Abstract
Background:
The prognosis of patients with diabetes mellitus (DM) is particularly determined by vascular comorbidities. A recent theory implies that DM could also promote aortic valve stenosis (AS). The present study investigates this association in a large collective of outpatients.
Methods:
This retrospective cohort study compared the incidence of AS in patients with an initial diagnosis of type 2 DM and a matched non-DM cohort in 809 general and diabetologist practices in Germany between January 2005 and December 2018. Cox regression models were performed to study the association between type 2 DM and AS incidence.
Results:
A total of 78,805 patients with type 2 DM and 78,805 patients without diabetes were analysed. Diabetes patients were more frequently diagnosed with obesity (52% vs 21%). Four percent of patients with and three percent of without diabetes were diagnosed with AS (p < 0.001). Diabetes was significantly associated with an increased incidence of AS (HR: 1.36, p < 0.001). This association was higher in men (HR: 1.41) versus women (HR: 1.30). The strongest association was observed in young patients (18–50 years, HR: 2.35, p < 0.001).
Conclusion:
In our study, diabetes patients had a higher incidence of aortic stenosis during their disease course.
Introduction
Type 2 Diabetes mellitus (DM) and aortic valve stenosis (AS) are both diseases that mainly affect older patients. Both have a high prevalence in this age group and have a significant impact on the quality of life and prognosis of those affected. Type 2 DM is associated with an increased incidence of cardiovascular disorders and cardiovascular events such as acute myocardial infarction and acute stroke.1–3 According to a large English registry study, peripheral arterial disease and heart failure are the most common initial cardiovascular manifestations of diabetes mellitus. 4 It has also been shown that diabetes worsens the prognosis in heart failure.3,5 There is evidence that diabetes mellitus worsens the progression of aortic valve stenosis.6,7 The development of aortic valve stenosis has not yet been fully elucidated on a molecular basis. Indeed, even the process that used to be called purely degenerative aortic valve stenosis seems instead to be a process that might involve active calcification and lipoprotein deposition.8,9 The ‘link hypothesis’ 6 has proposed several pathomechanisms that might be involved in this interaction between type 2 DM and AS: It has already been demonstrated that poorly-controlled type 2 DM can lead to accumulation of advanced glycation end products (AGEs), which may be associated with the severity of aortic stenosis in patients with concomitant type 2 diabetes. 10 Furthermore, it has been shown that transient hyperglycemia leads to excessive synthesis of proinflammatory phospholipids and an activation of coagulation, which might facilitate AS in patients with poorly-controlled diabetes. 11
By contrast, the question of whether type 2 DM also promotes the development of aortic valve stenosis, has not yet been investigated sufficiently.12,13 The aim of the present retrospective study on a large collective of patients with type 2 DM is to investigate whether type 2 DM is associated with an increased incidence of aortic valve stenosis.
Methods
Database
This study is based on data from the Disease Analyser database (IQVIA), which contains drug prescriptions, diagnoses and basic medical and demographic data obtained directly and in anonymous format from computer systems used in the practices of general practitioners and specialists. 14 The database covers approximately 3% of all outpatient practices in Germany. Diagnoses (according to International Classification of Diseases, 10th revision (ICD-10)), prescriptions (according to Anatomical Therapeutic Chemical (ATC) Classification system), and the quality of reported data are being monitored by IQVIA. In Germany, the sampling methods used to select physicians’ practices are appropriate for obtaining a representative database of general and specialised practices. It has previously been shown that the panel of practices included in the Disease Analyser database is representative of general and specialised practices in Germany. 14 Finally, this database has already been used in previous studies focusing on diabetes.15–17
Study population
This retrospective cohort study includes adult patients (⩾18 years) with an initial diagnosis of type 2 diabetes (ICD-10: E11) (index date) and antihyperglycemic therapy (ATC: A10) within 12 months after the index date in 809 general and diabetologist practices in Germany between January 2005 and December 2018 (Figure 1). An observation time of at least 12 months prior to the index date was a further inclusion criterium. Patients with nonrheumatic aortic valve disorder diagnoses (ICD-10: I35) prior to index date were excluded.
