Abstract
A novel and efficient method for the electroless silver plating polyimide fabric was developed with electromagnetic shielding properties. Firstly, polyaniline (PANi) was in situ polymerized with ammonium persulfate used as oxidizing agent. Secondly, Ag+ was in situ reduced to Ag0 by PANi, and the electroless silver plating was initiated by Ag0 used as active center. Fourier transform infrared, scanning electron microscopy, X-ray diffraction, contact angle analysis and energy dispersive spectroscopy were used to characterize the composite fabric. It could be observed that silver layer-coated polyimide fabric was compact and uniform with surface resistance about 0.02 Ω/sq. The thermal stability was evaluated by thermogravimetric analysis. The as-prepared fabric had excellent anti-corrosion, tensile strength and fastness. The shielding effectiveness of this fabric could reach 54–90 dB, which implied it was a good candidate as electromagnetic shielding materials in many fields.
Introduction
The wide development of conductive materials with the excellent electromagnetic interference (EMI) shielding effectiveness (SE) has attracted a great deal of attention, and the materials have been widely applied in smart clothing, electronic products, medical equipment, military industry and other fields [1–3]. When the electromagnetic wave penetrates the EMI SE materials, its energy is gradually attenuated through absorption efficiency and multiple reflection efficiencies and the remaining is transmitted [4–6]. Therefore, these EMI SE materials which can weaken the energy either by absorption, dissipation and reflection are the effective tools to prevent electromagnetic pollution or interference [7].
Recently, the electromagnetic shielding materials with excellent magnetic permeability and electric conductivity have been fabricated by using composite structure model [8]. However, the traditional shielding materials that contain metallic wires (Cu, Ni, Ag and Au) and polymers show certain shortcomings, such as poor ductility and easy oxidation [9,10]. Thus, the shielding materials based on textiles have gradually replaced the traditional shielding materials due to light quality, excellent ductility and low cost [11]. The shielding textiles can be prepared by coating with nickel [12], copper [13], gold [14] and silver [15], but the SE of composite fabric and the fastness between fabric and metal layer was poor.
Polyaniline (PANi) has attracted wide attention because of cheapness, good environmental stability, outstanding electrical conductivity and electrical activity [16–18]. But in the field of electromagnetic shielding materials, its combination with metal layers, especially using its reducibility to metal ions are rarely reported [19]. Therefore, we propose a method to prepare shielding textiles by the surface treatment wherein metal ions are reduced to metal species by PANi [20], and these metal species are used as catalyst to initiate the electroless plating (Figure 1) [21–23]. The advantages of this method are that the adhesion strength between metal layer and fabric can be improved and better SE can be obtained due to refection of metal layer and absorption of conductive polymers [24]. The as-prepared Ag/PANi/PI fabric not only had excellent SE and remarkable electrical conductivity but had also shown outstanding anti-corrosion resistance, thermal stability and fastness. In addition, as a novel-type high-performance fiber, PI fiber presents huge potential due to its outstanding thermal stability, high dimensional stability, corrosion resistance, low dielectric constants and electrical insulation. We believe our research will broaden its application in protecting textile markets [25–27].
The process of the preparation of Ag/PANi/PI fabric by PANi in situ reduction.
Experimental
Materials
The polyimide (PI) fabric was purchased from Jiangsu Aoshen Hi-tech Materials Co. Ammonium persulfate (APS), aniline (AN) monomer, nitric acid, sodium carbonate, sodium hydroxide and silver nitrate were purchased from Shanghai Chemical Reagent Co., Ltd, China. Ammonia, ethylenediamine, ethylalcohol, glucose, polyethyleneglycol were provided by Chinese Medicine Group. All reactants were of analytic purity grade and were used without further refinement.
In situ deposition of PANi and the reduction of silver on PI fabric
Firstly, PI fabric was immersed into 10 g/L NaOH solution at 80℃ for 10 min, liquor to material ratio 1:50; then it was completely rinsed by distilled water and dried in an oven. Secondly, PANi/PI fabric was prepared by in situ deposition. The treated PI fabric was dipped into the solution containing 0.25 mol/L AN monomer, 0.5 mol/L HNO3 and 0.25 mol/L APS at the room temperature for 2 h.
