Abstract
Little is known about the acceptability of wearing physical activity-monitoring devices. This study aimed to examine the compliance, comfort, incidence of adverse side effects, and usability when wearing the SenseWear Armband (SWA) for daily physical activity assessment. In a prospective study, 314 participants (252 people with COPD, 36 people with a dust-related respiratory disease and 26 healthy age-matched people) completed a purpose-designed questionnaire following a 7-day period of wearing the SWA. Compliance, comfort levels during the day and night, adverse side effects and ease of using the device were recorded. Non-compliance with wearing the SWA over 7 days was 8%. The main reasons for removing the device were adverse side effects and discomfort. The SWA comfort level during the day was rated by 11% of participants as uncomfortable/very uncomfortable, with higher levels of discomfort reported during the night (16%). Nearly half of the participants (46%) experienced at least one adverse skin irritation side effect from wearing the SWA including itchiness, skin irritation and rashes, and/or bruising. Compliance with wearing the SWA for measurement of daily physical activity was found to be good, despite reports of discomfort and a high incidence of adverse side effects.
Keywords
Introduction
Physical activity is defined as any bodily movement produced by skeletal muscles, which results in energy expenditure. 1 A myriad of methods have been used to assess physical activity and relatively strong associations have been found between physical activity and health. 2,3 Assessment of physical activity in people with COPD has been widely used to describe levels of physical inactivity 4,5 and, increasingly, to measure the effect of interventions to promote increased physical activity. 6 –9 People with COPD have lower levels of daily physical activity than healthy people, 4,10 –13 and this is related to an increased risk of hospitalization 14 –16 and mortality. 14,17,18 Physical activity levels have also been shown to be reduced in people with a dust-related respiratory disease compared to healthy people. 19
Accelerometers are a common method of measuring physical activity in daily living by measuring acceleration in one plane (uniaxial) or in two or three planes (biaxial or triaxial). These devices provide a valid and reliable objective measurement of physical activity intensity 20,21 and have been shown to be a reproducible and valid measure of energy expenditure in people with COPD in the free-living environment. 22,23 There are many different makes and models of accelerometers available for daily physical activity monitoring, and each varies in data storage capacity, size, shape, weight, and body placement location. 24
The SenseWear Armband (SWA; Bodymedia, Pittsburgh, Pennsylvania, USA) is a lightweight, metabolic monitor that uses a biaxial (Pro3 model) or triaxial (Model MF) accelerometer and non-invasive physiological sensors, measuring galvanic skin response, heat flux and skin temperature, to estimate energy expenditure. The galvanic skin response sensor consists of two hypoallergenic stainless steel electrodes that sit on the posterior aspect of the device and lie in direct contact with the skin. The SWA is small and lightweight, and it is easily applied to the upper arm, making it convenient for physical activity measurement in the free-living environment. The SWA is widely used in clinical practice and research studies and has been shown to be a valid and reliable measure of energy expenditure and physical activity in people with COPD 23,25 –27 and healthy people. 28 –30
The respresentativeness of the physical activity data obtained from any physical activity-monitoring device is reliant on the compliance of the person wearing the device. Compliance is required to ensure the device is worn when required and is correctly applied and removed. There is limited reporting of non-compliance of wearing physical activity monitors in older people. In a study of elderly people, 20% of the subjects did not comply with wearing an accelerometer. 31 A similar result was reported in a study of people with COPD where 19% of the participants’ data was excluded from the study due to non-compliance or technical issues. 4
The usability of multiple physical activity-monitoring devices has previously been reported in people with COPD. In a study of six different physical activity-monitoring devices available on the market, the SWA was rated by people with COPD as the least favourable for comfort and ease of use and was reported as the most bothersome device. 32 In healthy people, the physical feel of the SWA on the body was reported to be uncomfortable. 33 However, neither study reported the incidence of adverse events when wearing the physical activity monitors and no studies have reported on comfort, usability or adverse side effects in people with a dust-related respiratory disease. Furthermore, no studies have examined the effect of obesity on adverse side effects when wearing the SWA. People with a higher body mass index (BMI) have a larger upper arm circumference 34 which may adversely affect comfort, usability and incidence of adverse side effects when wearing the SWA. Therefore, the primary aim of this study was to examine the compliance, comfort, incidence of adverse side effects and usability when wearing the SWA for daily free-living physical activity assessment in people with COPD, people with a dust-related respiratory disease and healthy people. The secondary aim was to examine whether obesity affected the compliance, comfort, incidence of adverse side effects and usability when wearing the SWA for daily free-living physical activity assessment in people with COPD, people with a dust-related respiratory disease and healthy people.
