Abstract
This article reviews the preparation, development and characteristics of conductive polymer-based electro-conductive textile composites for electromagnetic interference shielding. Modification of ordinary textile materials in the form of electro-conductive composites makes them suitable for this purpose. Various metallic and non-metallic electro-conductive textiles have been explored here as the material for electromagnetic shielding. Different approaches of preparing textile electromagnetic shield have been described here. Recent advancements of application of conductive polymers in the field of textile electromagnetic shielding are described. Conductive polymer-coated textile materials showed superior electrical property as electromagnetic shield. Different methods of applications of conductive polymers onto textile surface are described here with their relative merits and demerits. Different conductive polymer-coated woven and nonwoven fabrics prepared by various researchers for electromagnetic shielding are taken into account. The effects of different process parameters of polymer processing on electromagnetic shielding are described.
Keywords
Introduction
Electromagnetic interference (EMI) shielding is a process of limiting the penetration of electromagnetic rays into a space by blocking them by a barrier made of conductive material. It is a very popular method of protecting electronic and electrical equipment and even people against electromagnetic radiation. The material or protector which protects a body, environment or a circuit from harmful electromagnetic radiation is called a shield. Shields are used either to isolate a space (a room, an apparatus, a circuit, etc.) from outside sources of electromagnetic radiation, or to prevent the unwanted emission of electromagnetic energy radiated by internal sources. It is well known that exposure to long-term or acute electromagnetic radiation can have harmful effects on human tissue [1–5], and furthermore, electromagnetic radiation can interfere with certain bio-electronic devices, such as pacemakers [6], and affect the lives of people. The recent proliferation of electronic devices, such as cell phones, and computer equipment, that emit low levels of electromagnetic radiation, or interference, has significantly increased the problem and created a need for everyday shielding garments. In this regard, a variety of electromagnetic shielding fabrics and garments are produced to prevent electromagnetic radiations [7–9].
Mechanism of electromagnetic shielding
When electromagnetic rays pass through a medium or object, they interact with molecules of the medium or object. This phenomenon of interaction can be divided into two major steps: (a) absorption attenuation and (b) attenuation due to reflection [8]. As the rays in forms of wave strike a conductive object, the charges in the object are forced to oscillate at the same frequency of the incident wave. These forced oscillating charges behave like antenna and cause reflection from the surface. The signal wave may reflect in many directions depending upon the pattern associated with a signal oscillating charge. Hence, the signal is scattered and there is some signal loss. This mode of signal loss is called attenuation due to reflection. There are also successive losses of signal in the depth of the layer of the medium. It is called attenuation due to successive internal reflections. Again, the forced oscillating charge loses some energy in the medium in terms of heat. This mode of signal loss is known as attenuation due to absorption. Thus, electromagnetic shields work on the basis of the above-mentioned two major electromagnetic mechanisms, viz. reflection from a conducting surface and absorption in a conductive volume. An electromagnetic wave striking a conductive surface encounters both types of loss [8,10,11]. The combined effect of these losses (reflection and absorption) determines the effectiveness of the shield as shown in Figure 1. Reflection from an electromagnetic shield occurs when the impedance of the wave in free space is different from the impedance of the electromagnetic wave in the shield. This phenomenon is independent of the thickness of shield and is a function of conductivity, magnetic permeability and frequency of the shield [11,12], whereas electromagnetic shields made of electromagnetic absorbers attenuate undesirable electromagnetic waves and substantially dismiss electromagnetic radiation. Shielding efficiency of this type of shield depends upon factors such as type of material, its thickness, size, shape and orientation of apertures [12–14].
Schematic representation of interaction between electromagnetic rays and a media or an object.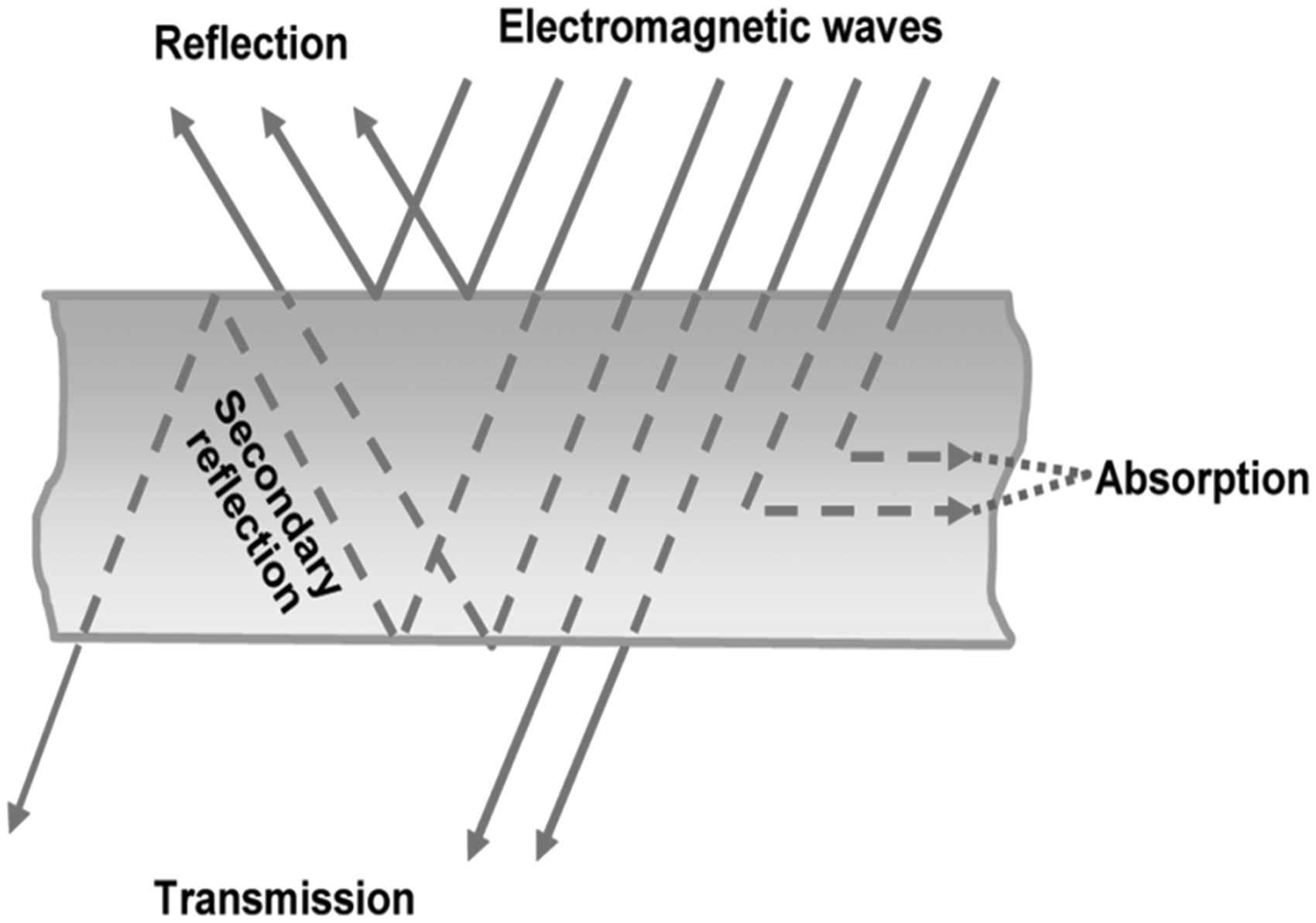
Electromagnetic shielding efficiency
Electromagnetic interference shielding efficiency (EMISE) value expressed in decibels (dB) is defined as the ratio of the incident to transmitted power of the electromagnetic wave [10,15]. Mathematically
By measuring the reflectance (Re) and the transmittance (Tr) of the material, the absorbance (Ab) can be calculated using following equation
Preparation of textile EMI shield
Protection from electromagnetic radiation can be achieved by covering ourselves with an electro-conductive media which can generate and transport free charges. It is known that synthetic textile fibers such as polyester, polyamide, acrylic and cellulose-acetate exhibit a very poor electrical conductivity and are hydrophobic in nature. When these fiber masses are rubbed, static electricity is generated and accumulates on the fibers [16,17]. Natural fibers, such as cotton, wool and silk, exhibit a relatively high hydrophilic property and have relatively higher electric conductivity in favor of static charge dissipation. They are suitable for static charge dissipation but do not serve as effective electromagnetic shield [18]. For this purpose, metallic fibers, or fibers coated with a metal, as an electrically conductive material are commonly used [8,11,19–23]. Also, metal fibers are used during staple spinning to manufacture electro-conductive yarns [23–26]. Those yarns are used in weaving or knitting to produce electro-conductive fabrics. But the processing of those yarns is difficult and they lose their