Abstract
Serum soluble receptor for advanced glycation end product (sRAGE) may reflect the activity of the advanced glycation end product (AGE)–receptor for advanced glycation end product (RAGE) axis, which has been proposed as a potential mechanism linking hyperglycaemia to vascular complications in diabetes. We have investigated whether serum AGEs, sRAGE and pentosidine levels were increased and correlated with microvascular complications in type 2 diabetes mellitus (DM). We included 30 healthy control subjects, and 200 diabetic patients were divided into two subgroups: 100 patients with diabetic retinopathy and 100 patients with diabetic nephropathy. AGEs, sRAGE and pentosidine were measured in serum by enzyme-linked immunosorbent assay (ELISA). Serum AGEs, sRAGE and pentosidine levels were significantly increased in diabetic patients with retinopathy and in diabetic patients with nephropathy compared to control subjects (p < 0.001). Serum AGEs, sRAGE and pentosidine levels are positively associated with microvascular complications in type 2 DM. Multiple regression analysis reveals serum pentosidine as an independent determinant of the presence of diabetic retinopathy (p = 0.004) and the presence of hypertension (p = 0.018) and hyperlipidaemia (p = 0.036). Pentosidine levels may be a biomarker for microvascular complications in type 2 diabetic patients.
Keywords
Introduction
Diabetes mellitus (DM) is increasing at an alarming rate in Tunisian population; patients are prone to the development of macrovascular and microvascular diabetic complications that represent a major cause of morbidity and mortality. While hyperglycaemia is a key factor in the pathogenesis of diabetic microvascular complications, such as retinopathy, nephropathy and neuropathy, it is only one of the multiple factors capable of increasing the risk of macrovascular complications. Hyperglycaemia induces vascular damage probably through a single common pathway, increased intracellular oxidative stress, linking four major mechanisms, namely, the polyol pathway, advanced glycation end products (AGEs) formation, the protein kinase C (PKC)–diacylglycerol (DAG) and the hexosamine pathways.1,2 Due to prolonged high glucose in diabetes, reactive derivatives are formed via nonenzymatic condensation reaction named Millard reaction between reducing glucoses and amine residues of proteins, lipids or nucleic acids that undergo a series of complex reactions such as rearrangement, dehydration and condensation to give an irreversible cross-linked complex group of compound termed AGEs.3–5 Examples of some of the best chemically characterized AGE-modified sites in humans with diabetes are carboxymethyllysine, carboxyethyllysine and pentosidine, which act as markers for the formation and accumulation of AGEs. 6 Increased AGE formation and accumulation have been found in retinal blood vessels of diabetic patients and animals and in human serum and vitreous of diabetic patients, which were found to correlate with the degree of diabetes retinopathy.7,8 These adducts also accumulate in the kidney at a rapid rate in diabetes 9 occurring in glomerular basement membrane, mesangial cells and renal tubules.10,11 Previous studies have confirmed that AGEs and soluble receptor for advanced glycation end products (sRAGEs) interaction elicits oxidative stress generation in various types of cells and subsequently evokes vascular inflammation, macrophage and platelet activation and thrombosis, thereby playing an important role in the development and progression of vascular complications in diabetes and heart failure.12,13 In view of these considerations, we have investigated whether serum AGEs, sRAGE and pentosidine levels were increased and correlated with microvascular complications in type 2 DM.
Subjects and methods
Subjects
The local ethics committee approved this study. Written informed consent was obtained from all patients before the enrolment. In this prospective cohort study, we measured serum concentration of AGEs, sRAGE and pentosidine in 230 participants (age range, 50–75 years). Our goal is to target specific patients with only one microvascular complication (retinopathy or nephropathy). The subjects were divided into three groups: Group I is composed of healthy volunteers (n = 30) with no DM or systemic or local eye lesion or renal disease. Group II is composed of diabetic patients who developed diabetic retinopathy without nephropathy (n = 100). All patients (Group II) were recruited from Ophthalmology Department and were diagnosed by ophthalmoscopy and fundus stereophotography after dilatation by specialized ophthalmologist in University Hospital Fattouma Bourguiba at Monastir (Tunisia). Group III is composed of diabetic patients with renal function impairment without retinopathy (n = 100), who were recruited from Nephrology Department at CHU Monastir. Chronic renal disease (CRD) defined as decreased estimated glomerular filtration rate (eGFR). The eGFR was calculated using the four-variable Modification of Diet in Renal Disease (MDRD) Study equation of Levery et al. 14 Reduced renal function was defined as eGFR < 90 mL/min/1.73 m2. For each patient, a data sheet was completed with the patient’s identification code, age, sex and duration of diabetes. All diabetic patients received traditional cardiovascular drugs [statins, thiazolidinediones, angiotensin-converting enzyme (ACE) inhibitors and angiotensin II type-1 (AT-1) receptor antagonists]. For cardiovascular risk factors, the following definitions were used: individuals were defined as hypertensive if their blood pressure was >140/90 mmHg or if they were receiving any antihypertensive treatment; individuals were deemed hyperlipidaemic when their total cholesterol concentration was ≥5.68 mmol/L or their triglyceride concentration was ≥2.28 mmol/L or they were receiving lipid-lowering drugs. Exclusion criteria included macrovascular complications.
