Abstract
Smart textiles are fabrics able to sense external conditions or stimuli, to respond and adapt behaviour to them in an intelligent way and present a challenge in several fields today such as health, sport, automotive and aerospace. Electrically conductive textiles include conductive fibres, yarns, fabrics, and final products made from them. Often they are prerequisite to functioning smart textiles, and their quality determines durability, launderability, reusability and fibrous performances of smart textiles. Important part in smart textiles development has conductive polymers which are defined as organic polymers able to conduct electricity. They combine some of the mechanical features of plastics with the electrical properties typical for metals. The most attractive in a group of these polymers are polyaniline (PANI), polypyrrole (PPy) and poly(3,4-ethylenedioxythiophene) (PEDOT) as one of the polythiophene (PTh) derivatives. Commercially available smart textile products where conductive polymers have crucial role for their development are medical textiles, protective clothing, touch screen displays, flexible fabric keyboards, and sensors for various areas. This paper is focused on conductive polymers description, mechanism of their conductivity, and various approaches to produce electrically conductive textiles for smart textiles needs. Commercial products of conductive polymers-based smart textiles are presented as well as the objective of a number of lab-scale items.
Introduction
Smart textiles are fabrics able to sense external conditions or stimuli, to respond and adapt behaviour to them in an intelligent way. The stimuli can be thermal, mechanical, chemical, electrical, magnetic, optical, etc. [1–9] There is no agreement about the definition of “smart”. Among other better known terms for the same purposes are connected, intelligent, interactive, responsive and adaptive [10]. Smart textiles are fabrics that interact with the surrounding environment or the user [11]. They can provide information needed in the current situations, help to master everyday life more efficiently [12,13] and they present a challenge in several fields of application such as health, sport, automotive and aerospace. Some of their first usage was in military and medical applications [14]. Advanced functionalized materials, such as breathing, fire-resistant or ultra-strong fabrics are not considered as smart, no matter how high-technological they might be [15]. Smart textiles have benefits such as non-invasive and continuous monitoring [16–20].
According to the applications developed during the last decades, smart textiles can be classified as: I. Passive smart textiles – the first generation of smart textiles integrates sensors which can sense (detect) environmental changes or stimuli (optical fibres, conductive materials, thermocouple, etc.), II. Active smart textiles – the second generation of smart textiles which can sense and react to stimuli from the environment or the user. The textiles consist of sensors and actuators that provide the ability to sense and actuate or move a part of their environment (chromatic materials, shape memory materials, phase change materials, hydrogels and membranes), III. Very smart textiles – the third generation of smart textiles which can sense, react and adopt to external conditions or stimuli (space suits, thermo regulating clothing, health monitoring apparel) [3–5,7,11,17,21–23].
“Smart” clothing (equipped with functionalized materials) provides a set of data that users must interpret themselves. Smart clothing integrates sensors into the garment and the sensors can track a user’s steps (e.g. calories burned, sleep rhythm and stress levels). Overall, smart clothing is powerless to automatically correct issues for the end user.
“Intelligent” clothing are garments that can interpret data automatically and adjust themselves to accommodate the wearers’ specific needs [24]. They are equipped with sensors, actuators that are flexible or textile based and with the decision unit able to make decisions regarding the sensors outputs and to control actuators.
The term “electrically conductive textiles” is used for a broad range of textile fibre-based products with widely differing specific electrical conductivity [20]. Electrically conductive textiles include conductive fibres, yarns, fabrics, and final products made from them [25]. Often they are prerequisite to functioning smart textiles. Their quality determines durability, launderability, reusability and fibrous performances of smart textiles [13].
Important part in smart textiles development has conductive polymers [26] which are defined as organic polymers able to conduct electricity, exhibiting a conductive or a semiconductive behaviour [27]. Today there are over 25 known conductive polymers [28,29]. They constitute an interesting class of materials which combine some of the mechanical features of plastics with the electrical properties typical for metals [30]. These polymers have become a popular choice of conductive materials because they are lightweight and cost-effective, show relatively high adjustable electrical conductivity (thanks to doping process), flexibility, biocompatibility, can be tailored to have a sensing and actuating function and are easy to prepare. The greatest advantage of conductive polymers is their processability, mainly by dispersion [13,28–34]. They can be applied in energy storage and energy conversion devices, fuel cells, supercapacitors, as adsorbents, conducting inks, heterogeneous catalysts, in metallic corrosion protection, antistatic packaging, electrostatic discharge (ESD) control, electromagnetic interference (EMI) shielding applications, smart membrane technology, etc. [34,35] Conductive polymers are suitable for fabrication of light smart materials without incorporation of any metals [36].
Commercially available smart textile products where conductive polymers have crucial role for their development are medical textiles, protective clothing, touch screen displays, flexible fabric keyboards, and sensors for various areas of application.
The future prospects for conductive polymers applications in textile area include growing usage in health, sport and fitness (health monitoring, clinical applications, sportswear), security (uniforms for firefighters), fashion (functional clothing), and non-clothing applications (automotive, home textile).
This article has three main parts. The first one is focused on conductive polymers description and mechanism of conductivity. Part two describes various approaches to produce electrically conductive textiles for smart textiles needs. The third one is focused mostly on commercial products of conductive polymers-based smart textiles as the objective of a number of lab-scale items.
Conductive polymers and mechanism of conductivity
Conductivity borderlines between electrical insulators, semiconductors and conductive materials are fluent and not precisely defined. Conductivity of conductive polymers is in the range of semiconductors’ conductivity [37]. The reason for the strong use of these polymers is their great potential for new applications. Conductivity of polymers compared to those of other materials are presented in Figure 1 [38,39]. Conductive polymers merge the positive properties of metals and conventional polymers. They conduct electrical charges and show great electrical properties [29].
Conductivity range of polymers compared to other materials conductivity [39].
There are two subgroups of conductive polymers [6]:
– intrinsically conductive polymers, – extrinsically conductive polymers.
