Abstract
Objectively measured severe physical inactivity (SPI) has been reported as the strongest independent predictor of mortality in patients with chronic obstructive pulmonary disease (COPD). Activity monitoring is not feasible in routine clinical practice; therefore, we set out to determine the utility of simple clinical measures for predicting SPI in patients with COPD. A total of 165 patients with COPD wore an activity monitor for 5 days to define the presence or absence of SPI. Logistic models were generated including the modified Medical Research Council (MMRC) dyspnea grade, spirometry and the age–dyspnea–airflow obstruction (ADO) index. Physical Activity Scale for the Elderly (PASE) and Stanford Brief Activity Scale (SBAS) were also tested for validity and reliability in a subgroup of 67 patients. The MMRC dyspnea grade, PASE score, ADO index and SBAS score were associated with SPI, but general self-efficacy and spirometry were not. An MMRC dyspnea grade ≥3 was the best independent predictor of SPI (AUC: 0.74; PPV: 0.83; NPV: 0.68) followed closely by a PASE score of <111. The combination of MMRC dyspnea grade and PASE score provided the most robust model (AUC: 0.83; Positive Predictive Value (PPV): 0.95; Negative Predictive Value (NPV): 0.63). The results were confirmed using 5000 bootstrapped models from the cohort of 165 patients. MMRC dyspnea grade ≥3 may be the best triage tool for SPI in patients with COPD. The combination of the MMRC and PASE score provided the most robust prediction. Our results may have significant practical applicability for clinicians caring for patients with COPD.
Introduction
Detecting severe physical inactivity (SPI) in patients with chronic obstructive pulmonary disease (COPD) is of the utmost importance. A recent study looking at a well-characterized cohort of patients with COPD showed that the SPI (determined by the activity monitoring and defined as a physical activity level (PAL) <1.40) was the strongest independent predictor of 48-month survival with a 31% mortality rate in this morbid category. 1 The PAL is obtained by dividing the total daily energy expenditure by resting energy expenditure as measured by an activity monitor, and it has been proposed by the World Health Organization as an objective measure of physical activity. A PAL of ≥1.70 defines an active person, 1.40–1.69 defines a sedentary person and <1.40 defines SPI. 2,3
While it is well known that physical inactivity is common among patients with COPD and that its prevalence increases with disease severity, 3 –5 the concept of stratifying patients into those having a lifestyle consistent with SPI is relatively novel and critically important given the associated mortality risk. The lack of dissemination of evidence demonstrating increased mortality associated with SPI in patients with COPD combined with the absence of a user-friendly tool to accurately assess physical activity may explain why it is not routinely addressed during clinical encounters. We speculate that the detection of SPI in patients with COPD may stimulate a dialog between patients and providers about possible interventions (pulmonary rehabilitation, health coaching, etc.) to modify this morbid lifestyle and potentially improve health care outcomes. Alternatively, for patients that do not respond to any intervention, persistent SPI likely signifies a very poor prognosis and may serve as a trigger to prompt conversations regarding advanced care planning and end-of-life goals, both important and useful endeavors in their own right.
Given the prognostic value and potential clinical importance of SPI in patients with COPD, we set out to determine the utility of simple metrics for predicting SPI including the modified Medical Research Council (MMRC) dyspnea scale, 6 percentage of predicted forced expiratory volume in 1 second (FEV1%pred), the Global Initiative for Obstructive Lung Disease (GOLD) stage, 7 and the multicomponent ADO (age, dyspnea and airflow obstruction (ADO)) index. 8 We also tested the Stanford Self-Efficacy for Managing Chronic Disease 6-Item Scale (SES6) 9 and two physical activity questionnaires: the Physical Activity Scale for the Elderly (PASE) and the Stanford Brief Activity Scale (SBAS). We also sought to determine the validity and reliability of the aforementioned physical activity questionnaires which is not previously tested in patients with COPD.
Methods
Physical activity questionnaire cohort
This study was approved by the Institutional Review Board of the Mayo Clinic College of Medicine (IRB# 09-002089). Patients with stable COPD were enrolled either by active recruitment from the outpatient pulmonary function laboratory or by direct referral from physicians within the Pulmonary Medicine Division. Patients recruited from the pulmonary function laboratory had been referred for pulmonary function testing at the discretion of attending physicians from all medical specialties, but were predominantly referred by specialists in Family Medicine, Internal Medicine and Pulmonary Medicine. This strategy created a cohort comprised of both local “primary” care patients and national “tertiary” referral patients. In addition to activity monitoring, all included patients had spirometry performed within the preceding year and completed the MMRC dyspnea scale, SES6, PASE and SBAS at the time of enrollment. The PASE and SBAS were repeated at the completion of activity monitoring (5–7 days), with the responses returned by mail. A total of 67 patients were enrolled during the recruitment period as a convenience sample. With a sample size of 67 patients, we had 80% power to detect a correlation coefficient of 0.34.
