Abstract
The present work deals with the development of electrically conductive cotton fabrics by in-situ deposition of copper particles. The dynamic light scattering, scanning electron microscope, and X-ray diffraction techniques were employed to study the morphology of deposited copper particles. The utility of conductive fabrics was analyzed for electromagnetic shielding ability over frequency range of 30 MHz to 1.5 GHz. The electromagnetic interference shielding was found to increase with increase in number of dips, which was attributed to increased reflection of EM waves due to dense, uniform, and percolated network of conductive copper particles on the surface. The sample produced from 100 and 150 dips exhibited the maximum shielding ability of 10 dB and 13 dB, respectively. Furthermore, the role of deposited copper particles on antibacterial properties was examined against pathogenic bacteria such as Staphylococcus aureus and Escherichia coli. The S. aureus showed more sensitivity towards copper particles as zone of inhibitions increased from 9.5 to 15.5 mm. At the end, the durability of fabrics was examined against washing after application of binder. The fabrics showed good retention of the copper particles, proved by scanning electron microscopic microstructures and small loss in the conductivity of the material after washing.
Keywords
Introduction
In recent years, research on electromagnetic interference (EMI) shielding materials has increased dramatically [1]. The EMI shielding can be defined as to prevent the propagation of electrical and magnetic waves from one place to another by using either a conductive or magnetic material. The effect of EMI shielding can be accomplished by minimizing the signals passing through a system either by absorption or reflection of waves [2]. The reflection of radiation from the shield is possible if the shield has mobile charge carriers [3]. From these aspects, flexible conductive textiles have gained the popularity due to their satisfactory electrical conductivity. The metal yarns or inorganic compounds are commonly used to develop EMI shielding fabrics. However, these materials cannot be widely used for personal protective clothing because of their heavy weight, cost inefficiency, poor flexibility, and poor scratch resistance [4]. Moreover, they exhibit only a single function (i.e. EMI shielding).
In order to render the textiles with electrical conductivity, there are various approaches such as surface metallization, coating of carbon materials, coating of conductive polymers, etc. [5, 6]. Among them, textile surface metallization is unique method which can provide multifunctional properties such as electrical conductivity, EMI shielding, anti-static and anti-bacterial properties, UV radiation screen, and radar reflectivity [7–9]. The commonly used metal coating techniques are metal foil and laminates, conductive paints, and lacquers. However, metallic fabric produced in such traditional manners consists of defects, such as stiffness, poor air permeability, and heavy in weight [10]. The sputter coating, vacuum deposition, flame and arc spraying, and electroless plating are some of the novel methods of surface metallization which can overcome the limitations of traditional methods [10, 11].
The electroless plating has advantages such as coherent metal deposition, excellent conductivity and shielding effectiveness, and applicability to complex shaped materials [9]. In previous research, fabrics plated with Cu showed more effective EMI shielding than those plated with Ni and Cu–Ni [12]. Each metal coated fabrics showed EMI SEs of 68–35 dB for copper, 37–32 dB for nickel, and 46–32 dB for copper–nickel, respectively, at the frequency of 100 MHz–1.8 GHz [4]. A number of techniques have been devoted to plating of copper using formaldehyde as reducing agent, however this process may release hazardous gases during their operation [3]. In another research, the ultrasonic-assisted electroless Ag plating of polyethylene terephthalate fabrics showed the SE of more than 32 dB at frequency ranging from 0.01 MHz to 18 GHz [13]. However, the plating of silver is costly than plating of copper. Therefore, further research is necessary to effectively modify the cotton fabric surface with controllable conductivity and high durability in safe conditions. Several papers have studied the electroless copper plating solutions using sodium hypophosphite as reducing agent due to its low pH, low cost, and relative safety features [3]. Those research focused mainly on the effect of additives on the properties of the deposits and the application of electroless copper plating to fabrication of printed circuit boards. However, no study on the EMI shielding, mechanical durability, wash ability or antimicrobial properties has been made.
Apart from EMI shielding property, personal protective clothing should also possess great antistatic, thermal insulation, antibacterial, and elastic properties to increase its hygiene and thermo-physiological comfort properties. The present work deals with the development of electrically conductive, multifunctional and durable cotton fabrics by in-situ deposition of copper particles using sodium hydrosulfite as the reducing agent. The morphology of in-situ deposited copper particles was studied by dynamic light scattering, scanning electron microscope (SEM), and X-ray diffraction (XRD) techniques. The utility of conductive fabrics was analyzed for electromagnetic shielding ability over frequency range of 30 MHz to 1.5 GHz. Furthermore, the role of deposited copper particles on antibacterial properties was examined against pathogenic bacteria such as Staphylococcus aureus and Escherichia coli. At the end, the durability of coated fabrics was examined against washing. In this way, the present research could serve to develop multifunctional cotton fabrics for applications in hospitals or biosensor electronics.