Methods
In all subjects, venous blood was collected in the morning after an overnight fast. The samples were stored at −80°C until analysis. Random plasma glucose and haemoglobin A1C (HbA1C) were measured using G7 HPLC Analyser (Tososh Europe N.V., Belgium); serum creatinine, uric acid and lipid levels [high-density lipoprotein (HDL), low-density lipoprotein (LDL), cholesterol and triglyceride] were measured using enzymatic methods by CX9 Auto-chemical analysis instrument (Beckman CX9, USA). AGEs, sRAGE and pentosidine were quantitatively determined in serum by enzyme-linked immunosorbent assay (ELISA) kits provided by ABO Switzerland Co., Ltd, China, according to the manufacturer’s instructions. Briefly, the microtiter plate has been pre-coated with an antibody specific to human AGEs (or sRAGE or pentosidine). The samples were added, and after incubation and washing, plates were incubated with horseradish peroxidase (HRP), developed with 3,3′,5,5′-tetramethylbenzidine (TMB) substrate, and OD450 was determined using an ELISA plate reader. Measurements were performed in duplicate, and the results were averaged. The intra-assay and inter-assay coefficients of variation were <7% and <9%, respectively.
Statistical analysis
All values are expressed as mean ± standard deviation (SD). A p value less than 0.05 was considered statistically significant. Significance between the two groups was determined by independent sample Student’s t test for continuous variables. Continuous data from more than two groups were compared with one-way analysis of variance (ANOVA). All analyses were performed using the SPSS program (version 17). Correlation was determined by a linear regression analysis. A multiple regression analysis was used to further explore the linear relationships between the variables. The equation was in the following form: [y = a + b1 × x1+ b2 × x2 + … + b p × x p ]. Regression variables were estimated as well as the correlation coefficient r. ANOVA was used to assess the significance of the regression with significance accepted at p < 0.05. As an alternative data evaluation method, a stepwise backward regression analysis was also used on the variables.
Results
Clinical characteristics
The clinical characteristics and laboratory data of controls and diabetic patients are shown in Table 1. The mean age of the study population with diabetes was 57 ± 12 years and that for control subjects was 52 ± 9 years. The mean duration of diabetes was 16.8 ± 9.6 years. In total, 36% of diabetic patients were hyperlipidaemia, and 48% were hypertension; 50% of all diabetic patients had retinopathy, and 50% had nephropathy. Significant differences in body mass index (p < 0.05), HbA1C (p < 0.001) and serum glucose (p < 0.001) were seen between the groups, and lipid profiles (p < 0.05) in diabetic patients were higher than in controls.
Clinical characteristics of control subjects and diabetic patients.
