Abstract
A highly conductive polyester fabric was fabricated by the two-step successive in situ polymerization of aniline. This was followed by silver electroless plating on the surface of the fabric. Also, a silver electroless-plated fabric and a PANI-coated fabric were prepared by the in situ chemical polymerization of aniline. The electrical conductivity measurements, scanning electron microscopy, thermal gravimetric analysis, water contact angle measurements, wide angle X-ray diffraction, Fourier transform infrared spectroscopy, and air permeability tests were conducted on the coated fabrics. It was found that the surface of the hybrid PANI/silver-coated fabric was coated with granular silver particles having the average diameter of 1.3 µm. Besides, the amount of the reduced silver on the surface of the fabric was much higher than that in the silver electroless-plated fabric. The emeraldine form of PANI was oxidized to pernigraniline and the silver ions were reduced to silver metal particles. The hybrid PANI/silver-coated fabric showed the electrical conductivity of 2.63 × 103 Scm−1; so, it could be applied in many smart wearable applications.
Introduction
Wearable electronics, which are used in many applications related to smart textiles, have received a lot of attention recently. The smart textile materials including conductive fibers and fabrics can be used in the conventional clothing with applications in various fields including sport, entertainment, education, medicine and military [1,2]. There are several approaches for producing conductive fibers and fabrics. Coating the fibers and fabrics with conducting polymers, such as PANI and polypyrrole, has been studied because of their potential applications in the smart textiles [3–6]. On the other hand, electroless plating of metals on the textile substrates has been carried out by several researchers [7–14]. The electroless silver plating is a non-galvanic chemical reaction in an aqueous solution through the chemical redox process. Jiang et al. [15–18], for example, conducted silver electroless plating on the surface of cotton and polyester fabrics . Lu et al. [19] also used the ultrasonic-assisted electroless deposition of silver on the polyester fabric for use in electromagnetic shielding applications. Further, Yu et al. [20] produced a conductive silk fabric with antibacterial properties by electroless silver plating on aramid fibers with good conductivity [21]. Besides, the composites of PANI/silver have been studied by many researches to produce the conductive materials [22]. The important aspect of PANI/silver composites is their electrical properties. PANI is a semi-conductive polymer and silver is a metallic conductor. Therefore, the hybrid composites of PANI and silver combine different electrical features of the parent ingredients. On the nanoscale, PANI-silver composites show a variety of morphologies, such as globules, nanofibers, nanotubes, nanobrushes and sheets [22]. The electrical properties of the composite are affected by the morphology and distribution of the constituents within the composite, as well as the interfacial phenomenon of the ingredients [22,23]. In some of the researches carried out to prepare the PANI/silver composites, aniline has been oxidized by silver salts, such as silver nitrate [24,25]. Another method for the preparation of the hybrid composites of PANI/silver is the use of PANI as the reductant of the silver ions to the metallic silver [26–28]. PANI can reduce several noble metal ions to their corresponding metals. The emeraldine form of PANI is oxidized to the pernigraniline form [29]. The nanostructure of both components, PANI and silver, can have a great impact on the electrical features and other properties of the composites [22].
PANI–silver composites intensely differ in their conductivity [30]. Due to the reaction between PANI salts and silver nitrate and the conversion of emeraldine to the pernigraniline form, the conductivity of PANI is reduced. The formation of the metallic silver, on the other hand, should lead to the increase in the conductivity. Therefore, both effects balance each other from the conductivity viewpoint. Although some papers have stated that the presence of silver in a PANI–silver composite increases electrical conductivity, in some other researches, the addition of silver to the composite has been found to reduce the conductivity, as compared with the PANI-coated fabric [31,32]. This could be due to the less electrical contact between the silver particles and/or the decrease in the conductivity of PANI because of its oxidation [32].