Experimental methods
Materials
Plain woven cotton fabric having areal density 150 g/m2 was used as substrate to produce the conductive fabrics. The chemicals used for the synthesis and deposition of copper particles had 99.99% purity and were obtained from Merck Co. Ltd. The organic–inorganic non-conductive binder for fixation of particles was obtained from Bezema, Switzerland with trade name of ISYS MTX.
Deposition of copper particles
Copper sulfate was used as the base material along with sodium hydrosulfite as reducing agent for in-situ deposition of copper particles on cotton fabrics. At first, different concentrations of copper sulfate from 6 g/200 mL, 4 g/200 mL, and 2 g/200 mL were dissolved in distilled water. Then, the cotton fabric (4 × 4 in.2) was dipped in the solution and dried at 100℃ for 3 min. This procedure of dipping and drying was continuously carried out for 50, 100, and 150 cycles. The dwell time of fabric in solution was about 30 s against each dip. Subsequently, the treated fabrics were transferred to the 6 g/200 mL sodium hydrosulfite solution. The concentration of sodium hydrosulfite was determined based on the concentration of copper sulfate. It should be always higher or at least equal to copper sulfate concentration. The reduction was continued for the duration of 20 min. The duration of reduction treatment was determined based on change in color of cotton. The cotton fibers changed their color from blue to blackish gray after 15 min of reaction with reducing agent, so 20 min of reduction treatment was allowed to complete the reaction.
Characterization of copper particles
The morphology of copper particles deposited on fabric surface was investigated by particle size analysis (Malvern Zetasizer), SEM analysis (Zeiss), energy dispersive X-ray spectroscopic (EDX) analysis (FEI Quanta 50), and XRD analysis (PANalytical X’pert PRO equipment). The dilute dispersion of copper particles was prepared in deionized water. It was ultrasonicated for 5 min with bandelin ultrasonic probe before characterization of particle size distribution on Zetasizer. The microstructure of copper coated fabrics was observed on SEM at accelerated voltage of 15 kV. The XRD pattern was measured in the 2θ range 10–80, at operating conditions of 40 kV and 30 mA using Cu-Kα X-ray source.
Characterization of copper coated fabrics
Electrical conductivity
The resistivity was measured using four probe meter according to ASTM D257-14 standard at 22℃ temperature and 40% relative humidity as shown in Figure 1. The specific voltage potential of 100 ± 5 V using direct current was applied across opposite ends of copper coated fabric sample and resultant current flowing across the sample was measured after 15 ± 1 s. The test was repeated five times for each samples and an average value was calculated from equation (1)
Measurement of electrical resistivity of conductive fabric.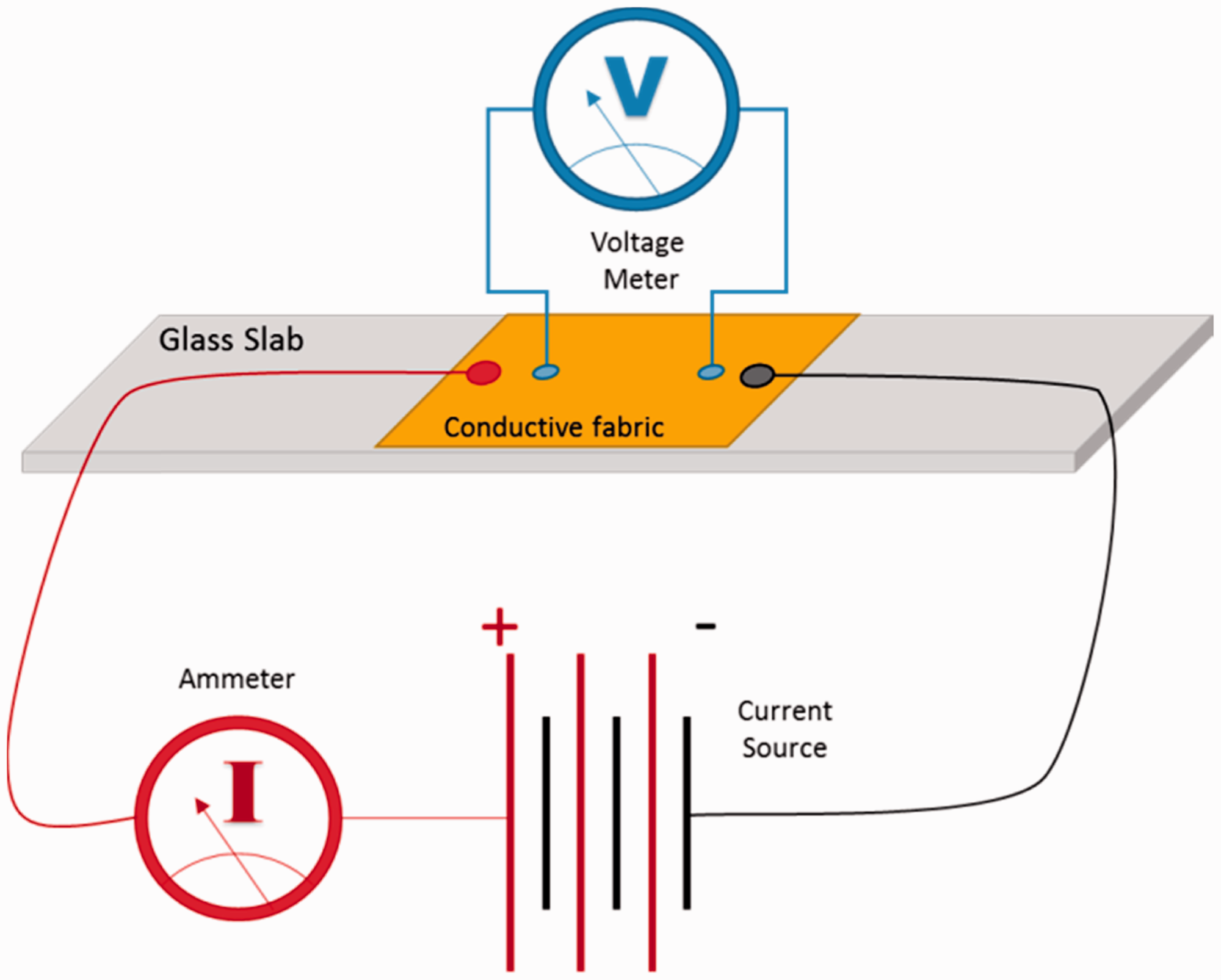
Electromagnetic shielding
It was measured by coaxial transmission line method using insertion loss principle according to standard ASTM D4935-10 over frequency range of 30 MHz to 1.5 GHz. The measurement set-up consisted of a sample holder with its input and output connected to the network analyzer. A shielding effectiveness test fixture (Electro-Metrics, Inc., model EM-2107A) was used to hold the sample. The network analyzer (Rohde & Schwarz ZN3) was used to generate and receive the electromagnetic signals. The ratio between transmitted to incidents power of the electromagnetic wave was calculated to express the effectiveness of EMI shielding (SE) in dB as depicted in equation (2)
Mechanical properties
The TIRA test 2300 Labor Tech machine was used to measure the tensile properties of copper coated fabrics as per ISO 1924-2 standard. Furthermore, Shirley crease recovery tester was employed to determine the crease recovery angle of the samples as per ASTM D-1296.
