are known as pseudo-capacitive materials and possess lower cost compared to CNT and graphene, are deposited on the activated carbon fiber fabric through a hydrothermal process. The resultant all-fabric solid-state flexible supercapacitor delivers a high device capacitance (13.24 mF cm−2), excellent cycle stability (capacitance retention of 87.6% at 5000 cycles), high power and energy density (4.6 × 10−7 Wh cm−2 at a power density of 3.31 × 10−6 W cm−2). Moreover, this facile, cost-effective and binder-free fabrication process is favorable for commercial production, showing great potential for use in new generation self-powered electronic devices.
Materials and experimental methods
Raw materials and modification
The activated carbon fiber fabric used in this study was produced from a viscose twill fabric precursor (denoted as VF) by carbonization and activation and is commercially available. This activated viscose fabric was labeled as ACVF. It had a fabric areal density of 154 g m−2 and fabric thickness of 0.432 mm. The carbon fiber fabric was a commercial plain woven fabric with fabric areal density of 212 g m−2 and thickness of 0.254 mm. The separator layer in the fabric supercapacitor was a commercial lightweight PET mesh fabric with fabric areal density of 44 g m−2 and thickness of 0.120 mm.
Two different suspensions were prepared by adding 10% CeO2 and ZnO nanoparticles by weight of the ACVF fabric into water. After ultrasonic dispersion of the suspension for 30 min, the ACVF sample was immersed in the suspension. The system was further ultrasonically dispersed for 30 min and then vacuum dried at 60℃ for 24 h. Once dried, the CeO2- and ZnO-modified ACVF fabric samples were obtained and labeled as CeO2-ACVF and ZnO-ACVF, respectively.
Fabrication of all-fabric supercapacitors
Five grams of PVA was slowly dissolved in 50 ml distilled water with agitation at 95℃ until a clear homogeneous mixture was achieved. Then 5 g H3PO4 was added, and the resulting solution was stirred continuously for 1 h. The mixture was used as electrolyte. Two pieces of ACVF were immersed in the electrolyte solution for 24 h. Then they were taken out and assembled into an all-fabric supercapacitor as electrodes, together with a piece of the PET fabric as a separator and two pieces of the carbon fiber woven fabric as current collectors.
Characterization
Nitrogen adsorption and desorption isotherms were measured at 77 K using a Micromeritics 3Flex automatic system. The specific surface area was determined via the Brunauer-Emmett-Teller (BET) method, and the pore size distribution (PSD) was estimated according to Horvath-Kawazoe model on the basis of slit-like pore geometry. The BET theory is widely used for the calculation of surface areas of solids by physical adsorption of gas molecules that do not chemically react with material surfaces. Nitrogen is the generally utilized gaseous adsorbate. The morphologies of the surface and cross-section of the ACVFs were observed by scanning electron microscopy (SEM) (Hitachi S5500), and the chemical element analysis of materials was also performed using this instrument equipped with energy dispersive spectroscopy (EDS). The electrochemical measurements were performed 3 h after the assembling of the all-fabric supercapacitors. Cyclic voltammetry (CV) measurements were carried out using an Autolab instrument (PGSTAT 128 N) from 0 to 1.0 V. Electrochemical impedance spectra (EIS) were measured using this instrument in a frequency range of 0.01 Hz to 100 kHz. Galvanostatic charge-discharge (GCD) tests were performed using a BT2000 Battery tester (Arbin Instruments) between 0 and 1.0 V.
Results and discussion
Porosity and morphology of materials
The schematic procedure of electrode fabrication is illustrated in Figure 1. Firstly, the precursor VFs were carbonized and activated into ACVFs to obtain great porosity in the fibers. The N2 adsorption/desorption isotherm of ACVF is shown in Figure 2. N2 uptake increases sharply at low relative pressure and approaches a plateau at high relative pressure. This suggests that the micropores are formed in ACVF [44]. Moreover, no apparent adsorption/desorption hysteresis loop is observed. This suggests that the isotherm of ACVF is typical type I and the micropores are dominating in ACVF. The inserted plot presents the PSD graph of ACVF. The abbreviated term DV/DW along the Y-axis of the graph represents differential pore volume. The PSD results reveal that ACVF consists mostly of micropores with a pore size range of 0.4–1.8 nm and two peaks at 0.46 and 0.54 nm respectively. The BET surface area of ACVF reaches 1179 m2 g−1.