Intrinsically (or inherently) conductive polymers (ICPs), also known as conjugated polymers and synthetic metals, exhibit interesting electrical and optical properties previously found only in inorganic systems [40]. Different types of ICPs can be prepared with a broad range of conductivities from 10−10 to 10+5 Scm−1 [31–35,41,42]. The most attractive in a group of these polymers are polyaniline (PANI), polypyrrole (PPy) and poly(3,4-ethylenedioxythiophene) (PEDOT) as one of the polythiophene (PTh) derivatives. They show high electrical conductivity and environmental stability, they are synthesized easily, but have poor mechanical properties [6,43–47].
Extrinsically conductive polymers (ECPs), or conductive polymer composites (CPCs) are obtained by blending (melt mixing) – an insulating polymer matrix, thermoplastic or thermosetting plastic, with conductive fillers. Three most important conductive fillers are carbon (carbon black (CB) and carbon nanotubes (CNTs)), metal powders and their compounds (indium tin oxide (ITO) and aluminum zinc oxide (AZO)), and ICPs (PPy, PANI). ECPs have special properties such as good electrical and thermal conductivity, corrosion resistance and good mechanical properties. They are used as conductive and semi-conductive polymer fibres, corrosion resistant coatings, ESD materials, electronics, solar collectors. Their conductivity values are much lower than the conductivity of ICPs, mostly in the range between 10−5 and 103 Scm−1 depending on the applications [6,41,43,48–52].
There are two conditions for polymers to become conductive. The first condition is that conductive polymers consist of alternating single and double bonds, called conjugated double bonds. Such bonds contain a localised “sigma” (σ) bond which form strong chemical bonds. In addition, each double bond also contains a less strongly localised “pi” (π) bond which is weaker (Figure 2).
Simplified schematic of a conjugated backbone: a chain containing alternating single and double bonds [29].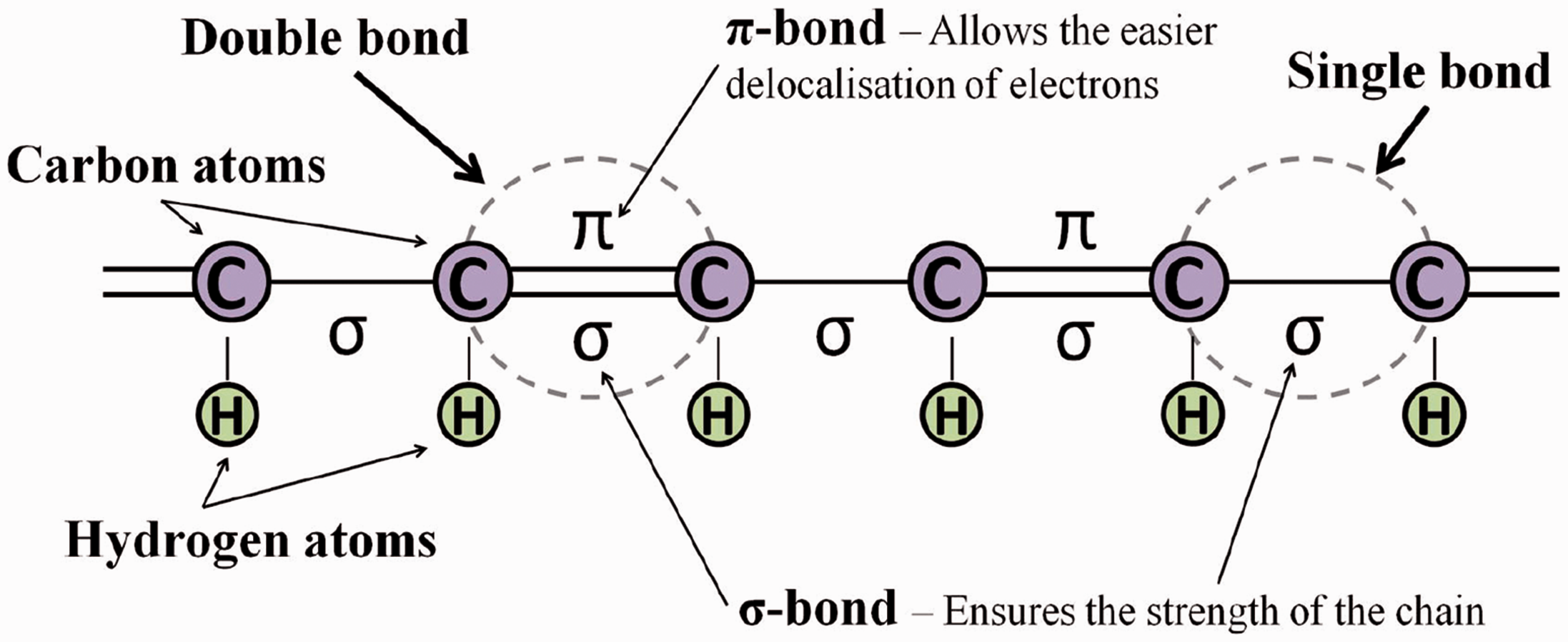
However, bond conjugation is not enough to make the polymer material conductive. Hence, the second condition is that polymer structure has to be disturbed – by removing electrons from (oxidation), or inserting them into it (reduction). The processes are termed as p-doping and n-doping. They can affect its surface and bulk structural properties (colour, porosity, volume) [28,29,53,54].
Properties changes of conductive polymers in response to redox transition.

Factors that affect to polymer conductivity are density of charge carriers, their mobility and direction, presence of doping materials and temperature [48,54–58].
Conductive polymers are in appropriate oxidized or reduced state conductors due to their unique, stretched, π-conjugation. Overlapped π-orbitals form the valence band, and π*-orbitals form a conductive band. Due to chemical or electrochemical oxidation of conductive polymers, the electrons are removed from the valence band, which leads to the presence of charge on the conductive polymer which is delocalized over several monomer units in the polymer and cause relaxation of the polymer geometry in the most stable form. Dopants can be incorporated in the polymer during synthesis or can be retrofitted. They can be anions or cations, e.g. ClO4−, Na+ or larger polymer particles such as polyelectrolytes, poly(styrene sulfonic acid) (PSS) and poly(vinyl sulfonic acid) (PVS). The degree of doping represents the ratio of counterion and monomers in polymer [59–61].