Textile fabrics have been coated with the composites of a conducting polymer and silver for different purposes, such as electromagnetic interference shielding [33–36], antimicrobial properties [33,37], and production of conductive fabrics [31]. In this regard, we can point to the coating of the polyester fabric with PANI that is followed by the electroless silver plating initiated by the in situ reduction of PANI [33], the composite coating of PANI/silver on polyimide and cellulosic fabrics [31,38], and the composite coating of polypyrrole/silver on poly(ethylene terephthalate), polyimide, cotton and wool fabrics [35,39–41]. According to the results of the mentioned investigations, it seems that further research into the production of composite-coated fabrics with the optimum electrical conductivity properties, as well as the properties related to the wearing performance, is needed. Therefore, the aim of this study, in the first stage, was to obtain the optimum conditions for producing a highly conductive polyester fabric with a PANI/silver composite coating. Secondly, the other important fabric properties such as air permeability and hydrophobicity were investigated. However, some of the properties of the coated fabrics, such as tensile properties, handle, stiffness, drape, etc., which need larger samples, will be investigated in the future studies as the continuation of this work. For this purpose, a polyester fabric was coated with PANI by the in situ chemical polymerization of aniline. Subsequently, the fabric was immersed in the AgNO3 solution as an activation step to reduce Ag(I) to Ag (0) by PANI; finally, electroless silver plating was performed. The study shows that the composite PANI/silver-coated polyester fabric could be used as a conductive material in many smart wearable applications.
Materials and methods
Materials
A plain polyester fabric (118 g/m2, warp: 100 den, 18 cm−1, weft: 177 den, 25 cm−1) was used in this study. Silver nitrate, sodium hydroxide, aniline, ammonium persulfate and aqueous ammonia were purchased from Merck (Darmstadt, Germany). All the fabric samples were pre-cleaned in a bath containing 0.5% nonionic detergent at pH=7 and at the temperature of 40℃ for 30 min, and then they were washed with deionized water.
Electroless silver plating (method A)
After pre-cleaning the fabrics, the sensitization process was performed with a solution of 5 g stannous chloride (SnCl2.2H2O) and 5 mL hydrochloric acid (HCl, 37%) in 1000 mL water at room temperature for 10 min. The samples were washed in deionized water. The electroless plating process was carried out with a mixture of the solution A (6 g silver nitrate, 4 g sodium hydroxide and a few drops of ammonium hydroxide solution (NH4OH, 28%) in 500 mL water and the solution B (1.3 g glucose (C6H12O6) in 500 mL water) for 10 min at room temperature (the sample Ag1) [17].
Electroless silver plating (method B)
The solution A consisted of AgNO3 solution (9 g/L), 6.25 g/L NH3·H2O (25%) and 10 mL/L ethane-diamine. The solution B was prepared with 1.56 g glucose, 40 mL/L ethyl alcohol, and 50 mL/L polyethyleneglycol. The solution A was added to the solution B with the same volume rapidly. The as-prepared samples were immersed in the mixed solution in an ultrasonic bath for 60 min at the range of 30–40℃ while stirring. The liquor ratio was 1:50. Finally, the samples were rinsed and dried (sample Ag2) [42].
In situ polymerization of aniline
In situ chemical polymerization of aniline was carried out in a bath by the following procedure. Distilled aniline (0.5 M) was dissolved in a bath containing the HCl solution (0.35 N) while stirring to achieve the homogenous mixing. The fabric samples were placed in the solution at 40℃ for 2 h to be soaked well with the monomer and the dopant solution. To initiate the polymerization reaction, the aqueous oxidizing agent (0.25 M ammonium persulfate dissolved in 035 N HCl) was slowly added into the bath. The oxidant to aniline ratio was kept constant at around 1.25. The whole polymerization reaction was carried out at 5℃ for 1 h. After the polymerization process, the fabrics were washed in distilled water containing 0.35 N HCl and dried at 60℃. Finally, the fabrics were washed with deionized water and dried (PAN1). For the PAN2 samples, the in situ polymerization of aniline was repeated one time; regarding PAN3 samples, it was repeated twice. All of the fabrics were conditioned at the relative humidity of 65% and 25℃ for at least 24 h before further evaluation.
The electroless plating of the PANI-coated samples was performed as described here. The samples were immersed in a 2 g/L AgNO3 solution at room temperature for 5 h. Then, the fabrics were washed with deionized water twice and dried. Afterwards, the silver electroless plating was carried out according to the method B, as described above. Finally, the samples were rinsed with distillated water and dried in air (the samples PAN1Ag, PAN2Ag and PAN3Ag). The process of the fabrication of PAN3Ag sample is shown schematically in Figure 1. Besides, the reduction of the silver ions by PANI is displayed in Figure 2.