Schematic procedure of electrode fabrication. N2 adsorption/desorption isotherm of activated viscose fabric with insertion of pore size distribution curve.

The morphology of the fiber surface in the ACVF sample is shown in Figure 3(a). The diameter of the fiber is approximately 7–9 µm. The cross-section of the fiber under larger magnification in Figure 3(b) shows a very uneven, rough surface, which can be ascribed to its great porosity. The EDS analysis of the cross-section of the fiber is shown in the inserted graph. The two main peaks including C and O appear in the range of binding energies from 0 to 1000 eV, suggesting that C and O are the major chemical elements that constitute the ACVF sample. By calculating the area of each peak, C element holds a dominant position. The carbon enrichment can be attributed to the carbonization and activation process at 800℃ [44]. Figure 3(c) and (d) present the SEM images of CeO2-ACVF and ZnO-ACVF samples, respectively. As shown in Figure 3(c) and (d), a layer of CeO2 and ZnO are deposited on the surface of fibers, respectively through the hydrothermal process.
Scanning electron microscopy images of (a) the activated viscose fabric (ACVF) surface; (b) the ACVF cross-section with an insertion of energy dispersive spectroscopy analysis; (c) the surface of CeO2-ACVF fiber; and (d) the surface of ZnO-ACVF fiber.
Structure and electrochemical performance of supercapacitors
Activated carbon has been widely used as an electrode material for supercapacitors because of its high porosity. In most cases, activated carbon is used as powder or fiber particle form [45–47]. In this case, the commercial fabric VF was carbonized and activated into ACVF with high porosity, and then was directly used as two electrodes to form an electrostatic type (Helmholtz Layer only) supercapacitor. The CeO2-ACVF and ZnO-ACVF fabrics were also used individually as electrodes to form two electrochemical type (redox reaction) supercapacitors. These three all-fabric flexible supercapacitors were all constructed using the commercial PET mesh fabric as a separator and the conductive carbon woven fabric as two current collectors. The construction of the all-fabric supercapacitors is illustrated in Figure 4.
Structure of the all-fabric supercapacitor.
To assess the electrochemical performance of the all-fabric supercapacitors, CV, EIS and GC measurements were conducted. Figure 5(a) shows the CV curves of the ACVF supercapacitor that use the ACVF as electrodes at different scan rates. The CV loops exhibit a quasi-rectangular shape indicating the capacitive behavior of double-layer electrostatic capacitor. Figure 5(b) and (c) present test results of the supercapacitors using the CeO2-ACVF and ZnO-ACVF as electrodes. Their CV curves reveal an increasingly enlarged loop upon an increase of the scan rates. For the purpose of comparison, the CV curves of the three supercapacitors at the scan rate of 20 mV s−1 are shown in Figure 5(d). Apparently, the curves of CeO2-ACVF and ZnO-ACVF exhibit a more rectangular-like shape and a larger size than that of ACVF, indicating that both the CeO2-ACVF and ZnO-ACVF supercapacitors possess a better capacitive behavior. Because both CeO2 and ZnO are electric active materials, a pseudofaradic reaction may take place during the charging and discharging process [48], as described below
Cyclic voltammetry curves of (a) activated viscose fabric (ACVF) supercapacitor; (b) CeO2-ACVF supercapacitor; (c) ZnO-ACVF supercapacitor at different scan rates; and (d) all supercapacitors at the scan rate of 20 mV s−1. (e) Calculated specific capacitances. (f) Nyquist plots of the supercapacitor electrochemical impedance spectra.
From Figures 5(b) and (c), a remarkable redox peak at 0.3 V and a less distinct one at 0.2 V are exhibited in the CV curves of ZnO-ACVF and CeO2-ACVF, respectively. The improvement in capacity behavior of the CeO2-ACVF and ZnO-ACVF supercapacitors should be due to the reaction between CeO2 or ZnO nanoparticles and the electrolyte. Although both CeO2 and ZnO nanoparticles indicate the improved capacitive performance of the ACVF supercapacitor for their pseudo-capacitive characteristics, the ZnO-ACVF supercapacitor shows a better capacitive behavior than the CeO2-ACVF supercapacitor. A major reason is probably because ZnO is a typical transition metal oxide having a better performance for redox reaction, while CeO2 is not. This phenomenon needs a further investigation.