In 1977, it was discovered that the conductivity of polyacetylene can be increased by doping with iodine. The 2000 Nobel Prize in chemistry was awarded to Heeger, MacDiarmid and Shirakawa for the discovery of conductive polymers [55].
Certain excitations occur in polyenes, which are related to solitary waves and to solitons [44,54–58]. It is interesting to compare three simple carbon compounds, diamond, graphite and polyacetylene. They may be regarded, as three-, two- and one-dimensional forms of carbon materials (Figure 3).
Three-, two- and one-dimensional carbon materials: (a) diamond, (b) graphite, (c) polyacetylene chain [38].
Diamond and graphite are modifications of pure carbon, while in polyacetylene, one hydrogen atom is bound to each carbon atom. Diamond contains only σ bonds and it is an insulator. High symmetry gives isotropic properties. Graphite and polyacetylene have mobile π electrons and for this reason they are highly anisotropic conductors [38].
Polyacetylene as the simplest possible conjugated polymer obtained by polymerisation of acetylene illustrates principles of conduction mechanisms in polymers (Figure 4) [57]. As a necessary consequence of the asymmetry of the polyacetylene ground sate, two equivalent polyene chains, L and R, are interconverted through the intervention of a soliton. The soliton as a mobile charge or neutral defect or a “kink” in the polyacetylene chain propagates down the chain and reduces the barrier for interconversion. The charge carrier in n-doped polyacetylene is a resonance-stabilised polyenyl anion of approximately 29–31 CH units in length, which the highest amplitude is at the centre of the defect. Soliton moving from one end of the sample to another is explained by bipolaron hopping mechanism [57].
Soliton intervention through the polyacetylene chain [57].
Conductivity and other properties of common conductive polymers.

Conductivity in PPy is mostly due to p-type conduction, the inter-chain hopping of electrons and the motion of anions or cations within the material. PPy can possess a conductivity of up to 7.5 × 103 Scm−1. The primary factor that limits the conductivity of PPy is the “disorder” in the PPy backbone. More of these defects can form as a result of redox switching or exposure to oxygen or water, resulting in the slow deterioration of conductivity [29]. PPy is one of the most important conductive polymers that is being applied in smart textiles due to its high conductivity, good environmental stability, good adhesion and non-toxicity. This polymer has been studied in microelectronics applications, microsurgical tools, biosensors, neural tissue engineering, etc. [29,62]
PANI has attracted much interest worldwide because of its thermal and chemical stability [23,52,63–67]. Its production cost is low and it can be easily doped with inorganic and organic acids to prepare the conductive form [52]. Thermal stability of PANI is superior to other ICPs. Emeraldine base PANI has low conductivity in the range of 10−10 Scm−1, while its salt created by modifying the base’s oxidative state is conductive with 30 Scm−1. In its base state the polymer chains are coiled, while in its salt form, the additional positive charges in the polymer repel each other extending the chains. In extended coil, form electrons are easier to delocalize, thus resulting in an increased conductivity [29]. PANI has many sensing applications such as in ammonia sensors, EMI shielding, and precious metals recovery [62]. PANI is investigated also for neural probes, controlled drug delivery and tissue engineering [29].
The solubility problem of PEDOT was circumvented with PSS, a water-dispersible polyelectrolyte [68]. PSS is used as a charge balancing counterion during oxidative polymerization of the 3,4-ethylenedioxythiophene (EDOT) monomer; this yielded polymer complex poly(3,4-ethylenedioxythiophene)-poly(styrenesulfonate) (PEDOT:PSS). The polyion complex can be easily dispersed in water as colloidal gel particles with diameters of several tens of nanometers and it has high stability [67–70]. The properties of this complex can be adjusted with the addition of organic co-solvents, surfactants, wetting agents, to the aqueous coating mixture to improve the coatability of the polymer on hydrophobic substrates and mechanical properties [68]. Secondary dopants are used in small amounts (glycerol, sorbitol, dimethyl sulfoxide (DMSO), N-methylpyrrolidone (NMP), ethylene glycol (EG)), etc.) [68–72]. The PEDOT:PSS polymer complex is used as electrode material in organic thin film transistors or as a hole transport layer in organic light emitting diodes. This complex can coat hard surfaces of microelectronics as well as fibres and fabrics and other stretchable substrates [62].
In general, there are several ways for polymers modification, via blending, grafting, and curing (Figure 5). Blending is the physical mixture of two (or more) polymers to obtain the requisite properties. Grafting is a method wherein monomers are covalently bonded onto the polymer chain, whereas in curing, the polymerization of an oligomer mixture forms a coating which adheres to the substrate by physical forces.