The process of PANI/silver coating of the polyester fabric. Reduction of the silver ions by PANI [30].

Weight and thickness change
The weight change percentage of the fabrics was calculated as follows
SEM micrographs
The surface morphology of the samples was investigated using a Philips scanning electron microscope (XL series, Netherlands) equipped with an energy dispersive X-ray (EDX) analyzer operating at an accelerating voltage of 20 kV and a field emission electron microscope (FESEM, Hitachi 4160, Japan).
Fourier-transform infrared spectroscopy
The Fourier-transform infrared spectroscopy (FTIR) spectra of the samples were taken using Fourier transforms infrared analysis (Bomem, MB100, Canada). KBr-sample pellets were prepared and the spectra were obtained in the range of 4000–400 cm−1 at room temperature.
Air permeability
The air permeability of a fabric is a measure of how well it allows the passage of air through it. So, it is defined as the volume of air in m3 which is passed in one second through 1 m2 of the fabric at a pressure difference of 1 cm of water. The air permeability of the fabrics was measured using the Shirley SDL-MO215 equipment, according to the ASTM D737 method.
Contact angle measurements
The hydrophobicity of the fabrics was evaluated by means of contact angle measurements. The contact angle was measured directly from the image at 3 s after a 5 µl droplet of distilled water was placed on the fabrics surface. The distance between the syringe and the sample was 5 mm. Each sample was tested 10 times and the average value was reported. The contact angles were calculated by the digimizer® software.
Electrical resistance
The surface resistance of the samples was measured using a four-probe square resistance tester (FPP-SN-554). This method is based on separate pairs of current-carrying and voltage-sensing electrodes to conduct more accurate measurements, in comparison to the simpler and more usual two-point method. The current was fed through the outer contacts and the voltage was measured across the inner electrodes, as shown in Figure 3. The electrical resistivity (ρ) of the samples was obtained using the following equation [43]
Four-point measurement of resistance. Voltage sense connections are 2 and 3, and current is supplied via the connections 1 and 4.
Thermal gravimetric analysis
A Rheometric Scientific 1998 analyzer was used for thermal gravimetric analysis. The tests were performed at nitrogen atmosphere. The heating rate was 10℃/min and the temperature ranged from 50 to 700℃.
Results and discussion
Weight and thickness change
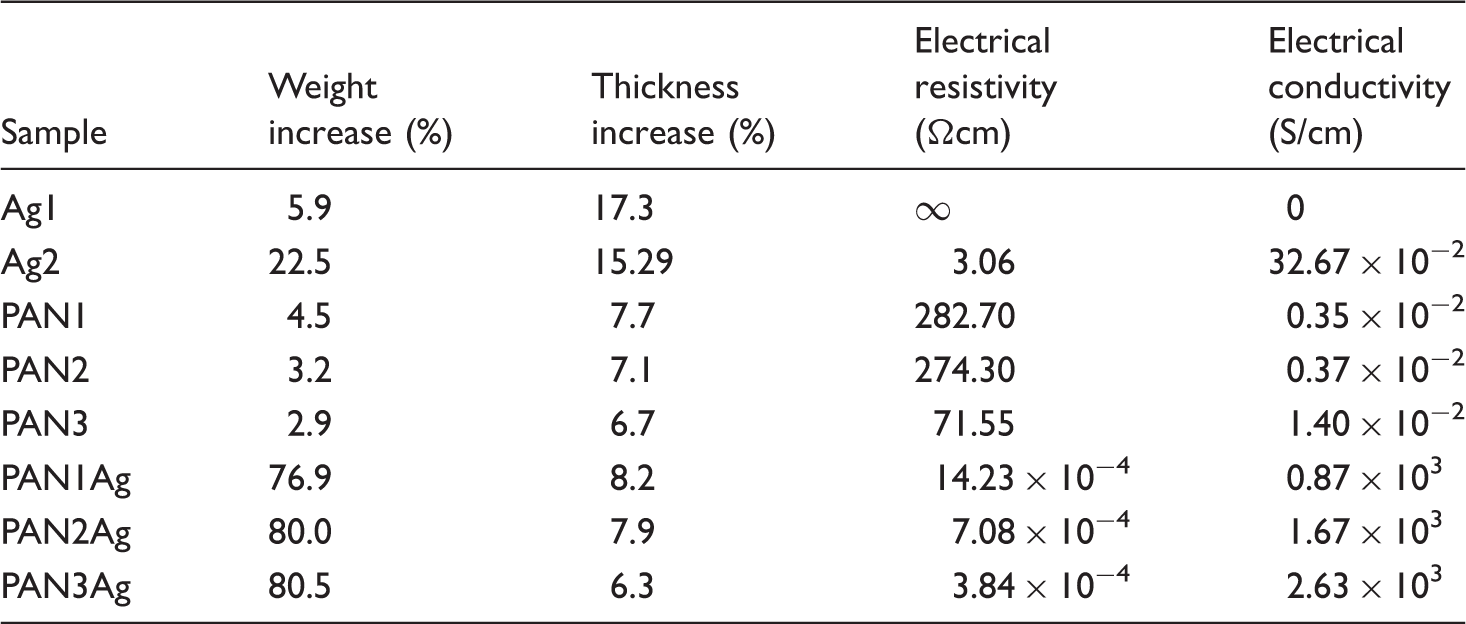