Spirometry
Spirometry was performed according to the current guidelines and established reference values. 10 Prebronchodilator values were used for all the statistical analyses.
Objective measurement of physical activity: the PAL
Physical activity was measured by a validated multisensor armband (SenseWear Pro ArmBand, BodyMedia Inc., Pittsburgh, Pennsylvania, USA). 11,12 Patients were instructed on the use of the activity monitor and were asked to wear it for 23 h/day (except when bathing) for 5 days. The data obtained for 5 days provide sufficient data for analysis, while maintaining maximal recruitment potential by minimizing burden to the patients. 3 The data from the device were downloaded to a dedicated software package (InnerView Research Software v.2.2, BodyMedia Inc., Pittsburgh, Pennsylvania, USA). Resting metabolic expenditure was calculated by multiplying a consistent value of energy expenditure during 1 min of consistent sleep by 1440 to estimate daily resting energy expenditure. The PAL was subsequently calculated by dividing the total daily energy expenditure by daily resting energy expenditure. 5
Physical activity questionnaires
The PASE is a validated 15-item questionnaire specifically designed to measure physical activity in individuals 65 years of age and older. 13 –17 It is divided into three components corresponding to leisure, household and occupational activities, respectively. The score is continuous and is reported as a total of the accrued points within each of the three components with a higher score reflective of increased physical activity. It was selected due to its inclusion of items addressing light physical activity, household work and occupational activities, all of which we felt were particularly relevant to our patient population.
The SBAS is a two-item physical activity questionnaire previously validated in older adults.
18,19
The respondent is asked to choose between various descriptions of physical activities (within leisure and occupational domains) that best represents his/her activity during the past year. The score is categorical and ranges from ‘
Self-efficacy
Self-efficacy was measured using the SES6.
9
The SES6 is comprised of six items asking the respondent to rate their confidence in dealing with various aspects of chronic disease management on a Likert scale ranging from 1
Dyspnea status
Dyspnea status was assessed using the MMRC dyspnea scale (range: 0–4) as described previously. 6
ADO index
The ADO index has been shown to be comparable to the body-mass index, airflow obstruction, dyspnea and exercise capacity (BODE) index and was utilized due to the lack of available exercise capacity data necessary for the calculation of BODE index. The ADO index was calculated for each participant as described previously. 8
Total cohort
In order to further validate our results from the validation cohort, we analyzed the data collected on an additional 98 patients with COPD (total of 165 patients) enrolled in prior and/or concurrent studies within our institution that included collection of the same measures including spirometry, the MMRC dyspnea grade, ADO index and PAL.
Statistical analysis
Descriptive summary statistics were reported for all included patients. Test–retest reliability of the PASE scores acquired 5–7 days apart was determined utilizing Pearson’s correlation coefficient and a Bland–Altman plot. The kappa coefficient was computed to determine the test–retest reliability between the two SBAS scores collected during the same time frame. Linear regression modeling was performed to determine the association between the PASE score and PAL as continuous variables. A Bland–Altman analysis was also used to determine the relationship between the PASE and PAL. In order to put the two variables on the same scale for comparison, both the variables were first transformed to Z-scores by subtracting their mean and dividing their standard deviation. The criterion validity of both the physical activity questionnaires for assessing SPI (binary variable: yes–no) was addressed by logistic regression models. The scores generated by the first administration of the PASE and SBAS, which were completed in a supervised clinical setting, were used for all the statistical analyses for the purposes of standardization.