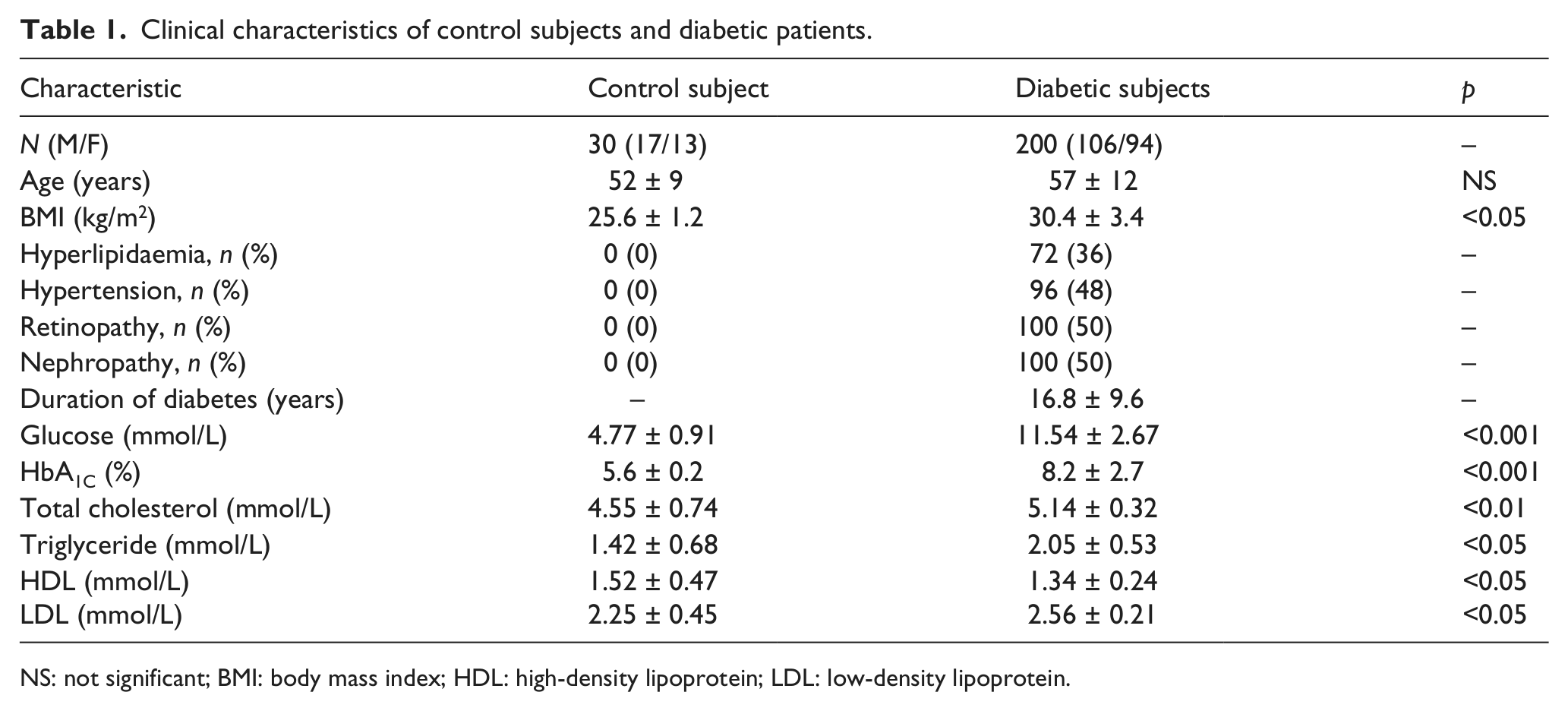
NS: not significant; BMI: body mass index; HDL: high-density lipoprotein; LDL: low-density lipoprotein.
Serum AGEs, sRAGE and pentosidine levels in diabetic microangiopathy
As shown in Table 2, the serum levels of AGEs, sRAGE and pentosidine were significantly increased in diabetic patients with retinopathy or with nephropathy than in controls (p < 0.001). Diabetic patients with retinopathy showed a significantly increased pentosidine levels than those with nephropathy (337.79 ± 63.16 pg/mL vs 287.58 ± 41.90 pg/mL, p = 0.001). No significant difference between diabetic retinopathy and diabetic nephropathy in serum AGEs and sRAGE levels had shown. These finding were illustrated in Figures 1 to 3.
AGEs, sRAGE and pentosidine levels in diabetic patients with microvascular complications compared to control subjects.