The weight increase, thickness increase, and electrical properties of the samples.
Surface morphology
The SEM micrographs of the samples are shown in Figure 4. In the samples Ag1 and Ag2, the silver particles were observed on the surface of the electroless-plated fibers. In the PAN3 sample, a uniform coating of PANI was observed on the surface of the fabric. On the surface of the PAN3Ag sample, a uniform layer of PANI/silver particles could be seen on the surface of the fabrics. The FESEM micrographs of the samples are shown in Figure 5. Granular silver particles could be seen on the surface of Ag1 and Ag2 samples. The average size of the silver particles coated on the surface of the fibers was 143 nm for Ag1 and 149 nm for Ag2. The comparison of the pictures of Ag1 and Ag2 samples with PAN3Ag illustrated that in PAN3Ag, the amount of the deposited silver on the surface of the fabrics was much higher than that observed for the other samples. The silver particles in the PAN3Ag sample were granular with the average size of about 1.3 µm. This indicated that both the amount and size of the silver granules were enhanced with Ag1 and Ag2 samples.
SEM micrographs of (a) Ag1, (b) Ag2, (c) PAN3, and (d) PAN3Ag. FESEM micrographs of Ag1 (a) (b) (c), Ag2 (d) (e) (f), and PAN3Ag (g) (h) (i) samples.

Wide angle X-ray diffraction
The wide angle X-ray diffraction (WAXD) patterns of the samples are illustrated in Figure 6. Three peaks observed at the diffraction angles (2theta) of 17.9 °, 22.56 °, and 25.8 ° were related to the crystalline structure of polyester [44]. In the sample PAN3, which was the polyester fabric coated with PANI, no other peak was observed for PANI. Therefore, it could be deduced that the PANI sample had no crystalline structure. It has been shown before that PANI could have a crystalline structure only at the very low temperatures, about −25℃, showing an amorphous structure at the higher temperatures [45]. In the diffraction patterns of the silver-coated samples (Ag1, Ag2, and PAN3Ag), five peaks at 2theta of 31.18 °, 44.37 °, 64.54 °, 77.46 °, and 81.43 ° were observed. These peaks corresponded to the X-ray diffraction from (111), (200), (220), (311) and (222) plans, respectively [33]. Table 2 presents the elemental compositions of electroless silver-plated and PANI-coated fabrics as obtained from the EDX analysis. The results confirmed the presence of silver on the surface of the electroless-plated silver fabrics (Ag1, Ag2 and PAN3Ag). PAN3Ag sample had the highest silver content (atom%) on the surface of the fibers.
WAXD patterns of the samples. EDX elemental analysis data for the samples.