In order to make the continuous variables simpler to use, we tested their performance as a binary variable. Cut-off points for the models were determined for each predictor using classification and regression tree analyses. The associations between SPI and the PASE score, MMRC dyspnea grade, SBAS score, SES6 score, FEV1%pred, GOLD stage and ADO index were estimated using logistic regression models. Multivariate logistic regression models including the variables that were significant in the univariate analysis were also constructed to further predict the probability of SPI. Performance characteristics were calculated for each predictor including sensitivity, specificity, positive predictive value, negative predictive value and likelihood ratios. Discrimination abilities were quantified with the concordance (
We performed a similar analysis on the larger cohort of 165 patients to confirm findings from the smaller physical activity questionnaire validation cohort regarding the simplest variables including MMRC dyspnea grade, ADO index and spirometry. In order to test the robustness of the models in the total cohort, 5000 bootstrapped models were generated. Bootstrapping is a recommended method for validating the predictive power of logistic models. 20,21
Results
Physical activity questionnaire cohort
Patient characteristics for the validation cohort are listed in Table 1. On an average, patients were 71 years of age and almost equally divided by sex. The patient cohort had moderately severe airflow obstruction (mean FEV1%pred, 46.7 ± 19.98) and SPI was prevalent as evidenced by a PAL of <1.40 in 46 (69%) patients (mean PAL: 1.37 ± 0.30). Patients were moderately dyspneic with a mean MMRC dyspnea grade of 2.5 ± 1.1 (scale 0–4) and had a mean ADO index of 6.1 ± 1.96 (scale 0–10) consistent with their advanced age, severe airflow limitations and moderate dyspnea.
Patient characteristics of physical activity questionnaire cohort.a

BMI: body mass index; FEV1: forced expiratory volume in 1second; %pred: % predicted; MMRC: modified Medical Research Council; ADO: age, dyspnea, and airflow obstruction; PAL: physical activity level; PASE: Physical Activity Scale for the Elderly; SBAS: Stanford Brief Activity Survey.
aData are presented as
Reliability analysis of the two physical activity scores
Different statistical methods were utilized due to the fact that the PASE score is continuous, whereas the SBAS is a categorical score.
The PASE scores acquired 5 days apart had a Pearson’s correlation coefficient of 0.75, consistent with good test–retest reliability. The SBAS scores acquired over the same time frame had a kappa value of 0.41 indicating only fair reliability.
Bland–Altman analysis
A Bland–Altman plot was utilized to confirm the test–retest reliability of the PASE score. The correlation coefficient between the mean and difference of the PASE scores 5–7 days apart was 0.13 (
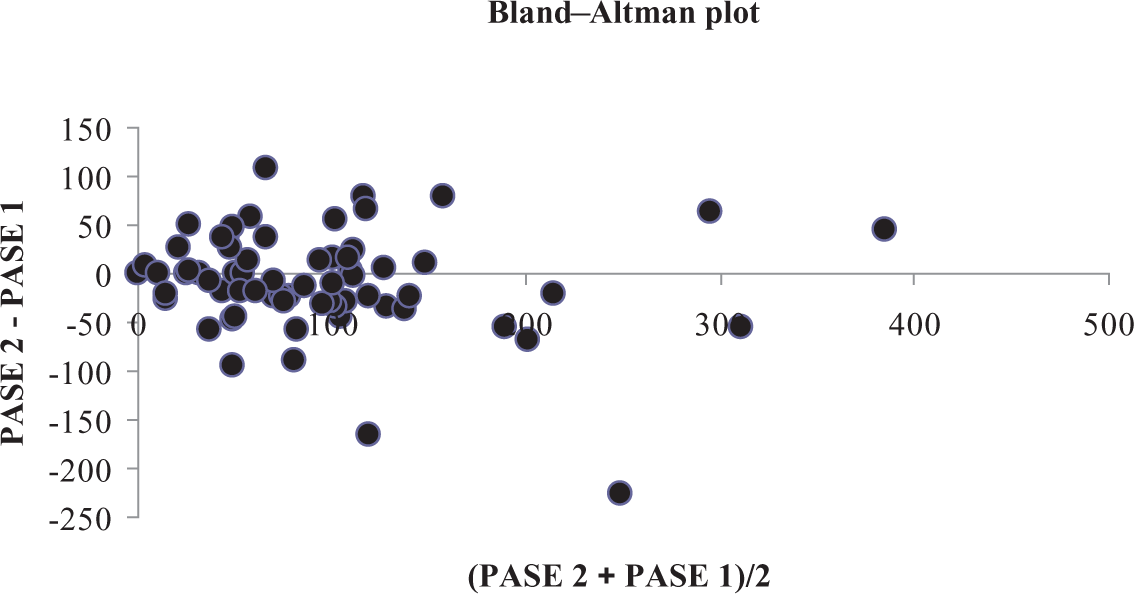
Scatter plot of Bland–Altman analysis of PASE scores acquired 5–7 days apart. The correlation coefficient between the mean and difference of the scores is 0.13 (
Linear regression model
Linear regression modeling demonstrated a significant linear relationship between the PASE score and PAL with an R 2 value of 0.38 (p < 0.001). The scatterplot (Figure 2) demonstrates significant variability around the linear model; however, when patients are dichotomized by a PASE score of <111 and/or a PAL of <1.40, the scatterplot demonstrates that the majority of patients with a PAL of <1.40 also had a PASE score of <111 (true positives) and, conversely, however less strongly, patients with a PAL of >1.40 had a PASE score of >111 (true negatives). Linear regression modeling was not performed for the SBAS due to its inferior reliability.