After the reaction, the fabric was washed with distilled water and dried at constant temperature. Ag+ particles were reduced to Ag0 particles by PANi [22] when the PANi/PI fabric was soaked into 2 g/L AgNO3 solution at room temperature for 5 h.
Preparation of silver plating PI fabric
The electroless silver plating was initiated by these Ag0 particles [20]. The reducing solution A was prepared with 8 g/L AgNO3; then 25% NH3·H2O was added into the solution to make it muddy, continued until it was clear again; after that, 10 ml/L ethylenediamine was added. The oxidant solution B was prepared with ethylalcohol, glucose, polyethyleneglycol (degree of polymerization 1540, 67,778 g/mol) which were 40 ml/L, 32 g/L and 75 mg/L, respectively. Solutions A and B were mixed in the same volume quickly. The as-prepared fabric was immersed into the solution, liquor to material ratio 1:50, stirred for 1.5 h at room temperature. Finally, the Ag/PANi/PI fabric was washed by distilled water and dried at room temperature.
Characterization
The FTIR spectra of the samples were obtained by Fourier transform infrared analysis (Nicolet 6700, Thermo Fisher, USA). Scanning electron microscope (SEM, TM-1000, Hitachi) was used to observe the surface morphology of samples. The hydrophilicity was measured by contact angle (CA) analysis (DSA30, Kruss, Germany). Thermogravimetric analysis (TGA) was performed using 209 F1 analyzer (Netzsch, Germany) at N2 atmosphere with a heating rate of 10℃/min, from 30℃ to 900℃. The chemical composition of the metal deposit on the fabric surface was determined using energy dispersive spectroscopy (EDS, IE 3000, UK) attached to the SEM. The crystal structure of samples was recorded using X-ray diffraction (XRD, D/Max-2550, Rigaku, Japan) at room temperature with 2θ ranges from 5° to 90°. The conductivity was measured by the surface resistance tester (MCP-T370, Loresta, Japan). The SE was evaluated using DR-913 anti-electromagnetic radiation tester (Wenzhou Darong Textile Co.). The washing fastness was evaluated by treating Ag/PANi/PI fabric in solution containing 5 g/L soap and 2 g/L Na2CO3 at 85℃ for 30 min according to Chinese standard GB/T3921-2008. The rubbing fastness was measured using rubbing fastness tester (CM-5, ATLAS, USA) according to Chinese standard GB/T3920-2008. To further measure the fastness of ultrasonic washing, the Ag/PANi/PI fabric was put into ultrasonic cleaner (SY-180, Shanghai, Chengxian Co., Ltd) containing 10 g/L soap solution and operated for 30 min [12]. Furthermore, the adhesion strength between fabric and metal layer was evaluated by a standard scotch tape method [28]. The corrosion resistance was tested by the changes of the electrical conductivity of the samples in different pH solutions with treatment for 24 h, and the tensile strength was measured according to Chinese standard GB/T3923.1-1997 (strip method) [29]. The weight increasing percentage was calculated by the following equation
Results and discussion
FTIR spectra
The FTIR spectra of untreated PI fiber, PANi doped HNO3 and PANi/PI fabric are presented in Figure 2. The FTIR spectra of PANi and PANi/PI fabric were similar to those reported previously [17,30]. As seen in Figure 2(c), the characteristic peaks at 1565 cm−1 and 1487 cm−1 were attributed to ring-stretching vibrations of the quinone ring skeleton (Q) and benzene ring skeleton (B) of PANi. The degree of oxidation of PANi could be obtained according to the intensity for these two peaks. The 1123 cm−1 absorption peak represented B-Q-B stretching vibration which was the characteristic band of the conductive form of PANi [31]. In Figure 2(a) and (b), the strong peak that appears in the 1726 cm−1 was associated to carbonyl groups of amide, which, due to the stretching vibration, was less disturbed by the other functional groups. The curve of PANi/PI fabric shown in Figure 2(b) contained both PI and PANi characteristic peaks and did not appear as new peaks, which indicated that PI fabric was successfully functionalized and the chemical structure of PI fabric was not damaged.