Air permeability
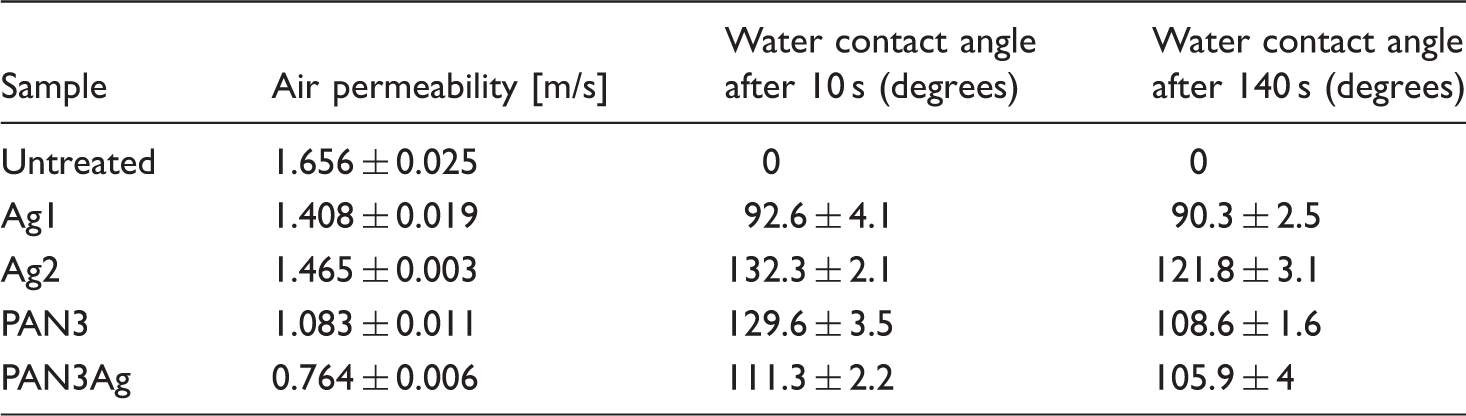
Air permeability and water contact angle of the samples.
Hydrophobicity
The results of the measurements of the contact angle for the samples are summarized in Table 3. The water contact angle of the sample Ag2 was higher than that of the others. In addition, the water contact angle of all treated samples was higher than that of the untreated sample. There was a correlation between the obtained results for the contact angles after 10 and 140 s. As the surface morphology of the sampled was investigated, it was observed that the surface of the treated samples had more roughness. This was in agreement with the previous studies, stating that a rough surface can efficiently improve the hydrophobicity of a surface [46–48]. This phenomenon can be expressed by the models developed by Wenzel [49] and Cassie–Baxter [50]. A roughened hydrophobic surface has a higher surface area in comparison to a smooth one, which increases its hydrophobicity (Wenzel model). On the other hand, air can be trapped in the roughness, which also improves its hydrophobicity since the drop then partially sits on air (the Cassie model). Therefore, it could be concluded that the hydrophobicity of the coated fabrics was higher than that of the untreated PET fabric.
Electrical conductivity
Table 1 summarizes the results of the electrical conductivity measurement of the fabrics. It could be observed that the conductivity of PAN3 was 1.4 × 10−2 Scm-1, which was higher than that observed for PAN2 and PAN1. Increasing the amount of the deposited PANI on the surface of the fabrics led to the enhancement of the conductivity of them, which was in agreement with the results of the weight increase as well as the observations of the SEM micrographs. The conductivity of the silver-plated fabric (Ag2) reached 47.75 × 10−2 Scm−1, which was much higher than that of PAN3. According to the references, silver is the most conducting metal and its conductivity is about 6.3 × 105 Scm−1 at 20℃ [24]. As described in the description of the surface morphology of the samples, the silver particles on the surface of the fibers were mostly separate (Ag2); as a consequence, the electrical conductivity of them was not high enough. The fabric Ag1 showed a very high electrical resistance (almost non-conductive) when compared with Ag1, which could be attributed to the lower content of the deposited silver particles, and hence the discontinuity of them on the surface of the fabric. On the other hand, a considerable increase in the conductivity of the samples was observed in the PANI/silver-coated fabrics (PAN1Ag, PAN2Ag and PAN3Ag). The highest conductivity value was obtained for PAN3Ag (2.63 × 103 Scm−1). The coated layer was indeed a hybrid composite of the two conducting components: PANI and silver. Therefore, its conductivity could be determined by the composition, morphology and interfacial contacts of the phases [22,23]. According to the FESEM images and TGA results, the amount of the deposited silver particles in these samples was much higher than that of Ag1 and Ag2. In other words, it seemed that in the composite PANI/silver coating, the silver content had a considerable effect on the conductivity of the composite. However, as discussed before, this increase in the silver content was because of the reducing effect of PANI [24]. This sample (PAN3Ag) with a high electrical conductivity could be, therefore, applied for many wearable conductive applications.