Antibacterial properties
The zone of inhibition test (AATCC 147) was conducted to confirm the antibacterial property of copper coated fabrics [14]. The zone of inhibition is a clear area of interrupted growth underneath and along sides of the test material and indicates the bioactivity of specimen. It is a qualitative test for the bacteriostatic activity by the diffusion of antibacterial agent through agar. The bacterial strains of Gram-negative E. coli (CCM 3954) and Gram-positive S. aureus (CCM 3953) used in this study were obtained from the Czech Collection of Microorganisms, Masaryk University Brno, Czech Republic. Bacterial suspensions were always prepared fresh by growing a single colony overnight at 37℃ in a nutrient broth. All agar plates were freshly prepared before the antibacterial tests.
Durability of copper coated fabrics
In order to improve the durability of coated fabrics without loss of functional properties, low concentration of binder was applied to hold the particles on the surface of fibers. At first, 1 g of ISYS MTX binder was dissolved in 700 mL of distilled water and pH of the solution was maintained at 6 with the help of acetic acid. Then, copper coated fabric was dipped in binder solution for 15 min and dried at room temperature. The washing durability of copper coated fabrics was studied to have an idea of their activity in service. It was examined according to ISO 105-C01 by vigorously stirring the copper coated fabrics in 5 g/L standard detergent with the liquor ratio of 50:1. Sample was rinsed at 40℃ with stirring speed of 800 r/min for 30 min. After washing, all samples were dried and conditioned in a standard atmosphere (65% humidity; 25℃) for 24 h before testing. Later, the performance was verified based on measurement of electrical conductivity and SEM observation of copper particles on the fabric surface.
Results and discussion
Electrical conductivity of copper coated cotton fabrics
The effect of copper sulfate concentration and number of dips were investigated for electrical resistivity of copper coated textiles. The development of electrical conductivity was verified by flow of electric current as shown in Figure 2.
Flow of electricity in copper coated cotton fabrics.
It is clear from Figure 3 that the higher concentration of copper sulfate solution resulted in higher electrical resistivity of coated fabric samples. This behavior can be attributed to the formation of big sized copper particles at higher concentration of copper sulfate solution. Surprisingly, lower concentration (2 g/200 mL) of copper sulfate produced more conductive fabrics due to formation of percolated network by creation of continuous connectivity between the small sized copper particles. This can be further justified from SEM images shown in Figure 4 ((a), (b)), where formation of a more percolated network of smaller particles can be found in case of 2 g/200 mL than 4 g/200 mL copper sulfate concentration. Therefore, the action of agitation or ultrasonication for disrupting the nucleation of copper particles is good topic for further research in order to obtain more percolated network at higher copper sulfate concentration. Furthermore, the electrical resistivity was found to reduce with increase in number of dips for all concentrations of copper sulfate solution. This indicated more dense and uniform deposits of copper particles at higher number of dips. This can be further justified from SEM images shown in Figure 5. As lower concentration of copper sulfate provided acceptable results for electrical conductivity, in further sections detailed discussion is made on samples coated with 2 g/200 mL of copper sulfate.