textile properties [27]. These metallic fibers are not proved to be satisfactory for practical use due to their poor recovery from bending and breakage [7]. The breakage results in a decrease in the conductive effect of the metallic fiber. Also, it is difficult to mix spin, mix weave or mix knit the metallic fibers with organic polymer fibers. In case of textile fiber, yarn or fabric coated with a metal layer by electro-plating technique, it is required to form a uniform and continuous layer on the textile surface. In order to satisfy the above-mentioned requirement, rather than other difficulties, the surface of the fiber is required to be smooth and highly polished. Also, intensive care is required during coating operation to coat a continuous layer of metal with uniform thickness. The requirement causes the cost of the coating operation to be very high. Also, the metal-coated fiber has demerits in terms of easy peeling off from the fiber during processing or use [28]. The fiber coated with a polymer dope containing an electrically conductive material, such as carbon black and silver particles, is also found unsatisfactory because the coating operation is expensive and the coated layer is easily peeled off from the fiber during processing [29,30]. When carbon black is used in dope mixing with polymer to spin a conductive filament then at least 15% of carbon black is required based on the weight of the fiber matrix polymer. This large amount of carbon black causes the fiber-forming process to be difficult, complex and expensive. Also, it is impossible to contain the carbon black inside the natural fibers. Many such limitations associated with processability, flexibility and durability could be successfully overcome by coating/applying conducting polymers such as polyaniline, polypyrrole (PPy) and polythiophene on textile substrates [31]. These conductive polymer-coated textile materials, owing to their flexibility, durability, ease of preparation and application, are considered promising for shielding of electromagnetic radiation [7,32–34].
Metallization of textile materials
Textile fabrics have been coated with metals such as aluminum, copper, nickel, silver and combination of them in industrial scale. The coating method employed mainly is chemical method [7,35]. Also, other methods such as laminating with aluminum foils, dyeing with copper sulfide or electroless plating with certain metallic components are used. Electroless metal plating is a non-electrolytic method of deposition of metal from solution by mixed potential theory of combination of oxidation and reduction processes [36]. This method has some advantages such as coherent metal deposition, excellent conductivity and shielding effectiveness. It is also applicable to complex-shaped insulating textile materials [36].
Surface modification and coating of fabrics using vapor deposition (PVD) technologies is relatively new [37]. PVD coating of woven or nonwoven fabrics is an important step for producing a variety of new thin-film composites for new applications. Depending on the evaporation conditions, PVD-coated fabrics can present quite different surface properties [37]. The shielding efficiency of these metalized textile fabrics mainly works by principle of energy reflection and not its absorption. In many cases, such a phenomenon is not satisfactory. Hence, there have been searches for materials with greater capability of absorbing electromagnetic radiation.
In recent years, some patents have appeared which deal with the problem of producing textile fabrics with ferromagnetic properties. Such products can be applied as flexible screens for attenuating electromagnetic radiation to produce cores in transformers, motors, generators, etc. and to produce filters to remove substances showing magnetic properties from air and water [7]. Nonwoven materials with ferromagnetic coating showed electromagnetic shielding effectiveness (EMSE) of about 30 to 35 dB in the frequency range of 1000 to 2000 MHz [7]. Those materials are proposed for applications of the camouflage of military objects, restricting the range of fields emitted by devices such as shortwave and microwave diathermy.
Textile materials embedded with metallic wires and fibers are also explored as EMI shield. The copper wire and stainless steel (SS) wire are wrapped with polyamide filaments to produce a hybrid conductive yarn which is found suitable for weaving and knitting [19]. The co-woven-knitted and knitted fabrics produced from these yarns show desired electromagnetic shielding properties [19]. Figure 2 shows the effect of single and multi-layer fabrics made of SS/polyester (40/60) spun yarn on shielding effectiveness. In another study, polypropylene (PP) filaments are wrapped on copper wire and polyamide filaments to produce the conductive yarn. This yarn is utilized for warp and weft identically for manufacturing woven fabric to obtain isotropic shielding behavior [38]. Shielding effectiveness of those isotropic fabrics and laminated composites have been measured for a wide range of frequencies from 30 to 1500 MHz. It is reported that the EMSE of woven fabrics increases with increase of warp and weft density at all frequencies of incident radiation. Also, the EMSE value of woven fabric increases with increase of conductive filler loading in yarn at all frequencies of incident radiation. The shielding effectiveness of a single layer is barely satisfactory for general applications and the multi-layer fabrics provide adequate shielding effectiveness (20–55 dB) when the wave is normally incident and fabric thickness is >1.6 mm [38]. The EMSE increases as fabric thickness increases and the tendency of EMSE keeps the similar shielding effectiveness at various frequencies as shown in Figure 3 [38]. The multi-layer structure of various thickness and SS fiber content are responsible for blocking the EMI at different frequencies [19]. The different compositions of Cu/Zn/Sn-coated polyester nonwoven (CNW) and Ni/Cu-coated PP nonwoven (SNW) show various shielding effectiveness (SE) depending on incident frequency as shown in Figure 4. The thickness of CNW is thicker than that of SNW. The fabric CNW having higher shielding effectiveness can be attributed to different metal composition and thickness [19].
