Abstract
The study aims to evaluate the relationship between skin autofluorescence, a marker of advanced glycated end-products accumulation in tissue, and high-sensitive cardiac troponin T, a cardiovascular biomarker, in Japanese subjects with diabetes. A total of 145 subjects with diabetes and 32 nondiabetic subjects as control attending the outpatient clinic were examined. Skin autofluorescence was measured using the AGE Reader™. Univariate and multivariate regression analyses were used to identify the factors associated with the high-sensitive cardiac troponin T and N-terminal pro-B-type natriuretic peptide values. Skin autofluorescence, high-sensitive cardiac troponin T, and maximum intima-media thickness values were significantly higher in subjects with diabetes than in nondiabetic subjects. Diabetic subjects with skin autofluorescence level⩾2.47 AU (median value) had higher levels of N-terminal pro-B-type natriuretic peptide (p = 0.006), high-sensitive cardiac troponin T (p < 0.0001), pentosidine (p = 0.011) and maximum intima-media thickness (p = 0.017) compared to those with skin autofluorescence level <2.47 AU. A multivariate regression analysis using variables that were significantly correlated with high-sensitive cardiac troponin T and N-terminal pro-B-type natriuretic peptide, revealed that estimated glomerular filtration rate (β = –0.364, p < 0.001) and skin autofluorescence (β = 0.254, p = 0.0022) were independent determinants of high-sensitive cardiac troponin T, but the variables that were significant in the univariate analysis were no longer predictors for N-terminal pro-B-type natriuretic peptide. Skin autofluorescence measured with the AGE Reader™ could be an easy and noninvasive surrogate marker for identifying diabetic subjects at high risk for subclinical cardiac injury.
Keywords
Introduction
Diabetes is one of the established risk factors for cardiovascular disease (CVD),1,2 and the increased risk of CVD in diabetes causes a growing economic and public health burden. Concerns about CVD have traditionally focussed on atherosclerotic vascular events, such as myocardial infarction, stroke, and peripheral artery disease. Heart failure is one of the earliest, most common, and most serious CVD in patients with diabetes, and therefore can no longer be ignored.3,4 N-terminal pro-B-type natriuretic peptide (NT-proBNP), which is indicative for myocardial stretch and volume overload has been shown to be a useful cardiovascular risk predictor in the general population and among patients with diabetes.5 –8 High-sensitive cardiac troponin T (hs-cTnT) is another cardiac biomarker, which represents subclinical myocardial injury of nonatherosclerotic origin with only a moderate association of risk of coronary heart disease. 9 Recently, several clinical studies have shown that circulating hs-cTnT levels are higher among subjects with diabetes than nondiabetic subjects and are associated with diabetic complications,10,11 providing incremental risk prediction beyond commonly known risk factors and biomarkers. 12 Poor glycemic control has been reported to be associated with increased risk of heart failure in diabetes, 13 and there is also evidence that cardiac damage is often present at the time of clinical diagnosis of diabetes. 14
Advanced glycated end-products (AGEs), which accumulate on long-lived proteins in tissues, play an important role in various diseases such as diabetes, chronic kidney disease (CKD), and CVD.15 –17 It was shown that skin collagen AGEs predict the risk of development or future progression of microvascular disease independent of HbA1c in the Diabetes Control and Complications Trial (DCCT) Skin Collagen Ancillary Study,18,19 suggesting that the tissue accumulation of AGEs is a key factor in the pathogenesis of diabetic complications,15,20 which can explain the so-called metabolic memory. Furthermore, increased deposition of AGEs and collagen in the myocardium have been reported in diabetes, contributing to excessive diastolic left ventricular stiffness and development of heart failure. 21
A new noninvasive device, the skin autofluorescence (AF) reader, has been developed to quantify skin AF, which has been shown to correlate with the accumulation of skin AGEs assessed by skin biopsy. 22 Skin AF is elevated in diabetes and end-stage renal disease 23 and is associated with diabetic microvascular complications24,25 and CVD.26,27 A recent study revealed that skin AF is an important determining factor for hs-cTnT elevation in Japanese hypertensive patients with no history of cardiovascular events. 28 However, there have been no studies regarding the relationship between skin AF and hs-cTnT in patients with diabetes. The aim of this study is to evaluate the association between skin AF and cardiovascular biomarkers including hs-cTnT in Japanese subjects with diabetes.