FTIR spectra of untreated PI fabric (a), PANi/PI fabric (b) and PANi powder (c).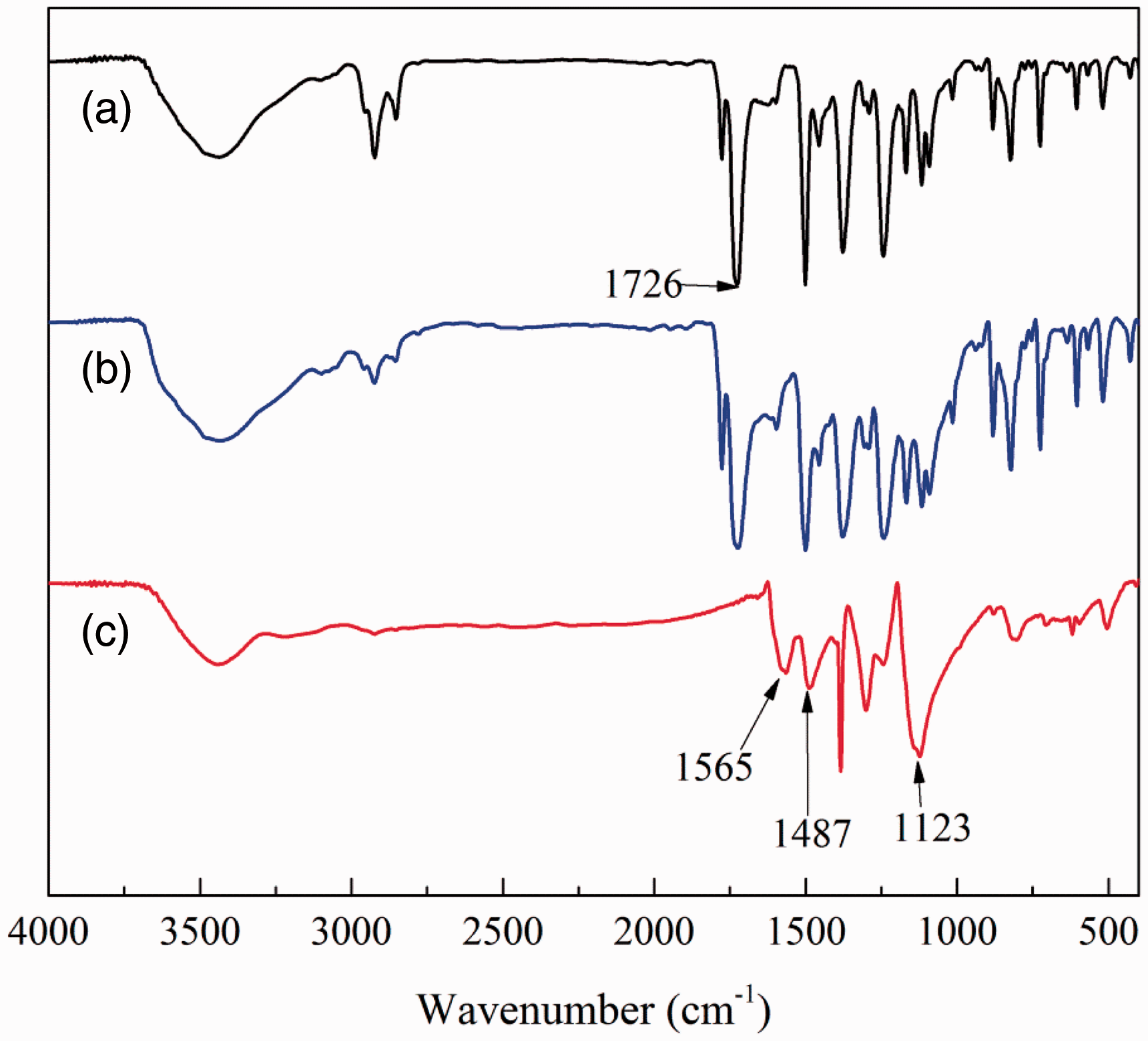
SEM/CA analysis
Neat PI textiles have so weak affinity with PANi that they must be treated by chemical etching to improve interfacial area and chemical activation. As seen in Figure 3(a), compared with the untreated PI fabric, the surface-treated PI fabric was smooth and clean with small pits which due to the chemical etching of NaOH solution. PANi had been in situ polymerized in aqueous solution, and the surface of treated PI fiber was successfully and completely coated by PANi could be observed in Figure 3(b). It could be noticed that Ag particles were absorbed on the surface of PANi/PI fabric which, due to Ag+, was reduced to Ag0 by PANi in Figure 3(c). Then, the electroless silver plating was initiated by the Ag0 particles used as active center, as shown in Figure 3(d); most of the surface of fibers was covered by compact silver layer.