Effect of copper sulfate concentration on electrical conductivity. SEM image of copper coated fabrics for different copper sulfate concentration. (a) 2 g/200 mL and (b) 4 g/200 mL. SEM: scanning electron microscope. SEM image with EDX spectra for copper coated cotton fabrics (a) 50 dips, (b) 100 dips, and (c) 150 dips. EDX: energy dispersive X-ray spectroscopic; SEM: scanning electron microscope.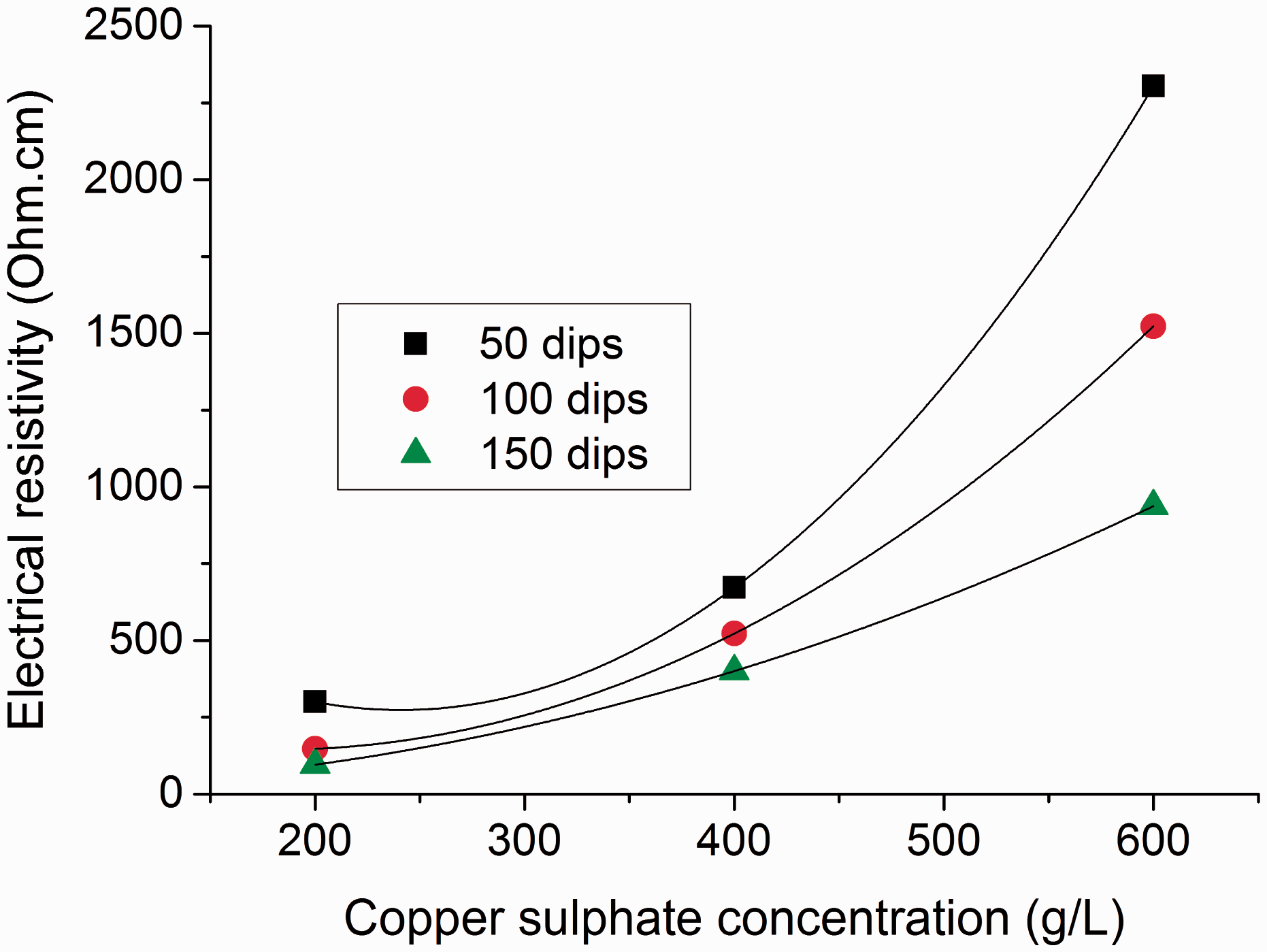


Morphology of copper coated cotton fabrics
SEM microstructure
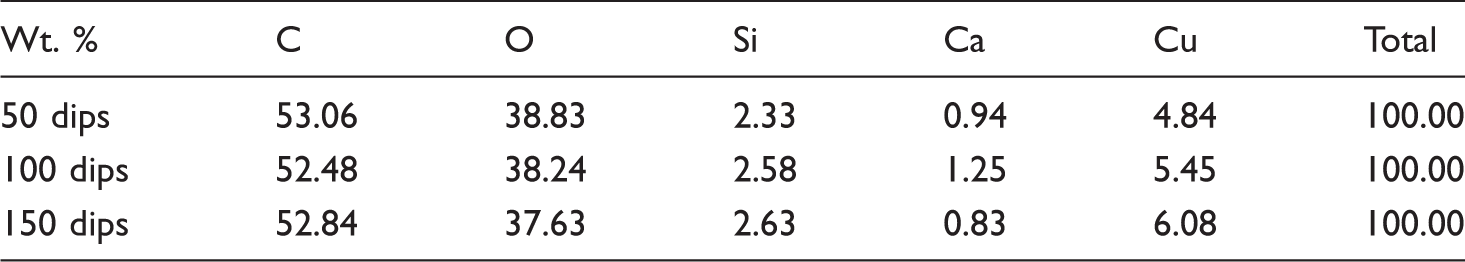
Elemental composition of copper coated cotton fabrics.