Materials and methods
A cross-sectional study of subjects with diabetes attending the outpatient diabetes clinic was performed during the period from January 2017 to December 2017. The inclusion criteria were age at diagnosis of diabetes is ⩾30 years and known duration of diabetes ⩾3 years. Patients were excluded if they were pregnant, had anaemia (haemoglobin <10 g/dL), infections, malignancies, and extreme sunburn, steroid therapy or were on steroid therapy or haemodialysis. No patients had a history of cardiovascular events such as ischaemic heart disease, stroke, peripheral artery disease or heart failure. As control subjects, 32 nondiabetic healthy subjects were also enrolled. Clinical assessments included a medical interview, a physical examination and laboratory tests. Smoking habits were classified as either current smoker or not. The study was conducted in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration. Informed consent was obtained from all participants before they were included in the study.
Blood samples were collected in the morning after 12 h of fasting. HbA1c was measured with the high-performance liquid chromatography method (ARKRAY, Kyoto, Japan). Serum creatinine, total cholesterol, high-density cholesterol (HDL-C), low-density cholesterol (LDL-C), triglyceride (TG), pentosidine concentrations were measured by SRL. Inc. (Tokyo, Japan). Urine albumin levels sampled from random voided urines were measured by immunoturbidimetric assay and expressed as urinary albumin-to-creatinine ratio (UACR). The estimated glomerular filtration rate (eGFR) was calculated using the following equation for the Japanese. 29 Hs-cTnT and NT-proBNP were measured by SRL. Inc. (Tokyo, Japan) with electrochemiluminescence immunoassays using commercial kits (Roche Diagnostics, Switzerland).
Nephropathy was diagnosed if patients had an UACR ⩾30 µg/g Cr or/and eGFR <30 mL/1.73 m² according to the Classification of Diabetic Nephropathy 2014 in Japan. 30 Diabetic retinopathy (DR) was diagnosed if patients had nonproliferative retinopathy (NPDR), or proliferative retinopathy (PDR), while patients without any abnormalities were denoted non-DR (NDR). 31 Diabetic peripheral neuropathy (DPN) was diagnosed according to the recent minimal criteria for typical DPN, 32 which was considered to occur if two or more of the following neuropathic symptoms were observed: decreased sensation, positive neuropathic sensory symptoms, symmetric decreased distal sensation and unequivocally decreased or absent ankle reflexes.
Skin AF was measured using the AGE Reader™ (DiagnOptics Technologies BV, Groningen, the Netherlands), a fully automated noninvasive device that uses the characteristic fluorescence properties of certain AGEs to estimate the level of AGEs accumulation in the skin. Technical and optical details have been described more extensively elsewhere.22,25 In brief, the AGE Reader illuminates a 1 cm² area of the skin using an excitation light source with a peak excitation of 370 nm. The skin AF was determined from the ratio between the emission fluorescence in the wavelength range between 420 and 600 nm and the reflected excitation light with a wavelength between 300 and 420 nm, which was measured using a spectrometer and software. Skin AF measurement was performed on the ventral side of the forearm in the sitting position at room temperature. A series of three consecutive measurements were carried out at three different skin sites of the same forearm, taking less than a minute of time. The mean skin AF was calculated from these three consecutive measurements and used in the analyses.
Carotid intima-media thickness (IMT) was evaluated by the same experienced physician using B-mode ultrasonography with a 7.5-MHz transducer according to the guidelines of the Japan Society of Ultrasonics. 33 In short, common carotid arteries, carotid bulbs, internal carotid arteries and external carotid arteries were scanned bilaterally in longitudinal sections. The carotid IMT was measured as the distance from the leading edge of the first echogenic line to the leading edge of the second echogenic line. The highest IMT value was defined as the max-IMT and was used in this study.
All values are presented as the mean ± standard deviation (SD) or number (in parentheses) for categorical variables. Between-group comparisons were performed using the Student’s t-test or Mann–Whitney U test, and the correlation coefficient was estimated using the Pearson’s correlation analysis. Categorical variables were compared using the Chi-square test. Univariate and multivariate multiple regression analyses were performed to determine the independent factors that contributed to increased levels of hs-cTnT and NT-proBNP. A p value < 0.05 was considered statistically significant. All analyses were performed using JMP®12.2 (SAS Institute Inc., Cary, NC, USA).