To compare the capacitance performance of different supercapacitors, their specific capacitances need to be calculated via the equation
To further evaluate the capacitive behavior of the ACVF-based supercapacitors, EIS measurements were conducted at a frequency range from 0.01 Hz to 100 KHz. As shown in Figure 5(f), the Nyquist plots exhibit an indistinct semicircle in the high frequency region, reflecting a very low charge interfacial transfer resistance at the electrode/electrolyte interface [38], which is benefited from the unique porous and open structure of the ACVF. At the low frequencies, the sloping lines demonstrate the capacitance characteristic. However, the Nyquist plots render a linear line slightly inclining to the Z′ axis, which is corresponding to Warburg resistance, the result of the frequency dependence of ion diffusion from electrolyte to electrode/electrolyte interface [55, 56]. The equivalent series resistance (ESR) values can be obtained at the first intercepting points of the EIS plots on Z′ axis. For the ACVF supercapacitor, ESR value is about 9.80 Ω. It is assumed that this large ESR is mainly determined by the relatively poor conductivity of the insulated ACVF electrodes. So it can be proposed that improving ACVF conductivity can reduce ESR value and enhance overall performance of the ACVF supercapacitors. Furthermore, a thick separator layer between two ACVF electrodes is also a cause to increase ESR value. Through the inclusion of CeO2 and ZnO nanoparticles into ACVF, the CeO2-ACVF and ZnO-ACVF supercapacitors exhibit lower ESR values of 5.06 Ω and 1.01 Ω, respectively, suggesting that the deposition of oxide on ACVF is capable of enhancing the electronic and ionic diffusion.
The capacitive performance of the all-fabric supercapacitors was also investigated by the GCD measurements. Figure 6(a) shows the GCD curves of the supercapacitors at a current density of 4 × 10−5 A cm−2. Compared with the ACVF supercapacitor, both the CeO2-ACVF and ZnO-ACVF supercapacitors show a longer discharge time, denoting a higher capacitance. This is consistent with the CV results. Symmetry of GCD curves is related to capacitive behavior. For the ACVF supercapacitor, the charge time and the discharge time are around 237 and 102 s, respectively, indicating