Abstract
In this study, polypyrrole was deposited separately on barium titanate, barium titanate-poly (acrylonitrile-co-methylacrylate) nanocomposite-coated textile fabrics by an in-situ chemical polymerization process. Electromagnetic shielding effectiveness, electrical conductivity, chemical structure, and morphology of fabrics were fully characterized and systematically studied for investigation of individual effects of barium titanate, pyrrole polymer, and barium titanate-poly (acrylonitrile-co-methylacrylate) on obtained fabrics’ conductivity and shielding behaviour. Electromagnetic shielding effectiveness of the fabrics was determined according to the ASTM D4935-10 protocol, by using a coaxial transmission line measurement technique in the frequency range of 15–3000 MHz. Electrical characteristics were measured by the two-end method. Electromagnetic shielding effectiveness data suggested that polypyrrole-coated fabrics had better electromagnetic shielding effectiveness than polypyrrole-barium titanate and barium titanate-poly (acrylonitrile-co-methylacrylate)-coated fabrics. On the other hand, conductivity increased due to the interaction between polypyrrole and barium titanate-poly (acrylonitrile-co-methylacrylate), with fabric conductivity values also increased with the use of barium titanate. Spectroscopic characterizations of coated fabrics were determined using Fourier transform infrared spectroscopy. Analyses demonstrated that there is a strong interaction between cotton and polypyrrole and barium titanate, and also with poly (acrylonitrile-co-methylacrylate)-barium titanate. Morphological characterizations of the coated fabrics were examined by scanning electron microscopy. Colour measurements of fabric samples were performed for determination of colour intensity as a function of polymerization efficiency.
Keywords
Introduction
Recently, due to the development of nanotechnology and surface science, numerous studies have been performed for determining different properties of textile-based substrates. These substrates exhibit several benefits, such as large surface area due to the porous structure of the textile fibers, as well as mechanical flexibility and stretchability. It is well-known that textile materials can act as insulators; however, they can be modified to produce conductive textile structures, for example, by using electrically conductive materials such as inorganic fillers or intrinsically conductive polymers like polypyrroles, polyanilines, and thiophenes [1–7].
Conducting polymers represent an important class of functional organic materials for next-generation electronic and optical devices. The most remarkable property of such polymers is their inherent electrical conductivity, which is substantially connected to charge transfer rate and electrochemical redox efficiency [8,9]. Among the various conducting polymers, polypyrroles (PPys) have generated great interest due to their reaction efficiency, chemical stability, thermal stability, and high electrical conductivity (up to 100 S/cm in doped form) [7,10]. However, they have limited industrial application due to their lack of processability to form fibres or filaments. This is due to the inherent structure of these polymers. The shielding efficiency of conducting polymers as a function of their conductivity properties, thickness, and temperature has been reviewed in detail [11,12].
Blends and composites of conductive polymers have been designed with a variety of ferro-electric/magnetic particles and insulator polymers to improve electrical, magnetic, and mechanical properties of these materials [13–18]. Among various fillers, ferroelectric properties and high dielectric constant make barium titanate (BaTiO3) [16,19,20] useful in an array of applications such as optics, optoelectronics, sensors, electromagnetic shielding applications [21,2], multilayer ceramic capacitors, gate dielectrics, waveguide modulators, IR detectors, and holographic memory. At room temperature, BaTiO3 adopts a tetragonal perovskite-type structure and among the many crystallographic forms, the tetragonal phase has the best dielectric properties [12,20,21].
Electromagnetic shielding materials have been an important research area for many multidisciplinary studies during recent decades, due to the negative effects of electromagnetic radiation on human health and electronic systems [4,5]. Electromagnetic shielding properties vary, and depend on the conductivity of the shielding material. Textile materials with electromagnetic shielding are used to produce multifunctional and interactive next-generation structures in electrical and electronic industries, as well as for the production of protective garments [1,4,5,22–26].