Results
Clinical characteristics
Among the 155 participants with diabetes screened, 10 subjects did not meet the inclusion criteria, and thus 145 were available for the study. Study subject characteristics are shown in Table 1. Systolic blood pressure, HbA1c, UACR, hs-cTnT, pentosidine, max-IMT and skin AF levels were significantly higher, and LDL-C and eGFR levels were significantly lower, in diabetic subjects than in nondiabetic subjects. Of all diabetic subjects, neuropathy was complicated in 48 subjects (33.1%), retinopathy in 49 (33.8%) and nephropathy in 65 (44.8%), respectively. Diabetic subjects received treatments with insulin (33.6%), calcium channel blocker (CCB; 33.1%), angiotensin converting enzyme inhibitor (ACEI)/angiotensin receptor blocker (ARB; 46.9%) and statin (53.1%). Table 2 shows the clinical characteristics of subjects with diabetes classified according to skin AF levels ⩾ median value (2.47 AU) and those <2.47 AU. Duration of diabetes, systolic blood pressure, HbA1c, UACR, pentosidine, NT-proBNP (93.4 ± 131.5 ng/L vs 49.7 ± 65.7 ng/L, p = 0.006), hs-cTnT (0.0126 ± 0.0080 µg/L vs 0.0078 ± 0.0058 µg/L, p < 0.0001), max-IMT (1.74 ± 0.74 mm vs 1.47 ± 0.56 mm, p = 0.017), skin AF (2.60 ± 0.46 AU vs 2.43 ± 0.39 AU, p = 0.016) levels, presence of microvascular complications and CCB and ACEI/ARB medications were significantly higher, and HDL-C and eGFR were significantly lower, in those with skin AF ⩾2.47 AU than in those with skin AF <2.47 AU.