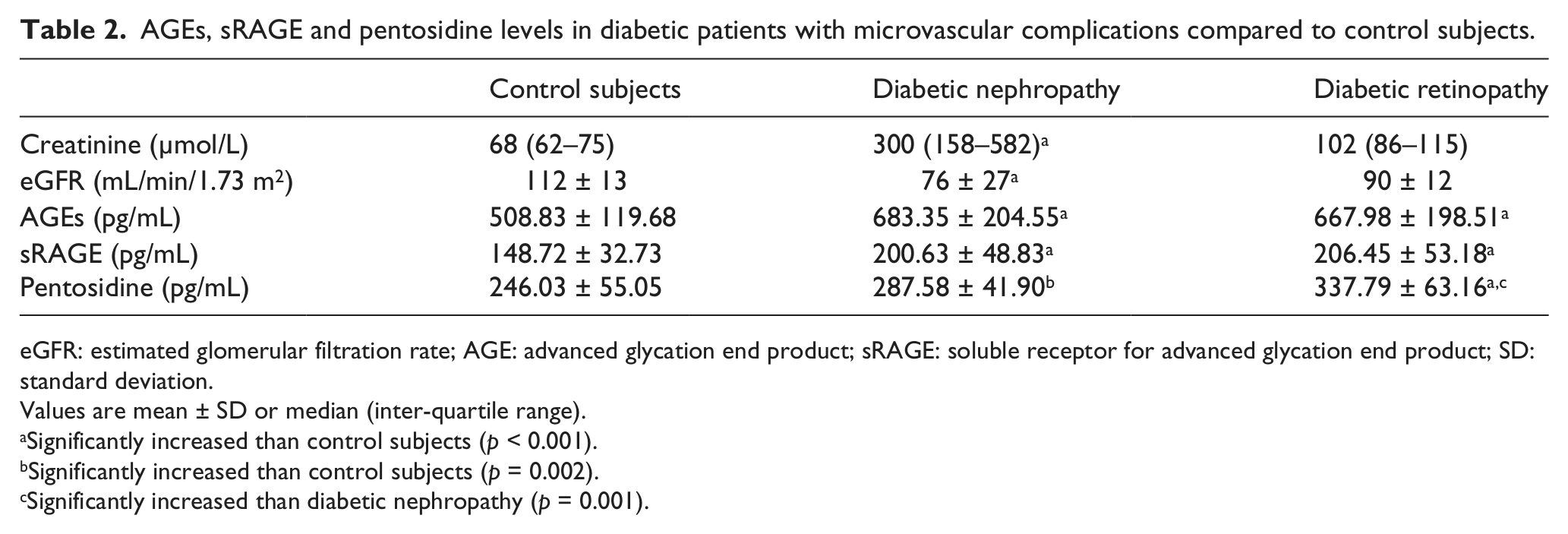
eGFR: estimated glomerular filtration rate; AGE: advanced glycation end product; sRAGE: soluble receptor for advanced glycation end product; SD: standard deviation.
Values are mean ± SD or median (inter-quartile range).
Significantly increased than control subjects (p < 0.001).
Significantly increased than control subjects (p = 0.002).
Significantly increased than diabetic nephropathy (p = 0.001).