SEM photos of PI fabric treated by NaOH (a), PANi/PI fabric (b), AgNO3 immersed PANi/PI fabric (c) and Ag/PANi/PI fabric (d).
From the results of CA tests, it can be clearly observed that the surface of treated PI fabric and PANi/PI fabric was hydrophilic with CA 0° (Figure 4(a) and (b)). On the one hand, the molecular chains of PANi were functionalized by doping nitrate. On the other hand, hydrogen bonds were easily formed between PANi and water molecules [32]. In Figure 4(c), the surface of AgNO3 activated fabric represented hydrophilic, but wetting time was longer than 10 s due to the existence of Ag particles on PANi/PI fabric surface. As shown in Figure 4(d), Ag/PANi/PI fabric performed hydrophobic with CA about 118° which illustrated surface property was changed and compact silver layer was formed after plating.
CA photos of PI fabric treated by NaOH (a), PANi/PI fabric (b), AgNO3 immersed PANi/PI fabric (c) and Ag/PANi/PI fabric (d).
EDS analysis
In the EDS spectra of untreated PI fabric, PANi/PI fabric, AgNO3 immersed PANi/PI fabric and Ag/PANi/PI fabric, the further information of fabric surface could be obtained. The component of silver had been found on the surface of PANi/PI fabric which, due to AgNO3, was reduced by PANi, as shown in Figure 5(c). In Figure 5(d), silver can be detected and the silver atomic percentage was 59.65% and mass percentage was more than 93.00%, which further confirmed the results shown in Figure 3(d).
EDS spectra of untreated PI fabric (a), PANi/PI fabric (b), AgNO3 immersed PANi/PI fabric (c) and Ag/PANi/PI fabric (d).
XRD and fastness analysis
In Figure 6, the XRD curves of untreated PI fiber, PANi/PI fabric and Ag/PANi/PI fabric were shown, respectively. Compared with the curve (b), the XRD spectra of untreated PI fabric (a) had a stronger peak at 2θ = 18.8°, which suggested that the crystallinity of PANi/PI fabric was decreased. The alkali treatment destroyed the structure and expanded amorphous region of PI fiber, which could promote that the distribution of PANi and PANi could easily penetrate the amorphous region of the PI fiber. In order to test the durability of metal layer and the substrate, the rubbing fastness was carried out and it reached grade 4. The adhesion strength between fabric and silver layer was also of four grades, indicating the good adhesion strength [5]. As shown in Figure 6(c), the diffraction peaks at 2θ = 38.2°, 44.5°, 64.7°, 77.6° and 81.7° represented (111), (200), (220), (311) and (222) planes of silver, respectively. The diffraction peak of Ag2O was not found.
XRD patterns of untreated PI fabric (a), PANi/PI fabric (b) and Ag/PANi/PI fabric (c).
Corrosion resistance and tensile strength properties
In order to study the corrosion resistance of plating silver layer, the electrical conductivity of Ag/PANi/PI fabric before and after treating with different pH solutions for 24 h was measured, and the results are shown in Figure 7. For each sample, 30 tests were conducted and the results were reported as the averages obtained. As seen in Figure 7, the surface resistance increased and the electrical conductivity decreased slightly after treatments, which was due to the improvement of the fastness between fiber and silver layer and the block of corrosive ions into dense silver layer. The results illustrated that silver-plated fabric had an excellent corrosion resistance.
Surface resistance of Ag/PANi/PI fabric treated in 0.2 mol/L HCl solution, 0.2 mol/L NaOH solution and 50 g/L NaCl solution.