The clinical characteristics of the study subjects.
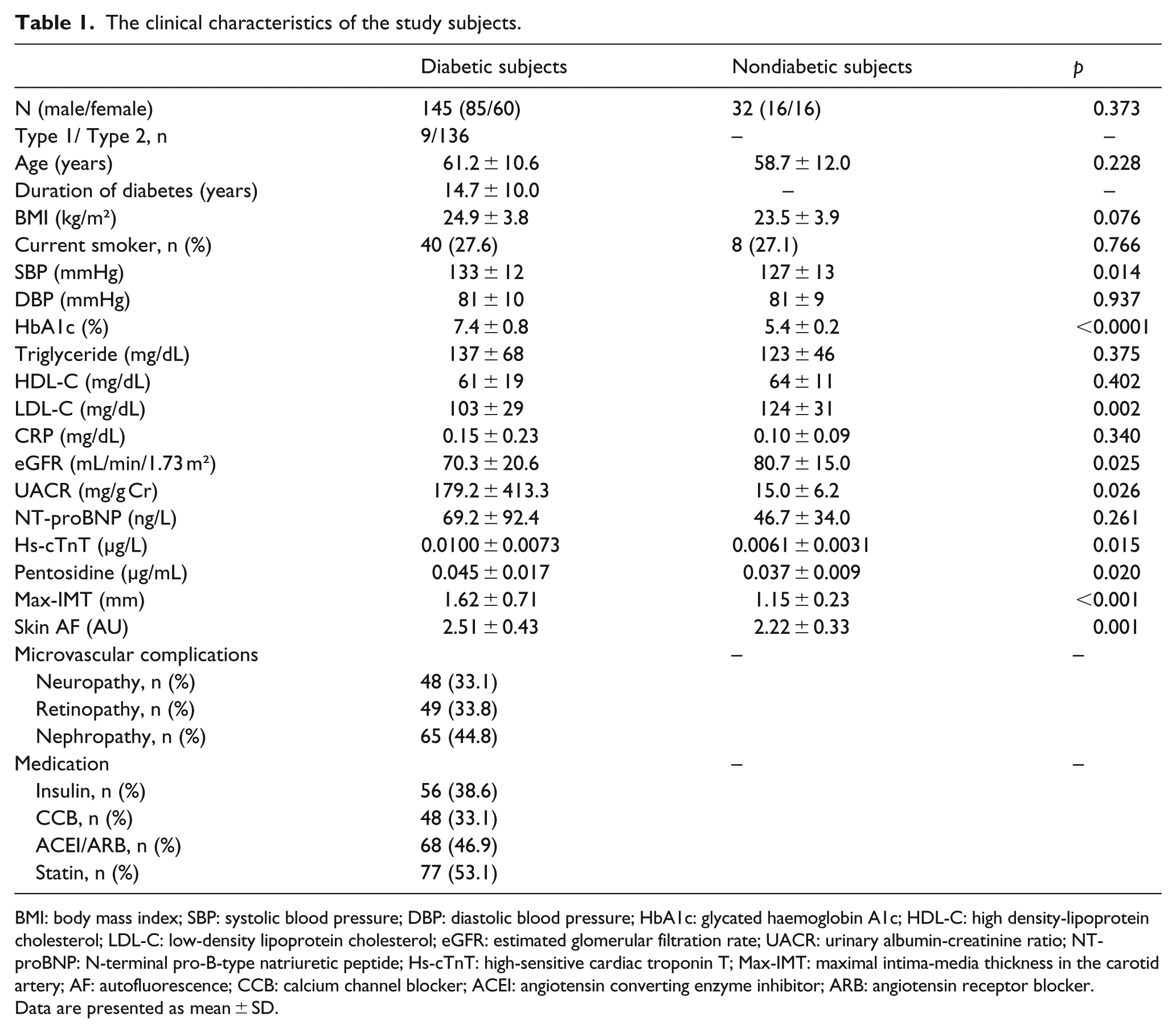
BMI: body mass index; SBP: systolic blood pressure; DBP: diastolic blood pressure; HbA1c: glycated haemoglobin A1c; HDL-C: high density-lipoprotein cholesterol; LDL-C: low-density lipoprotein cholesterol; eGFR: estimated glomerular filtration rate; UACR: urinary albumin-creatinine ratio; NT-proBNP: N-terminal pro-B-type natriuretic peptide; Hs-cTnT: high-sensitive cardiac troponin T; Max-IMT: maximal intima-media thickness in the carotid artery; AF: autofluorescence; CCB: calcium channel blocker; ACEI: angiotensin converting enzyme inhibitor; ARB: angiotensin receptor blocker.
Data are presented as mean ± SD.
The clinical characteristics of subjects with diabetes subdivided into two skin AF groups.
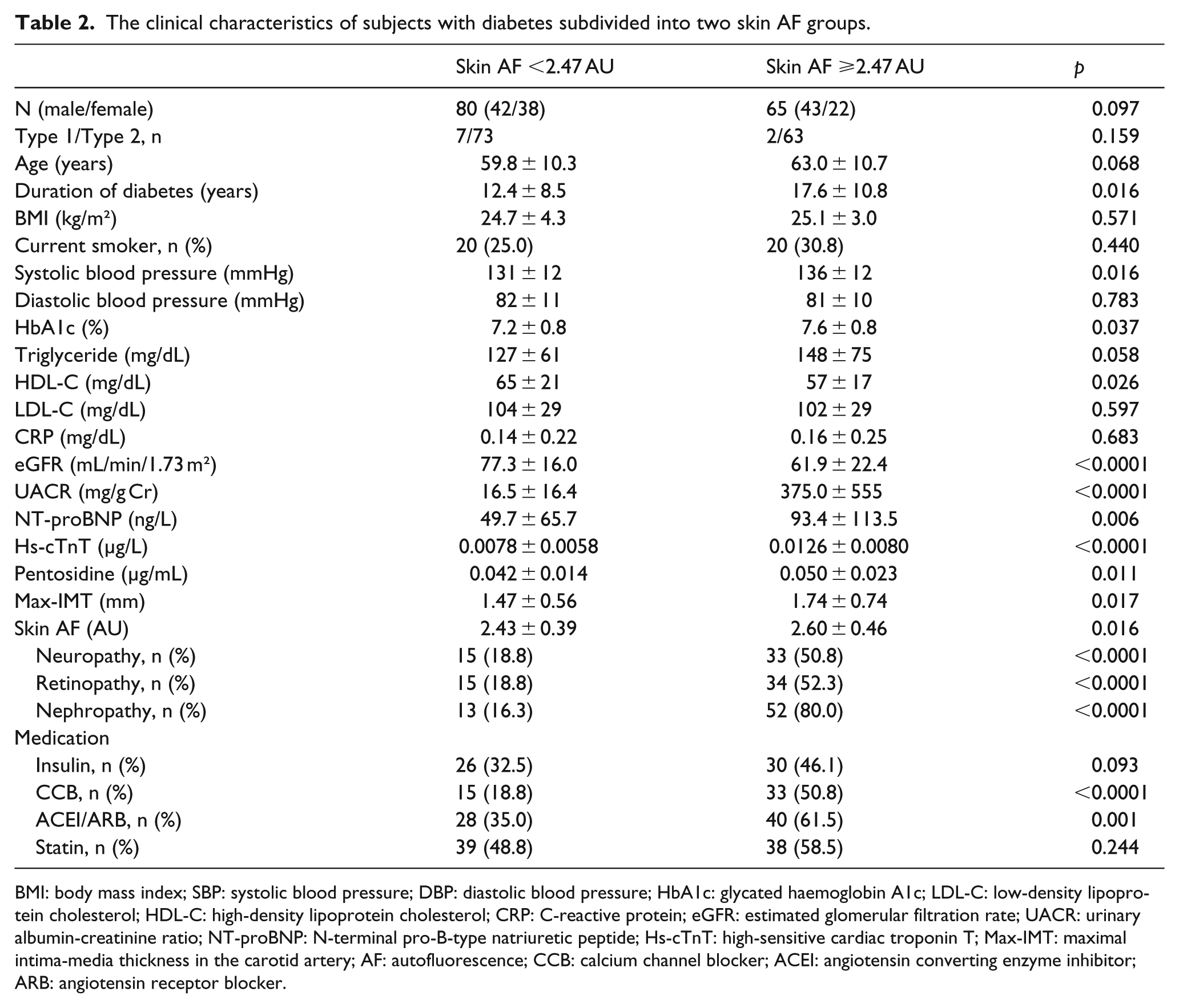
BMI: body mass index; SBP: systolic blood pressure; DBP: diastolic blood pressure; HbA1c: glycated haemoglobin A1c; LDL-C: low-density lipoprotein cholesterol; HDL-C: high-density lipoprotein cholesterol; CRP: C-reactive protein; eGFR: estimated glomerular filtration rate; UACR: urinary albumin-creatinine ratio; NT-proBNP: N-terminal pro-B-type natriuretic peptide; Hs-cTnT: high-sensitive cardiac troponin T; Max-IMT: maximal intima-media thickness in the carotid artery; AF: autofluorescence; CCB: calcium channel blocker; ACEI: angiotensin converting enzyme inhibitor; ARB: angiotensin receptor blocker.