Schematic representation of the methods of polymer modification [73].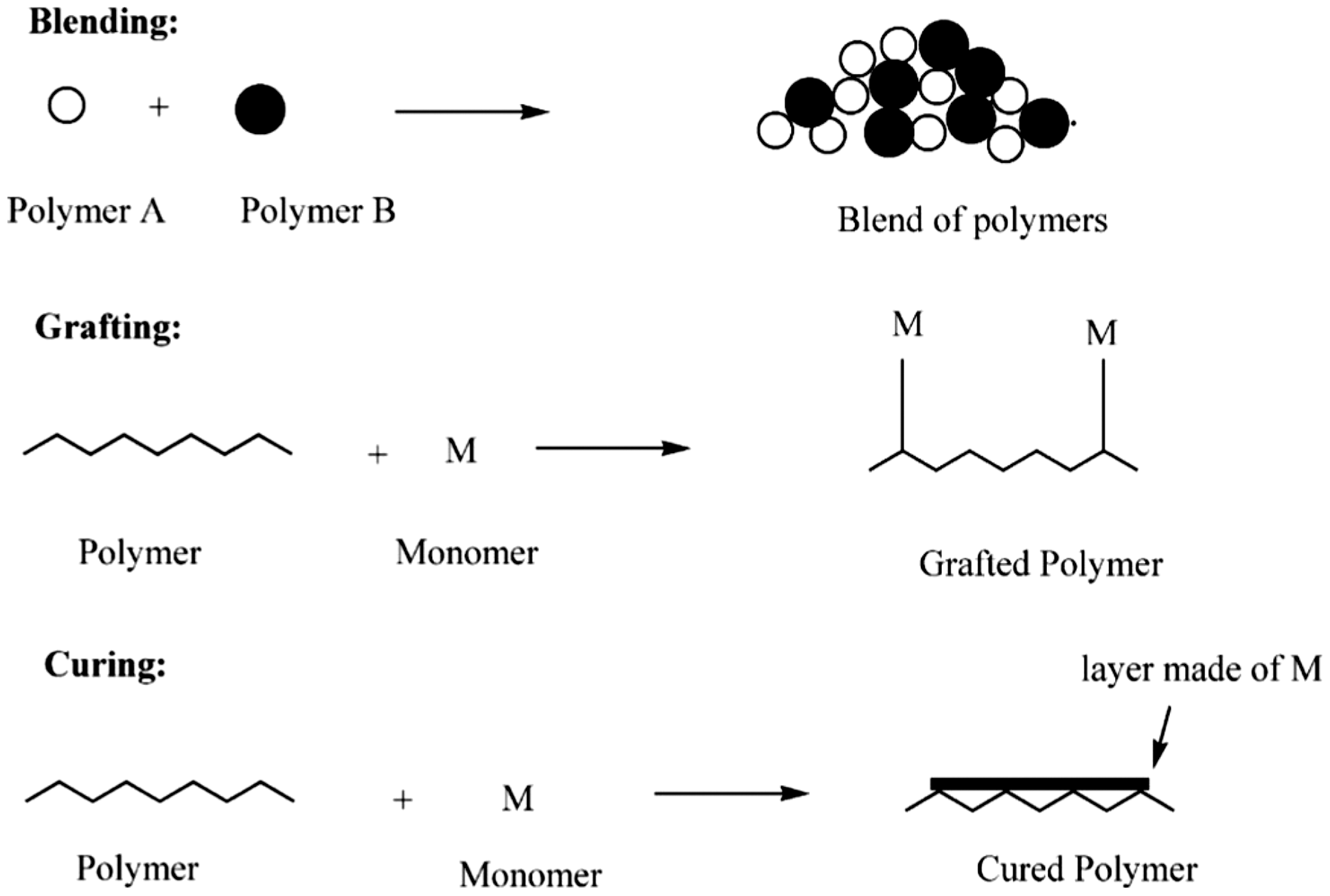
Considerable work has been done on techniques of graft co-polymerization of different monomers on polymeric backbones. These techniques include chemical, radiation, photochemical, plasma-induced techniques and enzymatic grafting.
In order to obtain a material suitable for applications in various technologies, the conductive polymers have to overcome certain limitations (poor mechanical properties and problems in processing, instability under the relevant ambient conditions). Grafting improves the processability of conductive polymers and provides possibility to obtain the particular use properties required for a target application.
Self-doped conductive polymers prepared by grafting techniques afford a water-soluble PPy. The grafting of pyrrole (Py) onto p-amino diphenylamine moieties of water-soluble 2-acrylamido-2-methyl-1-propane sulphonic acid-N-(4 aniline phenyl) methacrylate co-polymers produces the desirable property. PANI was also made soluble by polymerizing aniline in an aqueous solution of poly(p-amino styrene) to form a graft co-polymer (Figure 6(a)) that is soluble in some solvents. Grafting also can reduce the rigidity of the polymer chain, as a result, solvation can occur by the solvents, imparting solubility [73].
Reaction scheme for preparation of: (a) polyaniline based graft co-polymer, (b) pyrrole-styrene graft co-polymers [73].
A styrene-based composite has been prepared by co-polymerizing styrene with 4-chloromethyl styrene, using (azoisobutyronitrile) (AIBN) as initiator and refluxing the mixture overnight with added solution of potassium pyrrolate in tetrahydrofuran (THF) medium to graft the Py moiety on the co-polymer matrix. The reaction scheme is shown in Figure 6(b). The conductivity of the composite is ∼10 Scm−1.
The other reason for grafting is to modify the surface of the conductive polymer. Hydrophilicity of the PANI film surface can be enhanced by grafting hydrophilic monomers, e.g. acrylamide, acrylic acid, the Na salt of 4-styrene sulphonic acid. This can be done by graft co-polymerization of the monomers by treating the emeraldine films with Ar plasma or a mixture of ozone and oxygen. Grafted PPy can function as ion-sensor also, e.g. it can sense the presence of ions in the solution [73].
An inherently anisotropic graft copolymer was synthesized by grafting of PANI on to chitosan using persulfate as an initiator in acidic condition (Figure 7). The grafted biomaterial exhibited electrical conductivity with pH switching behaviour like PANI. PANI-grafted biopolymer provides good processability with improved solubility, mechanical strength and controlled electrical properties. Therefore, it can be used for chemical and biosensor applications [74].