Particle size distribution
The dynamic light scattering technique was employed to measure the particle size based on Brownian motion of particles. Figure 6 shows the particle size distribution of copper particles. The copper particles were found having multi-modal distribution with size varying from micrometer to the nanometer range. The average particle size was measured around 500 nm and it has advantages over those particles which are below 100 nm. The reduced inflammatory response and toxicity can be expected from the copper particles produced in this work as compared to smaller copper particles (i.e. below 100 nm) of the same chemical composition [15, 16].
Particle size distribution of copper particles.
XRD analysis
The XRD analysis was carried out to know the phase composition of deposited copper particles. The XRD spectra of the cotton fibers dipped and dried in the copper solution prior to reduction treatment are not given because it will only show the peaks of cotton due to absence of crystal structure of copper. The XRD analysis was performed on copper coated cotton fibers after reduction step. Figure 7 shows the XRD patterns of samples for the 2θ range of 10° to 80° with a step of 0.02°. The phase purity of the prepared copper particles can be clearly seen from perfect indexing of all the diffraction peaks to the copper structure. The diffraction peaks appeared at 2θ of 43.3o, 50.5o, and 74.2o represented (1 1 1), (2 0 0), and (2 2 0) planes of copper, respectively [3]. The crystalline nature of copper particles was confirmed from the sharp peak, whereas the broadening of the peaks indicated the formation of nanoscale copper particles [17].
XRD patterns for copper coated cotton fabrics. XRD: X-ray diffraction.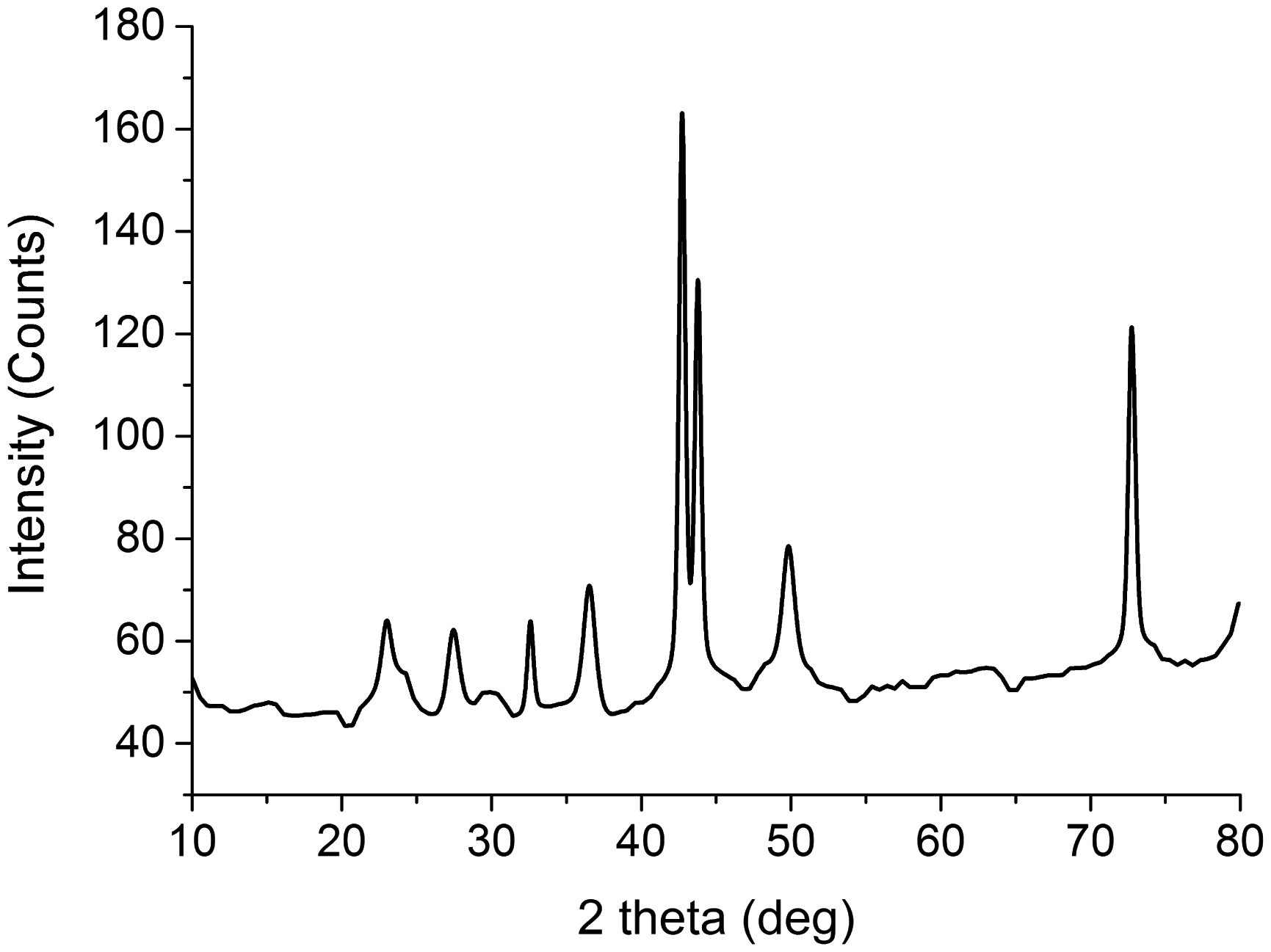
Mechanism of copper deposition
The mechanism of attachment of copper on cotton fabric surface can be explained from the schematic diagram shown in Figure 8. The dissolution of CuSO4 in water resulted into Cu+2 and Schematic of copper deposition on cotton fabrics.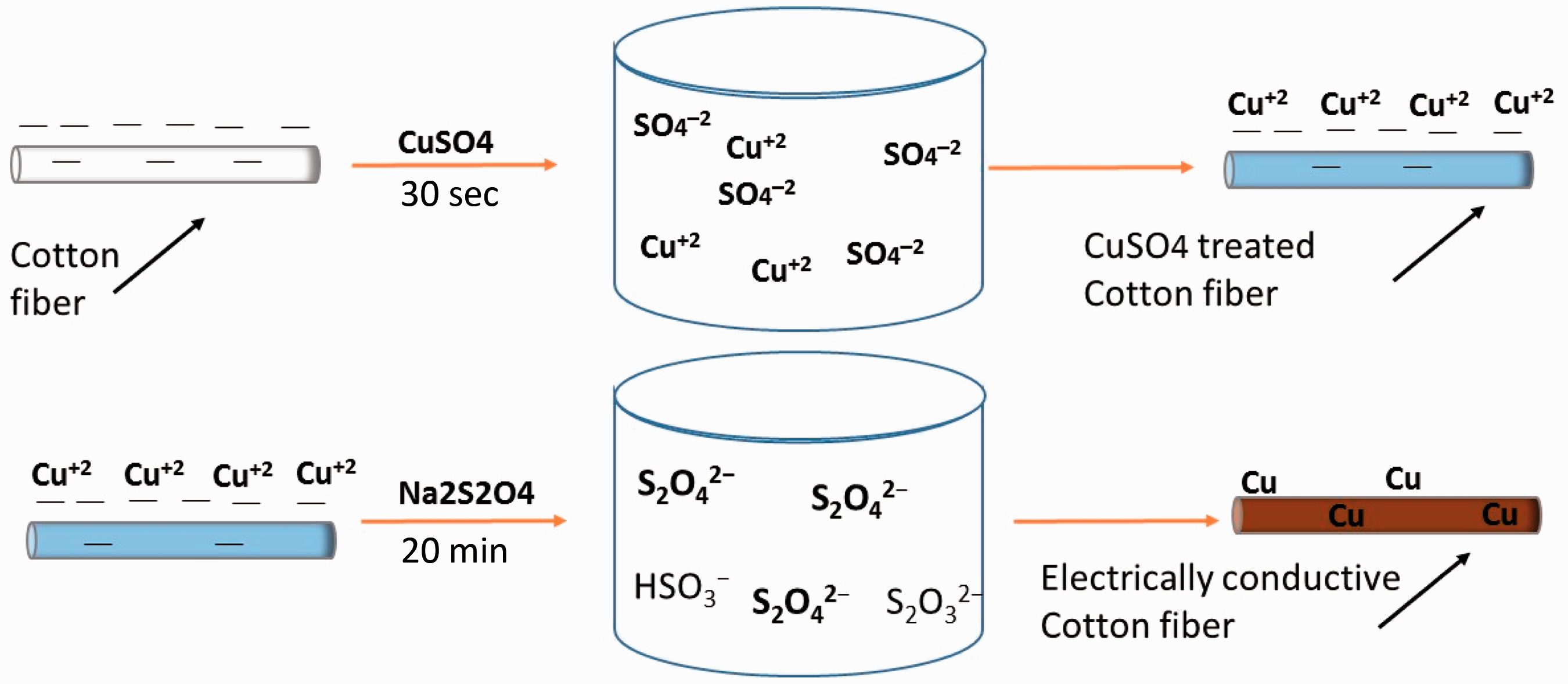
Due to small dwell time of fabric (i.e. 30 s) in solution and no preferential adsorption of copper ions on fabric with increase in number of dips, the concentration of copper sulfate did not change significantly after 50, 100, and 150 dips. However, the concentration of sodium hydrosulfite was reduced to greater extent for samples of higher dips. The color of cotton changed from white to blue after treatment with copper sulfate solution, and then from blue to dark brown after treatment with reducing agent sodium hydrosulfite.