Scatter plot demonstrating linear relationship between the PASE score and PAL. Vertical and horizontal coordinates indicate the cut-points used to identify severe physical inactivity for the PASE score (<111) and PAL (<1.40) respectively. PASE: Physical Activity Scale for the Elderly; PAL: physical activity level.
SPI prediction models
Logistic regression models utilizing SPI (PAL of < 1.4) as the dependent binary variable showed that the PASE score, MMRC dyspnea grade, SBAS score and ADO index were associated with SPI (Table 2). An MMRC dyspnea grade ≥3 was the best individual predictor of SPI in patients with COPD followed very closely by a PASE score of <111. A combined model including both the MMRC dyspnea grade and the PASE score was the most robust model. FEV1%pred, GOLD stage and SES6 score were not associated with SPI.
Performance characteristics for predictors of severe physical inactivitya in patients with chronic obstructive pulmonary disease (physical activity questionnaire cohort).
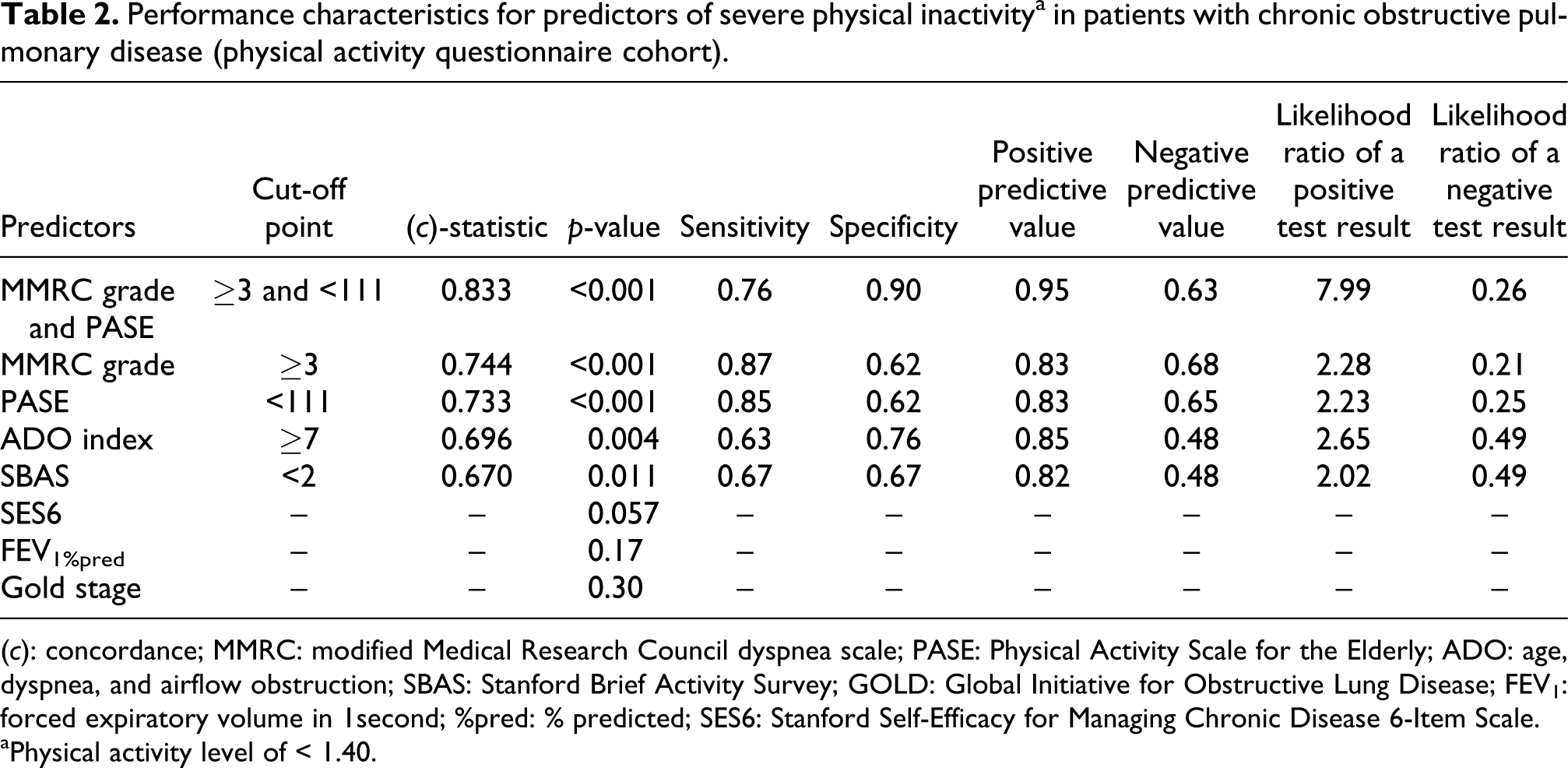
(
aPhysical activity level of < 1.40.