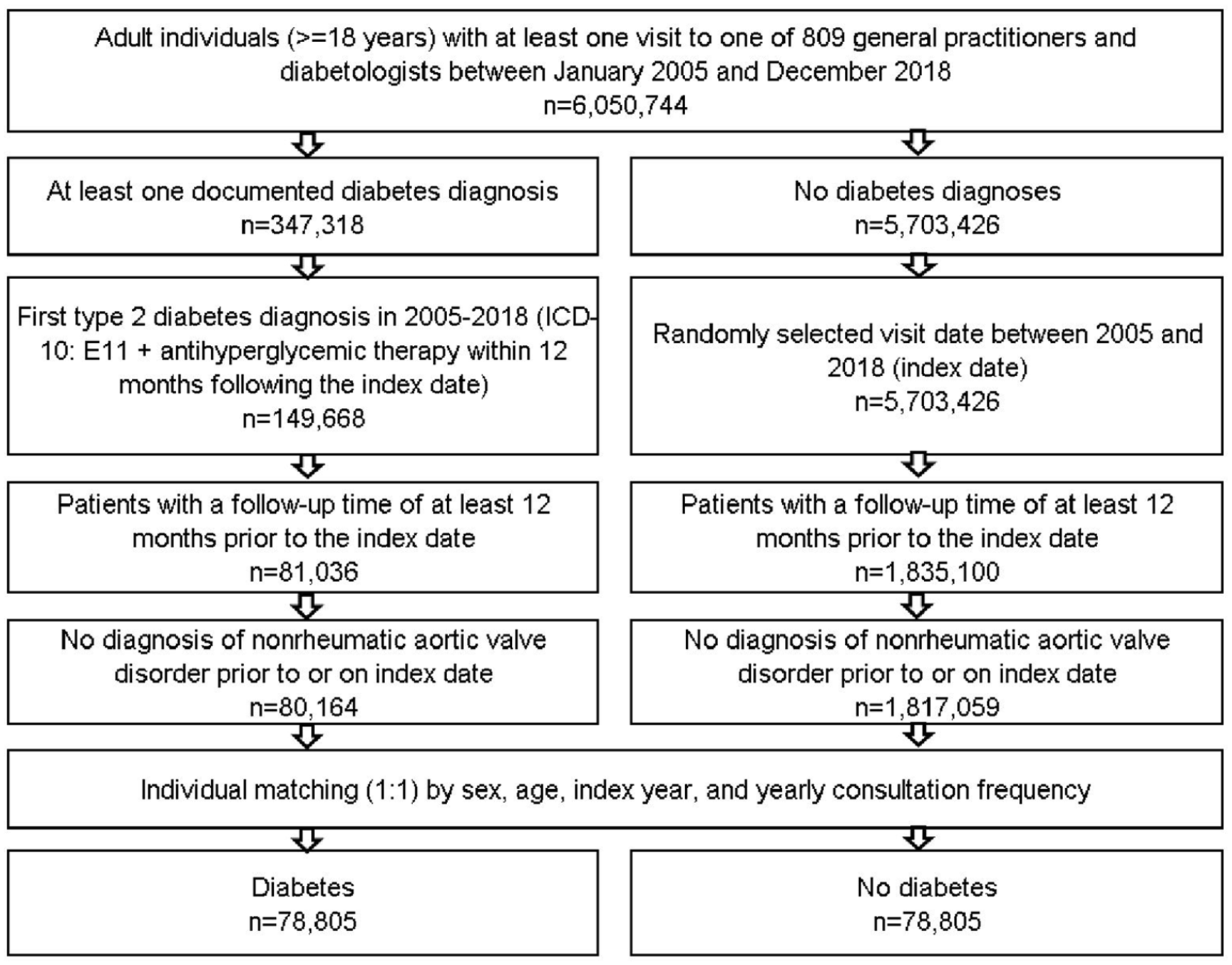
Selection of study patients.
Diabetes patients were matched to non-diabetes patients by sex, age, index year and yearly consultation frequency. As diabetes patients have much higher consultation frequency due to diabetes treatment, and higher consultation frequency can increase the probability of other diagnoses documentation, we included consultation frequency per year in the matching. For the non-diabetes cohort, the index date was that of a randomly selected visit between January 2005 and December 2018 (Figure 1).