Conductive fabrics from natural and synthetic [27–30] fibers have been elaborated by polymerizing pyrroles in situ, using ferric compounds as oxidizing agents. Recent research shows that polypyrrole can be coated to cellulose-based fibers [23–25,27,31–33]. Cotton fibers, one of the most common materials in the textile industry, are a porous and flexible material. These fibers have a hierarchical structure with high surface area, large porosity, and hydrophilic functional groups such as hydroxyl groups. This specific structure allows cotton fibers to be well wetted by water or other polar chemicals during the polymerization and coating procedure [34].
Electro-conductive textiles can be prepared by using in situ chemical, electro-chemical, and vapour phase polymerization processes. Among various processing methods, coating the textile surface is suitable for industrial scale processes, due to the simplicity and feasibility of the procedure. The coated textiles not only provide controllable electrical properties but also improve the physical properties of the textiles [1,2,35,36].
This study was designed to investigate the electrical and electromagnetic shielding properties of BaTiO3-P[AN-co MA]-PPy deposited fabrics. Such fabrics can be used as an alternative material with a polymer coating that can act as a conductive and adhesive layer, which could be used in various application areas, such as protective garments, biotechnology, electrical, and electronic industries.
Experimental
Material
Properties of cotton fabric.

Method
Preparation of BaTiO3-P(AN-co-MA) nanocomposites
An emulsion polymerization method was used to synthesize (BaTiO3-[P(AN-co-MA]) nanocomposites by free radical polymerization, by using ammonium persulphate as an oxidant in the aqueous medium. The nanocomposite was synthesized following the procedure reported in our previous study [12].
Dip coating of nanocomposite
The emulsion solution was deposited onto the cotton fabric through a dip coating process at room temperature. Cotton fabric (15 ×15 cm) was dipped in a well diluted solution of 10 mL nanocomposite emulsion (pH ∼6).
After the coating process, samples were dried (120℃, 8 minutes) in a box furnace to evaporate the remaining solvent. The wet pick-up (the weight of the liquid as a percentage of the substrates) was approximately100%.
Chemical deposition of polypyrrole onto nanocomposite-coated fabric
Polypyrrole was deposited on nanocomposite-coated textile fabric by a chemical polymerization process. In all, 100 mL aqueous surfactant solutions were prepared by dissolving 5 mL of DBSA in distilled water, and the solution was stirred (21℃, 15 minutes); 0.1 M pyrrole monomer was then added to the solution, which was then stirred vigorously (5–8℃, 15 minutes). Coated fabrics were then fully immersed into the solution, which was stirred (5–8℃, 30 minutes).
An oxidation bath was prepared by dissolving 5.20 g FeCl3, 6.08 g APS, and 5 mL DBSA in 125 mL of distilled water. The oxidation bath was cooled (5–8℃) and added drop-wise into the solution, in which the fabrics were then left (5–8℃, ∼pH=6, 45 minutes) for polymerization to develop. A 100:1 liquor ratio was used for polymerization solutions.
Cotton fabric samples with various treating conditions.

After the polymerization process, fabrics were removed from the solution. Fabric specimens were rinsed with 100 mL of distilled water to clear the excess molecules on the fabric, and finally they were dried (21℃, 24 hours) (Figure 1).