In Figure 8, the changes of elongation were basically consistent with the changes of tensile strength. Compared with the untreated PI fabric, the tensile strength of warp-tensile strength (a) and weft-tensile strength (b) both slightly decreased and the retention rate of tensile strength reached up to 80%–85%, which was mainly due to some etched effect on the surface of the treated PI fabric. However, compared with the treated PI fabric, the tensile strength of the PANi/PI fabric was slightly higher, which was due to the structure that PANi between yarn and yarn played the adhesive role, and this structure also could explain the enhancement of the tensile strength of Ag/PANi/PI fabric. According to the analysis of the experimental results, this silver plating process basically retained the tensile strength of PI fabric which could provide the potential possibilities in application.
Tensile strength and elongation of the untreated PI fabric (a), PI fabric treated by NaOH (b), PANi/PI fabric (c) and Ag/PANi/PI fabric (d).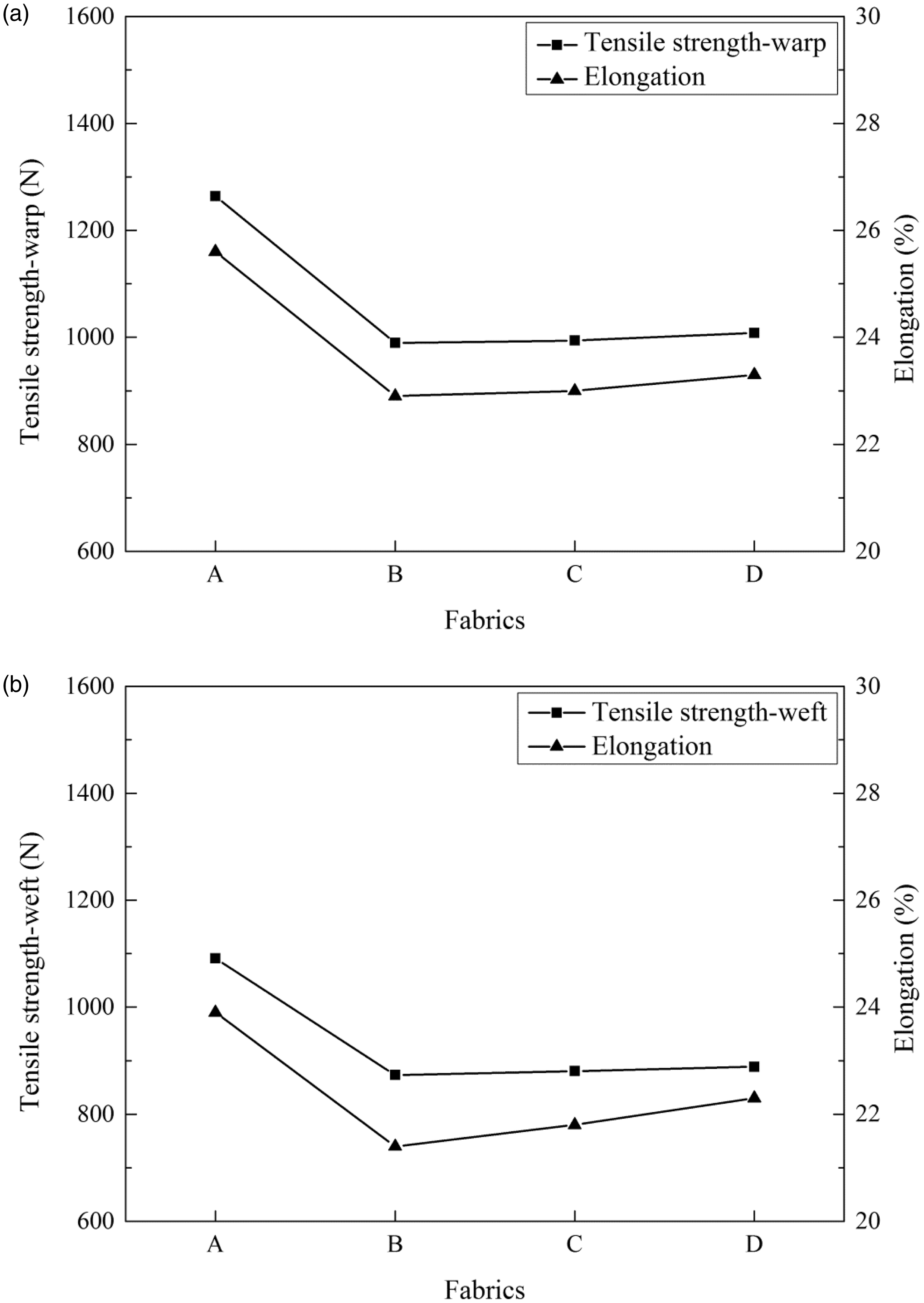
TGA analysis
To analyse the thermal stability of the untreated PI fabric, PANi/PI fabric and silver-plated fabric, TGA was used under N2 atmosphere with a heating rate of 10℃/min. In Figure 9(a), the first decomposition reaction of untreated PI fabric occurred in the range of 209℃–239℃, and the residual contents were more than 91.1%, which was due to the oligomers and impurities that may be decomposed at this temperature. For PANi/PI fabric (b) and Ag/PANi/PI fabric (c), there were no obvious difference in 209℃–239℃ which was due to the protective effect of PANi and Ag layer for PI fiber. And after treating in NaOH solution, the oligomers and impurities on the PI fiber surface were removed. As shown in Figure 9, these three samples all started thermal degradation from 595℃; with the rising of temperature, the decomposition became different