Shielding effectiveness (SE) of laminated SS/PET (40/60) woven fabrics [19]. Electromagnetic interference shielding efficiency of woven fabric at various layers [38]. EMSE: electromagnetic shielding effectiveness. Shielding effectiveness (SE) of various metalized nonwovens [19].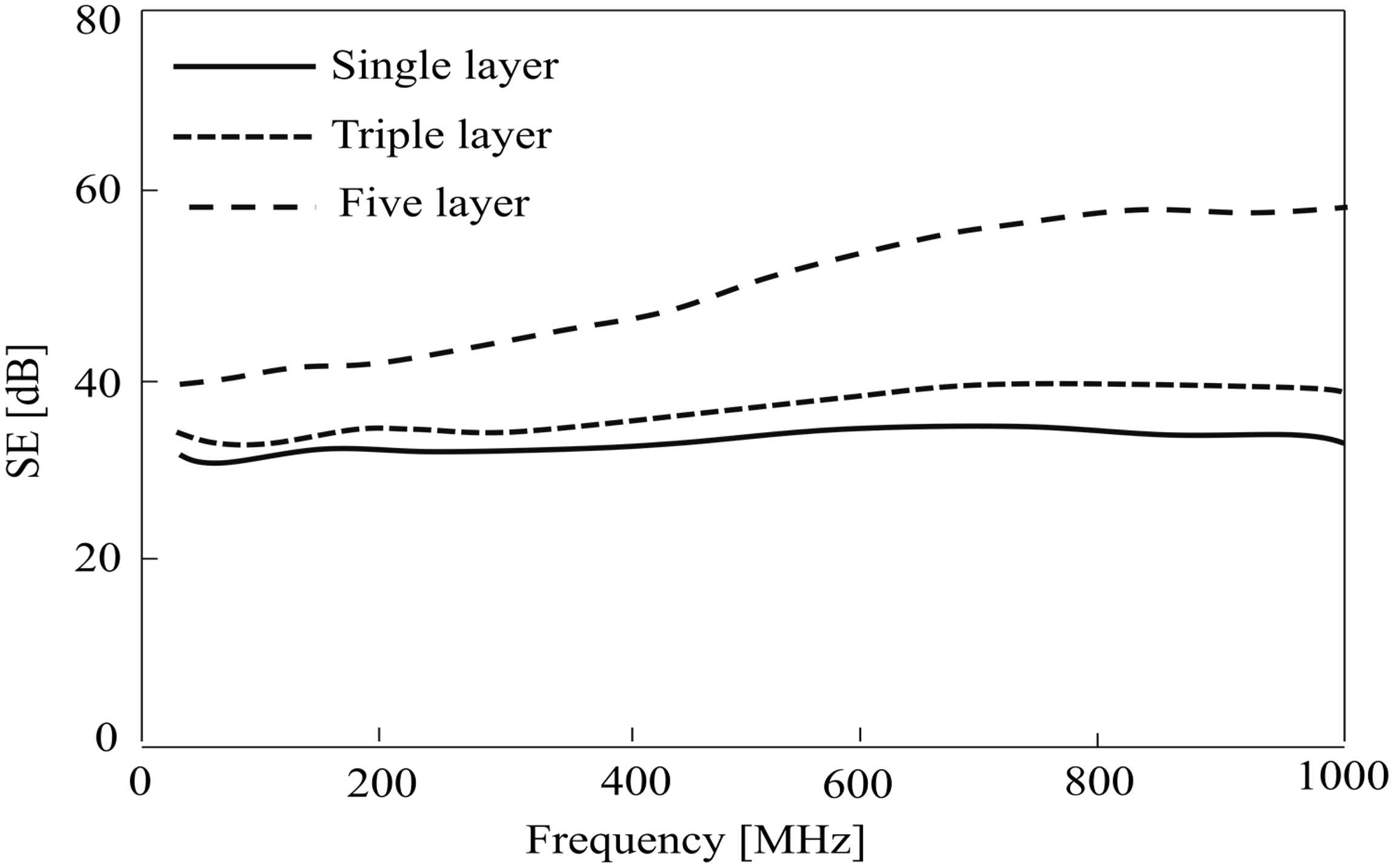


Knitted fabric forms a good conductive network which can better stop the electromagnetic radiation and achieve shielding effectiveness [9]. The literature indicates that knitted fabric reinforced polymer composites are suitable for making complex-shaped components. Moreover, they display good impact energy absorption characteristics. Electro-conductive composites are prepared by using knitted fabric as reinforcing material [39]. PP is chosen as the matrix material and knitted glass fibers as the reinforcement. Copper wires or staple yarns were incorporated as conductive fillers to provide the composite material with the desired EMISE properties. The EMISE of knitted composites is greatly influenced by the amount of copper in the composite material, which can be varied by changing the knit structure, stitch density and linear density of yarns used for knitting and inlaying [39–41].
Coating textiles with conductive polymer
Conducting polymers are a new class of organic materials which have extensive delocalization of π-electrons in a conjugated structure with wide applications in number of technologies such as energy storage, molecular recognition, EMI shielding, opto-electronic devices, corrosion protection, micro-wave absorption, gas separation, sensors and heat generation [15, 35, 42–49]. Conjugated structures are characterized by repeated units in which atomic valence is not satisfied by covalent bonds. The valence electrons of adjacent carbon hetero-atoms (atoms other than carbon and hydrogen) significantly overlap and form double bonds that give rise to π-bonds. The π-electrons are de-localized over large segments of the polymer chain, which are responsible for the electronic properties of the conductive polymers. The essential feature of the conducting polymers is that they provide p-bands of delocalized molecular orbital within which full range of semiconductors and metal behavior can be achieved through the control of the degree of band filling. Chemical structures of some of the commonly known conductive polymers are shown in Figure 5. Among conducting polymers, polyaniline, PPy and polythiophene have received wide spread attention because of their good environmental stability and conductivity [50–53]. The hetero atoms such as –N– or –S– present in the polymer chain play an important role in the unique conduction mechanism. Conducting polymers are difficult to process to form fiber or filament due to the extensive delocalization of π-electrons. So, different conducting composites are produced by blending conducting polymers with conventional insulating polymers/materials so that the conducting composites retain the mechanical properties of the conventional polymers and electrical conductivity of the conducting polymers [54].
Chemical structure of some conductive polymers.