Preparation scheme of the polypyrrole-deposited nanocomposite-coated fabrics.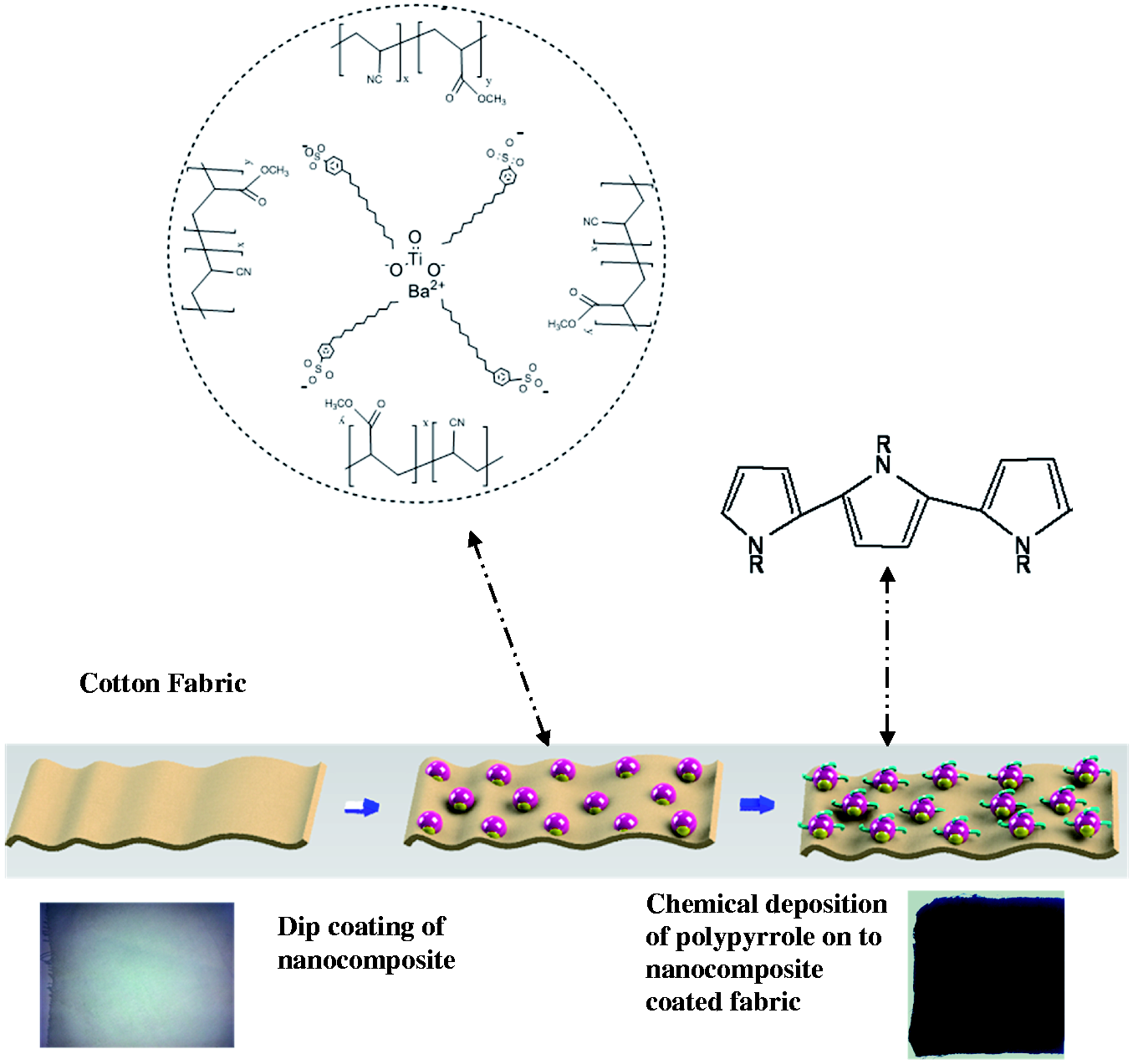
Characterization
The developed fabrics were individually tested and analyzed, in order to comprehensively understand their properties. These include surface resistivity, colour measurements, chemical functionality, surface morphologies, and electromagnetic shielding effectiveness (EMSE). Prior to testing, test specimens were conditioned (20 ± 2℃, 65 ± 2% relative humidity, 24 hours).
Surface resistivity measurements of fabric specimens were carried out in accordance with the ASTM D 257-07 standard, by using a Keithley 6517A Electrometer/High Resistance Meter instrument and a Keithley 8009 Resistivity Test Fixture.
Colour measurements of fabric samples were performed using a Datacolor 650 spectrophotometer, with illuminant D65 and 10° standards.
Fourier transform infrared (FTIR–ATR) spectrophotometric analyses of fabrics were carried out with an FT-IR reflectance spectrophotometer (Perkin Elmer, Spectrum One, using a Universal ATR attachment with a diamond and ZnSe crystal) at room temperature. Measurements were recorded at 650–4000 cm−1 at 4.0 cm−1 resolution. A background spectrum in air was obtained for each measurement.
