Abstract
Malondialdehyde (MDA), an end product of lipid peroxidation and biomarker for oxidative stress, and its soluble receptor (sRAGE) were evaluated in 42 patients with type 1 diabetes mellitus, but without chronic complications, during the early years after diagnosis (0–10 years) and through the further progression of the disease (10–20 and > 20 years after diagnosis). Clinical and biochemical parameters of the cohort of diabetic patients were compared with those determined in 24 healthy individuals. The median levels of MDA in plasma were similar in type 1 diabetes patients and in healthy subjects. In contrast, statistically significant increases were detected in the median values of sRAGE in patients with type 1 diabetes compared with healthy subjects (2423.75 versus 1472.75 pg/ml; p=0.001, Mann–Whitney test). However, no significant between-group differences (p>0.05) were observed in levels of sRAGE when diabetic patients were grouped according to time elapsed after diagnosis. It is concluded that increased plasma levels of sRAGE in type 1 diabetes may provide protection against cell damage and may be sufficient to eliminate excessive circulating MDA during early years after disease onset.
Keywords
Introduction
Vascular complications, typically caused by micro- and macro-angiopathies, are common in individuals with poorly controlled type 1 diabetes mellitus (T1DM), and their development increases significantly the risk of patient morbidity and mortality. 1 It is known that the formation and accumulation of advanced glycation end-products (AGEs) progress at an accelerated rate in T1DM, and a number of studies have indicated a role for AGEs and their receptor (RAGE) in the pathogenesis of diabetic complications.2,3
In healthy adults, RAGE is expressed at low levels in a range of cell types including peripheral blood-derived monocytes, lymphocytes, endothelial cells, vascular smooth muscle cells, glomerular epithelial cells or podocytes and neurons. 4 The interaction of AGEs with RAGE on macrophages causes oxidative stress and induces the activation of nuclear factor-κB, which modulates the transcription of genes coding for various pro-inflammatory molecules. 5 In addition, the binding of AGEs to RAGE is known to cause phenotypic changes in various cells, leading to the pathogenesis of diabetic retinopathy, nephropathy and macro-angiopathies. 4
Soluble forms of the RAGE receptor (sRAGE) have been detected in human plasma, 6 and these may comprise endogenous splice variants of RAGE lacking the transmembrane domain of the receptor, as well as proteolytically cleaved forms released into the blood stream by the action of extracellular metalloproteinases. Administration of murine recombinant sRAGE, consisting of the extracellular ligand-binding domain of RAGE, has been shown not only to suppress the development of atherosclerosis but also to stabilise established atherosclerosis in diabetic apolipoprotein E null mice. 7
However, little is known about the regulation and role of endogenous sRAGE in patients with type 1 diabetes, and even less concerning the evolution of this receptor as a function of time after diagnosis.
In view of the above, the objective of the present study was to investigate the levels of circulating malondialdehyde (MDA), an end product of lipid peroxidation, and sRAGE in patients with T1DM, but without chronic complications, during the early years after diagnosis (0–10 years) and through the further progression of the disease (10–20 and > 20 years after diagnosis).
Subjects and methods
Details of the study were submitted to, and received the approval of, the Ethical Committee of the Santa Casa Hospital, Belo Horizonte, Minas Gerais, Brazil. The investigation was carried out according to the principles expressed in the Declaration of Helsinki, and written informed consent was obtained from each participant prior to the commencement of the study.
Subjects
The study population comprised 42 patients with T1DM and 24 healthy subjects (non-diabetic). The exclusion criteria for the study were: (i) patients with T1DM presenting diabetic complications, (ii) diabetic patients receiving statin and/or metformin and/or vitamins, (iii) individuals suffering from infection, dementia, inflammation or malignant disease, (iv) individuals addicted to smoking or alcohol, and (v) pregnant females. Patients with T1DM who were selected for inclusion in the study presented histories of diabetic ketoacidosis and/or were positive for glutamic acid dehydrogenase (GAD) antibodies and C peptide (< 0.5 ng/ml), and all were receiving intensive insulin therapy through multiple daily injections. T1DM patients, all of whom had exhibited clinical onset of the disease when aged within the range 13.03 ± 9.42 years, were divided into three groups according to the time elapsed since diagnosis, namely, 0–10 years, 10–20 years and > 20 years. The healthy control population was not divided for analysis as the group of type 1 diabetics (divided by disease duration) has been analysed as a single group. All participants were subjected to clinical and biochemical evaluations performed according to standard procedures.
Preparation of plasma samples
Plasma was obtained by centrifugation (200 g, 15 min, room temperature) of 10 ml samples of heparinised venous blood (collected from participants between 07:00 and 08:00 h using a standard venipuncture technique), and was stored at −20°C until required for analysis.
Measurement of circulating malondialdehyde
Levels of MDA in plasma samples were assessed with the aid of OXI-tek TBARS assay kits (ZeptoMetrix Corporation). Measurements were performed in triplicate and the mean value for each participant reported.
Measurement of circulating sRAGE
Levels of sRAGE in plasma samples were determined by enzyme-linked immunosorbent assay (ELISA) using Quantikine (R & D Systems) kits according to the manufacturer’s instructions. Measurements were performed in triplicate and the mean value for each participant reported.