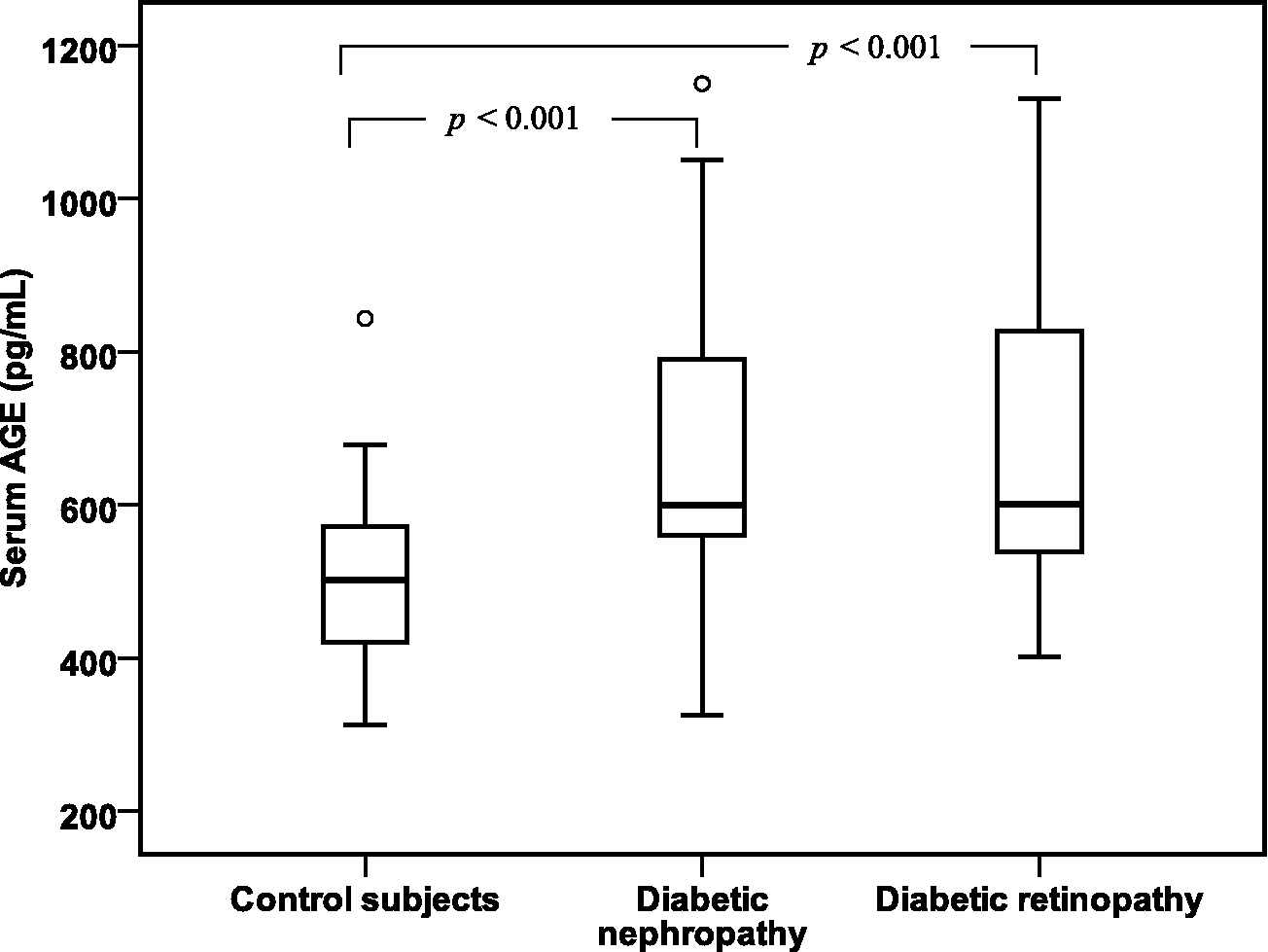
Box plots of serum AGE levels in control subjects and in diabetic patients with nephropathy and diabetic patients with retinopathy. The horizontal lines in each box plot represent (bottom to top) the 10th, 25th, 50th (median), 75th and 90th percentiles.
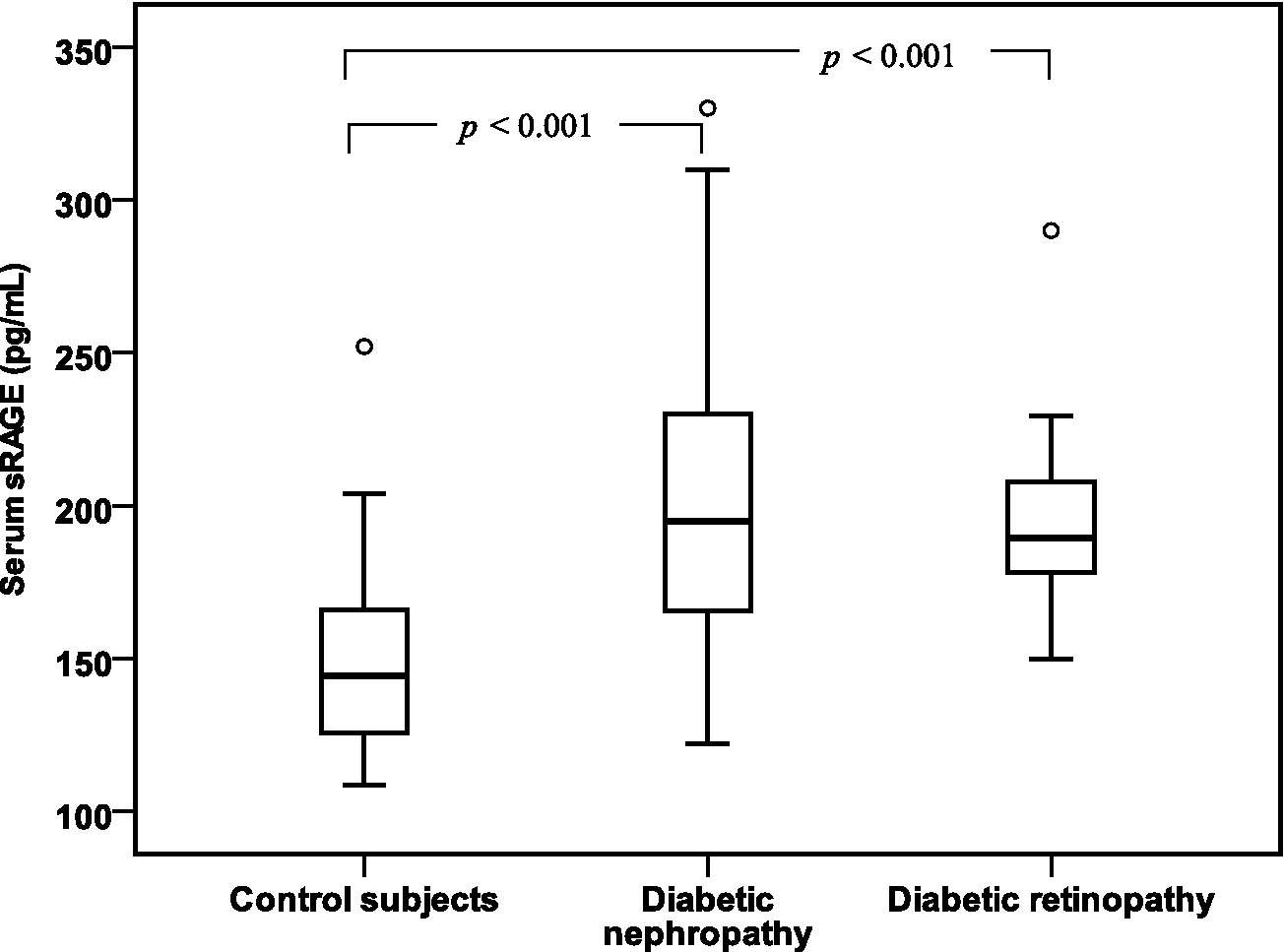
Box plots of serum sRAGE levels in control subjects and in diabetic patients with nephropathy and diabetic patients with retinopathy.