Study outcomes and covariates
The main outcome of the study was the cumulative incidence of nonrheumatic aortic valve disorder in diabetes patients compared to matched non-diabetes patients. Moreover, variables associated with a probability of nonrheumatic aortic valve disorder in diabetes patients were investigated.
Statistical analyses
Differences in the sample characteristics between those with and those without diabetes were tested using chi-squared tests for categorical variables and Wilcoxon tests for continuous variables. Kaplan–Meier-Curves were used to display the cumulative incidence of nonrheumatic aortic valve disorder in patients with and without diabetes.
Hazard regression models were conducted to study the association between diabetes and nonrheumatic aortic valve disorder. These models were created separately for five age groups, women and men. The multivariate Cox regression model was adjusted for co-morbidities documented within 12 months prior to the index date including lipid metabolism disorders (ICD-10: E78), hypertension (ICD-10: I10), chronic coronary heart disease (ICD-10: I24, I25 excl. I25.2), myocardial infarction (ICD-10: I21–I23, I25.2), stroke/TIA (ICD-10: I64, I64, G45), peripheral vascular disease (ICD-10: E11.5, I70.2, I73.9), and chronic renal failure (ICD-10: E11.2, N18, N19). To counteract the problem of multiple comparisons, p-values <0.01 were considered statistically significant in this model.
Finally, the association between age, sex, co-morbidities, body-mass-index (BMI), HbA1c values and nonrheumatic aortic valve disorder was analysed in the diabetes cohort. For this analysis, the average BMI and HbA1c values were calculated in the total follow-up time starting from 6 month prior to the index date to the date if the first nonrheumatic aortic valve disorder or end of the follow up, whatever occurred first. To counteract the problem of multiple comparisons, p-values <0.001were considered statistically significant in this model. Analyses were carried out using SAS version 9.4 (SAS institute, Cary, USA).
Ethical standards
Only aggregated, anonymised patient data were used in these analyses. This study was performed in accordance with the Declaration of Helsinki, the guidelines for Good Practice of Secondary Data Analysis 18 and the ICMJE Recommendations for the Conduct, Reporting Editing and Publication of Scholarly Work in Medical Journals. Since we used only anonymised data, which could not be traced back to individual persons, the research protocol did not have to be approved by the local ethics committee, and it was not necessary to obtain informed consent from individual patients to participate in the study. This was confirmed by the local ethics committee of the Christian-Albrechts-University (CAU) of Kiel, Kiel, Germany (File reference D413/21).
Results
Basic characteristics of the study sample-
The present study included 78,805 patients with diabetes and 78,805 patients without diabetes. The basic characteristics of study patients are displayed in Table 1. Mean age [SD] was 52.3 [13.3] years; 45.5% were women. On average, patients visited their general practitioner (GP) 4.5 (SD: 5.3) times per year during the follow-up time. Diabetes patients were more frequently diagnosed with obesity (52% vs 21%). Although other diagnoses differed significantly between diabetes and non-diabetes cohort, absolute differences were relatively small. In the diabetes cohort, the mean HbA1c value was 7.2% (55 mmol/mol); SD 1.7% (5 mmol/mol); 32% of the patients had an average HbA1c >7.5% (58 mmol/mol), 13% of the patients had an average HbA1c >8.5% (69 mmol/mol) and 6% of the patients had an average HbA1c>9.5% (80 mmol/mol).
Basic characteristics of the study sample (after 1:1 matching by sex, age, index year and yearly consultation frequency).
