Abstract
Objective:
To examine the relationship between inflammation and central arterial stiffness in a type 2 diabetes Asian cohort.
Method:
Central arterial stiffness was estimated by carotid-femoral pulse wave velocity and augmentation index. Linear regression model was used to evaluate the association of high-sensitivity C-reactive protein and soluble receptor for advanced glycation end products with pulse wave velocity and augmentation index. High-sensitivity C-reactive protein was analysed as a continuous variable and categories (<1, 1–3, and >3 mg/L).
Results:
There is no association between high-sensitivity C-reactive protein and pulse wave velocity. Augmentation index increased with high-sensitivity C-reactive protein as a continuous variable (β = 0.328, p = 0.049) and categories (β = 1.474, p = 0.008 for high-sensitivity C-reactive protein: 1–3 mg/L and β = 1.323, p = 0.019 for high-sensitivity C-reactive protein: >3 mg/L) after multivariable adjustment. No association was observed between augmentation index and soluble receptor for advanced glycation end products. Each unit increase in natural log–transformed soluble receptor for advanced glycation end products was associated with 0.328 m/s decrease in pulse wave velocity after multivariable adjustment (p = 0.007).
Conclusion:
Elevated high-sensitivity C-reactive protein and decreased soluble receptor for advanced glycation end products are associated with augmentation index and pulse wave velocity, respectively, suggesting the potential role of systemic inflammation in the pathogenesis of central arterial stiffness in type 2 diabetes.
Keywords
Introduction
Type 2 diabetes (T2DM) is a rapidly evolving global health issue. It has been predicted that T2DM prevalence will double from 7.3% in 1990 to 15% in 2050 in Singapore. Atherosclerosis is the leading cause of morbidity and mortality in patients with diabetes. Central arterial stiffness (CAS) can aggravate systemic vasculopathy by propagating elevated systolic and pulse pressures forward, thereby accentuating global vascular injury. Pulse wave velocity (PWV) is widely used as the ‘gold-standard’ index for CAS. 1 Augmentation index (AI) is a surrogate measurement of wave reflection and CAS derived from central arterial pressure waveform. 1 CAS measured by PWV or AI has been identified as a non-invasive indicator of atherosclerosis.
Inflammation is known to play an important role in the development and progression of atherosclerosis. Elevated level of high-sensitivity C-reactive protein (hs-CRP), the most extensively studied inflammatory biomarker in heart disease, has been frequently observed in Asians with T2DM. 2 Protein glycation and formation of advanced glycation end products (AGE) have been implicated in various diabetic complications, partially mediated through their interaction with the receptor for advanced glycation end products (RAGE). The activation of RAGE by AGEs is the main pathogenic cause of vascular complications in diabetic patients. 3 The soluble receptor for advanced glycation end products (sRAGE), a C-truncated isoform, could compete with full-length RAGE for ligand binding, resulting in antiatherogenic effects. sRAGE is significantly lower in T2DM patients with microvascular complications than in patients without complications. 3
However, studies on the association of CAS with hs-CRP and sRAGE are limited and inconsistent. In healthy subjects, Yasmin et al. 2 found positive association of hs-CRP with PWV but not with AI. Hs-CRP was not associated with AI in young healthy subjects, 4 but associated with AI in middle-aged subjects.5,6 Hs-CRP was not associated with PWV in Europeans with T2DM,7,8 but associated with PWV in Asians with T2DM.9,10 Yoon et al. 3 reported positive association of sRAGE with PWV in T2DM, while we found negative association in normalbuminuric T2DM in this cohort. 11 Therefore, we aim to examine the association between hs-CRP and sRAGE with PWV and AI in a large multi-ethnic T2DM Asian cohort.
Methods
Study population and design
The Singapore Study of Macro-angiopathy and Micro-vascular Reactivity in Type 2 Diabetes (SMART2D) is a cross-sectional study conducted between August 2011 and February 2014 including a total of 2057 adults aged 21–90 years with T2DM. Inclusion and exclusion criteria of SMART2D have been previously described. 12 This study has been approved by our institution’s domain-specific ethics review committee. Individual written informed consent was obtained prior to enrolment in the study.