an asymmetry of the GCD curve and a low charge efficiency, which may be attributed to a low electrical conductivity of ACVF. By incorporating CeO2 and ZnO nanoparticles into ACVF, the charge and discharge time become 228 and 211 s for the CeO2-ACVF supercapacitor, and 323 and 256 s for the ZnO-ACVF supercapacitor, their charge/discharge curves become fairly symmetric, demonstrating that both CeO2 and ZnO nanoparticles can improve the charge efficiency of the ACVF supercapacitor.
Performance of charge/discharge, energy density, and retention. (a) GCD curves at a current density of 4 × 10−5 A cm−2; (b) CeO2-activated viscose fabric (ACVF) cycling performance at the current density of 4 × 10−5 A cm−2 after 5000 charge/discharge cycles; (c) Ragone plots; and (d) CeO2-ACVF specific capacitances after 3 h and 4 months of fabrication.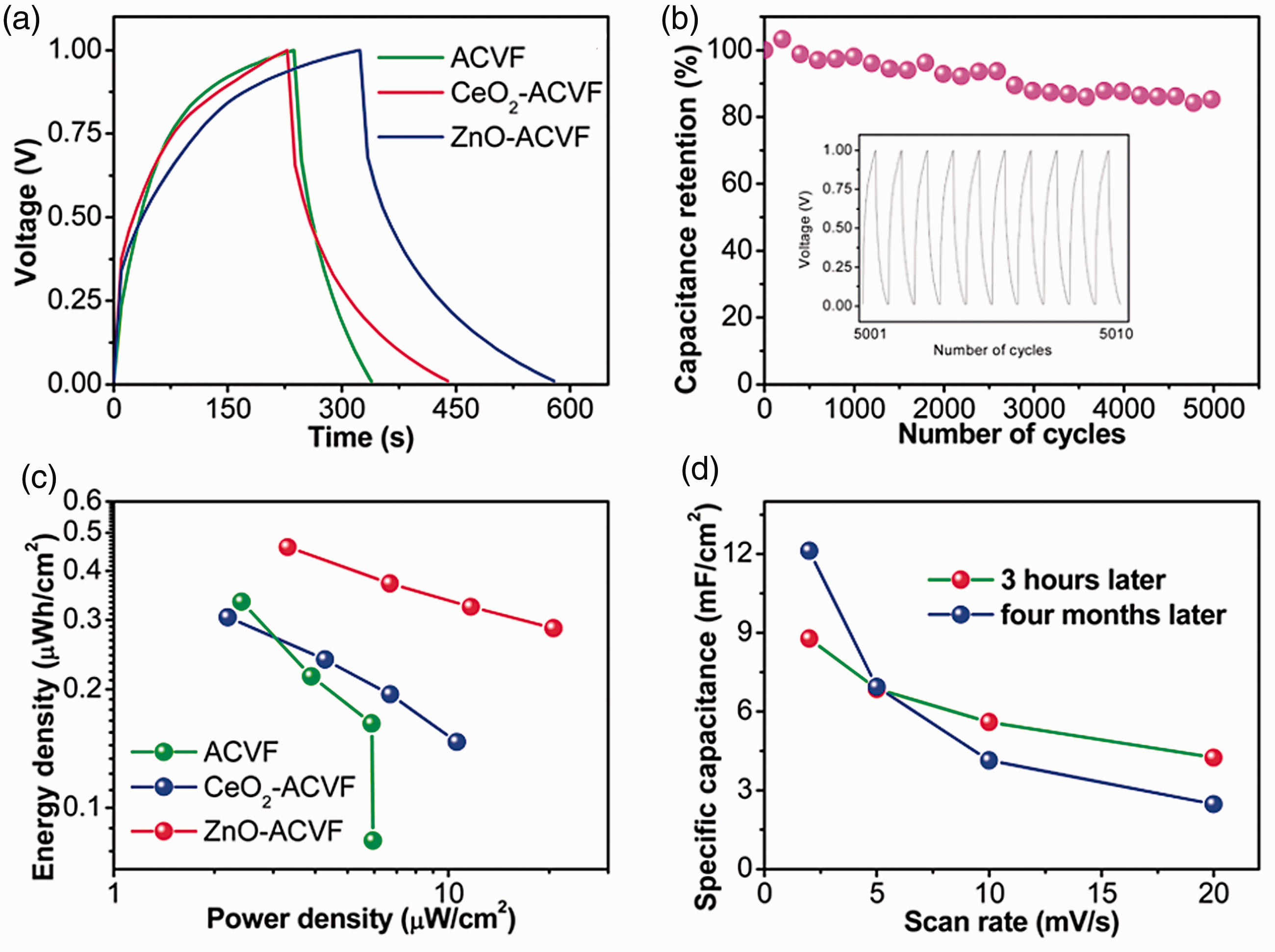
The stability of supercapacitors is an important factor for commercial applications. The cycling performance of the CeO2-ACVF supercapacitor was studied by measuring its charge–discharge curves for 5000 cycles. As shown in Figure 6(b), the specific capacitance slowly decays to 87.6% of the initial value at the 3000th cycle and remains almost constant for the remaining 2000 cycles. A recent research on a nonfabric solid-state supercapacitor with a PVA-based electrolyte reported that its capacitance retention after 3000 cycles of charge-discharge was around 90% [57]. In comparison to this, the present CeO2-ACVF supercapacitor also holds a good stability. It should be noted that this study did not include the stability test for the ZnO-ACVF supercapacitor because of the time-consuming measurement.
Figure 6(c) presents the Ragone plots of different supercapacitors. The energy densities of CeO2-ACVF and ZnO-ACVF are 3.0 × 10−7 Wh cm−2 and 4.6 × 10−7 Wh cm−2 while their power densities are 2.19 × 10−6 W cm−2 and 3.31 × 10−6 W cm−2, respectively. Generally, the energy densities increase with decreasing power densities for all types of supercapacitors. The energy densities of CeO2-ACVF and ZnO-ACVF are higher than that of ACVF especially for higher power densities. These demonstrated that CeO2 and ZnO nanoparticles can enhance the energy storing ability of the ACVF supercapacitor.