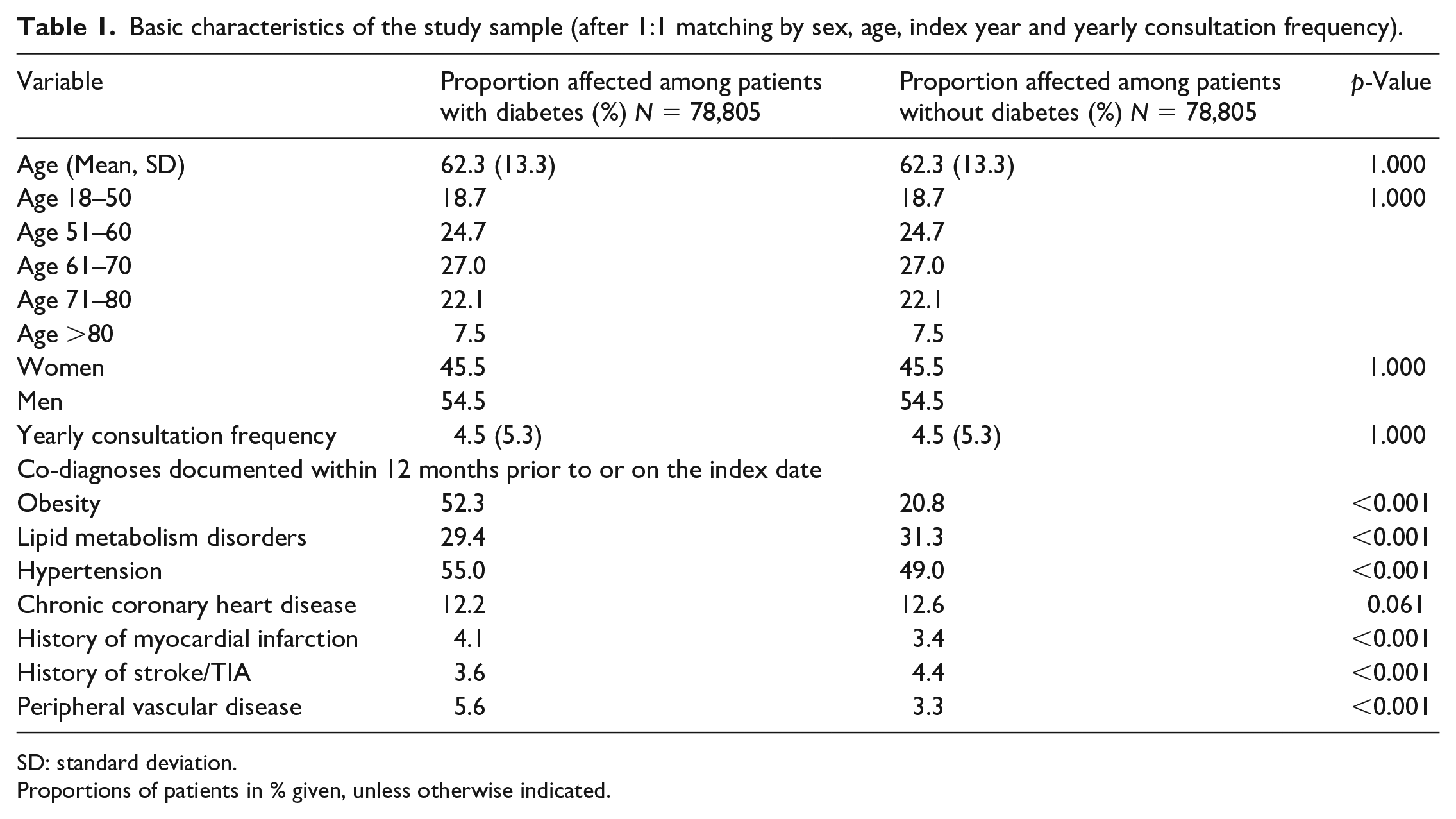
SD: standard deviation.
Proportions of patients in % given, unless otherwise indicated.
Association of diabetes and incidence of nonrheumatic aortic valve disorder
Within 10 years of the index date, 4.0% of patients with and 3.0% of without diabetes were diagnosed with AS (log-rank p < 0.001) (Figure 2).