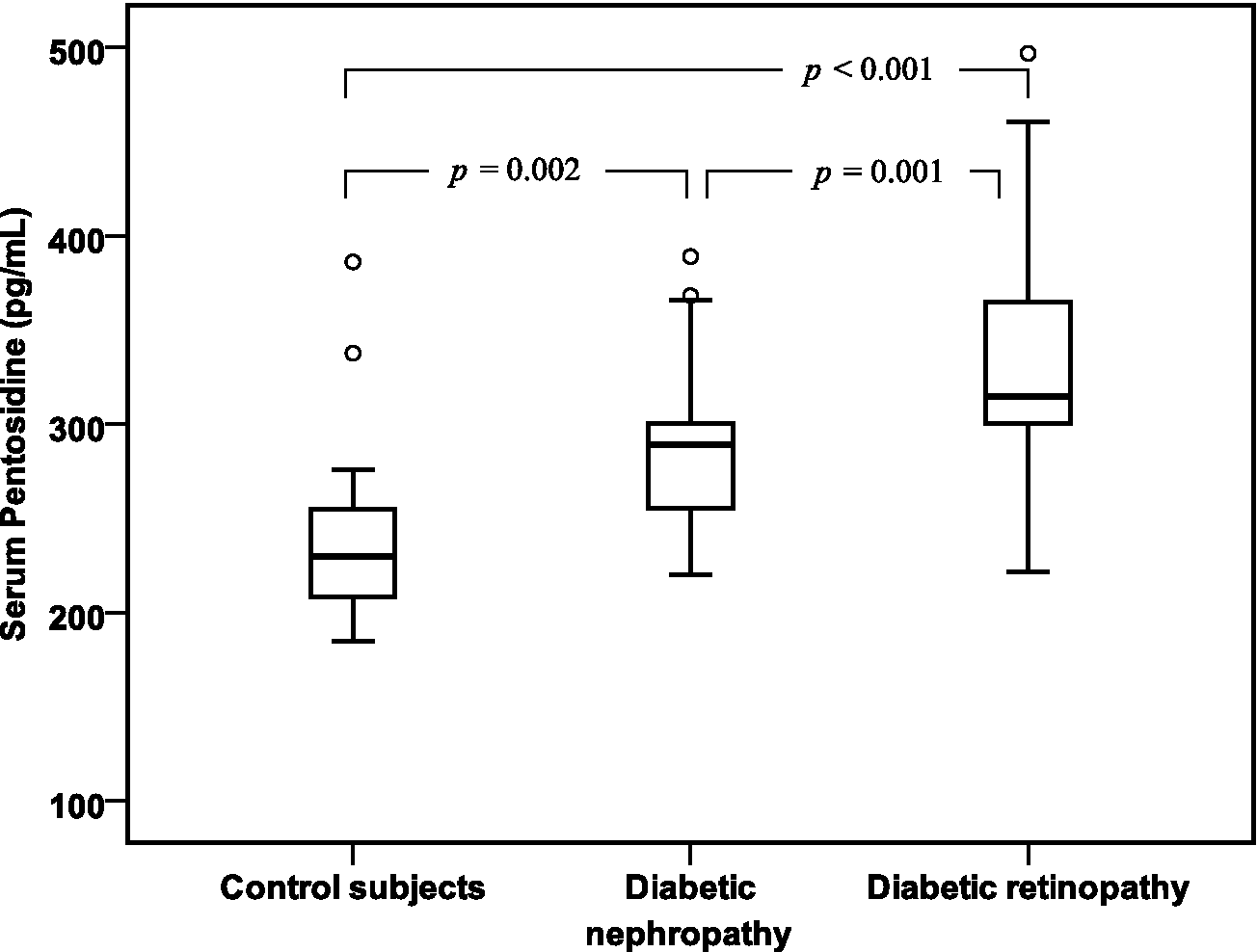
Box plots of serum pentosidine levels in control subjects and in diabetic patients with nephropathy and diabetic patients with retinopathy.
