Abstract
Objective
This study aimed to determine the importance of biomarkers of chronic heart failure (CHF) for assessing disease severity in euvolemic stable patients.
Patients and methods
N-terminal pro-B-type natriuretic peptide (NT-proBNP), growth differentiation factor (GDF)-15, galectin-3, cystatin-C, soluble suppression of tumorigenicity 2 (sST2), tissue type inhibitor of matrix metalloproteinase (TIMP)-1, and ceruloplasmin levels were measured in euvolemic patients with stable CHF. Severity of CHF was defined by echocardiographic and biochemical parameters.
Results
In 160 patients (123 men and 37 women, mean age: 65.8±12.2 years), we found strong associations between NT-proBNP and bilirubin levels (r = 0.434) and the estimated glomerular filtration rate (r = −0.321). GDF-15 and cystatin-C levels were significantly correlated with parameters of kidney function. In multivariable regression analysis, NT-proBNP levels were associated with the left ventricular ejection fraction and left ventricular end-systolic volume (coefficient of determination R2 = 0.777). Additionally, GDF-15 levels were correlated with urea levels (R2 = 0.742), and cystatin C levels were correlated with urea and bilirubin levels (R2 = 0.732).
Conclusion
Besides NT-proBNP, GDF-15 and cystatin C are promising biomarkers for establishing the severity of disease in euvolemic patients with stable CHF.
Keywords
Introduction
Natriuretic peptide levels are helpful in the diagnosis, prognostic stratification, and monitoring of treatment effect in acute and chronic heart failure (CHF). 1 Measurement of N-terminal pro-B-type natriuretic peptide (NT-proBNP) levels is recommended for making a diagnosis and prognostication in ambulatory patients with CHF. 2 Natriuretic peptide levels may be used for ruling out heart failure (HF) in untreated outpatients and cardiac causes of acute dyspnea. 3 NT-proBNP levels are useful in obtaining prognostic information in patients with CHF and have been tested as a guide for therapy in euvolemic subjects.3,4
Research has identified novel biomarkers that are involved in the processes associated with HF and they may be used together with NT-proBNP levels in prognostic stratification in CHF. 5 Several biomarkers have been investigated in the different HF populations as follows. 6 Galectin-3 is involved in fibrotic processes. 7 Galectin-3 levels are increased in patients with HF, both in ischemic and non-ischemic cardiac dysfunction, and are predictive for the risk of death.8,9 Soluble suppression of tumorigenicity 2 (sST2) protein is a member of the interleukin 33 receptor family and has a cardioprotective effect against myocardial fibrosis and hypertrophy. 10 Serum sST2 levels are increased in response to mechanical wall stress and are associated with the risk of death and hospitalization for HF.11,12 Levels of sST2 are additive to natriuretic peptide levels in prognostic stratification in patients with acute decompensation of CHF.13–15 A decrease in sST2 levels before and after treatment of HF may reflect the effect of specific therapy. 16 Growth-differentiation factor-15 is a cytokine, and its levels are increased in response to myocardial pressure and volume overload. Increased GDF-15 levels are predictive for the risk of death in HF.17,18 Matrix metalloproteinases (MMPs) and tissue inhibitor of metalloproteinase-1 (TIMP-1) levels in blood may reflect remodeling of the extracellular matrix. MMP levels are associated with an increased risk of death in HF populations. Serum MMP-2, but not MMP-3, MMP-9, and TIMP-1 levels, predict mortality of patients with CHF.19,20 Cystatin C is a biomarker that indicates kidney function and is also a marker of inflammation. 21 Cystatin C levels predict mortality and HF events in patients with stable CHF.22,23 Ceruloplasmin serves as an acute phase inflammation reactant and is involved in copper transport and in iron metabolism. 24 Ceruloplasmin levels in patients with CHF are correlated with the severity of HF and with NT-proBNP levels, and are predictive for mortality. 25
This study aimed to determine the role of biomarker levels in assessment of the degree of cardiac dysfunction and the severity of HF in euvolemic patients with stable CHF.