About 4 months after fabrication, the CV and EIS measurements were performed again on the CeO2-ACVF supercapacitor. The comparison of the specific capacitances of the supercapacitor at different scan rates before and after 4 months is illustrated in Figure 6(d). After 4 months, the specific capacitances decrease from 5.6 and 4.24 mF cm−2 to 4.14 and 2.47 mF cm−2 at the scan rate of 10 and 20 mV s−1, respectively. This may be because of moisture evaporation in the electrolyte which can possibly be improved by utilizing other electrolyte like ionic liquids. As a result, ESR of the supercapacitor increases.
Being a portable and wearable energy storage device, both flexibility and safety are very important. Figure 7(a) presents the CV curves of the CeO2-ACVF supercapacitor measured under various bending angles. The CV curves are in rectangular shape, and the sizes are nearly the same in various bending angles, demonstrating that the deformation would not affect the capacitive performance of the supercapacitor. As shown in Figure 7(b), the CeO2-ACVF supercapacitor is able to be bent in any angles without losing its performance, showing a high flexible property. The capacitive performance was also tested upon applied bending cycles. As shown in Figure 7(c) and (d), the specific capacitance of the supercapacitor does not decline but increases by 13.4% after 200 bending cycles, denoting that the bending cycles also would not affect the capacitive performance of the supercapacitor. These results demonstrate that this carbon-based flexible fabric supercapacitor is suitable for being a portable and wearable energy storage device.
Influence of bending deformation on CeO2-ACVF capacitive performance. (a) and (b) CV curves and specific capacitance under various bending angles; (c) and (d): CV curves and specific capacitances after different bending cycles. ACVF: activated viscose fabric.
Integration of ACVF supercapacitor with solar cell
Solar energy is probably the most clean and renewable energy in the world, but it is always limited by time, location, and weather. Integrating solar cells with energy storage devices such as supercapacitors and batteries may offer a solution to build stand-alone self-powered systems [16, 58, 59]. To demonstrate potential applications of the carbon-based all-fabric supercapacitors, a supercapacitor/solar cell integration was formed by using four fabric supercapacitors and four solar cells units each capable of charging a 1.2 V AA Ni-Cd rechargeable battery with a capacity of 400 mAh. When sunlight was available, each supercapacitor was connected with a solar cell, so the solar cell harvested solar energy and stored it in the supercapacitor. After 120 s charging under the illumination, the voltage of each supercapacitor reached approximately 0.9–1.1 V (Figure 8(a)). Then the solar source was turned off, and the four charged supercapacitors were connected to power a commercial red light-emitting diode (LED). The four series supercapacitors could illuminate the red LED for more than 130 min, and could still maintain a voltage output around 1.61 V after 130 min (Figure 8(b)).