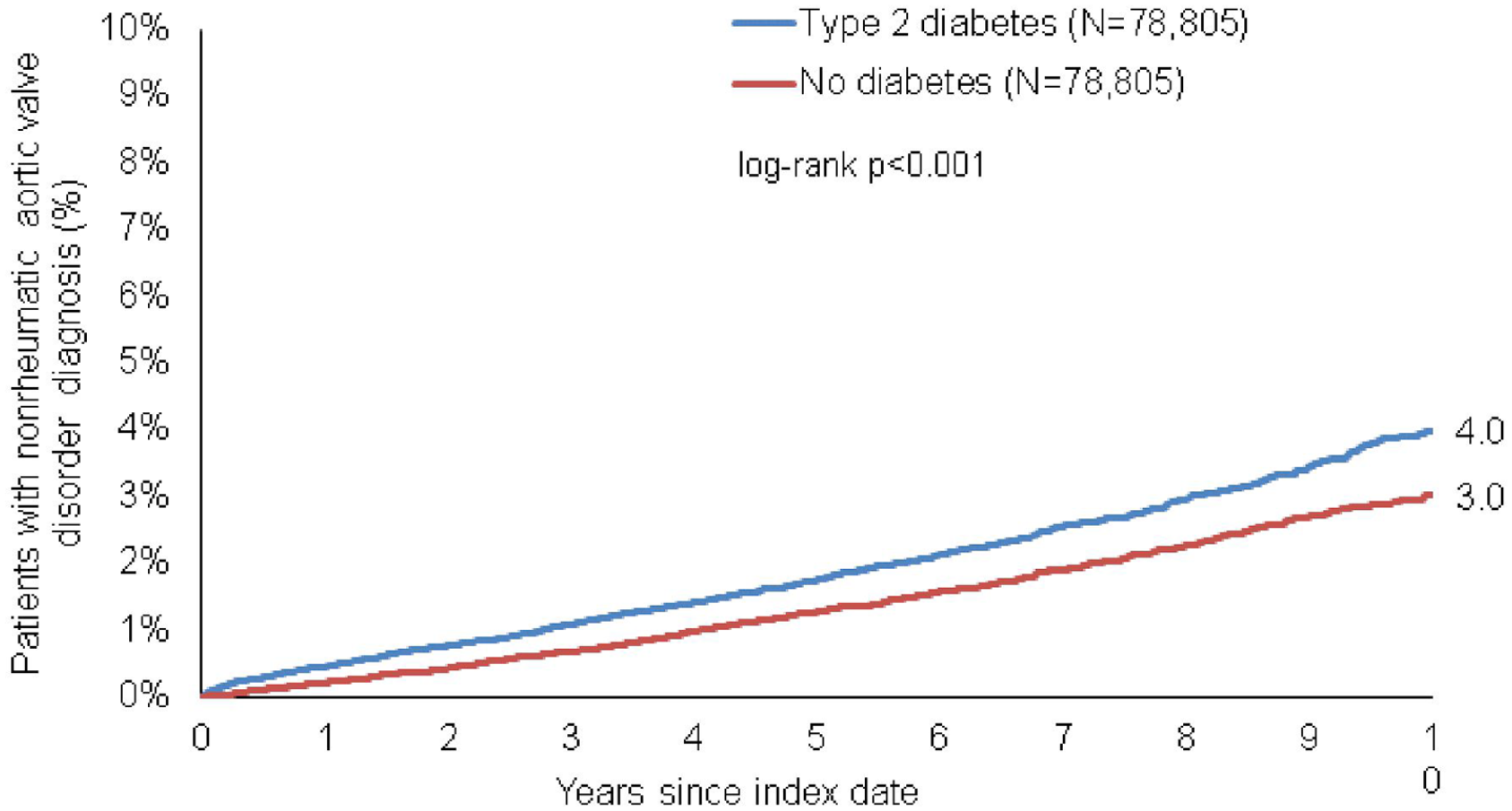
Kaplan–Meier curves for time to diagnosis of nonrheumatic aortic valve disorder in patients with and without type 2 diabetes.
The cumulative incidence of AS increased with age from 1.4% in the age group of 18–50 years to 9.7% in the age group >80 years in diabetes, and from 0.6% to 9.7% in the same age groups of non-diabetes patients.
Our regression analyses indicated a significant association between type 2 DM and the incidence of nonrheumatic aortic valve disorder (HR: 1.36, p < 0.001). This association was slightly stronger in men (HR: 1.41) than in women (HR: 1.30). The strongest association was observed in youngest age group (18–50 years, HR: 2.35, p < 0.001). Notably, the association was not significant in patients aged >80 years (Table 2).
Association between diabetes and the cumulative 10-year-incidence of nonrheumatic aortic valve disorder diagnoses in patients followed in general practices in Germany (Cox regression models).

Multivariate Cox Regression models adjusted for obesity, lipid metabolism disorders, hypertension, chronic coronary heart disease, history of myocardial infarction, history of stroke/TIA, peripheral vascular disease.