Patients and methods
Patients
We studied a cohort of consecutive euvolemic patients with stable CHF from a tertiary care HF clinic who had a clinic visit between 1 January 2016 and 30 September 2016. Only patients without signs of lung or systemic congestion on a physical examination were included in the study. The diagnosis of CHF was previously confirmed by the presence of symptoms, evidence of structural heart disease, elevated natriuretic peptide levels, and requirement for HF therapy. A stable course of HF was determined by an absence of hospitalization, no change in pharmacotherapy, and no change in diuretic dose by >50% within 12 months before study entry (inclusion criteria). Patients with clinical or laboratory suspicion of infection and patients with chronic inflammatory disease and with known cancer or other malignancies were not included in the study. All patients provided informed consent and Na Homolce Hospital Ethics Committee approved the study protocol.
Methods
Blood samples for biomarkers and biochemical parameters were collected in the morning in fasting patients.
The severity of HF was assessed by NT-proBNP levels and other biochemical parameters with a prognostic effect in CHF (parameters of renal function: levels of urea, creatinine, sodium, bilirubin, and hemoglobin, and the estimated glomerular filtration rate [eGFR]). The severity of cardiac dysfunction was assessed by echocardiography.
NT-proBNP levels were measured using a validated, commercially available sandwich electro-chemiluminescence immunoassay on a Cobas e411 analyzer (Roche Diagnostics, Mannheim, Germany) using established methodology. Normal NT-proBNP levels in healthy subjects are <125 ng/L. Cerulopasmin levels were measured by turbidimetric immunoassay on an AU 400 analyzer (Olympus Life and Material Science Europa GmbH, Hamburg, Germany). Normal ceruloplasmin levels in healthy subjects range from 0.220 to 0.400 g/L. To analyze serum levels of galectin-3, sST2, GDF-15, cystatin C, and TIMP-1 in patients with HF, we used the RayBio™ Custom Quantibody Array (Raybiotech, Inc., Norcross, GA, USA) according to the manufacturer’s protocol. Briefly, array glass slides were pre-treated with blocking buffer at room temperature for 30 minutes and incubated with two-fold diluted serum and standards overnight at 4°C. The array glass slides were then washed, incubated with biotin-conjugated detection antibodies at room temperature for 5 hours, washed again, and developed for 2 hours with Cy3 equivalent dye-conjugated streptavidin. The arrays were scanned with a GeneTAC UC4Microarray Scanner (Genomic Solutions Inc., Ann Arbor, MI, USA) and analyzed using RayBio Analysis Tool software (Raybiotech, Inc.). Signals were normalized using internal, positive, and negative controls included on the array. We obtained galectin-3 and ST2 levels by subtracting background staining, normalizing to positive controls on the same array glass slide, and calculating the standard curve. Standard biochemical analyses were performed on the Unicel DxC 800 analyzer (Beckman Coulter Company, Krefeld, Germany).
Echocardiographic studies were performed on the same clinic visit day by a broadband transducer with a transmitting frequency from 1.7 to 4.0 MHz on commercially available equipment (Vivid 7; GE Healthcare, Milwaukee, WI, USA). The Left ventricular ejection fraction (LVEF) was measured by Simpson’s method. Cardiac chamber dimensions, including left ventricular (LV) end-diastolic diameter, LV end-systolic diameter, right ventricular (RV) diameter, and left atrial (LA) diameter were measured by dual mode (2D) echocardiography. LV end-diastolic volume and LV end-systolic volume were derived from Simpson’s method.
Mitral early filling velocity (E wave) and atrial contraction velocity (A wave) were measured by pulse wave Doppler echocardiography and the E/A ratio was calculated. Mitral annular velocity (eʹ) was obtained by tissue Doppler imaging echocardiography and the mean septal eʹ and lateral eʹ values were calculated. The mean E/eʹ ratio was also calculated. 26 Systolic pulmonary artery pressure was obtained by continuous wave Doppler of tricuspid regurgitation velocity and adding estimated right atrial pressure. Right atrial pressure was assumed by the size and distensibility of the inferior vena cava. 27
New York Heart Association (NYHA) functional class was evaluated and recorded in each patient with CHF on the day of the clinic visit for HF.
