Abstract
Objective:
To characterize simplified meal bolus strategies in adults with insulin-treated type 2 diabetes using automated insulin delivery (AID).
Research Design and Methods:
In the 2IQP study, a 13-week randomized, controlled trial comparing Control-IQ+ AID to continuation of pre-study insulin regimen with continuous glucose monitoring, 201 participants in the AID arm were classified by meal bolus strategy. Glycemic outcomes were compared to baseline.
Results:
68 participants’ meal bolus strategies (33.8%) were classified as Carbohydrate Counting, 79 (39.3%) were classified as Preset Carbohydrate Amounts, 27 (13.4%) were classified as Fixed Insulin Doses, and 27 (13.4%) as Other Methods. All bolus strategies were associated with similar, significant improvements in HbA1c from baseline: −0.9% for Carbohydrate Counting (P < 0.001), −1.1% for Preset Carbohydrate Amounts (P < 0.001), −0.8% for Fixed Insulin Doses (P < 0.001), and −0.9% for Other Methods (P = 0.003). Hypoglycemia rates were low at baseline and remained low for all bolus strategies. As participants gained experience with the Control-IQ+ AID system, more participants opted to use a simplified bolus strategy in the second half of the study compared with the first half (63% vs. 52%).
Conclusion:
Simplified bolus strategies worked well for adults with type 2 diabetes using Control-IQ+ in the 2IQP trial. All bolus strategies led to substantial HbA1c improvements, without safety concerns.
Introduction
Carbohydrate counting is a main barrier to successful use of multiple daily injections (MDI), insulin pumps, or hybrid closed-loop automated insulin delivery (AID) systems with diabetes to achieve optimal glycemic control. 1 Yet many people with type 2 diabetes (T2D) have limited experience with more sophisticated insulin dosing strategies, including carbohydrate counting. 2 In addition, individuals with T2D using MDI often report missed meal boluses, contributing to higher HbA1c and lower time in range.1,3
In the 2IQP randomized controlled trial (RCT) of 319 adults with insulin-treated T2D, we found that AID therapy using the t:slim X2™ insulin pump with Control-IQ+™ technology (AID group) reduced HbA1c levels by 0.9% from baseline, compared with a reduction of 0.3% in the group that continued their pre-study insulin delivery method with continuous glucose monitoring (CGM) group, for a mean adjusted difference of −0.6% (95% CI: −0.8% to −0.4%, P < 0.001). Time in range 70–180 mg/dL increased by 14% (95% CI: 11%–17%, P < 0.001) with AID compared with CGM, and other outcomes reflective of hyperglycemia showed similar benefit. 4
Contrary to studies of AID in people with type 1 diabetes (T1D), 75% of 2IQP participants were not adjusting their mealtime insulin doses for meal size at the time of enrollment. Therefore, we designed the 2IQP study with the intention of teaching participants simplified bolusing strategies using teaching aids specifically developed for the trial. Herein, we report on a post-hoc analysis that characterizes the meal bolus strategies used by 2IQP AID group participants and assesses associated glycemic outcomes.
Methods
The 2IQP study design has been previously described. 4 Following a 3–4-week run-in period where baseline CGM data were collected, 319 participants at 21 clinical sites were randomly assigned in a 2:1 ratio to use either the t:slim X2 insulin pump with Control-IQ+ technology (AID group) or to continue their pretrial insulin-delivery method with the Dexcom G6 CGM (CGM group). The primary outcome was the change in HbA1c from baseline to 13 weeks, measured at a central lab. At baseline, participants were asked if they were adjusting their mealtime insulin doses for meal size prior to the study. Participant comfort level with mathematical concepts that might be important for carbohydrate counting was assessed with the Subjective Numeracy Scale. 5
AID group participants completed meal challenges (full bolus, half bolus, and no bolus) at lunchtime or dinnertime on three consecutive days during the trial with the same carbohydrate content (≥50 g) for each meal to assess safety for missed meal boluses. Meal challenges were not standardized to provide flexibility for carbohydrate or other macronutrient content, and carbohydrate quality, across participants. However, the same meal was consumed three times by each participant for consistency. Approximately 25% of challenges were performed in each of the following four periods from randomization to avoid a period effect: 1–2 weeks, 3–4 weeks, 5–6 weeks, and 7–8 weeks. Study staff ensured correct bolus entry into the study pump for each full bolus and half bolus challenge.