Clinical, biochemical and CAS measurement
Blood pressure, urinary albumin-to-creatinine ratio (UACR), estimated glomerular filtration rate (eGFR), haemoglobin A1c (HbA1c), high-density lipoprotein-cholesterol (HDL-C), low-density lipoprotein-cholesterol (LDL-C), total triglycerides and total sRAGE were measured as described previously. 12 Hs-CRP was assayed on a Roche/Integra 400 Analyser (Roche Diagnostics, Rotkreuz, Switzerland) by a particle-enhanced immunoturbidimetric method. Carotid-femoral PWV and AI were measured as described previously. 12 Only results passing the quality control algorithm by the manufacturer were used for the analysis.
Statistical analysis
For categorical analysis, hs-CRP levels were classified into low (<1 mg/L), average (1–3 mg/L) and high cardiovascular risk groups (>3 mg/L) based on the cut-off points recommended by the Centers for Disease Control and Prevention and the American Heart Association. Variables that are statistically significant in univariate analysis or with putative roles in the pathobiology of CAS were added into multivariable linear regression models. PWV or AI was entered as the dependent variable. Hs-CRP was analysed as a continuous variable and categories. sRAGE was analysed as a continuous variable. A two-tailed p value of <0.05 was considered as statistically significant.
Results
Based on the hs-CRP cut-off points, we did not observed significant difference in AI or PWV among the three categories. There are more males in moderate (47.6%) and high-risk groups (60.6%) than in low-risk group (36.5%, p < 0.001). Compared with the low-risk group, subjects in the high-risk group were younger (55.8 ± 11.2 vs 59.3 ± 10.5 years, p < 0.001), had higher BMI (29.9 ± 5.7 vs 25.4 ± 4.3 kg/m2, p < 0.001), shorter duration of diabetes (10.7 ± 8.5 vs 12.6 ± 9.3 years, p = 0.001) and higher HbA1c level (8.0 ± 1.4 vs 7.6 ± 1.2, p < 0.001). The median of hs-CRP and sRAGE levels in all subjects are 1.84 mg/L and 794.6 pg/mL, respectively.