Coating methods of conductive polymers onto textiles
A key requirement of synthesis of conducting polymers is that the conjugated nature of the polymer should be conserved during the synthesis process. Due to strong interaction within polymer chain, they are insoluble and do not melt. Hence, they are difficult to spin to filaments or fibers. Again, lack of thermal stability makes them unsuitable for hot molding process. So, they are very difficult to apply onto textiles. In situ polymerization methods are found as most suitable techniques in this regard. Electro-conductive textiles can be prepared by using in situ chemical, in situ electrochemical, in situ vapor phase polymerization, in situ polymerization in super critical fluid, solution coating processes, etc. [48,49,55–62].
In situ chemical polymerization
The chemical polymerization is very simple. One of the key requirements of this process is that the monomer should be soluble. For chemical polymerization, solutions of monomer and a suitable oxidant (e.g. FeCl3) are mixed together and subjected to constant stirring for prolonged duration. As a result, oxidative polymerization occurs and polymers form in bulk. The coating of different materials with conducting polymers, i.e. polyaniline, PPy, polythiophene and their derivatives, is possible by means of in situ chemical polymerization [24,48,49,58,62–69]. The in situ chemical polymerization can be performed in single- or a double-bath process. For single-bath process, monomer and oxidant solutions are mixed in a single beaker and simultaneously textile substrate is immersed into it as shown in Figure 6. For double-bath process, the textile substrate is treated with monomer solution first and then the monomer-enriched substrate is immersed into oxidant solution or vice versa as shown in Figure 7. As polymerization begins, some polymers deposit on the textile substrate due to adsorption and some present in the solution in bulk. As a result, the color of the substrate as well as solution changes to greenish black which is the color of the polymer [70]. In comparison to other in situ polymerization methods, this method is simplest. The experimental set-up is very simple. It is suitable for laboratory preparation as well as for mass production of conductive textiles. The only requirement is that PPy should have some affinity to the textile substrate used.
In situ chemical polymerization by single-bath process. In situ chemical polymerization by double-bath process.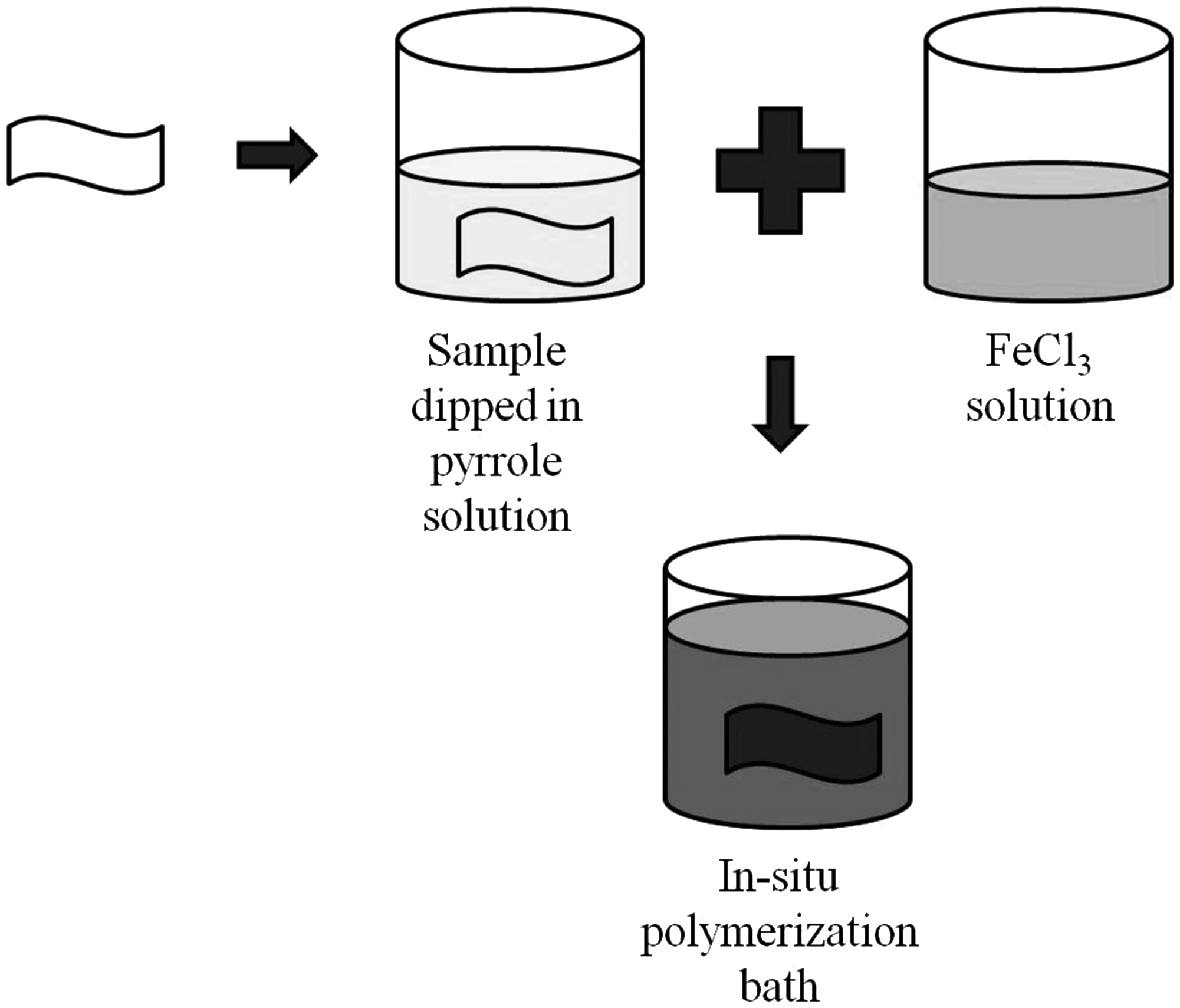

In situ electrochemical polymerization
Electrochemical polymerization is usually performed in a one-compartment cell where two electrodes such as anode and cathode are connected with an external power supply. The cell is provided with monomer solution with suitable electrolytes and dopant as shown in Figure 8. In most of the cases, the electrolytes act as dopants also. Electrochemical oxidation of monomer results in polymer films deposited on anode surfaces. If anode surface is covered with a textile substrate, then polymers will deposit on it [71]. The polymerization rate and yield depends upon the material of electrode, types of solvent, electrolyte, supply voltage, temperature, time, etc. [52,71]. Different electrolysis techniques can be used such as potentiostatic (constant potential), galvanostatic (constant current) and potentiodynamic (potential scanning, i.e. cyclic voltammetry) methods. Potentiostatic and galvanostatic methods are particularly suitable for mechanistic investigation nucleation and the macroscopic growth of polymers. Potentiodynamic techniques, such as cyclic voltammetry, correspond to a repetitive triangular potential waveform applied at the surface of the electrode. This method has been mainly used to obtain information about the redox processes involved in the early stages of the polymerization reaction and to examine the electrochemical behavior of the polymeric film after deposition [71]. The main limitation of this process is the size of the sample to be prepared which is the size of the anode.