To facilitate participant training in the study, clinical sites were given streamlined participant educational materials created by the study sponsor, Tandem Diabetes Care. The materials were offered by the clinical sites to participants based on investigator discretion and participant preference, as per their assessment of how the participants could best use them.
The materials included general overview information on pump therapy and carbohydrates and two documents on meal bolus strategies: “Managing Mealtime Insulin with Set Carbs” describing entry of preset carbohydrate amounts (e.g., 30 g for small, 60 g for medium, and 90 g for large meals); and “Managing Mealtime Insulin with Set Units,” describing direct entry of units of insulin into the pump for different meal sizes, skipping over carbohydrate entry entirely. Updated copies of the materials used in the trial have been posted by Tandem Diabetes Care at https://www.tandemdiabetes.com/providers/resources. Beyond providing general guidance to participants, the study did not monitor how providers configured pump settings or how often patients redefined their small, medium, or large meal sizes. Site investigators were directed to modify insulin delivery profiles only for safety concerns, aligning with real-world practice in which adjustments to pump settings are less frequent for people with T2D over a 13-week period.
For this analysis, participants with at least 21 days of Control-IQ+ technology use were evaluated using the data from user-initiated boluses throughout the 13-week study period. The t:slim X2 insulin pump with Control-IQ+ technology and the t:slim mobile app allow entry of either grams of carbohydrate or units of insulin for mealtime boluses on the bolus calculator screen (Supplementary Fig. S1). Using the criteria shown in Table 1, each participant was classified into one of four bolus strategies: Carbohydrate Counting, Preset Carbohydrate Amounts, Fixed Insulin Doses, or Other Methods.
Classification of Meal Bolus Strategies Used in the Trial Among AID Participants

Also commonly referred to as simplified dosing or small/medium/large.
Statistical Analysis
The cohort used in this analysis included participants randomized to the AID group who used Control-IQ+ AID on at least 21 days over the 13-week follow-up period. The participant’s bolus strategy was determined based on all available study pump data.
Paired t-tests were used to evaluate the changes in HbA1c and CGM metrics from baseline to 13 weeks separately within each bolus strategy. Insulin use, change in weight, and responses to patient-reported outcome (PRO) surveys were also analyzed. In addition, to evaluate if the changes in these outcomes differed by bolus strategy, a linear model was fit with change as the outcome, bolus strategy as the covariate, and adjusted for the baseline value of the metric. Skewed outcomes were winsorized at the 10th and 90th percentiles. For winsorized outcomes, a bootstrap was used to obtain confidence intervals for change within each bolus strategy, and a permutation test was used to evaluate if the change differed by strategy. A similar analysis was performed to evaluate HbA1c and CGM metrics by groups defined by the number of user-initiated boluses per day.
In addition, the following were tabulated by bolus strategy: the percentage of bolus events and percentage of bolus units over the 13-week follow-up period that were automated vs. user-initiated, and CGM metrics calculated over the study meal challenges.
P values and confidence intervals were adjusted to control the false discovery rate. Analysis was performed using SAS version 9.4.
Results
Among the 215 participants in the AID arm of the trial, sufficient data were available to classify 201 into one of the meal bolus strategy categories: 68 (33.8%) were classified as Carbohydrate Counting, 79 (39.3%) as Preset Carbohydrate Amounts, 27 (13.4%) as Fixed Insulin Doses, and 27 (13.4%) as Other Methods. The Other Methods category included participants mostly bolusing in units, using many different values for entry.