Chitosan-graft-polyaniline synthesized through the oxidative-radical graft copolymerization [74].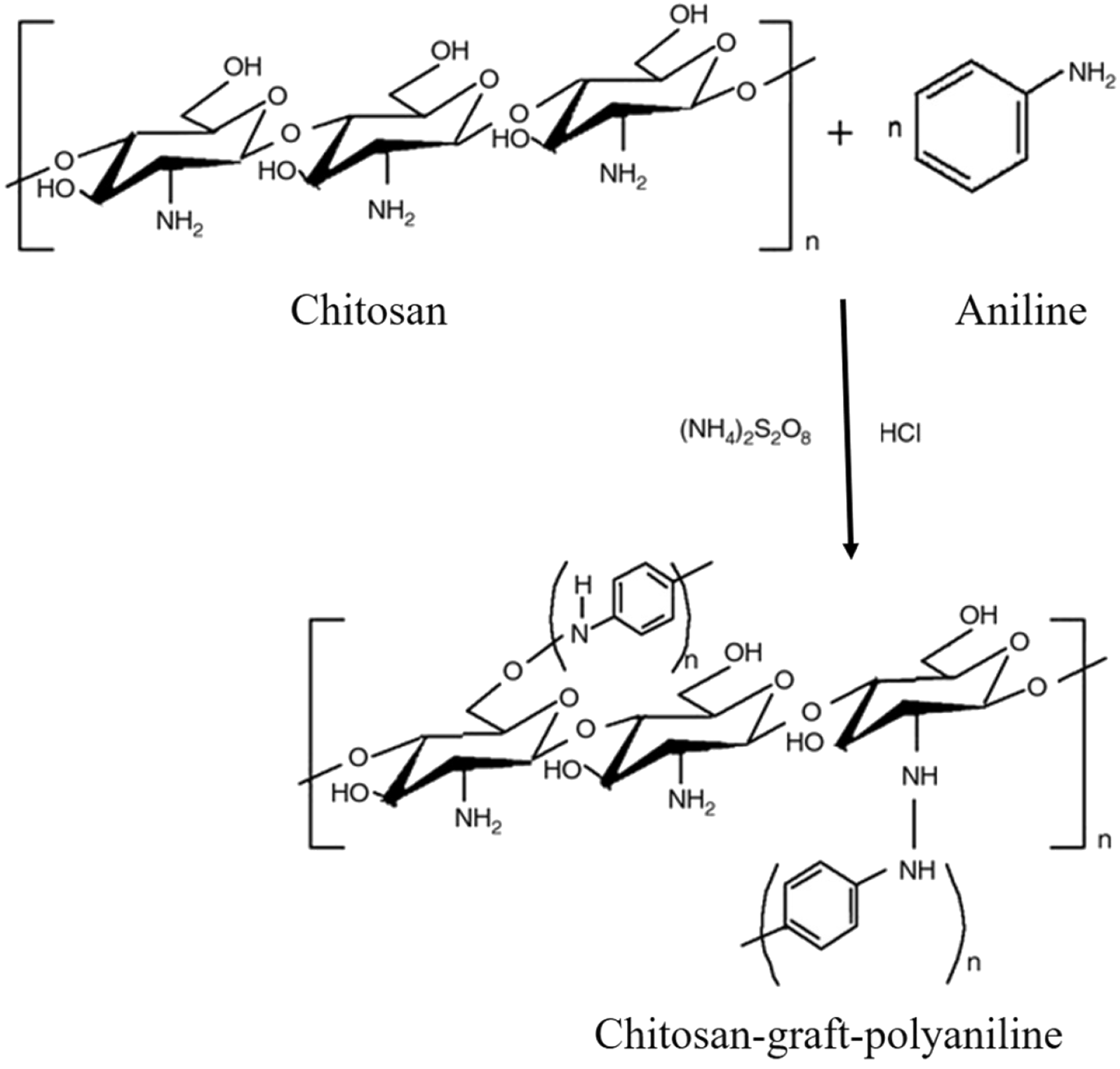
Production of electrically conductive textiles
Electrically conductive textiles have been considered in different applications due to their desirable properties in terms of electrostatic discharge, EMI protection, radio frequency interference protection, thermal expansion matching, etc. [75] Various approaches were developed to produce electrically conductive textiles including conductive fibres/yarns production, conductive yarns insertion during/after fabric manufacturing, coating textile techniques, embroidery techniques, etc. [13,21,34,75–79].
Conductive fibres/yarns production
Conductive fibres/yarns were initially used in technical areas such as medical applications, and electronic manufacturing. They have diverse functions, like antistatic applications, EMI shielding, electronic applications, infrared absorption, etc.
Following the manufacturing processes, the conductive fibres/yarns are divided into:
– intrinsic conductive fibres/yarns (naturally conductive),
Intrinsic conductive fibres/yarns
Intrinsic conductive fibres/yarns are made by materials featuring high electrical conductivity (wholly conductive substrate).
Metallic fibres are produced from electrically conductive materials such as ferrous alloys, nickel, stainless steel, titanium, aluminium, copper through a bundle-drawing or shaving process or shaved off the edge of thin metal sheeting. These fibres are highly conductive, but brittle and heavier than most textile fibres. The Sprint Metal Company (2015) discerns metallic fibres (2–40 µm) and fine metal wires (30 µm–1.4 mm) according to their diameter. Metallic yarns/threads made up of metallic fibres are very thin and may be knitted or woven into a textile or used to form interconnections between components [21,34,76–79].
Carbon fibres/yarns are fibrous carbon materials with carbon content more than 90%. They are transformed from organic matter by 1000–1500℃ heat treatment. These fibres/yarns have attractive properties (strength, electrical conductivity, stability, low density, low-to negative coefficient of thermal expansion, heat resistance). They are used as sorption materials, ESD materials, and reinforcement in composites. Variations in composition and structure, related to the conditions of their production and to impurities present in structure, are the cause of changes in their electrical resistivity, resulting in properties ranging from those of conductors to semiconductors [75].
Fibres/yarns made completely of ICPs are usually produced by melt spinning, wet spinning or electrospinning. The ICPs are mostly non-thermoplastic materials that decompose at a temperature lower than their melting point. As a result, melt spinning is not very good technique for ICPs fibres/yarns spinning. In case of electrospinning, instability and low concentration problems are the main limitations of this process [34,80,81]. Han et al. [82] prepared ICPs fibres by melt spinning low-cost thermoplastic trans-1,4-polyisoprene and doping with iodine, which can be as fine as 0.01 mm, and electrical resistivity can be as low as 10−2 Ω·m (Figure 8(a)). Drawing can improve the orientation of trans-1,4-polyisoprene crystals in the fibres and the conductivity of the fibres. Such fibres could be used in textile and other fields.