Predictors for hs-cTnT and NT-proBNP in subjects with diabetes
Table 3 shows which factors predicted hs-cTnT and NT-proBNP levels in diabetic subjects. In the univariate regression analysis, hs-cTnT was significantly correlated with age (r = 0.234, p = 0.008), duration of diabetes (r = 0.208, p = 0.020), eGFR (r = –0.581, p < 0.0001), UACR (r = 0.376, p < 0.0001), NT-proBNP (r = 0.304, p = 0.0005), pentosidine (r = 0.262, p = 0.0037), max-IMT (r = 0.242, p = 0.0062), skin AF (r = 0.404, p < 0.0001) and the medications CCB (r = 0.303, p = 0.0005) and ACEI/ARB (r = 0.329, p = 0.0002). NT-proBNP was also correlated with age (r = 0.355, p < 0.0001), duration of diabetes (r = 0.337, p = 0.0001), systolic blood pressure (r = 0.202, p = 0.023), eGFR (r = –0.343, p < 0.0001), UACR (r = 0.221, p = 0.013), hs-cTnT (r = 0.304, p = 0.0005), skin AF (r = 0.318, p = 0.019), and the medications CCB (r = 0.212, p = 0.0167), ACEI/ARB (r = 0.241, p = 0.0063), and statin (r = 0.191, p = 0.0315). A multivariate regression analysis using the variables, that were significantly correlated with hs-cTnT and NT-proBNP in the univariate analysis, revealed that eGFR (β = –0.412, p < 0.001) and skin AF (β = 0.191, p = 0.020) were independent determinants of hs-cTnT, but those variables significant in the univariate analysis were no longer predictors for NT-proBNP.
Predictors for hs-cTnT and NT-proBNP in subjects with diabetes.