Morphological characterizations of the fabrics were examined, and scanning electron microscope (SEM) images of considered fabric samples were obtained at various magnifications (×100 and ×5000), under an acceleration voltage of 15 keV using a Leo Supra 35 VPSEM. Fabric samples were coated with gold to a 30-nm thickness prior to SEM observations.
Fabric EMSE values were calculated in units of dB for 15–3000 MHz, in accordance with ASTM D 4935-10 standard, by using a RODHE & SCHWARZ ZVL-Network Analyzer and EM 2107A as sample holder. The measurement method is explained in the literature [4,5].
Results and discussion
Surface resistivity and thickness analysis
The thickness values of treated fabrics showed that thinner coatings were formed when using FeCl3 (Figure 2). The standard redox potential of FeCl3 (0.77) is lower than that of APS (2.00) [37,38], which suggests that the accretion or elongation process for samples oxidized by FeCl3 is much slower compared with those oxidized by APS. This is also consistent with results showing that the average diameter of coated polymers oxidized by FeCl3 was smaller than when oxidized by APS. Consequently, thickness values of coated fabrics were increased in the presence of the P(AN-co-MA)] latex, and decreased when using FeCl3.
Thickness values of fabrics after treatment.
The surface resistivity of treated fabrics is depicted in Figure 3. Results showed that conductivity values of nanocomposites were dependent on PPy, FeCl3, and BaTiO3 content (Figure 3a). The electrical surface resistivity value of PPy-BaTiO3-P(AN-co-MA)-nanocomposite-cotton with FeCl3 content decreased sharply compared to the control cotton fabric, due to the electrical conductive network consisting of PPy or PPy-BaTiO3-P(AN-co-MA) nanocomposites. Typically, the electrical resistivity of PPy cotton was higher than that of Ppy-BaTiO3-P(AN-co-MA)cotton specimens [39]. Also, the conductivity of the polymer coating oxidized by FeCl3 was higher than when oxidized by APS [38].
Surface resistivity values of fabrics after treatment. (PPy indicates Py+APS).
Whilst the conductivity of polypyrrole emulsions is of the order of 60 µS [12,21], this value is further increased for cotton coated with such an emulsion. Also, conductivity is dependent upon coating thickness (Figure 2) and PPy molar ratio (Figure 3b). Conductivity values increased with PPy and BaTiO3 content. This increase was thought to be a direct consequence of the improved compactness of the polymer layer, due to coupling between composite particles, explaining the reduced resistivity of the samples [21].
Colour measurement analysis
The colour intensity of fabric samples was investigated in order to compare the amount of PPy-BaTiO3 adhered onto the surface of the samples. Colour intensities were determined from colour strength, K/S, values, as a function of polymerization efficiency. For each case, three measurements were performed, with mean values being reported. K/S of samples was calculated using the Kubelka–Munk equation [1,27]
K/S values for control cotton fabric and treated fabrics.

The colour coordinates for control cotton fabric and treated fabrics.

FTIR–ATR spectrophotometric analysis
The FTIR-ATR spectrum of cotton fabrics coated with polypyrrole (Figure 5) exhibited typical polypyrrole peaks, as described in the literature [12,24,25,34,39]. The FTIR spectra of untreated cotton fabrics (Figure 5a) indicated the presence of a broad band at 3300–3500 cm−1 attributable to OH groups, with this absorption band disappearing from treated cotton fabric spectra. The IR spectra of treated fabric samples showed a strong absorption band at approximately 1566 cm−1, which is typically assigned to C=N and C=C groups [27,39]. Peaks at 1552 and 1471 cm−1 correspond to C=C and N—C stretching modes of the PPy ring, respectively [12,34].
(a) FTIR Spectrum of the (a–b) untreated cotton, PPy/cotton, BaTiO3-PPy/cotton, BaTiO3-P[AN-co-MA]-PPy/cotton, FeCl3 –PPy/cotton fabrics and (c) FeCl3 -BaTiO3-P[AN-co-MA]-PPy/cotton fabrics with (b) 0.1 M, (c) 0.2 M, (d) 0.3 M Py concentrations.