which was due to the diverse chemical bonds that were destroyed. The residual components of these three samples at 900℃ were 46.1%, 52.8% and 70.5%, respectively. It could be deduced from the results that the content of silver plating was about 37.5%, which indicated the result consisted with weight increasing value 36.7% according to equation (1).
TGA curves of untreated sample (a), PANi/PI fabric (b) and Ag/PANi/PI fabric (c).
Electrical conductivity and SE
Compared with PANi/PI fabric, Ag/PANi/PI fabric had a better electrical conductivity with the surface resistances of PANi/PI fabric and silver-plated fabric being about 1.10 KΩ/sq and 0.02 Ω/sq, respectively. It is known that SE consists of absorption efficiency and multiple reflection efficiencies from materials, and the reflection efficiency is related to conductivity of metal layer and electromagnetic wave frequency. The SE is determined by conductivity of metal coating when the frequency is changeless [33].
The Ag/PANi/PI fabric could reach favorable SE mainly by the absorption efficiency of PANi and the reflection efficiency of silver layer. The SE of samples was shown in Figure 10, the result of PANi/PI fabric (Figure 10(b)) was less than 17.7 dB and the result of Ag/PANi/PI fabric (Figure 10(c)) was more than 54 dB in the electromagnetic radiation range from 30 kHz to 3 GHz, which indicated that Ag/PANi/PI fabric had outstanding SE, as large-scale electromagnetic wave could be shielded by the reflection efficiency of silver layer. After ultrasonic washing in 10 g/L soap solution for 30 min as shown in Figure 10(d), the change of SE was less. Moreover, after 10 cycles washing in the solution containing 5 g/L soap and 2 g/L Na2CO3 at 85℃ for 30 min, the SE remained 30–70 dB, which suggested that the washing process of the Ag/PANi/PI fabric was satisfactory and it was a good candidate as electromagnetic shielding materials in many fields.