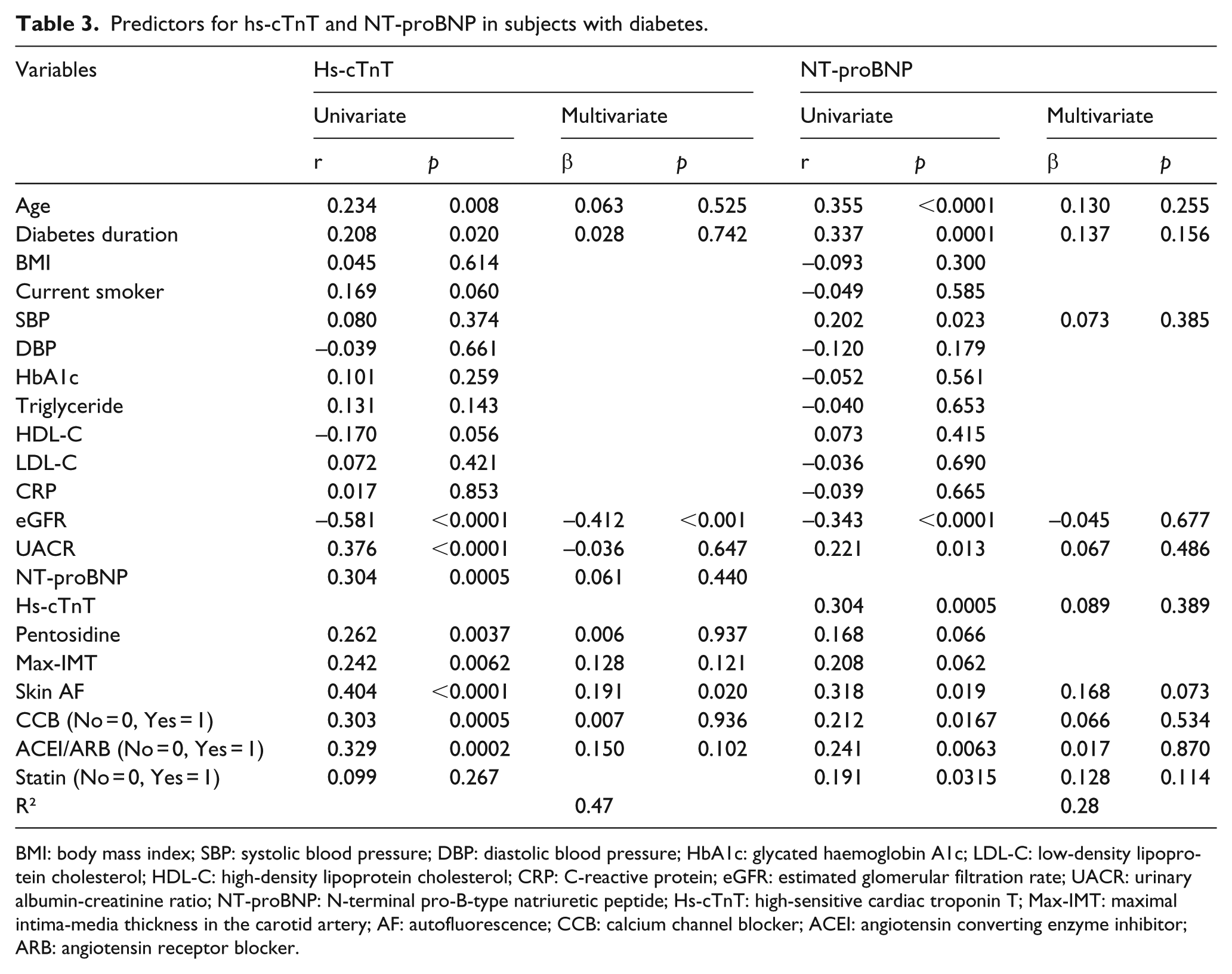
BMI: body mass index; SBP: systolic blood pressure; DBP: diastolic blood pressure; HbA1c: glycated haemoglobin A1c; LDL-C: low-density lipoprotein cholesterol; HDL-C: high-density lipoprotein cholesterol; CRP: C-reactive protein; eGFR: estimated glomerular filtration rate; UACR: urinary albumin-creatinine ratio; NT-proBNP: N-terminal pro-B-type natriuretic peptide; Hs-cTnT: high-sensitive cardiac troponin T; Max-IMT: maximal intima-media thickness in the carotid artery; AF: autofluorescence; CCB: calcium channel blocker; ACEI: angiotensin converting enzyme inhibitor; ARB: angiotensin receptor blocker.