Jalili et al. [83] reported a novel one-step fibre wet spinning route for the production of poly(3,4-ethylenedioxythiophene):poly(styrenesulfonate)-polyethylene glycol (PEDOT:PSS-PEG) fibres with enhanced electrical conductivity from 9 to 264 Scm−1 and redox cycling properties with respect to ethylene glycol (EG) post-treated PEDOT:PSS fibres (Figure 8(b)).
Quality of fibres was influenced by the spinning formulation and the choice of coagulation bath. The one-step PEDOT:PSS-PEG fibre approach represented advantage over the post EG-treated PEDOT:PSS fibres. Dependence of molecular ordering, charge delocalization, spinning parameters, and treatment conditions employed was evident to the resultant electrical, electrochemical, and mechanical properties of the fibres [83].
Extrinsic conductive fibres/yarns
Extrinsic conductive fibres/yarns are made by the combination between conductive and non-conductive materials and they show high electrical properties. Special treatment involves the mixing, blending or coating process.
Conductive-filled fibres/yarns are made by adding conductive fillers (metallic powder, metallic nanowires, CB, CNTs, ICPs) into nonconductive polymers (polypropylene, polystyrene or polyethylene). Melt spinning and wet spinning are the common processes to develop this kind of fibres/yarns. The wet spinning process guarantees fibres/yarns with improved electrical and mechanical properties over those produced by the melt spinning [79].
To synthesize CB-polymer fibres/yarns, a certain amount of CB particles, 20–100 mm, is directly added into melts or thermoplastic polymers. Then, the polymer melts can be melt-spun into conductive polymer fibres/yarns. The concentration of CB in the fibre is higher than 10 wt.%, while in the yarns is in the range from 10 to 40 wt.%. The mechanical properties of developed yarns are degraded with increasing CB concentrations, which limits yarn conductivity. The main advantage of CB-filled polymer yarns is low cost, commercial availability of CB and the easy of the synthesis process of the yarns. Due to the black colour of CB, produced yarns made have undesirable appearance for many applications [84].
CNTs feature a unique one-dimensional structure and extraordinary physical properties (high aspect ratio, light weight, good electrical and thermal conductivites). Due to these features, CNTs are considered a better conductive additive than CB for fabrication of fibres/yarns. The mechanical properties of CNTs-filled fibres/yarns are improved compared to the original fibres/yarns. These fibres/yarns can be spun by melt spinning, wet spinning or electrospinning techniques [84].
Soroudi and Skrifvars [85] performed studies on the conductive polyblend filaments prepared by melt spinning process (Figure 9). The ternary blend of PP/PA6/PANI complex showed a matrix/core-shell dispersed phase morphology with widely varying droplet size. The binary PP/PANI complex and the ternary PP/PA6/PANI complex blends showed that their conductivity depends on the fibre draw ratio. The ternary blend fibres exhibited a smoother surface and more even fibres.
Low vacuum SEM images: (a,b) binary blend fibres (PP/PANI complex), (c,d) ternary blend fibres (PP/PA6/PANI complex), prepared at draw ratio of 2 [85].
Conductive-coated fibres/yarns are produced by coating insulating fibres/yarns with conductive ones (metals, CB, CNTs or ICPs). The properties of these fibres/yarns depend on the type of conductive materials and the manufacturing process [79,81,84,86].
Various types of metals, such as silver, copper, aluminum and gold, can be deposited on polymer yarns made of PA6, PP, etc. Diverse coating techniques can be used including polymer-metal lamination, physical vapor deposition, metallic-paint brushing, and electroless plating. Disadvantage of the metal-coated yarns is that the metal layer may be peeled off due to washing or other types of mechanical abrasion [84]. Lee at al. [87] demonstrated the electroless plating of aluminum on cotton threads, and the resistance of the conductive cotton threads was measured to be ∼0.2 Ω/cm.
Coating of CNTs or CB on polymer yarns is usually carried out by a simple dipping-and-drying technique. Nauman et al. [9] demonstrated coating cotton, nylon, and polyethylene yarns with a CB-Evoprene layer (a copolymer of styrene-butadiene-styrene units). The experimental volume resistivity of the yarns or filaments was 0.2–1 kΩ·cm. They also used these conductive yarns to develop a smart fabric that can be used as a piezoresistive strain sensor [84].
Yarns coated with intrinsically conductive polymers.

Conductive yarn insertion into fabric
Conductive yarns are usually integrated in textile structures by weaving, knitting or braiding technology. However, their integration in structures is a complex process [20,86].
E-glass/polypropylene (GF/PP) commingled yarn produced by Textile sensor insertion into fabric: (a) PEDOT:PSS yarn coated by roll to roll coating, (b) 2D weaving fabric with integrated textile sensors [88].
Silver-plated nylon and elastomeric yarns were used for the sensing knitted fabrics manufacturing by flat-bed knitting technology. Developed sensors are suitable for the measurement of human body articulations or physiological signals. Electro-mechanical tests were performed on the specimens. The conductive yarn was positioned only on the technical face of the knitted fabric (Figure 11) [89].
Sensing knitted fabric: (a) technical face with conductive yarns, (b) contact points between conductive loops [89].
The contact pressure and the number of contact areas between the conductive loops demonstrates their high values prior to extension of the knitted sensor but during the force loading stage. Increasing the number of conductive courses creates a greater propensity to buckle and this has a measureable effect on the starting point of the linear working range [89].
Coating textile techniques
Most methods for imparting conductivity of textiles and other flexible substrates rely on a coating of metals, CB, conductive polymers and paints onto their surfaces. Coating textile techniques include electrodeposition (electroplating process), electroless (chemical or autocatalytic) plating, sputtering of thin films, vapor deposition, printing, spraying, knife-over-roll coating. Thick coating can be applied manually or by masking techniques, dip-coating, soft lithography, embossing, or imprint. Other types of coating applied to fabrics are visible light sensitive, pH sensitive, humidity sensitive, electrochromic, etc. The parameters that influence the consistency of coating include the viscosity and uniformity of the coating material, and the tension, flexural rigidity, porosity and coating factor of the substrate. The last factor gives a measure of the fabric openness, air permeability, degree of moisture resistance and adhesion. With a surface pre-treatment, even low-surface energy materials can be made conductive on the surface with a good coating adhesion (polyolefins, fluoropolymers and silicones) [61,90,91].