Total cohort
The total cohort consisted of 165 patients with COPD with similar characteristics when compared with the physical activity questionnaire cohort used for the validation of PASE and SBAS (Table 3).
Patient characteristics of total cohort.a

aData are presented as
BMI: body mass index; FEV1: forced expiratory volume in 1second; %pred: % predicted; MMRC: modified Medical Research Council; ADO: age, dyspnea, and airflow obstruction; PAL: physical activity level; PASE: Physical Activity Scale for the Elderly; SBAS: Stanford Brief Activity Survey.
SPI prediction models
Logistic regression models utilizing SPI (PAL < 1.4) as the dependent binary variable were generated using this larger sample size and again demonstrated that the MMRC dyspnea grade and ADO index were associated with SPI (Table 4), although with a small reduction in their
Performance characteristics for predictors of severe physical inactivity in patients with chronic obstructive pulmonary disease (total cohort).

(
Discussion
The results of this study suggest that an MMRC dyspnea grade ≥3 on a scale of 0–4 is the best independent triage test for SPI in patients with COPD. The utility of this finding is not simply the confirmation that more dyspneic patients are less physically active, which is ultimately intuitive, but rather than we have provided a benchmark that offers respectable predictive capabilities for SPI in patients with COPD. In addition, the MMRC dyspnea grade was superior to other common measures. This method may be all that is needed to recommend interventions to increase physical activity in these patients. Options include (but are not limited to) formal pulmonary rehabilitation, health coaching, enrolment in a wellness program and/or one-on-one counseling regarding physical activity options that may fit the individual patient’s lifestyle and goals.
We found a modest (but significant) linear relationship between the PASE score and the PAL indicating that the PASE showed poor performance in predicting the overall spectrum of physical activity in patients with COPD. However, we found the PASE to be a valid and reliable tool for predicting SPI in patients with COPD when using a dichotomized cut-point of <111. To our knowledge, this is the first study showing evidence for the utility of a physical activity questionnaire for predicting SPI in this patient population. The PASE score performed nearly as well as MMRC dyspnea grade; however, administration and interpretation of the PASE is more time consuming. The ADO index was inferior to both the PASE score and MMRC dyspnea grade. FEV1%pred, GOLD stage and the SES6 were not predictive of SPI at all. The SBAS, while valid, was not meaningfully reliable.
The testing of various combined models proved fruitful. The model including an MMRC dyspnea grade ≥3 and a PASE score of <111 was the most robust overall for the prediction of SPI (Table 2). The positive predictive value of the combined model was excellent while maintaining a more than acceptable negative predictive value. For the purpose of ‘ruling out’ SPI using the MMRC dyspnea grade alone, consideration must be given to the fact that the patients with ‘negative’ testing may still be severely inactive in up to 28% of cases (NPV 0.72). We believe our results are the most informative for the clinicians seeking simple tools to support a clinical suspicion of SPI in patients with COPD.