Variables associated with nonrheumatic aortic valve disorder in diabetes cohort
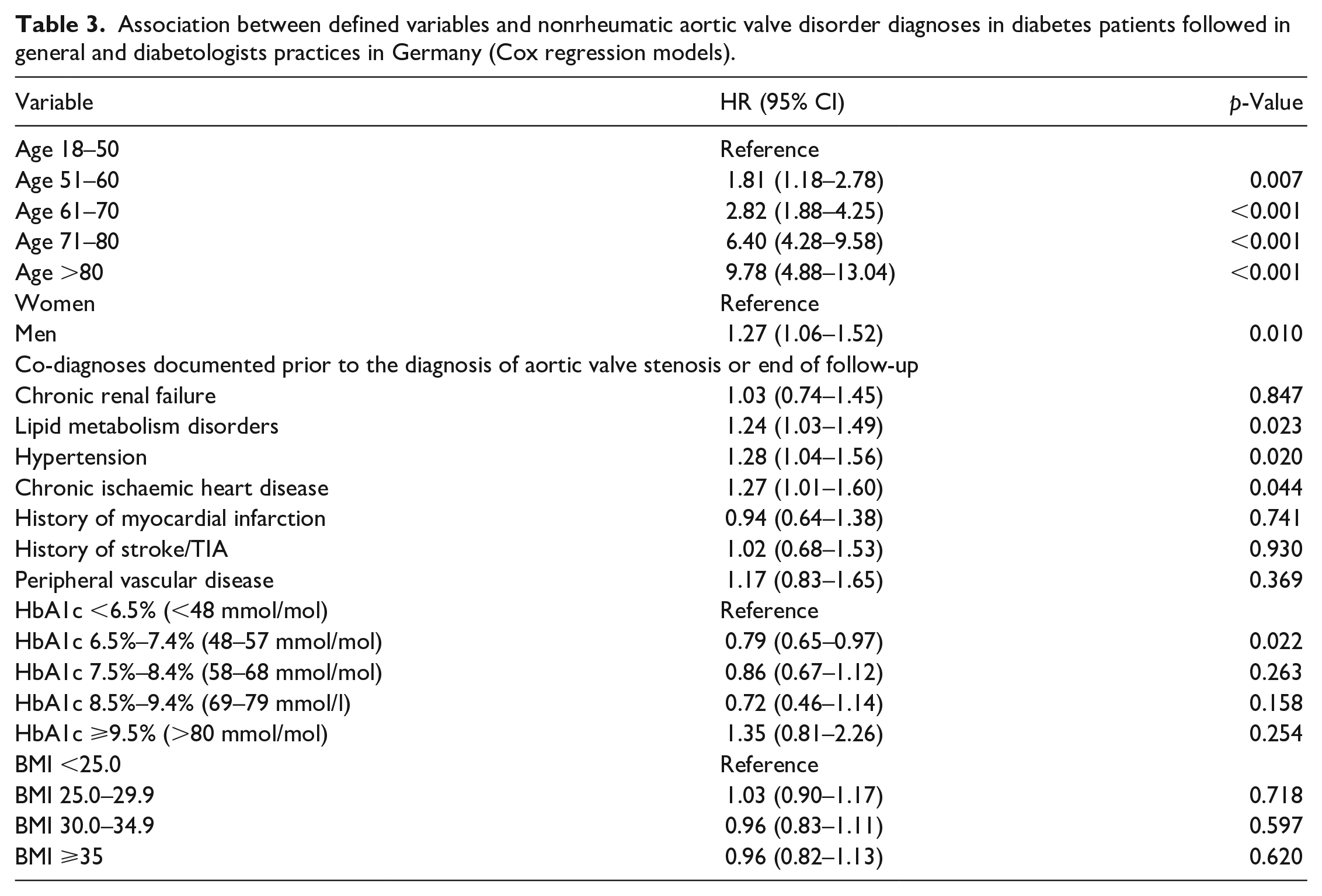
In the diabetes cohort, there was a strong association between age and incidence of nonrheumatic aortic valve disorder (HR: 6.40, p < 0.001 for age 71–80 years, and HR: 9.78, p < 0.001 for age >80 as compared to 18–50 years). Interestingly, neither higher HbA1c values nor a higher body mass index (BMI) were significantly associated with the incidence of nonrheumatic aortic valve disorder in diabetes patients (Table 3). Other comorbidities such as ischaemic heart disease, chronic renal failure, lipid metabolism disorders or peripheral vascular disease were not significantly associated with an increased incidence of AS (Table 3).