Discussion
This study found that skin AF, a marker of AGEs accumulation in tissues, is significantly corelated with cardiac biomarkers of hs-cTnT, but not NT-proBNP in subjects with diabetes and suggested that skin AF is a noninvasive surrogate parameter for identifying diabetic subjects at high risk for subclinical cardiac injury.
It is known that hyperglycaemia and glucotoxicity can induce glycation of lipids, lipoproteins and amino acids leading to the accumulation of AGEs in tissues.15,20 Since pathologic changes of diabetic complications occur gradually over a long period, tissue AGEs that accumulate on long-lived proteins have been implicated in the pathology of diabetic complications, while short-lived glycated proteins such as HbA1c or glycated albumin are not necessarily the best predictors of these complications. The DCCT/Epidemiology of Diabetes Interventions and Complications (EDIC) study clearly demonstrated that intensive glycemic control maintained during the DCCT period continued to have a beneficial effect on diabetic complications during the EDIC follow-up study even though glycemic control worsened during the EDIC period.34,35 This phenomenon was termed ‘metabolic memory’. Furthermore, the DCCT Skin Collagen Ancillary Study18,19 showed that skin collagen AGEs predict the risk of development or future progression of microvascular disease independent of HbA1c, suggesting that the tissue accumulation of AGEs is a key factor in the pathogenesis of diabetic complications.16,22 AGEs bind to cell surface receptor for AGE (RAGE) and induce production of reactive oxygen species (ROS), which promote inflammation and fibrosis via maladaptive inflammatory gene expression such as mitogen-activated protein kinase (MAPK) and Janus kinase (JAK) pathways. 36 Thus, engagement of AGEs–RAGE interaction elicits oxidative stress generation and results in evoking inflammatory reactions. In this study, serum pentosidine levels, one of AGEs, were significantly elevated in diabetic subjects compared with nondiabetic subjects and were correlated with skin AF (r = 0.335, p = 0.0002). The cohort profiles, concerning age, smoking and male gender, which are known as conventional cardiovascular risk factors did not differ between diabetics and nondiabetic subjects. Diabetes has been regarded as a cardiovascular risk equivalent as traditional risk factors. There is a growing body of evidence that skin AF is associated with age and HbA1c, and there is evidence that skin SF increases with the degree of the presence and severity of diabetic microvascular complications.24,25
Regarding the relationship between AGEs and cardiovascular complication in diabetes, in a mouse model of type 1 diabetes, diabetes increased AGEs and RAGE levels in the heart, especially in cardiomyocytes, leading to myocardial injury; however, the administration of a RAGE antagonist prevented AGEs/RAGE signalling–mediated increases in myocardial collagen, fibrosis, stiffness and diastolic dysfunction. 37 In patients with early diabetic heart failure, elevated serum levels of AGEs have been associated with prolongation of left ventricular relaxation time. 38 Therefore, AGEs deposition and potential AGEs–RAGE interactions in diabetes may contribute to diabetic cardiomyopathy by developing myocardial fibrosis and reducing cardiac compliance. It was reported that skin AF is associated with AGE-modified tissue collagen levels 38 and cardiac tissue glycation in a human study. 39 Recently, Hitsumoto 28 has shown that skin AF is an important determining factor for the elevation of the cardiac biomarker, hs-cTnT, in hypertensive patients with no history of cardiovascular events. This study found for the first time that skin AF was significantly associated with hs-cTnT but was not associated with NT-proBNP in patients with diabetes. The prognostic cardiac biomarkers hs-cTnT and NT-proBNP have been shown to be correlated with the incidence of the development of heart failure, coronary heart disease and all-cause mortality in community-based populations and diabetes.5 –13 The Atherosclerosis Risk in Communities (ARIC) study indicated a cross-sectional association between HbA1c and hs-cTnT 9 and extended these findings to show that prediabetes and diabetes were significantly associated with the 6-year incidence of subclinical myocardial damage as assessed by the elevation of hs-cTnT, and that the incident elevations in hs-cTnT were linked to future clinical outcome, particularly heart failure and death. 12 Hs-cTnT is known to be related to age, eGFR and hypertension in a general population. In this study, systolic blood pressure, hs-cTnT and skin AF levels were significantly higher, and eGFR levels were significantly lower, in diabetic subjects than in nondiabetic subjects. A univariate regression analysis showed that hs-cTnT was significantly correlated with age, duration of diabetes, eGFR, UACR, NT-proBNP, pentosidine, max-IMT, skin AF and the medications, CCB and ACEI/ARB in diabetic subjects. After the multivariate regression analysis, skin AF and eGFR remained independent predictors for hs-cTnT.