Methods
Background and participants
This study was performed prospectively in two Australian cities. People participating in this study were enrolled in one of a number of research studies being conducted in Australia where measurement of daily free-living physical activity was performed. 35 –38 Participants were people diagnosed with either COPD or a dust-related respiratory disease and healthy age-matched people. The people with COPD were recruited either from a respiratory ward during a hospital admission for an acute exacerbation of COPD or following referral to outpatient pulmonary rehabilitation. People with a dust-related respiratory disease were recruited following referral to outpatient pulmonary rehabilitation. The people with COPD were included if they had a diagnosis of COPD according to GOLD criteria (forced expiratory volume in 1 second/forced vital capacity <70%). The people with a dust-related respiratory disease were included if they had a diagnosis of non-malignant dust-related pleural or interstitial respiratory disease including asbestosis, silicosis and asbestos-related pleural disease (defined as diffuse pleural thickening and/or rounded atelectasis) which was diagnosed based on occupational history, clinical examination by a physician, radiological findings on chest X-ray and computed tomography scans confirming dust-related pleural and/or interstitial disease and lung function (spirometry and lung volumes via plethysmography). The healthy people were recruited following advertisement in the local community. BMI was calculated for all participants, with a ‘lower BMI’ classified as <32 kg/m2 and a ‘higher BMI’ classified as ≥32 kg/m2. Ethics approval was obtained and written informed consent was provided by participants.
Measurement of free-living physical activity
Free-living daily physical activity was measured using either the SenseWear Armband Pro3 model or the SenseWear Armband Model MF (SenseWear, BodyMedia, Pittsburgh, PA, USA). Participants were requested to wear the SWA on their upper arm 24 h a day, with the exception of swimming or showering/bathing, for a period of seven consecutive days. Participants were instructed by an experienced physiotherapist on how to apply and remove the device and a written instruction sheet was provided.
Study procedure
At the end of the wear period, participants completed a short purpose-designed questionnaire. The questionnaire was developed by one of the researchers and reviewed by a further two researchers for clarity and completeness. To determine the ease of reading and comprehension, a pilot group of four people with COPD completed the questionnaire, with no changes required. The Flesch reading ease score was 78 on a 100-point scale, indicating that the text was easy to read. 39 The questionnaire consisted of five questions with a combination of both closed and open-ended responses (Appendix 1). The first question asked the participant to indicate whether they were compliant wearing the SWA for the complete data collection period as instructed. Compliance was defined as wearing the device for the duration of the data collection period with the participant not removing the device and ceasing to wear it any longer. If they answered ‘no’, they were asked to give their reasons for non-compliance. Level of comfort wearing the SWA during both the day and night was rated on a 5-point Likert-type scale with responses ranging from ‘very comfortable’ to ‘very uncomfortable’. Participants were asked to report whether they suffered from any adverse side effects whilst wearing the device using both an open-ended response and four predetermined responses (itchiness, skin redness/rash, broken skin/wound/infection and bruising). Level of ease of applying and removing the SWA (usability) was also rated on a five-point Likert-type scale with responses ranging from ‘very easy’ to ‘very hard’. The final question asked the participants whether they referred to the instructions provided to them and whether the instructions were helpful using a ‘yes’/‘no’ response.