In the IR spectrum of BaTiO3/cotton fabric composites (Figure 5b), strong peaks at 855 and 810 cm−1 demonstrate a strong interaction between PPy-cotton and BaTiO3, associated with localization effects [12]. In addition, some other strong characteristic bands of PPy at 1536, 1450, 1156, 1000, and 700 cm−1 signify the formation of PPy-BaTiO3 on the surface of the cotton fabric. These IR spectra confirm the presence of polypyrrole polymerized or deposited onto the surface of the treated cotton fabric. In the spectra of fabrics coated with polypyrrole, BaTiO3, and latex (Figure 5a–c), the peaks are well distinguished.
Surface morphology analysis
The deposition of polypyrrole on cotton is clearly visible to the naked eye. The originally white textile becomes black after polypyrrole coating. Colour differences between samples were also determined (Table 3). SEM was used to depict the morphology of nanocomposite-treated cotton fabrics. From the low magnification version (100×) in the images of each case, the interlaced structure of warp and weft yarn can be clearly observed, with the deposited PPy- BaTiO3, BaTiO3-P(AN-co-MA)-PPy, and PPy (Figures 6 and 7). Surfaces of PPy-coated cotton fabrics differed significantly from those coated with PPy-BaTiO3 and BaTiO3-P(AN-co-MA)-PPy.
SEM images of (a) untreated cotton, (b) PPy/cotton, (c) BaTiO3-PPy/cotton, (d) BaTiO3-P[AN-co-MA]-PPy/cotton fabrics. SEM images of (a) FeCl3 –PPy/cotton, FeCl3 -BaTiO3-P[AN-co-MA]-PPy/cotton fabrics with (b) 0.1 M, (c) 0.2 M, (d) 0.3 M Py concentrations.

Considering the morphological structure of nanocomposite-coated cotton fabrics (Figures 6 and 7), granular particles dispersed uniformly onto fabric surfaces coated with BaTiO3-P(AN-co-MA)-PPy nanocomposites. Figure 6(b) reveals that uniform layers of globules or cauliflower-like pure PPy are present on the surface of cotton fibers. The addition of BaTiO3 during the polymerization of PPy resulted in the formation of a nanosheet PPy/BaTiO3 coating on the surface of the cotton fabric (Figure 6c). Presented images suggest that BaTiO3 NPs strongly impact the polymerization process of PPy, and consequently on the morphology of PPy/BaTiO3-coated cotton fabrics.
It has been reported that the aggregated construction of polypyrrole granular particles could severely weaken the electrical ability of conductive networks [39]. The high surface area of BaTiO3-P(AN-co-MA) nanosheets helps to enhance the uniform dispersion of polypyrrole nanoparticles on cotton fabric surfaces (Figure 6d). Therefore, it can be concluded that BaTiO3-P(AN-co-MA) coatings on cotton fabric surfaces can improve the fabric’s electrical conductivity.
Furthermore, the morphology of coated cotton fabrics is significantly affected by the initial Py mole ratio and BaTiO3-P(AN-co-MA) content (Figure 7). In the sample with the lowest mole ratio, the coating is not completely uniform [22]. Coatings with FeCl3 were notably thicker than other samples (Figure 7). It is well known that during the polymerization of conducting polymers, in the presence of surfactant or water-soluble polymers, thinner films are produced on available surfaces, due to additional formation of colloidal polymer in the reaction mixture [34].
As seen in SEM observations (Figure 6c and d), Ppy, BaTiO3, and BaTiO3-P(AN-co-MA) were uniformly deposited on cotton fabric surfaces through in-situ chemical polymerization of pyrrole. A suitable deposition of PPy, BaTiO3, and BaTiO3-P(AN-co-MA) onto the fabric surface is an important parameter for achieving adequate electrical conductivity and electromagnetic shielding properties of the fabrics.