The voltage profile for (a) the fabric-based supercapacitor when being charged by the solar cell and (b) the device of four series supercapacitors in powering a red LED.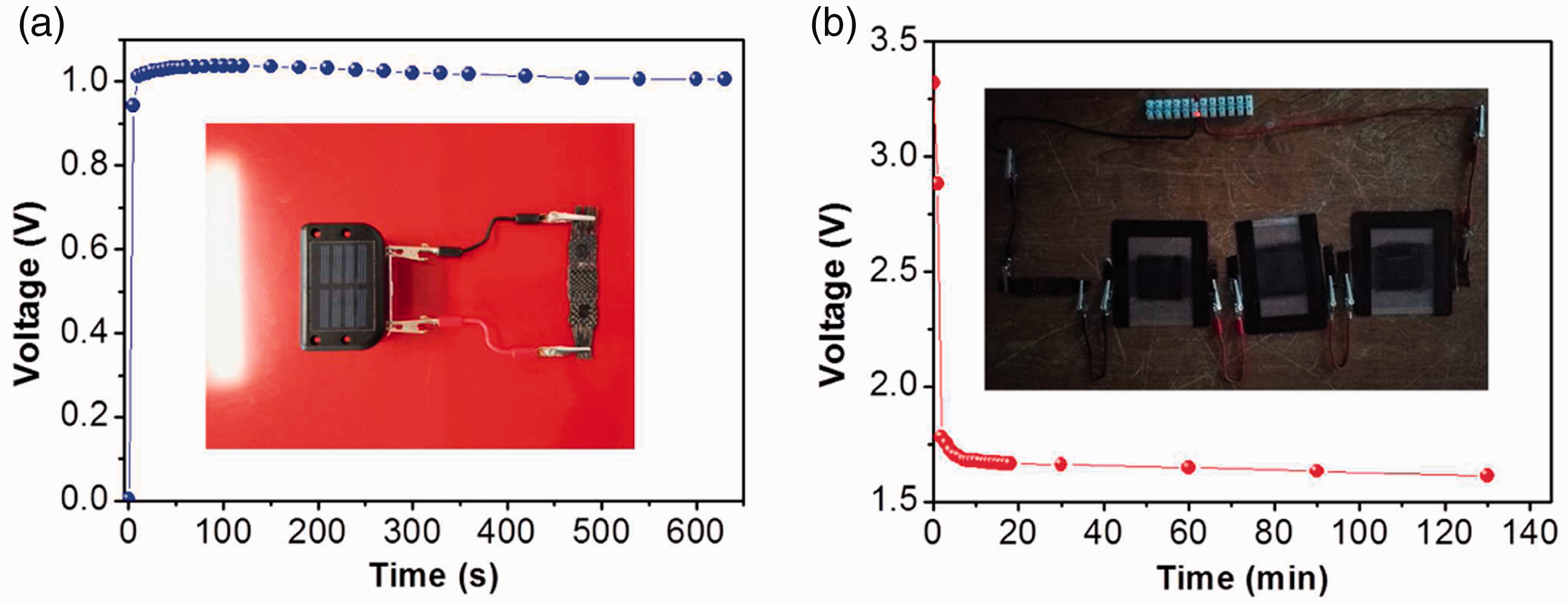
Considering the flexibility of the fabric, it is feasible to integrate flexible solar cells and the fabric-based supercapacitors into a self-powered system. This self-powered device can adopt a flexible multilayer sheeting structure. Its area and shape can be defined according to different end uses. Thus, the fabric supercapacitor can be directly integrated into apparel, bag, tent, and other consumer products, making these fabric-based products a mobile and portable solar power supply. Furthermore, the technology does not need any new yarn spinning and fabric-forming infrastructures. A simple short supply chain would be expected.
Summary
Overall, the facile, cost-effective carbon-based fabric supercapacitor exhibits high capacitive performance with a specific capacitance of 13.24 mF cm−2 at scan rate of 0.2 mV s−1, a capacitance retention of 87.6% at 5000 charge/discharge cycles, and an energy density of 4.6 × 10−7 Wh cm−2 at power density of 3.31 × 10−6 W cm−2. The supercapacitor is susceptible to cyclic bending in 0–180° angles without losing its performance. CeO2 and ZnO nanoparticles can enhance the energy storing ability of the ACVF supercapacitor. Compared with the ACVF supercapacitor, both the CeO2-ACVF and ZnO-ACVF supercapacitors show a longer discharge time, denoting a higher capacitance.
Conclusion
The commercial woven viscose fabrics were firstly carbonized and activated into capacitive fabrics, and then impregnated with CeO2 and ZnO nanoparticles individually through a hydrothermal process. The as-prepared activated carbon fiber fabrics were utilized as electrodes to fabricate all-fabric solid-state flexible supercapacitors, in which the commercial PET fabric and conductive carbon fiber fabrics were used as separator and current collector, respectively. The as-constructed all-fabric supercapacitors possess a high specific capacitance, an excellent cycling retention of capacitance, a high energy density and power density, and could maintain their performance even under harsh bending conditions. They hold great potential for many energy storage applications, such as smart clothing and mobile and portable self-powered systems. The method to fabricate these carbon-based all-fabric flexible supercapacitors is also facile and cost-effective, favorable for mass production.
Footnotes
Acknowledgements
The authors would like to acknowledge their thanks to China Scholarship Council for sponsoring Dr Weihua Luo and Ms Xinxin Li as visiting scholars. Thanks are also extended to Micromeritics Instrument Corporation for helping the test of ACVF surface and micropore properties.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.