Baseline characteristics of the 201 participants are shown in Table 2. Seventy-four percent of participants reported they were not adjusting their mealtime insulin doses for meal size prior to the study, meaning they had not been dosing insulin based on carbohydrate amounts. The mean subjective numeracy score at screening was 4.7 (on a 6-point scale) for those classified during the study as Carbohydrate Counting, 4.3 for those who used Preset Carbohydrate Amounts, 4.4 for those who used Fixed Insulin Doses, and 4.5 for Other Methods, suggesting that participants who opted for simplified strategies had a slightly lower comfort level with mathematical concepts.
Participant Characteristics at Baseline
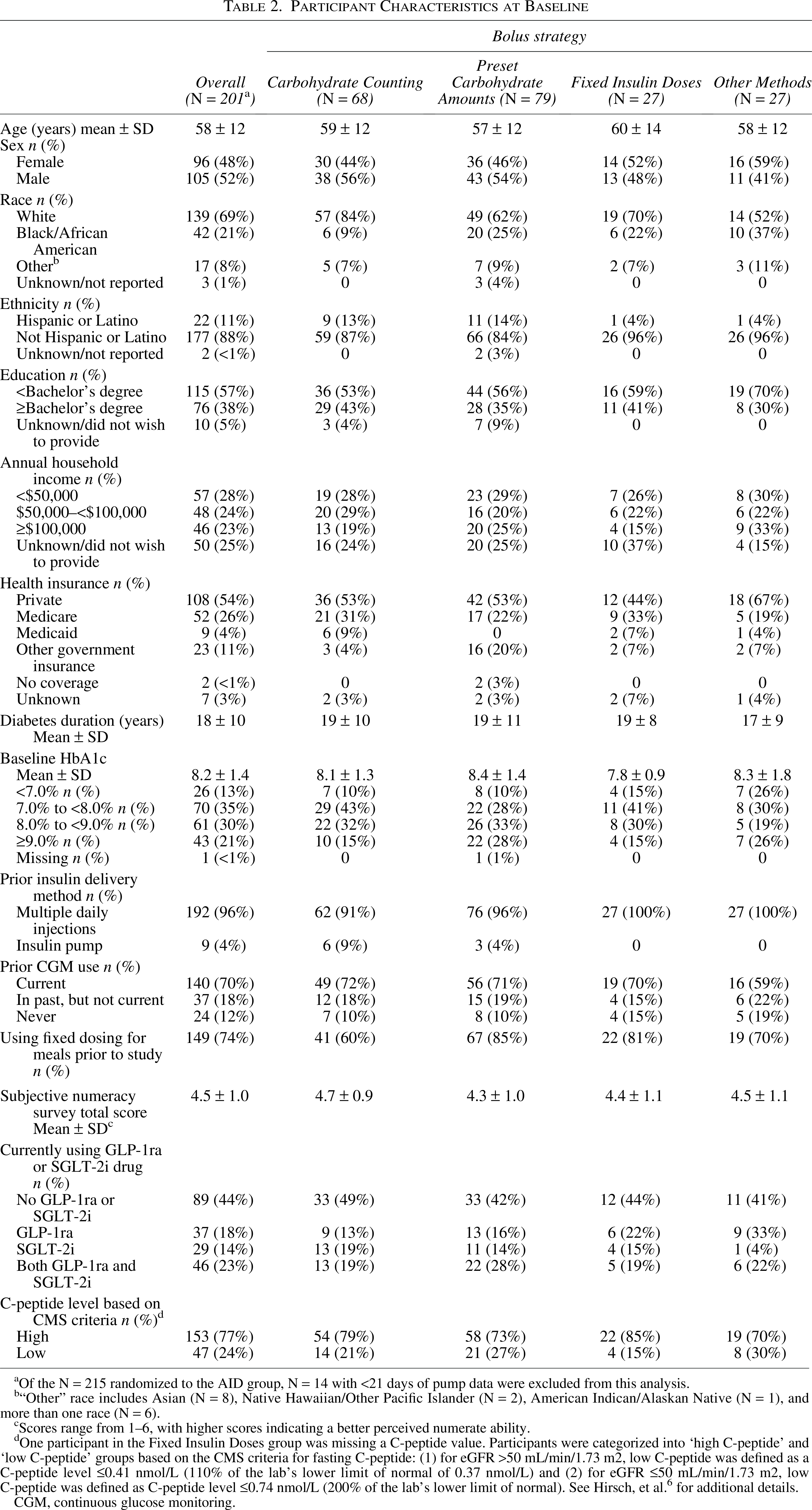
Of the N = 215 randomized to the AID group, N = 14 with <21 days of pump data were excluded from this analysis.