Association between defined variables and nonrheumatic aortic valve disorder diagnoses in diabetes patients followed in general and diabetologists practices in Germany (Cox regression models).
Discussion
In this study, we analysed the association between Type 2 DM and AS. We followed a population-based approach using data from the Disease Analyser database (IQVIA), which collects drug prescriptions, diagnoses and basic medical and demographic data directly from general practitioners and specialists in Germany. 9 Our results demonstrate that the incidence of aortic valve stenosis is significantly higher in patients with DM than in matched patients without DM. Remarkably, this association was independent of HbA1c values and the patients’ BMI.
The prevalence of type 2 diabetes mellitus and its main risk factor, obesity, has reached epidemic proportions in recent years.3,19 Many cardiovascular complications of type 2 DM are well known. These include Macrovascular complications, including coronary heart disease, stroke and peripheral vascular disease and microvascular complications, such as end-stage renal disease (ESRD), retinopathy and neuropathy, along with lower-extremity amputations. 19 In addition to these ‘classic’ complications, type 2 DM is also increasingly associated with age-related diseases such as dementia. 20 Aortic valve stenosis is also a typical disease of the elderly. 21 In our study, there was a strong association between age and incidence of nonrheumatic aortic valve disorder. Interestingly, the increased association of diabetes was no longer detectable in patients >80 years. The younger the patients, the stronger the association between diabetes mellitus and the incidence of aortic valve stenosis. The association was highest in the group of 18–50 year olds.
While coronary risk factors like male sex, hypertension, elevated levels of LDL cholesterol and smoking have formerly been identified as risk factors for the emergence of AS, 9 association between DM and AS has not been fully established before.
It must be made clear again that we have not demonstrated a causal relationship between diabetes mellitus and the occurrence of aortic valve stenosis, but have instead only illustrated a statistical accumulation. The very well-established criteria of Sir Hill, 22 which make an important contribution to the conclusion of a causal relationship in the presence of statistical association, may help to classify our results. These criteria call for caution when interpreting one’s own results.
Some of them are mentioned here as examples: The effect has to take place after the occurrence of the presumed cause. This is one reason why we only included patients in whom aortic valve stenosis was already present when diabetes was diagnosed. Another postulate concerns the biological gradient: stronger exposure to a risk factor should lead to a more frequent occurrence of the disease. Applying this postulate to our study at least establishes that (as shown in Table 3), patients with particularly poorly-controlled diabetes may be at a particular risk of aortic valve stenosis. The group of patients with poorly-controlled diabetes seems to be the driver of the outcome for the total population. The observation of patient groups with better-controlled diabetes mellitus does not show a similarly strong effect. In particular, the hazard ratio of the patient group with an HBA1c of 6.5%–7.4% (48–57 mmol/mol), is actually significantly lower than that of the control group. We cannot provide a clear explanation for the findings for this subgroup. It might be possible that this subgroup with lower average HbA1c has more frequent episodes of hypoglycaemia. Severe hypoglycaemia has been identified as one of the strongest predictors of macrovascular events, adverse clinical outcomes and mortality in people with type 2 diabetes. 23 As no data are available regarding aortic valve stenosis, however, this assumption remains speculative. Overall, the application of the Bradford Hill criteria urges caution in the interpretation of statistical associations. Nevertheless, association studies like ours might trigger research about possible mechanisms causing aortic valve stenosis in patients with type 2 DM:
The pathophysiology and genesis of aortic valve stenosis is actually not yet well understood. For decades, aortic stenosis was thought to represent a passive, degenerative process that causes a progressive narrowing of the valve. 9 Increasing calcification of the valve exacerbates narrowing. There is increasing evidence that the changes in the development of AS is not a passive degenerative process, but rather an active biological process. Several studies have described an ‘early lesion’ in this context that has many similarities with the early lesion of an atherosclerotic plaque, suggesting that aortic valve stenosis is an active process, resembling the development of an atherosclerotic plaque.24,25