Statistical analysis
All quantitative variables are shown as mean and standard deviation. Medians and ranges are also shown because the majority of variables did not have a normal distribution. To determine the relationship between biomarkers and biochemical and echocardiographic parameters, non-parametric Spearman’s correlation coefficients were calculated. For comparison of novel biomarkers among NYHA stages (1 vs. 2 vs. 3), non-parametric Kruskal–Wallis ANOVA with multiple comparisons of mean ranks was used.
To investigate predictors of novel biomarkers, including GDF-15, galectin-3, cystatin C, sST2, TIMP-1, and NT-ProBNP, multivariable regression analysis using the “whole model” technique for each individual biomarker separately was performed. For dependent variables, we used demographic (age, sex), clinical (NYHA), echocardiographic (LVEF, LV end-diastolic volume, LV end-systolic volume, LA, RV, E/eʹ, and estimated pulmonary artery pressure), and biochemical (Na, K, urea, creatinine, bilirubin, hemoglobin and eGFR) parameters. Correlations were rated according to Chan. 28
STATISTICA version 9 (Statsoft, Tulsa, OK, USA) was used for statistical analysis. A p value < 0.05 was considered to be statistically significant.
Results
A total of 160 patients (123 men and 37 women) met the criteria for the study. A total of 47% of patients had ischemic etiology of cardiac dysfunction, 39% had an implantable defibrillator, and 36% were treated with cardiac resynchronization therapy. Of 160 patients, 31% had diabetes mellitus, 63% had arterial hypertension, 14% had atrial fibrillation, and 15% had chronic pulmonary obstructive disease. The patients were treated with guideline-directed medical therapy, including beta blockers in 98%, angiotensin-converting enzyme inhibitors or angiotensin receptor blockers in 84%, and mineralocorticoid receptor antagonists in 69%. The median NYHA class was 2. The median NT-proBNP level was 121 pmol/L, mean LVEF was 30.0%, the E/A ratio was 1.0, and the E/eʹ ratio was 9.60. The patients’ characteristics are shown in Table 1. Of 149 (93%) patients who had HF with reduced ejection fraction (LVEF: ≤40%), 4 and 7 patients had HF with mid-range ejection fraction (LVEF: 40%–49%) and HF with preserved ejection fraction (LVEF: ≥50%), respectively.
Main characteristics of euvolemic patients with stable chronic heart failure (n = 160).
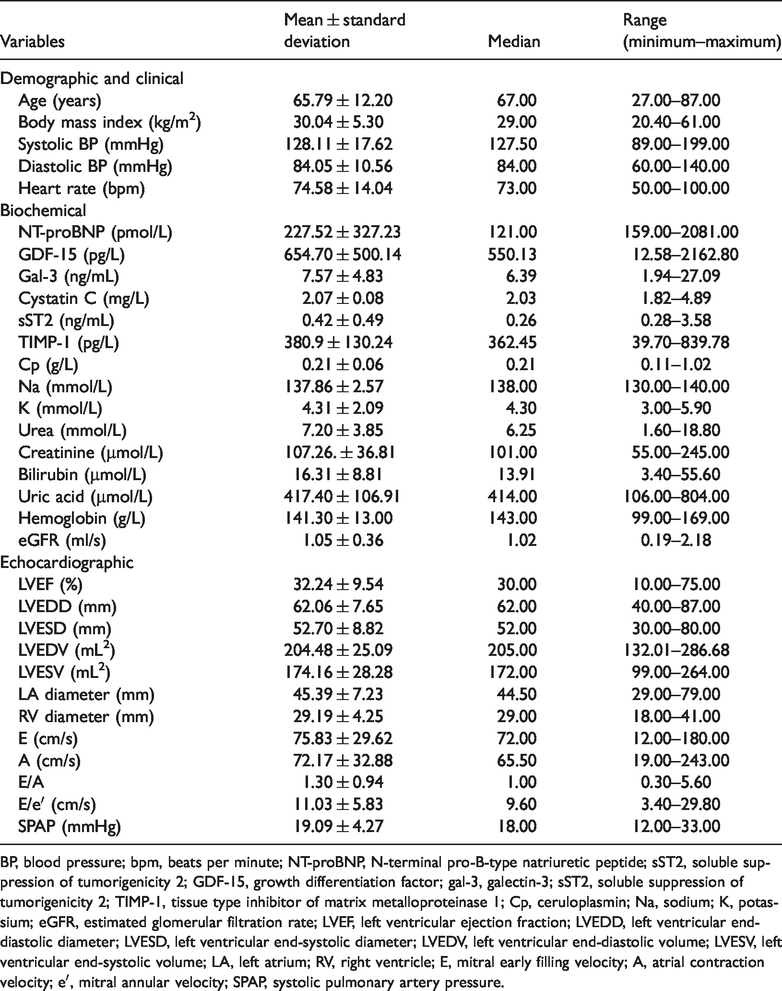