Schematic diagram of experimental set-up for electrochemical polymerization: (1) polymerization bath, (2) monomer and electrolyte solution, (3) anode, (4) cathode, (5) electrical wire, (6) power supply and (7) textile fabric affixed on anode surface.
In situ vapor phase polymerization
Chemical vapor phase deposition of conducting polymers is a suitable process for producing electro-conductive textile in two steps as follows. Impregnation of textile is done in an aqueous solution of oxidant and dopant and followed by drying. After that the fabric is exposed to monomer vapor for in situ polymerization. A schematic diagram of in situ vapor polymerization technique is shown in Figure 9. Vapor phase prepared fabrics show a high uniform polymer coating on the fiber surface. As a result, the variability in surface resistivity minimized and fastness to light and washing improved. But controlling of the add-on% is difficult and equipment set-up is complicated [72–77].
Experimental set-up of in situ vapor phase polymerization.
Solution coating process
Conducting polymers are insoluble in most of the solvent available. But by grafting, some functional group in the polymer chain can be made soluble in some selected solvent. Synthesis of soluble PPy can be possible in the form of alkyl PPys. Solubility increases with the increase of length of the alkyl chain attached to the pyrrole ring without significantly affecting the conductivity. These soluble alkyl pyrroles can be pre-packaged in aerosol cans or as commercially available paints and applied directly to any surface in any desired pattern for intelligent textile applications [78]. The main advantage of soluble conducting polymers is that they can be directly applied to any substrate, which avoids exposure of the surfaces to damaging oxidizing agents and fulfill the requirements for controlled laboratory conditions [55].
In situ polymerization in super critical fluid
As discussed above, textile materials can successfully be covered by conducting polymers. The washing of conductive polymer-coated textiles is still problematic because after washing polymers lose conductivity. Improved washing conductivity has been achieved using in situ polymerization of polyaniline and PPy in supercritical CO2 onto polyester and polyamide fibers [79,80]. The main advantage of using this technology is that the environment of super critical fluid during polymerization helps the substrate polymer to expand so that the pyrrole monomer can be able to penetrate into it. As a result, a much durable electro-conductive textile can be prepared. But, this process is costly and set-up is complicated.
Conductive polymer-coated textiles
Due to the metallic conductivity and many other unusual properties, conducting polymers have become the focus of attention in material science. Coating of conducting polymers on insulating fabrics makes them suitable for their possible application as a novel material for EMI shielding [47,61,81,82]. Various textile fibers, such as cotton, wool, viscose, cupro and lyocell, are made electro-conductive by means of in situ polymerization [55,72–76,83–89]. These fibers showed better performance than metal fiber and powder-filled composites which are susceptible to galvanic corrosion or loss of conductivity due to friction [35]. The chemical polymerization of pyrrole in the presence of textile fabric results in the formation of electro-conductive fabrics which are reported suitable for EMI shielding [81,82,90]. The conducting polymers reflect and absorb radiation and generally have larger absorption levels than reflection [35]. It is reported that the desired value of electrical resistivity of an electromagnetic shield is less than 100 Ω/cm2 [15,91,92]. The specific resistance of wool yarn (40 tex, 400 twist per meter) is achieved 1.69 Ωg/cm2 and that of for cotton yarn (40 tex, 500 twist per meter) is achieved 1.53 Ωg/cm2 by using continuous vapor phase polymerization of pyrrole with a FeCl3 solution concentration of 80 g/l [73]. A number of polymers containing the “ether link,” such as polyethers, polylactones and polyetherlactones, form coordination complexes with FeCl3. These complexes are placed in the vapor of pyrrole to initiate polymerization. PPy composites obtained in this way have superior conductivity of 2–3 × 10−3 S cm−1 [93]. Also, polyaniline-coated polyester fabric of resistivity 5 kΩ/ϒ has been found suitable for EMI shielding [33]. In another study, PPy is coated over MnZn ferrite (MZF), nickel coated over PPy and PPy coated over Ni-MZF magnetic core particles, and these materials are found suitable for EMI shielding [47]. These textile materials can be used as EMI-shielded cloth for concerned persons, RADAR barrier shield for military purposes and other devices [83].