In multivariable linear regression model, there is no association between PWV and hs-CRP. AI was associated with hs-CRP both as a continuous variable [β = 0.328, 95% confidence interval (CI): 0.001–0.654, p = 0.049] and categories (β = 1.474, 95% CI: 0.394–2.554, p = 0.008 for hs-CRP: 1–3 mg/L and β = 1.323, 95% CI: 0.216–2.429, p = 0.019 for hs-CRP >3 mg/L; Table 1).
Linear regression analysis of hs-CRP and sRAGE with CAS.
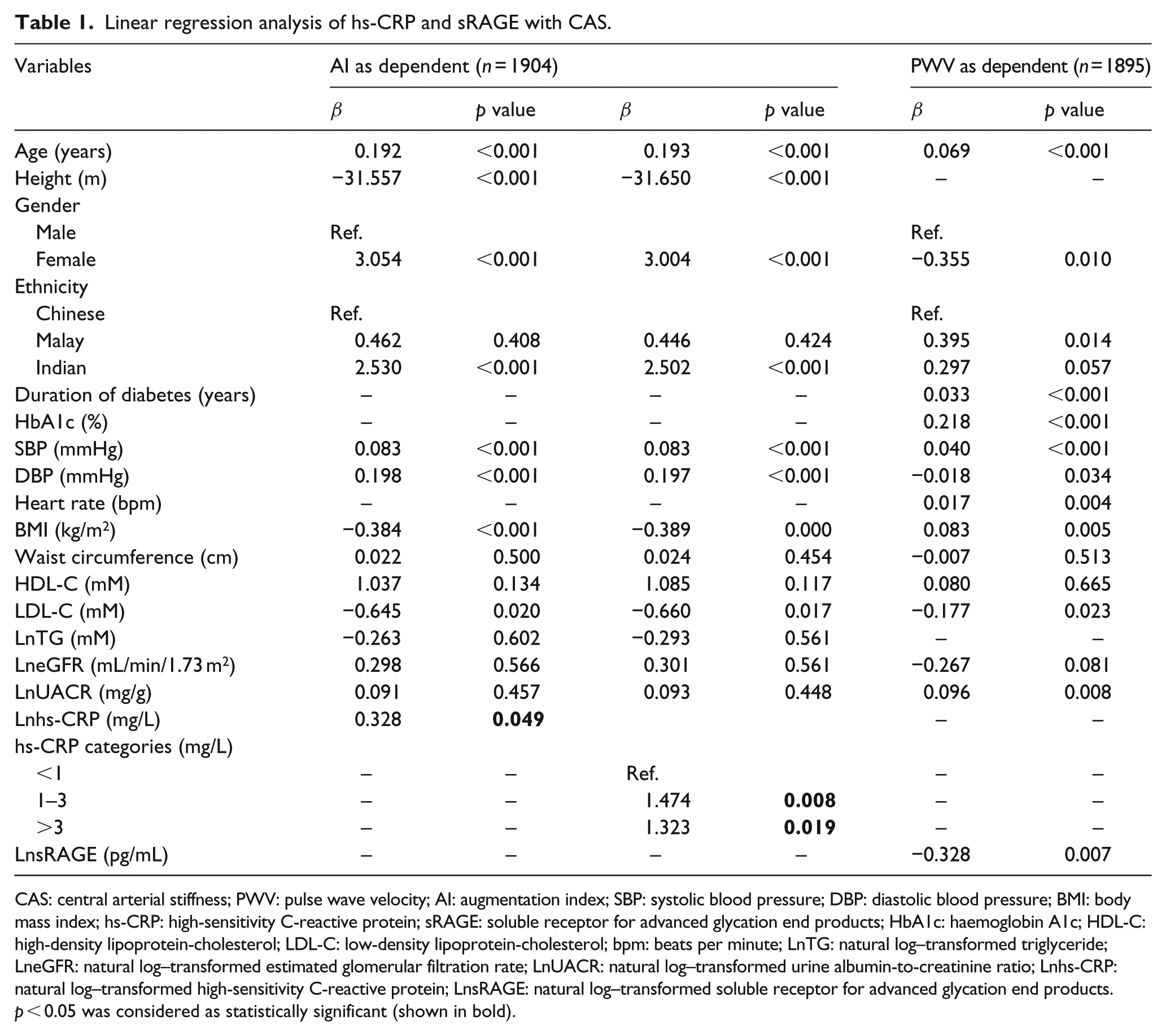
CAS: central arterial stiffness; PWV: pulse wave velocity; AI: augmentation index; SBP: systolic blood pressure; DBP: diastolic blood pressure; BMI: body mass index; hs-CRP: high-sensitivity C-reactive protein; sRAGE: soluble receptor for advanced glycation end products; HbA1c: haemoglobin A1c; HDL-C: high-density lipoprotein-cholesterol; LDL-C: low-density lipoprotein-cholesterol; bpm: beats per minute; LnTG: natural log–transformed triglyceride; LneGFR: natural log–transformed estimated glomerular filtration rate; LnUACR: natural log–transformed urine albumin-to-creatinine ratio; Lnhs-CRP: natural log–transformed high-sensitivity C-reactive protein; LnsRAGE: natural log–transformed soluble receptor for advanced glycation end products.
p < 0.05 was considered as statistically significant (shown in bold).
No association was observed between AI and sRAGE. Each unit increase in natural log (Ln)-transformed soluble receptor for advanced glycation end products (LnsRAGE) was associated with 0.328 m/s decrease in PWV (95% CI −0.564 to −0.091, p = 0.007; Table 1). Because of the possible dual roles of sRAGE as inflammation biomarker 13 and endogenous protection factor 14 under different inflammation status, we further performed linear regression analysis stratified by hs-CRP categories. We found no association in low and moderate risk groups, but significant association in high-risk group (β = −0.620, 95% CI −1.014 to −0.226, p = 0.002).