On the other hand, BNP or NT-proBNP, released in response to ventricular volume expansion and pressure overload, has been another predictive biomarker for the subsequent risk of CVD in the general population.5 –7 The Framingham study firmly identified the epidemiologic link between diabetes and left ventricular (LV) dysfunction, and the measurement of BNP or NT-proBNP can help reliably detect the presence or absence of LV dysfunction in patients with diabetes, independently of conventional cardiovascular risk factors. 40 NT-proBNP is reported to be associated with eGFR and urinary albumin excretion. A recent report of the Action in Diabetes and Vascular Disease: Preterax and Diamicron Modified Release Control Evaluation (ADVANCE) trial found that several circulating biomarkers, such as C-reactive protein (CRP), interleukin (IL)-6, hs-cTnT, and NT-proBNP were independent predictors of the incidence of heart failure in patients with diabetes, but only NT-proBNP materially improved the predictive performance for heart failure beyond that of clinical factors. 8 In this study, a univariate regression analysis showed that NT-proBNP was significantly correlated with age, duration of diabetes, systolic blood pressure, eGFR, UACR, hs-cTnT, skin AF and the medications, CCB, ACEI/ARB and statin. However, significant associations between these variables and NT-proBNP were no longer observed after the multivariate regression analysis. The different backgrounds of study subjects may explain the different results between the ADVANCE trial and this study.
Carotid IMT is a useful surrogate marker for the presence and progression of atherosclerosis41,42 and is an excellent predictor of future cardiovascular events,43,44 while skin AF has been demonstrated to be associated with CVD.45,46 There was an association between skin AF and carotid IMT, whose value was only evaluated with max-IMT in this study because max-IMT represents a better indicator for the assessment of atherosclerosis in patients with diabetes. 47 However, a significant relationship between carotid IMT and the cardiac biomarkers of hs-cTnT and NT-proBNP was not observed in this study, indicating that the presence and progression of atherosclerosis may not necessarily coexist with myocardial injury or LV dysfunction. Since physicians have long ignored heart failure in their focus on glycemic control and their concerns about the ischaemic macrovascular complications of diabetes, it is clinically important that in patients with diabetes, heart failure is not only common but can also be prevented and treated, as recently reported.3,4 Recently, Bachmann and Wang 48 have reviewed the contributions of cardiovascular biomarkers to risk prediction for CVD in individuals with diabetes. At present, certain biomarkers and biomarker combinations, including those related to cardiac stress (hs-cTnT and NT-proBNP), inflammation (hs-CPR, IL-6 and IL-15), matrix remodelling, lipids, endothelial dysfunction (albuminuria) and diabetic pathophysiology (AGEs) can lead to modest improvements in the prediction of CVD in diabetes beyond traditional risk factors. This study also suggests that measurements of skin AF and combinations with cardiac stress biomarkers may provide a helpful risk prediction for subclinical cardiac injury in diabetes.
This study has some limitations. First, because this is a small-sample size and cross-sectional study without longitudinal assessment, the results do not necessarily indicate causal relationship between skin AF and hs-cTnT levels. Second, imaging examinations with echocardiogram, angiography and computed tomography were not performed; therefore, cardiac function and presence or absence of atherosclerotic disorders could not be evaluated. Third, administration of anti-hypertensive drugs including ACEI/ARB or statins may have influenced the results, although these medications had no significant effects in this study.
In conclusion, this study demonstrates that compared with nondiabetics, skin AF and the cardiac biomarkers of hs-cTnT and NT-proBNP are elevated in subjects with diabetes, and skin AF is significantly associated with hs-cTnT. Skin AF measured with the AGE Reader could be an easy and noninvasive surrogate marker for identifying diabetic subjects at high risk for subclinical cardiac injury. Additional prospective studies including imaging examinations are needed to validate the findings of this study.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