Conductive polymer-coated woven fabrics for electromagnetic shielding
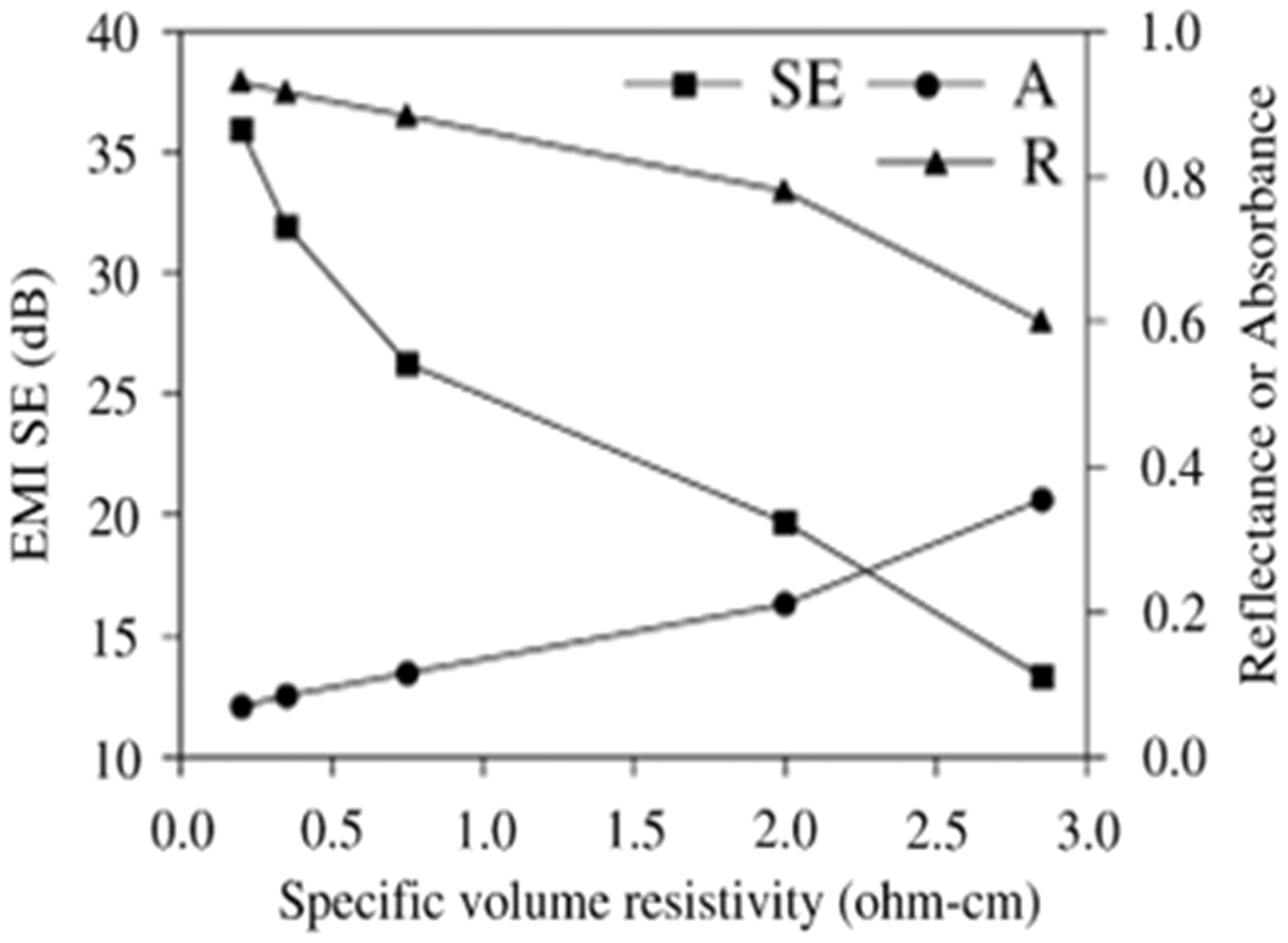
The polymerization of aniline and pyrrole on insulating fabrics like polyester cloth, glass fabrics and high silica cloth is carried out by in situ chemical polymerization. In the radio frequency range from 100 to 1000 MHz, conducting polyaniline-coated polyester fabrics show a shielding effectiveness in the range of 30–40 dB as shown in Figure 10 [15]. The reflectance studies of conducting polyaniline-coated fabric show that 98% of the energy is absorbed in the UV–Vis–NIR range and 2% is reflected back. In PPy-coated fabric, 96% of energy is absorbed and 4% is reflected back, whereas in polythiophene-coated fabric, 82% of the energy is absorbed and 18% is reflected back [15]. An effective shield should absorb all the energy and nothing reflects back. The shielding effectiveness of PPy-coated nylon/lycra fabric prepared with an anthraquinone-2-sulfonic acid dopant is reported as 89.9% at 18 GHz [94]. PPy is polymerized chemically and electrochemically in sequence on a polyester woven fabric, for preparation of a composite with very low electrical resistivity of 0.2 Ωcm. EMISE measured with this composite is about 36 dB over a wide frequency range up to 1.5 GHz as shown in Figure 11. It is found that the composite shielded EMI by absorption as well as reflection and that EMI shielding through reflection increases with the electrical conductivity [74] and 7% of the power of the incident electromagnetic wave is absorbed by the composite [75]. Conducting PPy films with high conductivity and good adhesion are successfully polymerized on the surface of insulating epoxy resin substrates using chemical polymerization. Those films show EMISE in the useful range of about 30 dB over a wide frequency range from 30 to 1500 MHz as shown in Figure 12 [95]. PPy-coated polyester and silica fabric show EMISE of 21.48 and 35.51 dB, respectively, at a frequency of 101 GHz [96]. The conductive fabric produced from PPy coating on glass fiber fabrics having resistivity range below 500 Ω/ϒ showed 98.67% to 99.23% signal loss in the frequency range of 800–2400 MHz [83]. Those samples with low resistivity or high conductivity show high shielding effectiveness compared to those samples with high resistivity as shown in Figure 13. These fabrics are proposed for applications as shield for household appliances, FM/AM radio broadcast sets, wireless phones, cellular phones, computers, buildings, secret rooms and various electronic gadgets that operate up to 2.4 GHz frequency [83].
Shielding effectiveness of polyaniline-coated fabrics in the frequency range 100–1000 MHz [15]. EMISE absorbances (A) and reflectances (R) of PET fabric/PPy composites with various specific volume resistivities [97]. EMISE: electromagnetic interference shielding efficiency. Shielding effectiveness of PPy film [95]. EMSE of PPy-coated glass fiber fabric as a function of electrical resistivity [83]. EMSE: electromagnetic shielding effectiveness.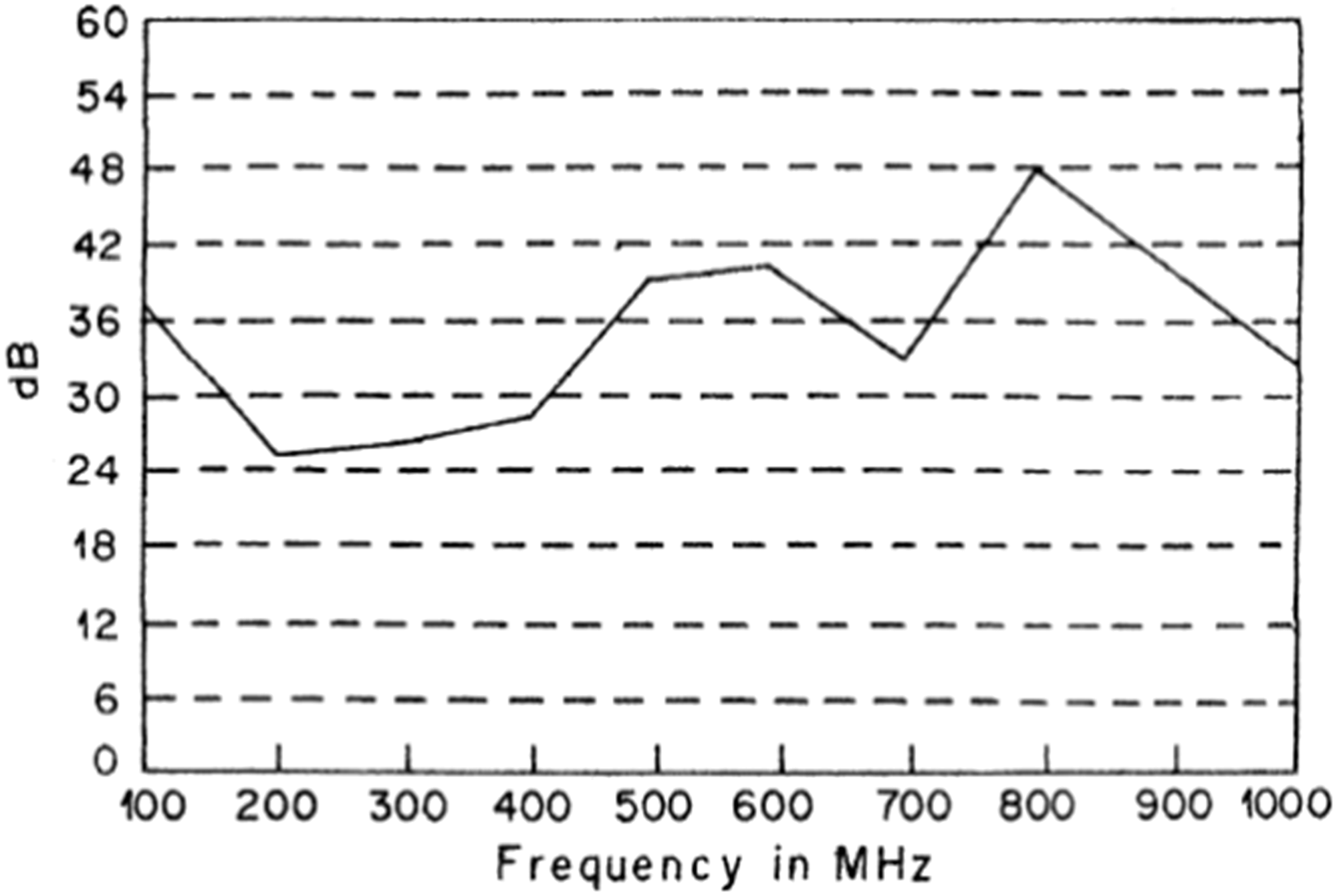


In another study, silver (Ag) is thermally vacuum-evaporated on the surface of PPy–polyester complexes. The EMISE of fabric complexes increases as the area of Ag evaporation layer increases. When the Ag is partially (approximately 37%) evaporated on one side of fabric complexes, the EMISE is 29 dB at 0.5 GHz, and while Ag is evaporated on total area of the sample the EMISE obtained is 33 dB at the same frequency range. The EMISE and the absorbance/reflectance of the shielding material can be controlled by the area and the array of the high conducting Ag layer and low conducting PPy layer as shown in Figure 14 [98]. PPy/Al2O3 textile nanocomposite is found suitable for effective EMI shield in the range of 8–12 GHz frequency and could be able to absorb more than 53% of microwave radiation [60].