SE curves of untreated sample (a), PANi/PI fabric (b), Ag/PANi/PI fabric before (c) and after (d) ultrasonic washing and after 10 cycles washing (e).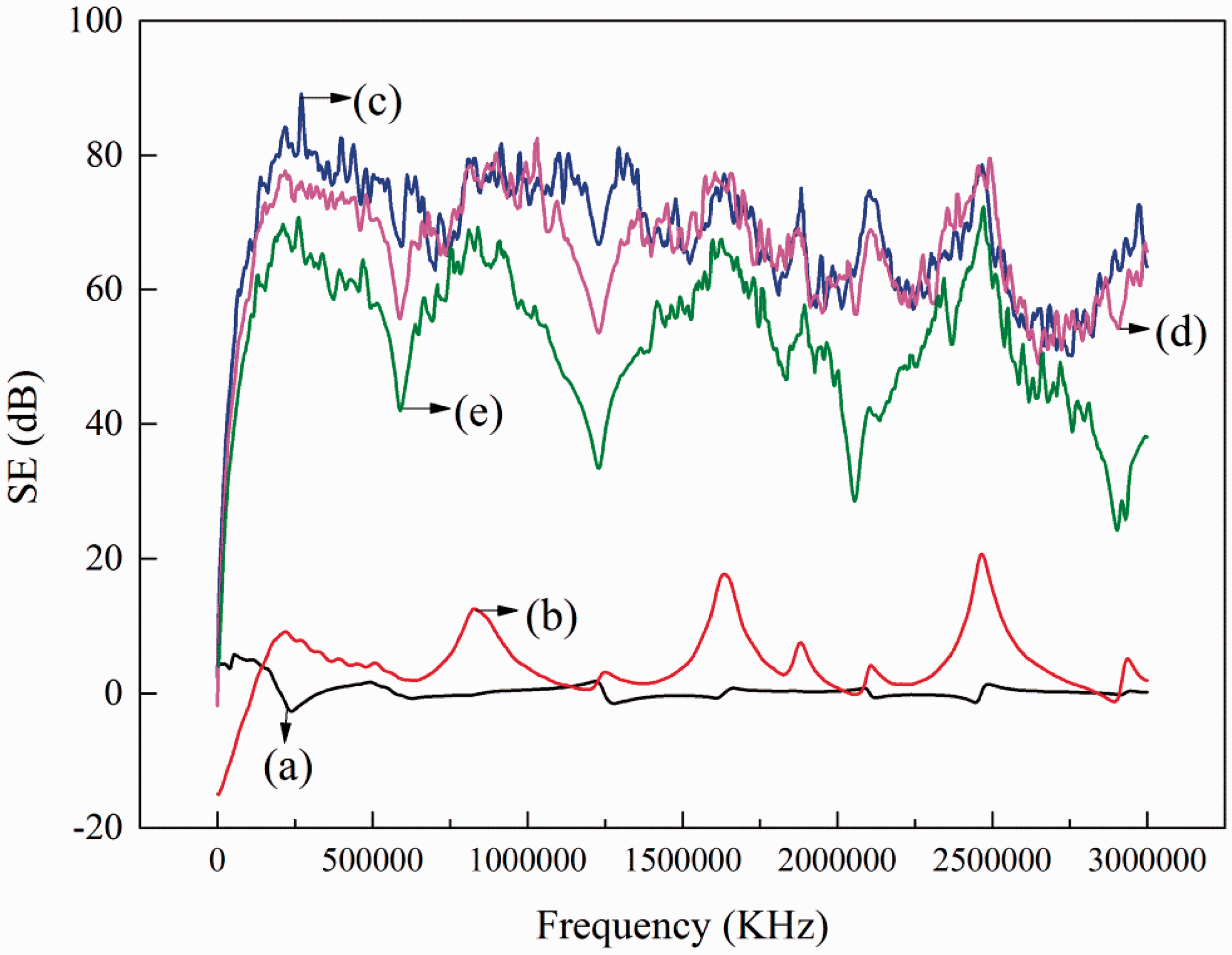
Conclusions
Silver-plated textiles have been widely investigated in many fields such as SE textiles for protective clothing, for their high SE and fastness, excellent electrical conductivity and outstanding thermal stability. In this study, the PANi/PI fabric could be prepared by in situ chemical deposition in the presence of PI fabric. The aim of the work was to prepare Ag/PANi/ PI fabric with high SE by electroless silver plating, and the Ag/PANi/PI fabric had satisfactory fastness. By SEM, CA and EDS tests, it could be noticed that Ag+ was in situ reduced to Ag0 by PANi on the surface of PANi/PI fabric. From TGA, XRD and conductivity of Ag/PANi/PI fabric, we found that a compact and uniform silver layer was coated on the PI fiber with surface resistance of about 0.02 Ω/sq. The tests of corrosion resistance, tensile strength, TGA and fastness demonstrated that the silver-coated fabric had shown excellent anti-corrosion resistance, tensile strength, thermal stability, washing and rubbing fastness. Furthermore, the SE of Ag/PANi/PI fabric could reach 54–90 dB. All the above-mentioned results show that this is a facile method to fabricate multifunctional electromagnetic shielding textiles.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research was supported by “the Fundamental Research Funds for the Central Universities” (No. 16D110518) and “the Funded Project for Zhejiang Postdoctoral Science Preferential Foundation” (No. 152342).