Data analysis
Data were analysed using SPSS Statistics 17.0 (SPSS Inc., Chicago, IL, USA). Descriptive statistics were used to summarize participant characteristics and outcome data. Independent samples
Results
There were a total of 314 participants who wore the SWA and completed the questionnaire (252 participants with COPD, 36 participants with a dust-related respiratory disease and 26 healthy participants). The SWA Pro3 model was worn by 69% (
Baseline characteristics of study participants.a
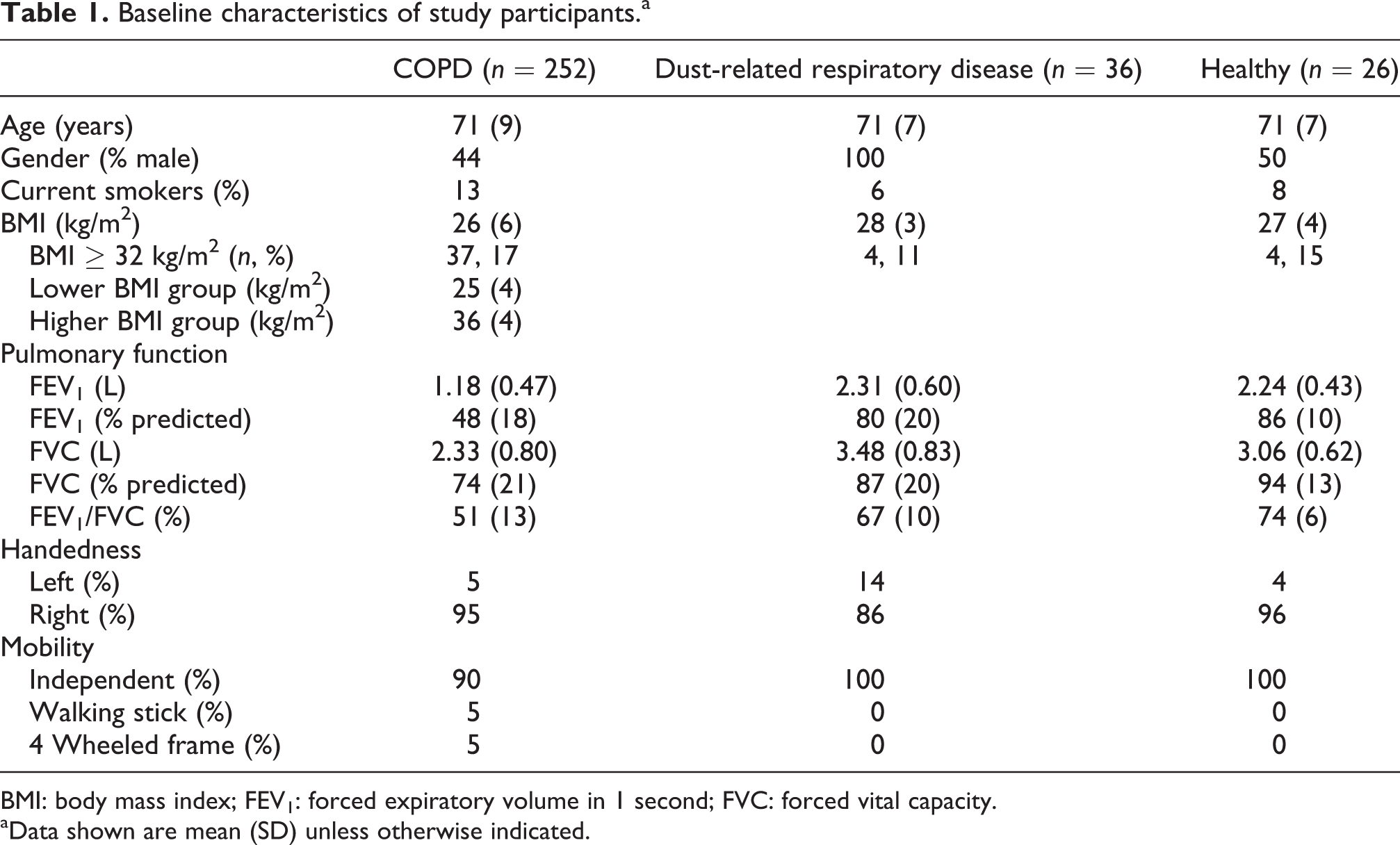
BMI: body mass index; FEV1: forced expiratory volume in 1 second; FVC: forced vital capacity.
aData shown are mean (SD) unless otherwise indicated.
Compliance
According to the questionnaire responses, the SWA was worn for the complete data collection period by 92% of all participants (Table 2). There was 100% agreement between this self-report response and the data captured by the SWA. There was no significant difference in compliance between participants with COPD who were recruited from a respiratory ward during a hospital admission for an acute exacerbation of COPD or following referral to outpatient pulmonary rehabilitation (
Questionnaire results.