BP, blood pressure; bpm, beats per minute; NT-proBNP, N-terminal pro-B-type natriuretic peptide; sST2, soluble suppression of tumorigenicity 2; GDF-15, growth differentiation factor; gal-3, galectin-3; sST2, soluble suppression of tumorigenicity 2; TIMP-1, tissue type inhibitor of matrix metalloproteinase 1; Cp, ceruloplasmin; Na, sodium; K, potassium; eGFR, estimated glomerular filtration rate; LVEF, left ventricular ejection fraction; LVEDD, left ventricular end-diastolic diameter; LVESD, left ventricular end-systolic diameter; LVEDV, left ventricular end-diastolic volume; LVESV, left ventricular end-systolic volume; LA, left atrium; RV, right ventricle; E, mitral early filling velocity; A, atrial contraction velocity; eʹ, mitral annular velocity; SPAP, systolic pulmonary artery pressure.
Levels of sST2, GDF-15, cystatin C, TIMP-1, and ceruloplasmin were significantly correlated with NT-proBNP levels (all p < 0.01) (Table 2). The strongest association was for GDF-15 with NT-proBNP levels. NT-proBNP levels were correlated with biochemical and echocardiographic parameters reflecting the severity of heart failure, such as urea (r = 0.401, p < 0.001), creatinine (r = 0.333, p < 0.001), bilirubin (r = 0.434, p < 0.001), eGFR (r = −0.321, p < 0.001), LVEF (r = −0.259, p < 0.01), and estimated systolic pulmonary artery pressure (r = 0.392, p < 0.001), and parameters of LV diastolic function (E/eʹ ratio, r = 0.254, p < 0.01). The strength of all of these correlations was fair (Table 3a, b). Among other biomarkers, only GDF-15 and cystatin-C levels were significantly correlated with parameters of kidney function (GDF-15 vs. creatinine, r = 0.439, p < 0.001; GDF-15 vs. eGFR, r = −0.418, p < 0.001). The strength of these correlations was also fair. We found a moderate significant correlation between GDF-15 and urea levels (r = 0.475, p < 0.001). Cystatin C levels were significantly correlated with creatinine levels (r = 0.440, p < 0.001), urea levels (r = 0.471, p < 0.001), and eGFR (r = −0.451, p < 0.001). The strength of these correlations was a mixture of moderate and fair. GDF-15 levels were weakly significantly correlated with bilirubin levels (r = 0.271, p < 0.01) and hemoglobin levels (r = −0.267, p < 0.001) (Table 3b). Levels of sST2 were weakly significantly correlated with bilirubin levels (r = 0.297, p < 0.001). sST2 levels were weakly associated with the parameters of cardiac structure and function, such as the LVEF (r = −0.173, p < 0.05), LV end-diastolic diameter (r = 0.241, p < 0.01), and estimated systolic pulmonary artery pressure (r = 0.204, p < 0.05).