Metal-coated textile materials are breathable and lightweight compared with metal fibre fabrics. Their electrical conductivity can be tailored by controlling the coating thickness and choosing the right metals. They provide functions such as antistatic properties, shielding against EMI and radiofrequency interference. These fabrics are used for the military, camouflage and antimicrobial applications [91].
Gan et al. [92] coated polyethylene terephthalate (PET) fabrics (51 × 38 count/cm2, 64 g/cm2, taffeta fabric) with CU-Ni-P alloy by using the electroless plating method (Figure 12). Conductive fabrics with high EMI shielding effectiveness could be prepared at an optimum condition. A Cu-Ni-P alloy deposit weight of 40 g/m2 produced a shielding effectiveness of more than 85 dB over the 100 MHz to 20 GHz frequency range.
SEM images of the alloy-coated fabrics obtained at different K4Fe(CN)6 concentration in the bath: (a) 0 ppm; (b) 1 ppm; (c) 2 ppm; (d) 4 ppm [92].
Gültekin and Usta [93] investigated properties of CB coated-fabrics (Figure 13). The dipping-drying process was repeated for several times to increase the CB loading in plain weaved cotton (30 warp/cm, 22 weft/cm). The basis weight of fabric is 113 g/m2. Agglomeration of CB nanoparticles was observed on fabric surface. The electrical resistivity decreased from 1.4 E+09 Ω·cm (neat fabric) to 5.8 E+07 Ω·cm at 5 mg/ml CB concentration. At higher CB loads, the resistivity decreases, suggesting that CB nanoparticles act as electrically conductive bridges.
Optical microscopy images of CB coated cotton fabrics with different amount of CB loading: (a) 0.5 mg/ml, (b) 1 mg/ml, (c) 2 mg/ml, (d) 5 mg/ml [93].
Characteristics of conductive polymer-coated textile materials include deposition of conductive polymers onto various textile forms; uniform and coherent coating, surface resistivity in a range between 10 Ω and a billion Ω, fabric thicknesses range from 0.1 mm to a few mm, and development of lightweight, flexible and durable materials. These fabrics have found use in applications such as antennas, radar-absorbing materials, non-radar-reflective, camouflage netting, etc. Conductive polymers can be applied also to polyurethane, silicone, and polyimide foams. These foams are finding use in EMI suppression, against high intensity radar absorption, static dissipation, etc. [1,35,90]
Abbasi and Militky [94] coated PPy on E-glass fabric (3/1 twill having 54 × 30 warp and filling per inch) by vapor deposition of pyrrole monomer in the presence of tetraethyl ammonium p-toluene sulfonate called as TsO- (tosylate) as doping agent with FeCl3 as an oxidizing agent. The conductive fabric provides 98.67%–99.23% loss in power at the frequency range of 800–2400 MHz (Figure 14). These fabrics can be used to shield the house hold appliances, cellular phones, buildings, various electronic gadgets that operate up to 2.4 GHz frequency, etc.
SEM images of GF fibres after PPy coating: (a,b,c) concentration of doping agent from 0.05 M to 0.1 M, (d) two coatings of PPy at low concentration of tosylate [94].
In addition, several printing processes are utilized for electrically conductive textile fabric formation: screen printing, gravure printing, roll to roll method, and inkjet printing. One of the problems with printing of conductive polymers is the resolution of the paste. Their thickness varies depending on the process applied. Different conductivity values can be obtained which are suitable for applications such as keyboards and notebook PCs, touch screens, electrocardiogram (EKG/ECG) electrodes, etc. [34,76,77]
Calvert et al. [95] used electroless plating method and ink-jet printing to deposit sensing lines of PEDOT:PSS polymer complex on weave fabrics and to print silver lines to connect the strain sensors developed to the monitoring equipment (Figure 15(a)). They also investigated the strain response of printable conductive composites. In this research, the fabric type was crucial to obtain greater sensors response. Fabrics showed a higher gauge factor for lines printed across the fabric axes.

Weremczuk et al. [96] reported that humidity sensors can be printed directly on textile using the ink-jet printing technology. Silver nanoparticles or gold were used for electrodes (Figure 15(b)). A humidity sensitive layer was formed by mixing PEDOT:PSS, poly(2 hydroxyethylmethacrylate) (pHEMA), cellulose acetate butyrate (CAB) and sulfonated tetrafluoroethylene (Nafion®solution). Sensors were fabricated mainly on Kapton®polyimide sheets and then woven into a textile.
Embroidery techniques
Another possibility to achieve a conductive fabric is to attach a conductive structure (thin stainless steel, copper or other metal wires) to a ground structure by using the embroidery techniques. Usually such techniques are largely integrated in textile industries to create interconnections between sensors and output electronic systems. These techniques allow precisely specifying the circuit layout and stitching pattern in a computer-aided design environment, under machine control and integration of yarns with different electrical properties [21,97].
Electrically conductive textiles and smart textiles applications
A large number of lab-scale electrically conductive textiles for smart textiles needs could be found in literature, but still not-enough number of them is present in market. Focus of interest of this paper is to present commercially available conductive polymer-based smart textile products for health, sport and fitness, and automotive applications. Furthermore, great potential in a group of smart textile materials show superamphiphobic electrically conductive textiles, electrochromic textiles and EMI shielding textiles for targeted applications.
Health, sport and fitness applications
Takamatsu et al. [97] developed textile-based wearable devices inspired by Japanese kimono dyeing technique (Figure 16(a)) to record high quality EKG in clinic and ambulatory conditions, and to determine heart rate. Polydimethylsiloxane (PDMS) stencil was used due to its hydrophobic nature to confine the spreading of the aqueous PEDOT:PSS dispersion onto interlock knit polyester fabric. PEDOT:PSS electrodes fabricated in this way and coated with an ionic liquid gel showed a low impedance contact with the skin.