“Other” race includes Asian (N = 8), Native Hawaiian/Other Pacific Islander (N = 2), American Indican/Alaskan Native (N = 1), and more than one race (N = 6).
Scores range from 1–6, with higher scores indicating a better perceived numerate ability.
One participant in the Fixed Insulin Doses group was missing a C-peptide value. Participants were categorized into ‘high C-peptide’ and ‘low C-peptide’ groups based on the CMS criteria for fasting C-peptide: (1) for eGFR >50 mL/min/1.73 m2, low C-peptide was defined as a C-peptide level ≤0.41 nmol/L (110% of the lab’s lower limit of normal of 0.37 nmol/L) and (2) for eGFR ≤50 mL/min/1.73 m2, low C-peptide was defined as C-peptide level ≤0.74 nmol/L (200% of the lab’s lower limit of normal). See Hirsch, et al. 6 for additional details.
CGM, continuous glucose monitoring.
All bolus strategies led to significant improvements in HbA1c from baseline with Control-IQ+ use: −0.9% for Carbohydrate Counting (P < 0.001), −1.1% for Preset Carbohydrate Amounts (P < 0.001), −0.8% for Fixed Insulin Doses (P < 0.001), and −0.9% for Other Methods (P = 0.003). HbA1c improvement was similar among the 4 groups (P value across groups = 0.77) (Fig. 1 and Table 3).
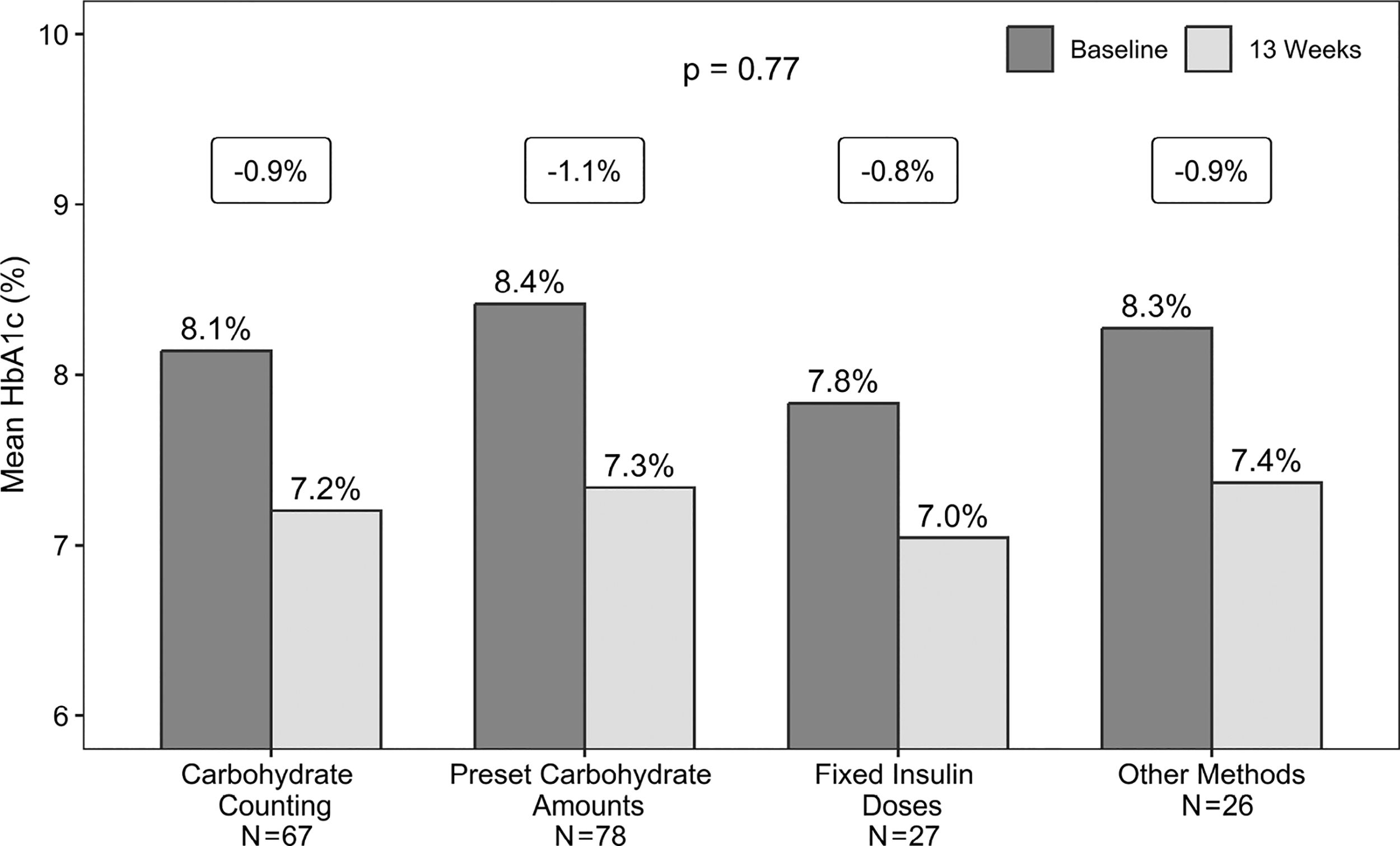
Change in HbA1c from baseline to 13 weeks, by bolus strategy. The numbers above each bar are the mean HbA1c. The numbers in the boxes above each pair of bars are the mean change from baseline to 13 weeks. The P value represents the comparison of the change across bolus strategies.
Glycemic Outcomes by Bolus Strategy
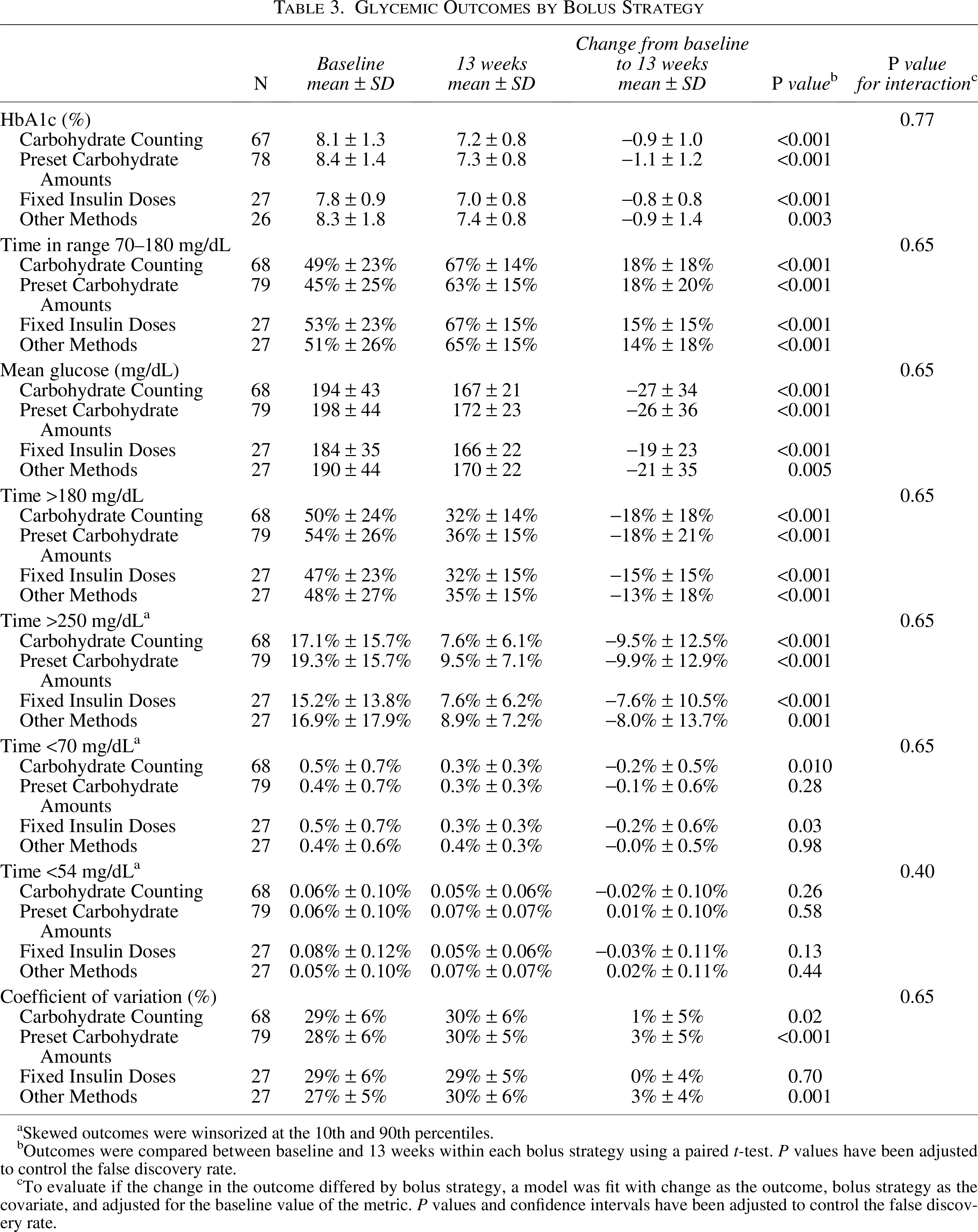
Skewed outcomes were winsorized at the 10th and 90th percentiles.
Outcomes were compared between baseline and 13 weeks within each bolus strategy using a paired t-test. P values have been adjusted to control the false discovery rate.
To evaluate if the change in the outcome differed by bolus strategy, a model was fit with change as the outcome, bolus strategy as the covariate, and adjusted for the baseline value of the metric. P values and confidence intervals have been adjusted to control the false discovery rate.
Hypoglycemia rates were low at baseline and remained low for all bolus strategies (Supplementary Fig. S2 and Table 3), showing that using a simplified bolus strategy did not increase hypoglycemia rates.
Participants were also characterized by the bolus strategy used at different times in the study. As participants gained experience with the Control-IQ+ AID system, more participants opted to use a simplified bolus strategy in the second half of the study compared to the first half (63% vs 52%) (Supplementary Fig. S3). For the 22 participants who shifted from Carbohydrate Counting in the first half of the study to a simplified strategy (Preset Carbohydrate Amounts or Fixed Insulin Doses) in the second half, time-in-range 70–180 mg/dL was essentially the same comparing the first half to the second half of the study (mean difference = −0.7% ± 13.8%).