Correlations of novel heart failure biomarkers in euvolemic patients with stable chronic heart failure (n = 160).

*p < 0.01, **p < 0.001.
†Strength of correlations was determined according to Chan. 28
GDF-15, growth differentiation factor; gal-3, galectin-3; sST2, soluble suppression of tumorigenicity 2; TIMP-1, tissue type inhibitor of matrix metalloproteinase 1; Cp, ceruloplasmin; NT-proBNP, N-terminal pro-B-type natriuretic peptide.
Correlation coefficients and strength of correlations of novel biomarkers of heart failure with echocardiographic parameters in euvolemic patients with stable chronic heart failure (n = 160).

*p < 0.05, **p < 0.01, ***p < 0.001.
† Strength of correlations was determined according to Chan. 28
LVEF, left ventricular ejection fraction; LVEDD, left ventricular end-diastolic diameter; LVESD, left ventricular end-systolic diameter; LA, left atrium; RV, right ventricle; E, mitral early filling velocity; eʹ, mitral annular velocity; SPAP, systolic pulmonary artery pressure; NT-proBNP, N-terminal pro-B-type natriuretic peptide; GDF-15, growth differentiation factor; gal-3, galectin-3; sST2, soluble suppression of tumorigenicity 2; TIMP-1, tissue type inhibitor of matrix metalloproteinase 1.
Correlations of novel biomarkers of heart failure with biochemical parameters in euvolemic patients with stable chronic heart failure (n = 160).

*p < 0.05, **p < 0.01, ***p < 0.001.
†Strength of correlations was determined according to Chan. 28
Na, sodium; K, potassium; eGFR, estimated glomerular filtration rate; NT-proBNP, N-terminal pro-B-type natriuretic peptide; GDF-15, growth differentiation factor; gal-3, galectin-3; sST2, soluble suppression of tumorigenicity 2; TIMP-1, tissue type inhibitor of matrix metalloproteinase 1.
Patients were divided into three groups according to NYHA class and analyzed using non-parametric Kruskal–Wallis ANOVA (class 1: n = 31, 19.38%; class 2: n = 88, 55.00%; and class 3: n = 41, 25.62%). Significant differences in NYHA class were found for the following biomarkers: GDF-15 (class 1 vs. class 3; p = 0.010), cystatin C (class 1 vs. class 3; p = 0.029), TIMP-1 (class 1 vs. class 3; p = 0.023), and NT-proBNP (class 1 vs. class 3; p = 0.010).
To identify predictors for biomarkers of HF, multivariable regression analysis for each individual biomarker was performed. Models for NT-proBNP, GDF-15, galectin 3, and cystatin C were significant and adequately interpolated the data (all p < 0.05). The coefficients of determination (R2) and significant parameters, as well as their unstandardized (B) and standardized (beta) coefficients for all models, are shown in Table 4.
Multivariable regression analysis of demographic (sex – M vs. F, age), clinical, echocardiographic (NYHA classes – 1 vs. 2 and 1 vs. 3, LVEF, LVEDV, LVESV, LA, RV, E/é, SPAP), and biochemical (Na, K, urea, creatinine, bilirubin, hemoglobin, eGFR) parameters associated with novel biomarkers in euvolemic patients with chronic heart failure (n = 160).

Only statistically significant parameters are shown. R2 expresses the percentage variation of the matrix of the dependent variable explained by the independent variables.
The beta coefficient expresses the relative contribution of each independent variable in prediction of the dependent variable. The p values represent the statistical significance of each independent variable.
NT-proBNP, N-terminal pro-B-type natriuretic peptide; LVEF, left ventricular ejection fraction; LVESV, left ventricular end-systolic volume; Na, sodium; K, potassium; GDF-15, growth differentiation factor; NYHA, New York Heart Association; RV, right ventricle; sST2, soluble suppression of tumorigenicity 2; TIMP-1, tissue type inhibitor of matrix metalloproteinase 1.