EMISE of fabric complex with various Ag evaporation layer [98]. EMISE: electromagnetic interference shielding efficiency; PPy: polypyrrole.
Conductive polymer-coated nonwoven fabrics for electromagnetic shielding
Shielding effectiveness of various conductive textiles.

Note: PPy: polypyrrole.
Effects of different process parameters on EMI shielding
EMSE values of the PPy-coated cotton fabrics at different FeCl3 concentrations can be seen in Figure 15. For each concentration, the highest value was at a frequency of 200 MHz. A remarkable decline in shielding values can be seen in the frequency range of 200–800 MHz. The shielding behavior of the samples shows almost a linear shielding effect between 800 and 1800 MHz. Afterwards, a slight increase from 1800 to 2500 MHz and then a slight decrease to 300 MHz were observed [103].
EMSE of PPy-coated fabrics with various FeCl3 concentrations [103]. EMSE: electromagnetic shielding effectiveness.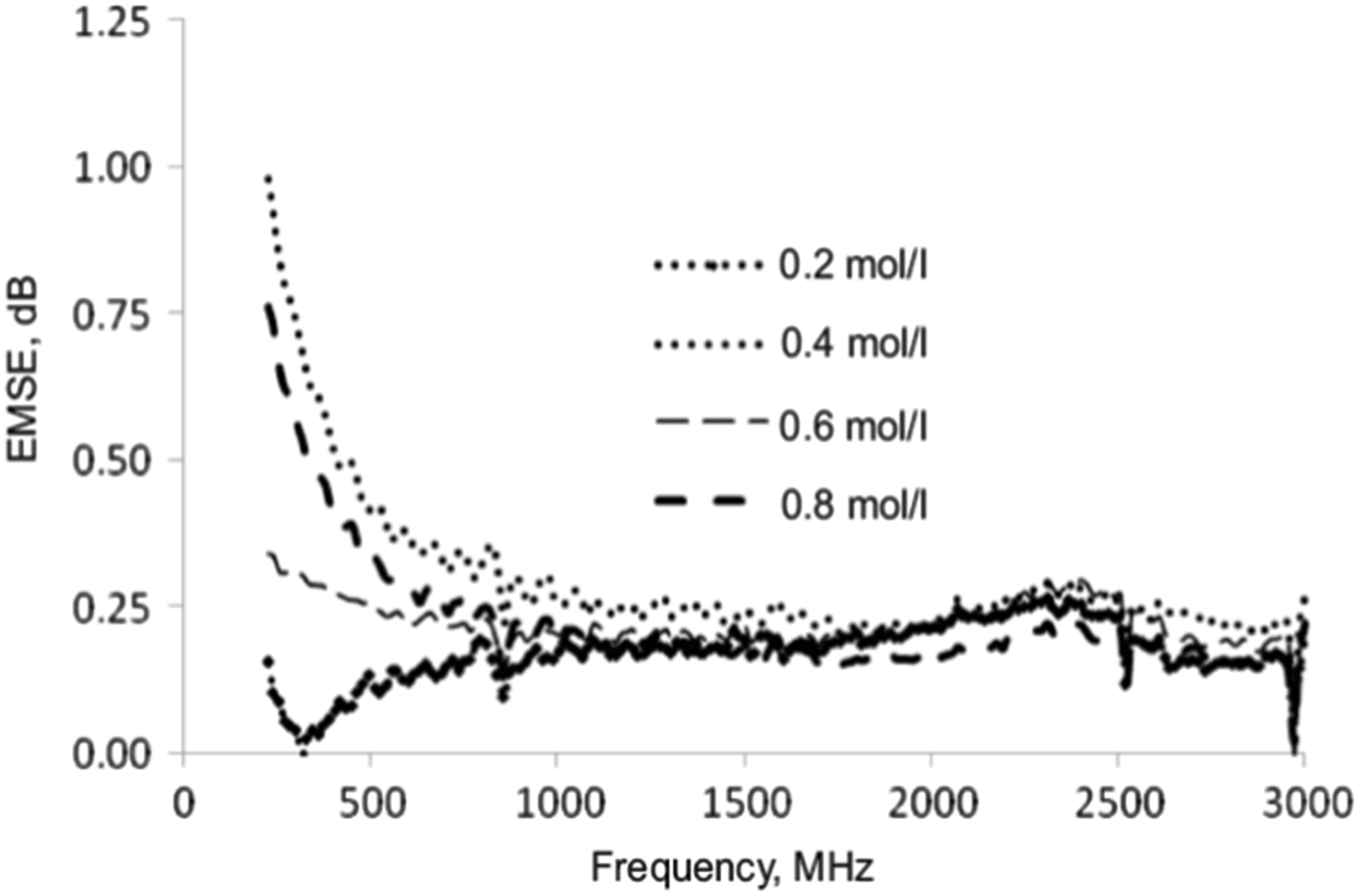
The effects of concentration of p-toluene sulphonic acid (PTSA) as dopant and polymerization time on EMSE of conducting polymer-coated textiles in the frequency range 1–18 GHz are investigated and result the is shown in Figure 16. The levels of absorption are higher than the levels of reflection in all the tested samples and absorption is relatively even throughout the frequency range for different concentrations of dopants and polymerization times. Reflection increases with increase in dopant concentration and polymerization time due to the increase in conductivity. The reflection values are more dispersive than the absorption and at the long polymerization times, the reflection increases throughout the frequency range [67]. It is reported that the shielding effectiveness of the PPy samples prepared in the presence of surfactant increases with the increment of that surfactant concentration [106].