Regardless of bolus strategy, participants in each bolus category averaged over 50% of their bolus events given as automated boluses. However, the total amount of insulin delivered via auto-boluses was modest, representing ∼10% of the total daily dose (Supplementary Table S1). Differences between baseline and 13-week HbA1c and CGM metrics were similar regardless of the frequency of user-initiated boluses during the study period (Supplementary Table S2).
Each participant had their glycemic responses evaluated while using Control-IQ+ during the three meal challenges. Post-meal differences in time above range between a full meal bolus and half-meal bolus were modest (Supplementary Table S3).
Total daily insulin use was lower than baseline for all bolus strategies (Supplementary Table S4). There were no differences in weight change or PRO results with the different bolus strategies (Supplementary Tables S5, S6, Table and S7).
Discussion
The 2IQP study enrolled a diverse cohort of adults with T2D, spanning the range of socio-economic status and approaches to meal bolusing. 4 Prior to entering the trial, only a quarter of the study participants adjusted their mealtime insulin bolus for meal size, and more than half of the participants had a highest education level less than a bachelor’s degree, indicating the study included a broad population of individuals who derived benefit from system use. 7 The main trial results showed that the benefit of Control-IQ+ compared with the control group was not influenced by participants’ pre-study approach to insulin bolusing or their score on a survey assessing their numeracy capability.
We have now taken these analyses one step further by evaluating the effect of different meal bolus strategies used with Control-IQ+ on glycemic outcomes. Our results show that simplified bolus strategies worked well for adults with T2D using Control-IQ+, with all bolus strategies leading to substantial improvement in HbA1c, TIR, and other CGM metrics reflective of hyperglycemia; as was seen during the formal meal challenges, even a partial premeal insulin dose was effective. Importantly, there was almost no hypoglycemia regardless of the bolus strategy used, suggesting the automation in Control-IQ+ technology was effective in managing postprandial hyperglycemia.
We believe there were two primary factors that explain these results. First, the broad array of options for bolusing provided the opportunity for a customized approach to training based on a participant’s prior experience, participant preference, and the site’s assessment of what approach would work best for the participant. There was also the opportunity to change approaches as desired while the study progressed. Over time, more users throughout the study moved toward a simplified bolus strategy, suggesting they gained experience with the system’s ability to administer auto-boluses to support their glycemic control, when needed, around meals. Second, the Autobolus aspect of the Control-IQ+ algorithm can compensate for certain approaches to bolusing that may not be optimally effective. With the Control-IQ+ algorithm, in addition to every 5-minute automated basal rate modulation that can substantially raise basal rate, the system can deliver an Autobolus up to once an hour with a calculated correction down to a target of 110 mg/dL.
Some prior studies of AID systems in T1D have shown that individuals performing carbohydrate counting achieved higher time-in-range than using simplified meal announcement, including an RCT using a non-commercial system with basal rate modulation only 8 and an RCT using the MiniMed 780G system with basal rate modulation plus auto-bolusing. 9 In contrast, several recent T1D studies have shown no meaningful differences in postprandial time in range with simplified meal announcement versus carbohydrate counting, including a retrospective real-life study of the Diabeloop DBLG1 system with basal rate modulation plus automated boluses 10 and an RCT using the CamAPS FX system with basal rate modulation only. 11
A prior meta-analysis of studies using Control-IQ in T1D showed Autoboluses had a considerable impact on improving HbA1c levels, where high baseline HbA1c levels likely reflected missed meal boluses and lack of correction boluses. 12 While our study cannot confirm the impact of the Autoboluses due to the size of the subgroups, differences in the counterregulatory response in people with T2D 13 may allow for a safer response to the multiple Autoboluses Control-IQ+ can deliver to correct hyperglycemia. Therefore, we hypothesize that the Control-IQ+ Autoboluses may have contributed to the improvements in glycemic control for some participants.