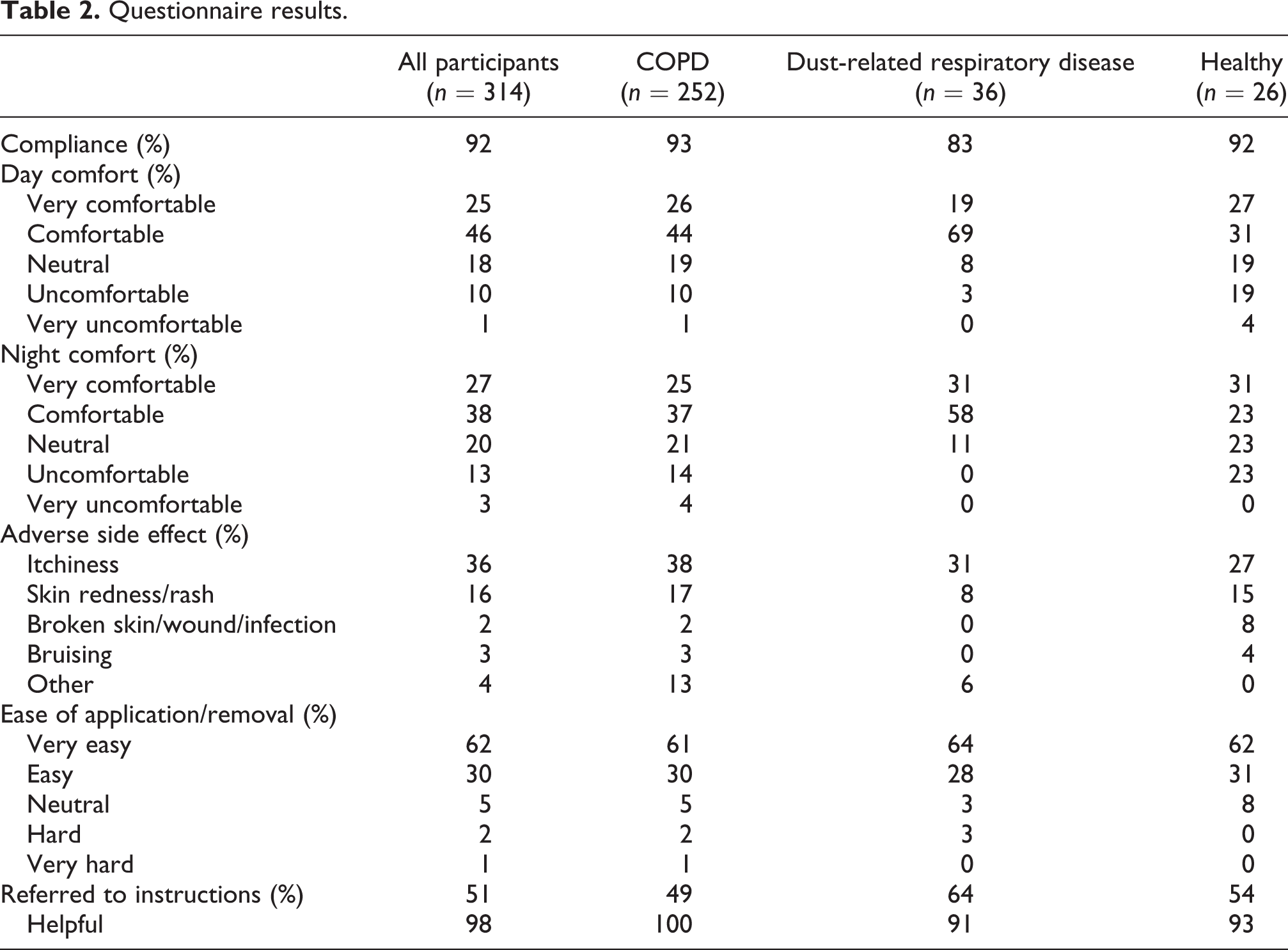
Questionnaire results for COPD participants according to BMI group and SWA model.

a
b
c
d
Comfort
The comfort level ratings for day and night for all participants are presented in Figure 1. There was a significant difference for all participants for rating the SWA being more ‘comfortable’ during the day compared to night (

Comfort level of SenseWear Armband during the day and night for all participants. *
Adverse side effects
There was a high incidence of adverse events related to wearing the armband. At least one adverse event was reported by 46% of all participants (

Incidence of adverse side effects from wearing the SenseWear Armband in all participants.
Usability
Rating of ease of application and removal of the SWA was high (Table 2). Eight participants (3%) reported the application and removal of the SWA to be ‘hard’ or ‘very hard’ (of which four participants reported that they did not read or refer to the instructions information sheet;
Discussion
To our knowledge, this is the first study to examine the compliance, comfort, usability and incidence of adverse side effects when wearing the SWA for daily free-living physical activity assessment in a large group of participants with COPD, participants with a dust-related respiratory disease and healthy participants. Good compliance with wearing the device over a 7-day period was found, despite the high incidence of adverse side effects such as itchiness, skin irritation and bruising. No significant difference was found between participants with COPD with a higher BMI compared to those with a lower BMI for any of the outcomes. Furthermore, no significant differences were found for any outcomes between the Pro3 model and the Model MF of the SWA.