Discussion
In a large multi-ethnic T2DM Asian cohort, we demonstrated a positive association of hs-CRP with AI, and a negative association of sRAGE with PWV after controlling for major vascular risk factors and renal function. Our findings suggest the potential role of inflammation in the pathogenesis of CAS, which may contribute to the increased atherosclerosis risks in T2DM. To the best of our knowledge, this is the first and largest study on association of inflammation with CAS in Asians with T2DM.
Our finding of positive association between hs-CRP and AI is consistent with previous studies in middle and older age subjects.2,5,6 Ageing is known to be associated with activation of innate immunity, resulting in a pro-inflammatory milieu. Therefore, middle and older aged subjects may be associated with increased inflammatory tone, serving as a cause for vascular changes. Several potential mechanisms have also been proposed on the association of increased hs-CRP level and CAS, including production of adhesion molecules, pro-inflammatory cytokines and endothelial nitric oxide, vascular calcification, and structural change in the arterial wall.2,5,6
Our finding of no association between hs-CRP with PWV is consistent with a previous study in T2DM patients with a comparable hs-CRP level (1.84 mg/L). 7 In two studies reporting positive association, the median hs-CRP level of 4.5 mg/L or <1 mg/L is markedly different from our study.9,10 The difference in hs-CRP levels ascribed to subject-selection (i.e. sampling-frame) and arterial stiffness measurement may all contribute to the discrepancy in the results.
Evidence on the association of sRAGE and PWV in diabetes is very limited. In hypertensive patients with low grade of inflammation (mean hs-CRP: 2.0 mg/L), Yoon et al. 3 found positive association of PWV with sRAGE in diabetic but not in non-diabetic patients. It is possible that there is more active reaction between AGE and RAGE in diabetic patients, so sRAGE was elevated corresponding to AGE–RAGE response through a positive feedback mechanism. 3 In contrast, we found negative association of PWV with sRAGE in subjects with high grade of inflammation (hs-CRP >3 mg/L), indicating that higher sRAGE level may attenuate the deteriorating effect of inflammation on PWV. It should also be noted that the sRAGE level in Yoon’s study is much higher than in our study, thus direct comparison may not be feasible. Taken together, sRAGE level may be a marker for low-grade systemic inflammation that is associated with arterial stiffness, as well as an endogenous protection factor to compete with RAGE for ligand binding, thus preventing the adverse effect of RAGE signaling. 14 Such protective effect has been supported by the suppression of accelerated diabetic atherosclerosis with the treatment of sRAGE in animal study. 14
PWV and AI reflect different properties of the arterial tree and may have shared and unshared pathogenic drivers, such as inflammation or glycation. Previous studies found that in response to inflammation, PWV increased while AI remained or decreased in healthy subjects and patients with rheumatoid arthritis. 12 Therefore, the differential pathogenic substrates of PWV and AI may, at least partially, explain different findings when CAS is estimated using PWV and AI in our study.
The strengths of our study include a large sample size, multi-ethnicity and evaluation of CAS by PWV, the gold-standard measurement. We also recognize several limitations. All subjects were recruited from a secondary hospital and a community medical centre. Whether our findings can be extended to the general population remains to be determined. The cross-sectional design precludes any causal-inference between inflammation and other risk factors with CAS. Although we used matching and multivariable models to control potential confounders, we cannot exclude residual confounding from unmeasured variables that have been associated with CAS, such as social economics, dietary and genetic factors.
In conclusion, in a multi-ethnic T2DM Asian cohort, increased hs-CRP and decreased sRAGE are associated with AI and PWV, respectively, suggesting the involvement of inflammatory process in the pathogenesis of arterial stiffness. Therefore, amelioration of inflammatory load, for instance, through lifestyle modification or pharmacotherapy, may contribute to reduction in cardiovascular burden.
Footnotes
Acknowledgements
The authors thank Dr Darren E. J. Seah for his assistance in subject recruitment. They also thank staff from the Singapore Clinical Research Institute (SCRI) for their contribution to study protocol and database design. Study data were collected and managed using REDCap electronic data capture tools hosted at SCRI.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
This work was supported by Singapore National Medical Research Council Grant [PPG/AH(KTPH)/2011].