Electromagnetic shielding of copper coated fabrics
Figure 9 shows the results of shielding effectiveness for the fabric samples coated from 2 g/200 mL of copper sulfate solution after 50, 100, and 150 dips. The number of dips showed significant effect on shielding effectiveness. The samples showed increase in shielding effectiveness with more number of dips in copper sulfate solution. The sample produced from 50 dips revealed the lowest electromagnetic shielding effectiveness of about 6 dB in frequency range of 600 MHz–1.5 GHz. On the other hand, the sample produced from 100 and 150 dips exhibited the maximum shielding ability of 10 and 13 dB, respectively. This behavior was attributed to increased reflection of EM waves due to formation of dense, uniform, and percolated network of conductive copper particles with higher number of dips [2]. This can be further explained by SEM images shown in Figure 5, where dense network of copper particles can be found for more number of dips.
Shielding effectiveness of copper coated cotton fabrics.
Mechanical properties of copper coated fabrics
Mechanical properties of copper coated cotton fabrics.


Load–elongation curve of copper coated fabrics.
Antibacterial properties of copper coated fabrics
The anti-bacterial property is important because EMI shielding fabrics are excellent media for microorganism growth, particularly when used in hospitals or working environments with unhealthy indoor air quality [23]. The anti-bacterial properties are studied in this work to find suitable applications of coated fabrics as personal protective clothing in hospitals. The antibacterial activity of copper coated fabrics was tested against Gram-negative E. coli and Gram-positive S. aureus. Figure 11 shows the zones of inhibition around fabric samples after 24 h of incubation in dark at 37℃. The test was repeated three times and the average value of zone of inhibition presented in Figure 12. From Figure 11, the virgin cotton fabric without copper coating showed no antibacterial activity. However, the zone of inhibitions was evidenced against both types of bacteria S. aureus and E. coli after the copper coating. Further, S. aureus depicted the highest sensitivity as compared to E. coli. The zone of inhibitions for S. aureus increased from 9.5 to 15.5 mm, while for E. coli it increased from 7.5 to 12 mm with increasing number of dips (Figure 12). The antibacterial property of coated fabrics can be attributed to the combination of chemical and physical interactions of bacteria with copper particles [24, 25]. The mechanisms associated with the antibacterial behavior of copper nanoparticles can be summarized as shown in Figure 13. It shows catalyzed radical formation, release of metal ions, and Trojan-horse effect due to endocytosis processes [26].
Antibacterial properties of copper coated cotton fabrics. (a) S. aureus and (b) E. coli. Zone of inhibition for copper coated cotton fabrics. Mechanism of antibacterial performance for copper particles. Electrical conductivity of copper coated cotton fabrics before and after washing .



Durability of copper coated fabrics after binder application
When the intake of copper exceeds the range of biological tolerance, it can cause adverse effects, including hemolysis, gastrointestinal distress, and liver and kidney damage in humans [27]. Therefore, the removal of copper particles and their durability was verified against washing. A low concentration of binder was applied on previously discussed samples to improve the adhesion of copper particles with fabric surface. The electrical conductivity of samples was measured before and after application of binder. From Table 3, there was no significant difference in change in conductivity of fabrics before and after binder application. Due to application of less concentration of binder (i.e. 1 g/700 mL), the conductivity of fabrics was found mostly the same. Therefore, similar EMI shielding properties can be expected for the fabrics with binder included. Furthermore, no significant decrease in the conductivity of the binder applied fabrics was found before and after washing (see Table 3). This indicated efficient working of binder to firmly fix the copper particles on cotton fabric surface without deterioration of electrical conductivity. The SEM images shown in Figure 14 also confirmed the presence of copper particles on fabric surface after washing. This indicated strong attachment of copper particles with fabric surface and therefore reduced toxicity in routine applications.