Results of univariate and multivariate analyses
A univariate analysis showed a close relationship between AGEs, sRAGE and pentosidine in all diabetic patients with retinopathy and/or patients with nephropathy. More interestingly, the multiple regression analysis revealed an independent influence of serum pentosidine on the presence of diabetic retinopathy (p = 0.004) (Table 3). Furthermore, the univariate analysis showed a close relationship between serum pentosidine and hypertension (p = 0.01) and a relationship between pentosidine and hyperlipidaemia (p = 0.03) in all diabetic patients. The multiple regression analysis revealed an independent influence of serum pentosidine on the presence of hypertension and hyperlipidaemia (p = 0.018 and p = 0.036, respectively) (Table 4).
Parameters affecting diabetic nephropathy and diabetic retinopathy: univariate and multivariate regression analyses.
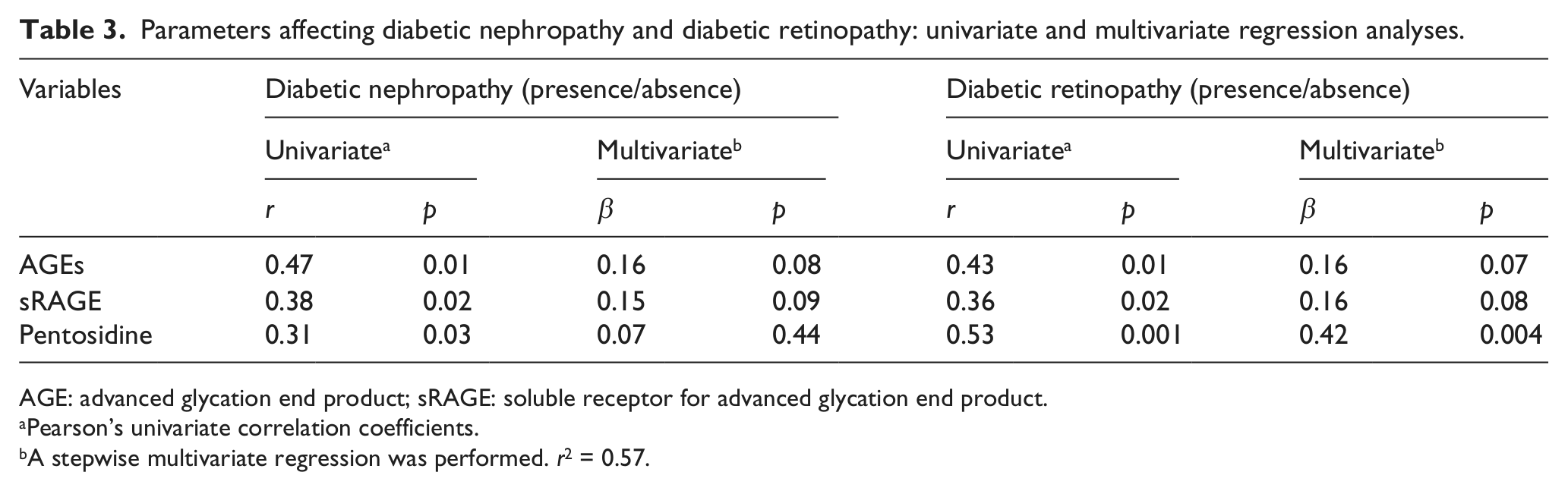
AGE: advanced glycation end product; sRAGE: soluble receptor for advanced glycation end product.
Pearson’s univariate correlation coefficients.
A stepwise multivariate regression was performed. r2 = 0.57.
Correlation between pentosidine and hypertension and hyperlipidaemia: univariate and multivariate regression analyses.

Pearson’s univariate correlation coefficients.
A stepwise multivariate regression was performed. r2 = 0.45.