Compliance in wearing the SWA over a 7-day period was very high in this study (92%) which concurs with another study examining adherence with the use of physical activity monitors. 32 The feasibility of physical activity monitoring relies heavily on the compliance and encounter of the participant with the physical activity monitor. Despite two or more adverse side effects being experienced by 15% of participants, this did not necessarily lead to discontinuation of wearing the device. It appears additional factors aside from the occurrence of adverse side events may have led to participants deciding to remove the SWA before the end of the wear period. Future studies could investigate whether factors such as arm circumference, mental health issues or mood, device size and weight or length of wear time lead to non-compliance. These issues need to be examined and addressed, if indicated, in future studies to further enhance compliance.
The SWA was reported by 17% of participants in this study to be uncomfortable to wear at night and 11% of participants reported the SWA to be uncomfortable to wear during the day. There was no difference in comfort levels between the Pro3 model and the Model MF of the SWA. Only one previous study was found which examined the participants’ comfort when wearing a physical activity monitor in people with COPD. 40 In this study, 6 out of 11 participants participating in a focus group reported that the physical activity-monitoring device worn (GT3X+) was uncomfortable due to its placement around the waist and the pressure of the elastic securing the device. 40 ‘Scratching’ and ‘burning’ were words used to describe the unpleasant sensations experienced when wearing the device, and the participants provided suggestions to improve the design of the device for greater comfort which included using a softer material for the strap and changing the size and dimension of the device. 40 To the best of our knowledge, the present study provides the first insights into the adverse side effects of wearing a physical activity-monitoring device around the upper arm. The findings of our study should be considered for the future design and redesign of physical activity monitors worn around the upper arm and may also be considered for devices worn around the wrist.
Participants were asked to wear the SWA for 7 days. The appropriate wear time of physical activity-monitoring devices to produce the most reliable results reported in the literature is variable. In healthy adults, studies have reported wear time from 3 to 7 days. 41,42 In people with COPD, evidence has emerged that appears to be similar, ranging from 3 to 6 days. 13,22,43,44 In one study wearing a physical activity monitor for 7 days was reported by participants to be too many days with the optimal duration suggested by participants in this study to be 3 days. 40 However, willingness of participants to wear physical activity-monitoring devices for more than a week was reported in another recent study. 32 Potentially, wearing the SWA in the present study for fewer days may have led to a different result in terms of comfort and adverse side effects. Future studies using the SWA should carefully consider the number of days required for physical activity monitoring to ensure the device is not overly burdensome or causing discomfort and/or the incidence of adverse side effects for participants.
A limitation of this study was the use of a purpose-designed questionnaire that had not been validated. Although open-ended responses in the questionnaire were used to enable participants to give a greater amount of detail than the closed-ended options, an alternative or additional data collection method such as using a qualitative interview format may have better elicited reasons for non-compliance. An interview may also have provided more in-depth feedback from the participants on the experience of wearing the SWA and the specific component/s of the device causing the adverse side effect/s, and it would have allowed the participants to give their opinion on the design of future models of the SWA. BMI was used as a measure of obesity, however, potentially a more accurate measure that might influence compliance, comfort, adverse side effects and usability of the SWA is arm circumference, which was not measured in this study.
The results from this self-reported study examining compliance, comfort, adverse side effects and usability support the use of the SWA for monitoring daily free-living physical activity in participants with COPD, participants with a dust-related respiratory disease and healthy participants, despite a high incidence of adverse side effects. The provision of written instructions on the physical activity-monitoring device is beneficial for participants. The ability to compare reports on comfort, usability and incidence of adverse side effects between different physical activity monitors in the future will help guide researchers to ensure their chosen physical activity-monitoring device is well accepted by participants.
Footnotes
Appendix 1
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding to purchase the SenseWear Armbands was obtained from the following research groups: National Health and Medical Research Council (grant number 570814); Physiotherapy Research Foundation (grant number S07-011); and Workers’ Compensation Dust Diseases Board of NSW.
Ethics approval
Ethics approval was provided by South Eastern Sydney Area Health Service Northern Network Human Research Ethics Committee; Human Research Ethics Committee of Sydney South West Area Health Service; The University of Sydney; Curtin University; Sir Charles Gairdner Hospital; and Bentley Hospital.