SEM image of copper coated cotton fabrics after washing. (a) 50 dips, (b) 100 dips, and (c) 150 dips. SEM: scanning electron microscope.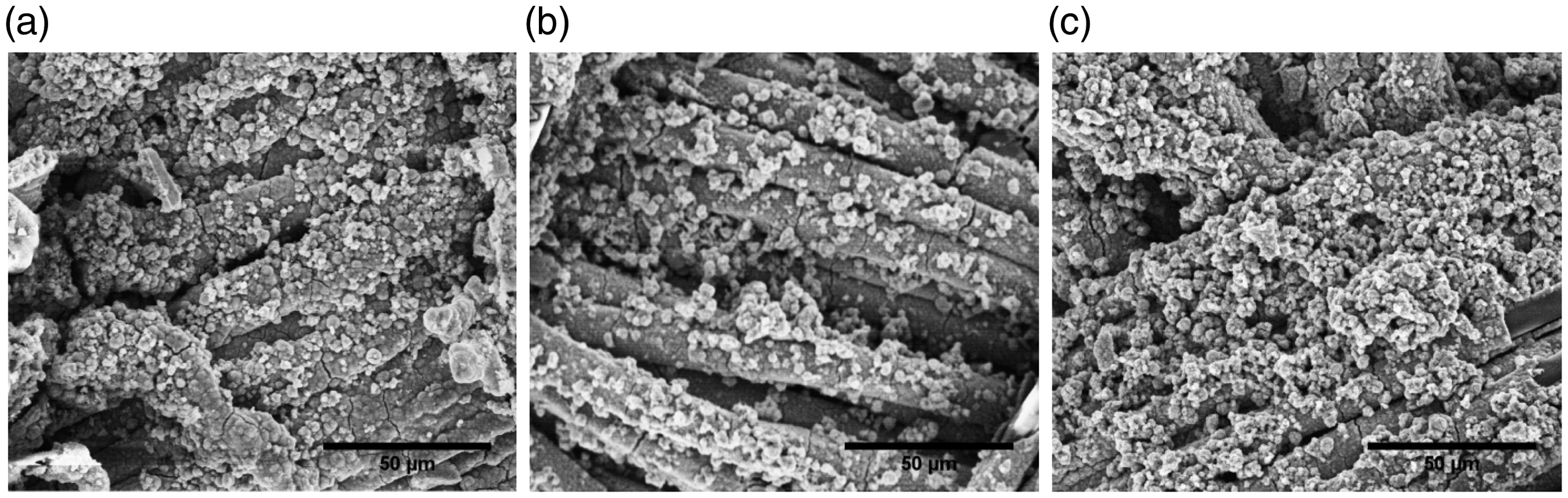
Conclusions
The present study was focused on development of electrically conductive, multifunctional and durable cotton fabrics by in-situ deposition of copper particles. The copper particles were incorporated into the structure of cotton by sequential dipping in copper sulfate and then sodium hydrosulfite solutions. The amount of deposition of copper particles was controlled by several numbers of dips (i.e. 50 to 150) in copper sulfate. The concentration of copper sulfate was decided at 2 g/200 mL based on acceptable level of achieved electrical conductivity. Further, the morphology of coated fabrics and copper particles was studied from dynamic light scattering, SEM, and XRD techniques. The utility of conductive fabrics was analyzed for electromagnetic shielding ability over frequency range of 30 MHz to 1.5 GHz by coaxial transmission line method. The EMI shielding was found to increase with increase in number of dips, which was attributed to increased reflection of EM waves due to dense, uniform, and percolated network of conductive copper particles on the surface. The sample produced from 50 dips revealed the lowest electromagnetic shielding effectiveness of about 6 dB in frequency range of 600 MHz to 1.5 GHz. On the other hand, the sample produced from 100 and 150 dips exhibited the maximum shielding ability of 10 dB and 13 dB, respectively. For multifunctional behavior, the copper coated cotton fabrics were further examined for antibacterial properties against pathogenic bacteria such as S. aureus and E. coli. The zone of inhibitions for S. aureus increased from 9.5 to 15.5 mm, while for E. coli it increased from 7.5 to 12 mm with increasing number of dips. When mechanical properties were tested, the load bearing capacity of cotton fabrics was found to improve due to coating of copper particles. The copper particles improved the crease recovery angle and rigidity of cotton fibers to some extent. Towards the end, the durability of coated fabrics was examined against washing after application of binder on the fabric. The fabrics showed good retention of the copper particles, proved by SEM microstructures and small loss in the conductivity of the material after washing. Therefore, the outcome of this work could provide alternative inexpensive and easier ways to obtain copper coated electrically conductive, EMI shielding and antibacterial textiles.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: the research project of Student grant competition of Technical University of Liberec no. 21197 granted by Ministry of Education, Youth and Sports of Czech Republic.