Various signalling pathways have been identified as being involved into the development of this condition, for example, signalling pathways that cause proliferation of the extracellular matrix.8,9,26 Several cell lines have been identified to regulate the calcium load of the valve leaflets, including valvular interstitial cells (VICs), endothelial cells and cardiac leukocytes. 27 Under pathological conditions such as inflammation, VICs can differentiate into myofibroblasts (causing fibrosis) and osteoblast-like cells (causing calcification). 27 There is growing evidence that low-grade inflammation is a common feature in subjects with type 2 diabetes. The majority of type 2 DM patients show chronically elevated inflammatory markers such as CRP. 28 This slightly increased inflammation could play a role in the development of AS: In this context, Natorska et al. 29 demonstrated that diabetes promotes the expression of proinflammatory markers such as CRP in patients with severe aortic valve stenosis. These data are supported by a new study by Tucureanu et al., 30 who demonstrated echocardiographic changes in the aortic valve in an animal model of diabetes mellitus. This was accompanied by an increased expression of cell adhesion molecules, extracellular matrix remodelling and osteogenic markers. Significant correlations were found between tissue valve biomarkers and plasmatic and haemodynamic parameters. The changes in this animal model were detectable from an early stage. This may reflect the fact that differences in the incidence of AS between patients with and without type 2 DM are most evident in the youngest age group, 18–50 years, as we found in our study. Another interesting mechanism that may be involved is an accumulation of advanced glycation end products (AGEs) at the aortic valve, which has been shown to be associated with the severity of aortic stenosis in patients with concomitant type 2 diabetes. 10 As this is thought to promote the incidence of AS, this may at least in part explain why we found an increased rate of AS especially in the subgroup of type 2 DM patients with very poorly-controlled DM.
Our study is subject to a number of limitations, which are due to the study design and cannot be completely avoided in our database analysis, as recently described. 31 Our analysis is based on diagnoses according to the ICD-10 coding system. The possibility of misclassifications and missing coverage of certain diagnoses cannot be excluded completely. For data protection reasons, not all laboratory parameters could be assigned to individual patients in the database. There is also a lack of data on the socioeconomic status of the patients and on lifestyle-related risk factors. Furthermore, our database is geared towards outpatients, meaning that inpatient treatment cases are not recorded. The disease analyser database only provides information on outpatient cases and not hospital data, which might give rise to a selection bias. Due to the study design and goal of the incidence investigation, patients with AS diagnosis prior to the index date were excluded, and the proportion of these patients was higher in the diabetes cohort than in the non-diabetes cohort. This may bias the study results, as the proportion of patients with initial diagnosis of AS after the index date may be underestimated in the diabetes cohort. We have not recorded any auscultation findings, as our database is based on the entry of ICD codes from treating physicians. Therefore, we cannot exclude the possibility of certain effects on the diagnosis of the patients, for example that patients with type 2 diabetes were auscultated more frequently and/or more precisely during their consultations with their general practitioners and aortic valve stenosis was thus detected earlier. Our database does not offer any information regarding the intake of dopamine agonists, which have been shown to induce valvular heart disease. 32 The diagnosis of diabetes itself or the higher rate of myocardial infarctions in the diabetes group might have led to an increased probability of seeing a cardiologist and receiving an echocardiogram and thus incidental detection of AS. Similarly, the fact that a guideline-compliant diagnosis of aortic valve stenosis was carried out is not documented for every single patient. We do not report treatment courses or correlate the incidence of aortic valve stenosis with symptoms. Our data do not allow us to grade the severity of aortic valve stenosis and do not provide any information about possible surgical/minimally invasive interventions on the aortic valve. It is not possible to trace back and test the reliability of individual data entries. Although we normalised both groups by frequency of visiting their GP, bias cannot be completely ruled out here either. Nevertheless, we would like to point out that the German Disease Analyser database has been extensively validated in various studies and has proven to be representative for the German outpatient sector.15,33
In summary, we present data from a large primary care database showing that diabetes mellitus is associated with an increased incidence of aortic valve stenosis, independent of other comorbidities and patient characteristics. Our data strengthen the theory that diabetes and aortic valve stenosis are closely associated. Further studies investigating underlying pathomechanisms are needed to further confirm the relationship between these two highly relevant diseases.
Footnotes
Acknowledgements
The authors would like to express their gratitude to Ms. Claudia Jones for critically revising the manuscript in terms of style and language.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