Discussion
There were several important findings in this study. The main finding is that not only NT-proBNP may be used as a biomarker of severity of HF and cardiac dysfunction in the population of euvolemic patients with stable CHF. The role of NT-proBNP in assessing the severity of HF was confirmed in our study by multivariable regression analysis. A significant association was found for NT-proBNP levels and the LVEF and LV end-systolic volume. Some of these associations have been reported in many previous studies.29–31 Our results also confirmed these associations in the population of euvolemic patients with stable CHF on optimal medical therapy. We also found that the levels of some of biomarkers of cardiac remodeling, myofibrosis, and inflammation were correlated with biochemical and echocardiographic parameters.
GDF-15 is probably involved in myocardial fibrosis and remodeling. 17 In our study, GDF-15 levels were correlated with NT-proBNP levels and were associated with biochemical parameters of kidney function in multivariable regression analysis. However, another promising biomarker of fibrosis, galectin-3, was not correlated with NT-proBNP levels and parameters of cardiac function. A previous study showed that galectin-3 levels in patients with compensated CHF were not correlated with LVEF and functional status. 32 Furthermore, galectin-3, which promotes proliferation of cardiac fibroblasts, collagen deposition, and ventricular dysfunction, has a low diagnostic capability and better prognostic capability in patients with CHF.7–9,32–34 In our study, galectin-3 levels were associated with NYHA functional class in multivariable regression analysis.
Soluble ST2 is a protein, which serves as an interleukin-33 receptor. ST2 gene expression in cardiomyocytes is upregulated in mechanical wall stress of the LV in CHF. Levels of sST2 are predictive for mortality and morbidity and are additive to natriuretic peptides.10–12 Several studies have shown a correlation of sST2 levels with natriuretic peptide levels. 35 Our study supports this finding in euvolemic patients with stable CHF. Our study showed that sST2 levels were weakly correlated with levels of bilirubin, which is another prognostic indicator in CHF.36,37 Levels of sST2 were associated with urea levels in multivariable regression analysis, but the model was not significant.
Cystatin C levels predict cardiovascular events in patients with stable CHF. However, cystatin C levels are not correlated with parameters of cardiac function, including the LVEF. 22 In our study, cystatin C levels were correlated with NT-proBNP levels in euvolemic patients with stable CHF, and they were associated with parameters of kidney function and bilirubin levels in multivariable regression analysis. Previous studies have investigated the role of cystatin C in a broad unselected HF population with different degrees of volume status and congestion, including patients with acute decompensated HF. 23
MMPs and TIMPs are involved in cardiac remodeling. MMP levels are lower and TIMP levels are higher in patients with HF than in healthy control subjects.38,39 TIMP levels have not been tested as a marker of the severity of HF. In our study, TIMP levels were correlated with NT-proBNP l, GDF-15, and sST2 levels. TIMP levels were also associated with urea in multivariable regression analysis, but the model was not significant. Ceruloplasmin is a marker of inflammation. Ceruloplasmin levels have been shown to be correlated with NT-proBNP levels in patients with CHF, 25 which is consistent with our study.
Biomarkers of cardiac remodeling, myofibrosis, and inflammation in addition to natriuretic peptides have been implicated in HF. The strongest evidence has been reported for biomarkers of myofibrosis, including sST2 and galectin-2. Levels of sST2 and galectin-2 are predictive for mortality and morbidity and have additive prognostic value when assessed together with natriuretic peptides.40,41 However, the role of these biomarkers in assessing disease severity in euvolemic patients with CHF is limited. NT-proBNP levels remain the main parameter for assessing severity of HF in patients with stable CHF. Among others, GDF-15 and cystatin C are promising biomarkers for HF.
Conclusion
In addition to NT-proBNP, GDF-15 and cystatin C are promising biomarkers for establishing the severity of the disease in euvolemic patients with stable CHF.