Discussion
The main finding of this study is that serum AGEs, sRAGE and pentosidine levels are positively associated with microvascular complications in type 2 DM. Statins and ACE inhibitors have been shown to increase sRAGE levels in human subjects. However, our study showed that no difference on sRAGE levels between diabetic patients received only antidiabetic drugs and diabetic patients received both statins and ACE inhibitors (data not shown). Recently, similar results showed by Tam et al. 15 found that statins can increase circulating endogenous secretory receptor for advanced glycation end product (esRAGE) but not sRAGE. In this large group of diabetic patients, serum AGEs, sRAGE and pentosidine levels were significantly increased in diabetic patients with retinopathy and in diabetic patients with nephropathy. More interestingly, serum pentosidine levels were an independent risk factor for the presence of diabetic retinopathy. Pentosidine levels were increased markedly with the severity of microangiopathy (data not shown). The multiple regression analysis reveals serum pentosidine as an independent determinant of the presence of hypertension and hyperlipidaemia. The reason why pentosidine indicated microvascular complications needs to be explained. At the molecular level, AGEs including pentosidine might promote oxidative stress and endothelial cell dysfunction16,17 and induce the growth factor according to previous studies. 18 As a consequence, AGEs are related to a wide variety of systemic pathologic conditions, including ageing, hypertension, renal failure, diabetes or diabetic retinopathy and nephropathy.19,20 A number of clinical studies have reported that the formation and accumulation of AGEs have been found in retinal blood vessels of diabetic patients and animals and in human serum and vitreous of diabetic patients, which were found to correlate with the degree of diabetic retinopathy. 7 Retinal pericytes that play an important role in the maintenance of microvascular homeostasis have been shown to accumulate AGEs during diabetes, which are implicated in endothelial cell injury and blood–retinal barrier dysfunction. 21 In addition, AGEs increase vascular endothelial growth factor (VEGF), monocyte chemoattractant protein-1 (MCP-1) and intercellular adhesion molecule-1 (ICAM-1) expression in microvascular endothelial cells through cellular reactive oxygen species (ROS) generation. 22 AGEs also activate nuclear factor-kappa B (NF-κB) and nicotinamide adenine dinucleotide phosphate hydrogen (NADPH) oxidase with an increase in ROS and apoptosis of pericytes and other retinal cells.23,24 AGEs disturb microvascular homeostasis through interaction with receptor for advanced glycation end product (RAGE). Furthermore, Sun et al. 25 recently reported that specific AGE combinations (carboxyethyllysine and pentosidine) were strongly associated with complications indicating a link between AGE formation and processing with the development of diabetic retinopathy. Because the kidney is the main elimination site for pentosidine, 26 several studies have examined serum pentosidine concentrations in patients with diabetes with overt nephropathy or those with CRD, reporting elevations.27–29 Especially, a dramatic increase in plasma pentosidine was reportedly found in patients with end-stage renal disease. 30 Renal insufficiency thus may lead to accumulation of pentosidine in the blood because renal is the major determinant of serum pentosidine. 31 We speculated that pentosidine might accelerate the development of microvascular complications both by accumulation of AGEs in the vessel walls and by causing endothelial dysfunction, as mediated by the AGE–RAGE axis activity.
Numerous studies have focused on the effect of renal function on AGE levels. In agreement with a study done in type 2 diabetic patients, we found that pentosidine levels increased with renal function impairment. 32 In experimental study, Waanders et al. 33 found that pentosidine accumulates in non-diabetic proteinuric kidney in damaged tubules. As reviewed previously, AGE-modified proteins initiate a range of cellular responses including enhanced growth factor expression, cellular proliferation and apoptosis, angiogenesis and tissue remodelling by binding to AGE-specific receptors that might be involved in nephrotoxicity. Receptor for AGEs contributes to mesangial activation and transforming growth factor-β production, processes that converge to cause albuminuria and glomerulosclerosis. 34 These data suggest that renal AGE accumulation that has resulted from a primary renal disorder, once present, can itself become a perpetuating factor in ongoing renal damage. Recently, Yamagishi et al. 35 showed the role of AGEs and oxidative stress in vascular complications in diabetes. The authors demonstrated that AGEs induce mesangial cell loss and dysfunction, glomerulosclerosis and tubulointerstitial fibrosis. Taking into account the fact that patients with diabetes are prone to microvascular disease, our observations suggest that serum pentosidine might be a useful biomarker of diabetic microvascular complications.
Footnotes
Acknowledgements
We thank Professor Mohsen Hassin and his colleagues of the Regional Blood Transfusion Centre in Monastir (Tunisia) for recruiting the healthy controls. Kerkeni expresses his sincere appreciation to Professor Francois Trivin (Saint-Joseph Clinical Laboratory Medicine, Paris, France) for his continued direction and encouragement throughout the study.
Conflict of interest
The authors declare that they have no conflict of interest.
Funding
This research was supported by a grant from the ‘Ministère de l’Enseignement Supérieur et de la Recherche Scientifique’ UR03ES08 ‘Nutrition Humaine et Désordres Métaboliques’ University of Monastir.