Thermal gravimetric analysis
It can be seen in Figure 7 that the starting temperature for the degradation of all of the samples was in the range of 450℃ to 500℃. However, the coated samples showed a higher thermal stability when compared with the untreated one; for instance, PAN3Ag began to undergo thermal decomposition at about 400℃, while the untreated fabric had an initial decomposition temperature of nearly 380℃. This observation was consistent with the results obtained by other researchers, indicating the increase in the thermal stability of the composite due to the presence of silver nanoparticles [51,52]. However, Mu et al. [33] reported no change in the starting decomposition of the neat polyester, PANI-coated polyester and silver-plated polyester fabrics. The comparison of the remaining weight percentage at 700℃ confirmed that a great amount of silver had been deposited on the surface of the PANI-coated/silver-plated fabric (PAN3Ag). At 700℃, the residual content of the untreated fabric, Ag1, Ag2, PAN3 and PAN3Ag samples was 9.1%, 15.1%, 15.3% 20% and 56.2%, respectively. Therefore, it could be concluded that the silver content of the PANI/ silver electroless-plated fabric (PAN3Ag) was much higher than that of the other ones. The conclusion might be that the attachment of PANI/silver on the surface of the polyester fabric enhanced the thermal stability of it [51]. The initial decomposition temperature of the PANI/silver-coated polyester fabric was slightly higher than that of the untreated one, implying that the PANI/silver coating caused the slight improvement in the thermal stability of the polyester fabric.
TGA thermograms of the samples.
FTIR
The FTIR spectra of the untreated and coated fabrics were obtained, as presented in Figure 8, to ensure the formation of PANI. The absorbance peak at 1705 cm−1 corresponded to the carboxyl groups of the polyester chains. The peak at 822 cm−1 was attributed to the C=C band and those at 1293 cm−1, 1236 cm−1 and 1094 cm−1 referred to the stretching vibrations of the C–N band in PANI [53]. However, the absorbance bands of PANI overlapped with the absorbed bands of polyester. In the PAN3Ag sample, the peaks at 1474 cm−1 and 1550 cm−1 were attributed to the C–N and C=N groups in the benzenoid and quinoid structures in PANI [38]. Therefore, the spectrum contained both PANI and polyester peaks, indicating that aniline was polymerized on the surface of the polyester fabric to a certain degree.
FTIR spectra of the untreated (a), Ag1 (b), Ag2 (c), PAN3Ag (d), and PAN3 (e) samples.
Conclusion
In this work, polyester fabrics were coated using three different methods: electroless silver plating, in situ polymerization of aniline, and the hybrid composite coating of PANI/silver. For the fabrication of the hybrid PANI/silver-coated fabric, the polyester fabric was first coated using the in situ polymerization of aniline; then silver electroless plating was performed. SEM images, EDX analysis, X-ray diffraction and TGA results confirmed the presence of the reduced silver metal on the surface on the fabrics. However, the content of the silver on the surface of the hybrid PANI/silver (PAN3Ag) was much higher than that of the other samples. This was because of the reduction of the silver ions to the metallic silver by PANI. The emeraldine form of PANI was oxidized to the pernigraniline form. The average size of the silver particles on the PANI/silver hybrid samples was about 1.3 µm, which was much larger than that of the silver particles on the electroless-plated samples (about 149 nm). The coated fabrics showed lower air permeability and a higher water contact angle when compared with the un-treated fabric. The electrical conductivity of the PANI/silver hybrid sample reached 2.63 × 103 Scm−1, which could be mainly attributed to the high amount of the deposited silver on the surface of this sample. The high conductivity of the hybrid PANI/silver fabrics could, therefore, make it a very good candidate for use in many smart wearable applications.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.