New monitoring Edema Stocking device (Figure 16(b)) produced by Edema ApS enables measurements of volume changes in the lower limbs. The system provides supplementary knowledge about fluid retention and efficiency of treatment for drainage via objective scientific measurement [98].
Sensoria company developed smart socks (Figure 16(c)) infused with textile sensors that can detect foot pressure. Conductive fibres in the sock relay data to an anklet which uses Bluetooth to communicate with a mobile app. Production of reliable and predictable textile sensors capable of sustaining machine wash cycles is key in the development of effective smart fabrics [99].
Adidas company integrated conductive yarns into stretchy garments by knitting technology. Textile electrodes, undetectable and friendly for skin (Figure 16(d)), can pick up signals from the heart and other muscles. The data gathered by these electrodes are transmitted through the garment to a small gadget snapped into sport top [100].
DuPont Microcircuit Materials launched the first products of stretchable conductive inks printed onto the textile (Figure 16(e)). The inks showed strong performance after 100 washing cycles when they were printed onto the PU film [101].
Seymourpowell company designed a new smart three-in-one jacket known as the Life Tech jacket (Figure 16(f)) for Korean outdoor sportswear brand KolonSport. This jacket keeps users alive in the hard conditions [102].
Paul et al. [103] developed textile electrodes suitable for human biopotential monitoring (Figure 17(a)). They used screen printing to create encapsulated conductive tracks. Different pastes were taken for the electrode networks: a screen printable polyurethane, a screen printable silver conductor and a stencil printed conductive rubber. Woven textile called “Escalade” was used as a printed substrate (density of 295 g/m2, woven in a 3 × 1 twill, thickness of 410 µm). Conductive encapsulation was stencil printed at electrodes sites maintaining an electrical connection to the skin surface.

Carvalho et al. [104] described a shirt for use in health, risk environments and sport monitoring. Therefore, basic weft structures were developed (jersey, simple pique, locknit). The electrode areas (Figure 17(b)) were knitted with a textured multifilament polyamide yarn with a thin silver coating (less than 10 nm). The ECG connections were routed to a single line, located on the back of the shirt and from there to a single place to connect to the specific acquisition circuit.
Automotive applications
One shot manufacturing of complex shaped parts may be considered as one of the best solutions for the fast production of composite pieces dedicated to the automotive and railway industries. Replacement of metallic cross stiffeners with their composite parts made of GF/PP commingled yarns was reported by Risicato et al. [105] (Figure 18(a)). The PEDOT:PSS sensors based on glass yarns were inserted into braided fabrics coupons (before thermal consolidation) and provided the ability to monitor the structural health of these composite parts in a real time in order to choose the appropriate composite architecture for the final automotive applications.

T-Ink company developed conductive inks (Figure 18(b)) so robust that circuits, sensors and switches can be printed on flat plastic and than molded into three-dimensional components that control overhead lights and sunroofs in cars. Parts made with this technology can replace thicker assemblies reducing the weight and dimensions of wired components [106].
Other applications
Wang et al. [13] prepared durable superamphiphobic (superhydrophobic and superoleophobic) electrically conductive fabric by one-step vapor phase polymerisation of EDOT with the presence of fluorinated decyl polyhedral oligomeric silsesquioxane (FD-POSS) and fluorinated alkyl silane (FAS) (Figure 19(a)). Commercial polyester, (plain weave, 168 g/m2) was used as the substrate. The addition of FD-POSS and FAS to the PEDOT showed little influence on the surface resistance. It can impart the PEDOT coating with durable liquid repellency and self-healing ability to autorepair from chemical damages. FD-POSS was found to play an important role in enhancing the washing and abrasion durability and self-healing function of the coating.

Chromic materials are very challenging in terms of their integration into textiles. Metallic materials may damage the textile structure or affect the handling and comfort properties of it. The replacement of inorganic materials with organic materials can be a solution to these problems [13,34,107]. Invernale et al. [107] developed electrochromic flexible textile electrodes by PEDOT:PSS impregnated spandex (Lycra) spray-coated with electrochromic polymer (gel electrolyte usage) (Figure 19(b)).
Bajgar et al. [35] presented the cotton fabric (bleached, plain weave, specific mass 120 g/m2, sett: 51.2 n/cm (warp), 28.0 n/cm (weft), yarn count 7.4/2 tex (warp), and 14.5 tex (weft)) coated with conducting polymers, PANI or PPy, in situ during the oxidation of respective monomers in aqueous medium (Figure 19(c)). Modified fabrics were coated again with PPy or PANI in order to investigate synergetic effect between both polymers with respect to conductivity and its stability during repeated dry cleaning. The resulting fabrics were used as electrodes to collect the electrical response of a
Conductive polymers are considered also for EMI shielding applications, e.g. PPy-coated woven polyester twill fabric (Figure 19(d)), EeonTex™ T-PI-365 (223 g/m2), is used to make artificial horizon, radar barriers for military aerospace purposes [27,34,76,77,90].
Future prospects
Based on many advantages of smart textiles, the growth in medical and healthcare applications is obvious. More end-user applications need to be launched to market to satisfy customer’s needs with significant focus on these areas. Sport and fitness applications should be daily basis consumer products. Due to the rapid rhythm of life in many regions, smart textiles should be directed to other applications, specially to smart home textile applications development.
Conclusion
Various approaches are used in this article for the development of electrically conductive textiles as a part of smart textiles area. Only a few commercial smart textile products based on conductive polymers usage are present today although a huge number of lab-scale structures can be found. It is necessary to look for new ways of their development, while the washability issue of these textiles needs to be taken into greater consideration for diverse commercialized applications. Therefore, good research strategy and industry plan have the most important role following the market needs and new technologies.
Footnotes
Acknowledgement
The authors would like to thank the European Commission for the funding of the project.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the EU project “MAPICC 3D” within the call NMP-FP7- 2010-3.4-1 (Grant Number 263159) entitled: One-shot Manufacturing on large scale of 3D up graded panels and stiffeners for lightweight thermoplastic textile composite structures.