Interestingly, in contrast to our study, Watz et al. found GOLD stage to be superior to BODE index, FEV1% pred, 6MWD and MMRC dyspnea grade for predicting SPI in a cohort of European patients with COPD. 3 One possible explanation is the fact that the Europeans are generally more physically active than Americans at baseline based on cultural differences. It is plausible that the functional class predictors would be less robust in an American population, where individuals with less severe disease may exhibit physical inactivity due to personal and cultural factors rather than disease severity. For example, it is a more common place in Europe to walk or a ride a bicycle to work and within the local community for routine daily activities. 22 Therefore, a functional class predictor, like GOLD stage, may work well in Europe, where the disease severity translates into the limitations of normal activities thereby reducing physical activity. In the United States, on the other hand, prevalent obesity coupled with the fact that the majority of Americans utilize motor vehicles for travel within their local communities likely contributes to a more inactive lifestyle. 23,24
The PASE is a well-validated tool that was specifically created for the purpose of measuring physical activity in the elderly population. 13 –17 It is somewhat unique in that it includes items assessing activities requiring minimal physical exertion. The examples include light walking, washing dishes, vacuuming, dusting, gardening and occupational endeavors including volunteerism. The PASE could theoretically confer a benefit compared with the MMRC dyspnea grade when screening for SPI (given that they performed relatively equally) in situations where the patient’s dyspnea is driven by cognitive and emotional factors. We postulate that, in these cases, SPI might be more reliably assessed by the PASE score. In addition, knowing the physical activity routine of a patient’s life as measured by the PASE can have tremendous implications for the prescription of activities in pulmonary rehabilitation and coaching programs for the patient. The routine practice of ‘mundane activities’ such as washing dishes or mopping floors may be just what is needed to prevent the patient from falling into the morbid SPI category. Patients with COPD have a significant reduction in their total daily energy expenditures. 3,4,25,26 A small change in the physical activity, such as washing widows or vacuuming the house, may therefore have a significant effect on their PAL. Based on our results, the PASE seems to be a good tool for identifying SPI, but a poor tool for assessing the overall spectrum of physical activity in the COPD population. The latter may be due to the recall bias leading to erroneous results and only modest correlation with objectively measured physical activity. 27
It is worth noting that only 13 (19%) of 67 respondents reported occupational activities indicating that the employment (paid or volunteer) is relatively uncommon among our population. However, 7 (54%) of 13 patients reporting active occupational activities had a PAL of >1.4 suggesting that their occupational activities may have been important for preventing SPI. Therefore, we feel it is appropriate to recommend for patients with COPD who are currently employed to maintain their occupational activities as long as possible, and for individuals who are unemployed that they consider active volunteerism as a means to increase physical activity.
We found that the Stanford Self-Efficacy for Chronic Disease Scale did not correlate with SPI in this study. Self-efficacy (an individual’s intrinsic confidence in their ability to perform a specific task) is a construct that has been associated with successful behavioral change, including increasing physical activity. 28 –33 Self-efficacy is by its very nature a task-specific construct. 34 The Self-Efficacy scale used in this study is a general scale designed to broadly assess self-efficacy for dealing with chronic illness. 18 It is not surprising, therefore, that this tool was not associated with physical activity given its lack of specificity for the task at hand. It is unlikely that any self-efficacy tool will show an association with physical activity until a tool specific for physical activity in patients with COPD is generated and validated.
The limitation of this study is that our results are not useful for predicting the whole spectrum of physical activity in COPD, since the study was aimed specifically at detecting SPI.
In summary, given recent evidence regarding the clinical relevance of SPI in COPD, we believe that a focus of routine screening in this population should be directed toward this morbid condition. In this study, we have shown that an MMRC dyspnea grade ≥3 is likely the simplest way to identify patients with COPD that are severely inactive. The PASE score improves minimally upon the MMRC dyspnea grade but offers the advantage of providing more specific information about routine physical activities. This additional information may be helpful when counseling patients about increasing their physical activity. Our results provide useful benchmarks for the utilization of the MMRC dyspnea grade and PASE and are informative to the clinicians wishing to identify their severely inactive patients with COPD. Identifying SPI in these patients may promote meaningful collaborative plans for increasing physical activity. Alternatively, identifying persistent SPI despite efforts to modify this significant morbid risk factor may serve to prompt conversations regarding advanced care planning and end-of-life goals when appropriate.
Footnotes
Acknowledgments
We would like to thank Mrs Marnie Wetzstein and Mrs Johanna Hoult for their assistance with logistical management of the study and Mrs Xinghua Zhao for her assistance with statistical analysis related to this study.
Author’ contribution
ZSD: contributed to data analysis and writing the manuscript; ACG: contributed to the design of the study and data collection; PJN: contributed to the statistical analysis and writing the manuscript; RPB: (guarantor) contributed to the design of the study, data collection, data analysis, and writing the manuscript.
Declaration of Conflicting Interests
The authors declare no conflicts of interest.
Funding
This work was supported by the National Institutes of Health (Grant NIH NHLBI 1R01CA163293-01 (RPB)).