There are limited data available on the use of different bolus strategies from other AID systems in larger T2D trials. The SECURE-T2D single-arm trial enrolled a population of adults with T2D, similar to the 2IQP cohort with the majority using some form of fixed dosing for meal boluses prior to the study. 14 However, different categories reported for bolus strategies in the SECURE-T2D trial make it challenging to compare results. 15 Glycemic improvement with AID in adults with T2D has also been demonstrated in the IMPACT2D single-arm trial, but limited details are available regarding user strategies for meal dosing from that trial since all participants in IMPACT2D entered carbohydrates before meals. 16
Strengths of this analysis include a diverse study population that largely lacked experience with carbohydrate counting and had varying comfort with numeracy, as well as the focused and flexible training designed not to overwhelm the participants. The main limitation is that this analysis was retrospective rather than being part of an RCT specifically designed to evaluate the efficacy of different meal bolus strategies.
In conclusion, for people with T2D, the glycemic benefits of Control-IQ+ AID are largely independent of meal bolus strategy, with low risk of hypoglycemia regardless of what bolus strategy was used. The results show that simplified bolus strategies can be used safely and effectively, which may reduce user burden and reduce staff time needed for training. Providers should strongly consider these findings when addressing strategies to optimize glycemic control and reduce care burden for people with T2D. Having options and flexibility for meal bolus strategies may increase uptake of AID in T2D.
Authors’ Contributions
C.J.L.: Investigation, writing-original draft, review and editing. L.K.: Formal analysis. S.A.B.: Investigation, writing—review and editing. S.M.: Investigation, writing—review and editing. T.S.: Investigation, writing—review and editing. D.W.S.: Investigation, writing—review and editing. V.S.L.: Writing—review and editing. P.Z.: Writing — review and editing. J.W.L.: Project administration, writing—review and editing. R.W.B.: Supervision, writing—original draft, review and editing. J.E.P.: Funding acquisition, writing—original draft, review and editing.
Footnotes
Author Disclosure Statement
C.J.L. reports grant support from the National Institutes of Health, The Leona M. and Harry B. Helmsley Foundation, Abbott Diabetes, Dexcom, Insulet, Tandem Diabetes Care, Mannkind, DEKA/Sequel, and serving on advisory boards for Dexcom, Mannkind, and Tandem Diabetes Care. L.K. reports no financial disclosures. S.A.B. reports grant support to her institution from the National Institutes of Health, Dexcom, Insulet, Roche, Tandem Diabetes Care, Tolerion and has served on a data safety monitoring board for MannKind. S.M. reports grant support to her institution from Tandem Diabetes Care. T.S. reports grant support from Eli Lilly and Novo Nordisk, and speaking fees from Sanofi. D.W.S. reports grant support from the National Institutes of Health, Abbott Diabetes Care, Insulet, Tandem Diabetes Care, DEKA/Sequel Med Tech, Novo Nordisk and consultancy payments from Abbott Diabetes Care. V.S.L, P.Z., and J.E.P. are employees and shareholders of Tandem Diabetes Care. J.W.L. reports no financial disclosures. R.W.B. reports no personal financial disclosures but reports that his institution has received funding on his behalf as follows: grant funding, study supplies, and consulting fees from Insulet, Tandem Diabetes Care, and Beta Bionics; grant funding and study supplies from Dexcom and Abbott; grant funding from Bigfoot Biomedical, Sequel Med Tech, and MannKind; study supplies from Medtronic; consulting fees and study supplies from Novo Nordisk; consulting fees from Vertex, Hagar, DreaMed, Ypsomed, Abata Therapeutics, Eli Lilly and Zucara.
Funding Information
Trial funding and automated insulin delivery systems were provided by Tandem Diabetes Care, which provided comments on the article but did not have approval authority. Insulin aspart (NovoLog®) was provided in kind by Novo Nordisk A/S. Continuous glucose monitor sensors and transmitters were purchased from Dexcom, Inc. at a discounted price.
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