Reflection (bottom three) and absorption (top three) percentages for three selected samples. PTSA concentrations and polymerization times are indicated [67].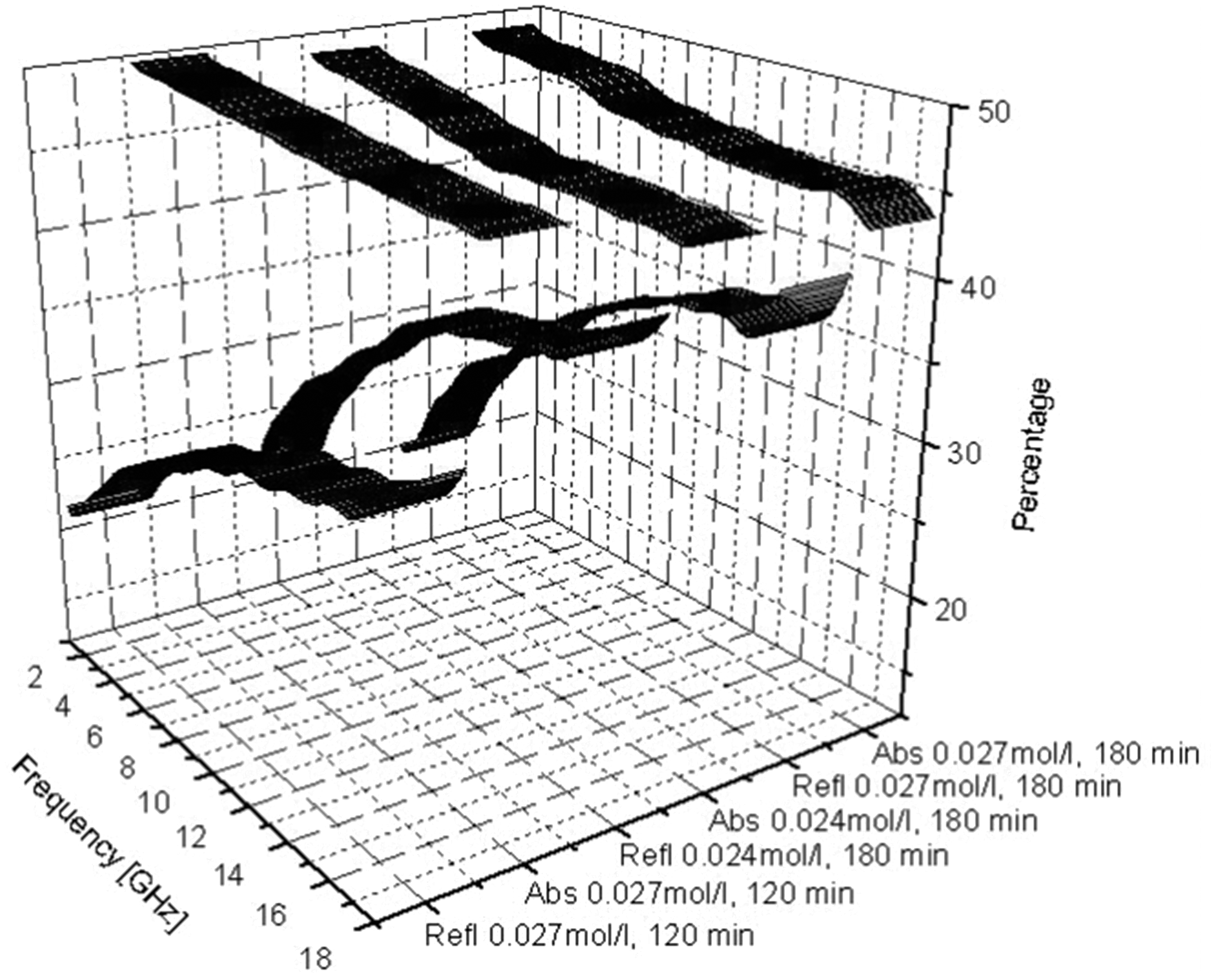
Chitosan when used as an additive can improve EMISE, electrical conductivity and thermal diffusivity of the PPy film [43]. The optimum concentration of Chitosan for the best conductivity is 0.7% (w/v) but after that, the conductivity decreased with further increase in Chitosan concentration. This is due to the fact that in the concentrations more than 0.7% (w/v) of Chitosan is highly viscose solution and for that the rate of polymerization was very slow and the amount of deposited polymer was much lower which led to low conductivity.
The shielding effectiveness (SE), absorbance (Ab) and reflectance (Re) of PPy–Chitosan composite films with various electrical conductivity and different concentration of Chitosan [43].
Conclusions
The electronic devices, such as cell phones and computer equipments emit electromagnetic radiation. Exposure to long-term or acute electromagnetic radiation can have harmful effects on human tissue, brain and can interfere with certain bio-electronic devices, such as pacemakers. Even electronic instruments, microprocessors and devices are also affected by electromagnetic rays and produce erroneous results. In this regard, a variety of electromagnetic shielding composite fabrics are developed to get protection from electromagnetic rays. Various approaches have been adopted by researchers to prepare electro-conductive textile materials for effective electromagnetic shield. Conductive particles such as copper, silver or even carbon are applied onto textile material as surface coating by suitable techniques to prepare electro-conductive textiles for electromagnetic shielding. The shielding efficiency of these metalized textile fabrics mainly works in principle of energy reflection. In many cases, such reflection phenomenon produces interference which is not desirable and the performance of the shielding material is unsatisfactory. Hence, there have been searches for materials with greater capability of absorbing electromagnetic radiation. In this regard, metal fibers are incorporated in woven, knitted and nonwoven structures in various forms and thickness and tested for EMISE in the view of absorption attenuation. Later, conductive polymers such as polyacetylene, PPy and polyaniline are applied to textile materials and hence prepared textile composites are proven to give satisfactory results. Conductive polymer-coated electro-conductive textiles can be prepared by various techniques such as in situ chemical, in situ electrochemical, in situ vapor phase polymerization, solution coating and in situ polymerization in super critical fluid. It is found that these conductive polymer-coated composites work on the principle of absorption of electromagnetic rays rather than reflection. EMISE of these conductive polymer-coated composite textiles depends upon the concentrations of FeCl3, dopant, surfactant, etc. during processing. Various researchers reported that these textiles are suitable as effective EMI shield for various range of frequencies.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.