Electromagnetic shielding analysis
EMSE of different samples was measured to study the effect of PPy, BaTiO3, and BaTiO3-P(AN-co-MA) coating on the fabrics’ final properties. EMSE values of coated fabrics as a function of frequency (15–3000 MHz) are given in Figure 8.
EMSE values of treated fabrics.
For uncoated cotton there was no shielding plot, due to giving zero conductivity. From Figure 8(a), it is evident that PPy-coated fabrics showed the highest EMSE performance, with 7.8 dB. This fabric also displayed better EMSE than PPy-FeCl3-coated ones. As mentioned in our previous work, PPy-BaTiO3-P(AN-co-MA) nanoparticles are conductive [21]. Additionally, conductivity results showed that FeCl3-BaTiO3-P(AN-co-MA)-PPy-coated fabrics had the highest values (Figure 3). In contrast, shielding values of these samples were lower than PPy-coated ones. This can be explained by the wave-absorbing properties and interactions of coated BaTiO3 particles and PPy on the fiber’s surface. Recent research shows that EMSE parameters of coated materials depend on the coating materials’ intrinsic conductivity and dispersion [1,19,20,21,36,42].
The EMSE of all samples exhibited an ascending tendency until the region of 1800–2100 MHz, except for PPy-BaTiO3-P(AN-co-MA)-coated fabrics, then increased from 2150–2400 MHz, followed by a sharp decrease until the region of 2700 MHz. Between 2700 and 3000 MHz, the EMSE of coated samples again displayed an increasing tendency.
Variations in the EMSE of the treated fabrics at 0.1 M, 0.2 M, and 0.3 M pyrrole concentrations are depicted in Figure 8(b). The highest shielding values were obtained at 0.3 M pyrrole concentration. The shielding value was measured as 1.8 dB at 0.1 M pyrrole concentration, and was found to be 3.9 dB at 0.3 M pyrrole concentration. The polymerization efficiency and conductivity value of 0.2 M pyrrole concentration was higher than 0.1 M; however, SEM images (Figure 7c) indicated that a coating uniformity of 0.2 M is lower than 0.1 M. Therefore, EMSE values may have been lower due to coating uniformity, although is seen in Figure 8(b), the difference between 0.1 M and 0.2 M is negligible(∼0.4 dB).
Conclusions
In this study, conductive fabrics were developed through the deposition of PPy, PPy-BaTiO3, and BaTiO3-P(AN-co-MA)-PPy onto cotton fabrics, using a chemical polymerization method. Oxidation of pyrrole with iron (III) chloride yields a conductive thin coating, compared to oxidation with APS, but EMSE was decreased with FeCl3. On the other hand, conductivity values of treated fabrics increased, and EMSE values decreased, in the presence of BaTiO3 nanoparticles. The lowest surface resistivity was determined for FeCl3 and BaTiO3-P(AN-co-MA)-PPy-coated cotton. The highest shielding value was obtained as 7.8 dB at 2000–2300 MHz in PPy-treated fabrics. It was observed from SEM images that PPy, BaTiO3, and BaTiO3-P(AN-co-MA) can be uniformly deposited onto cotton fabric surfaces under appropriate preparation conditions, through in-situ chemical polymerization of pyrrole. The surface of PPy-coated cotton fabrics significantly differed from that of BaTiO3 and BaTiO3-P(AN-co-MA)-coated fibers. The SEM images suggest that BaTiO3 NPs have a strong impact on the polymerization process of Py, and consequently on the morphology of PPy/BaTiO3-coated cotton fabrics. BaTiO3-P(AN-co-MA) coatings and Py polymerization occurrence on cotton fabrics was determined using FTIR analysis. Colour intensity analyses of different samples revealed that samples prepared with PPy, BaTiO3, latex, and FeCl3 show significant differences compared to those samples which were prepared without them. At higher concentrations of pyrrole, a better yield of conductive polymer and a better K/S were achieved.
Footnotes
Acknowledgement
The authors thank Prof. Dr Ismail Usta from Marmara University Textile Engineering Department for support and for use of the electromagnetic shielding effectiveness and conductivity facilities.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Istanbul Technical University Scientific Research Support Foundation (No.38242).