Statistical analyses
Normality of distribution of the data was assessed using the Shapiro-Wilk test and, where applicable, mean values were compared using Student’s paired sample t-tests. Between-group comparisons of median values of circulating MDA and sRAGEs were performed using the non-parametric Mann–Whitney test. Within-group correlations were performed using Spearman rank correlation tests. In all cases, statistical significance was accepted at p values <0.05.
Results
Baseline characteristics of the study populations
Patients with T1DM and healthy subjects presented clinical characteristics that were very similar, as shown in Table 1. With respect to the tested biochemical characteristics, fasting plasma glucose levels of diabetic patients were significantly higher (p < 0.05) than those of healthy subjects and glycated haemoglobin (HbA1c) values in patients with T1DM (8.84%) were somewhat higher than the accepted norm for healthy subjects (< 7%). Table 2 presents the clinical and biochemical characteristics of T1DM patients distributed according to the time elapsed since diagnosis, and reveals significant between-group differences for some parameters, although these were without significance in clinical practice.
Clinical and biochemical characteristics of the studied population.
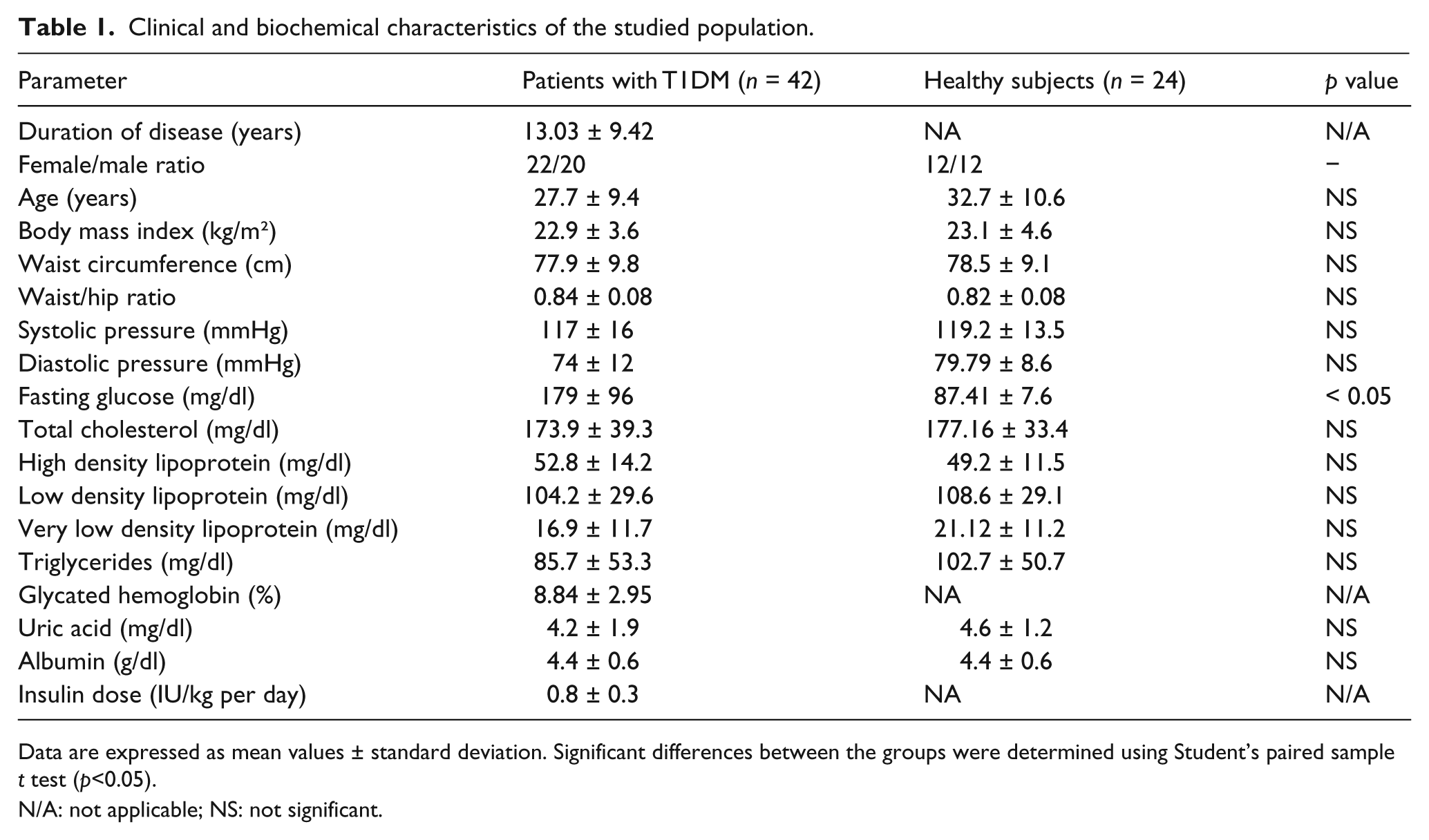
Data are expressed as mean values ± standard deviation. Significant differences between the groups were determined using Student’s paired sample t test (p<0.05).
N/A: not applicable; NS: not significant.
Clinical and biochemical characteristics of type 1 diabetes patients stratified according to time elapsed after diagnosis.
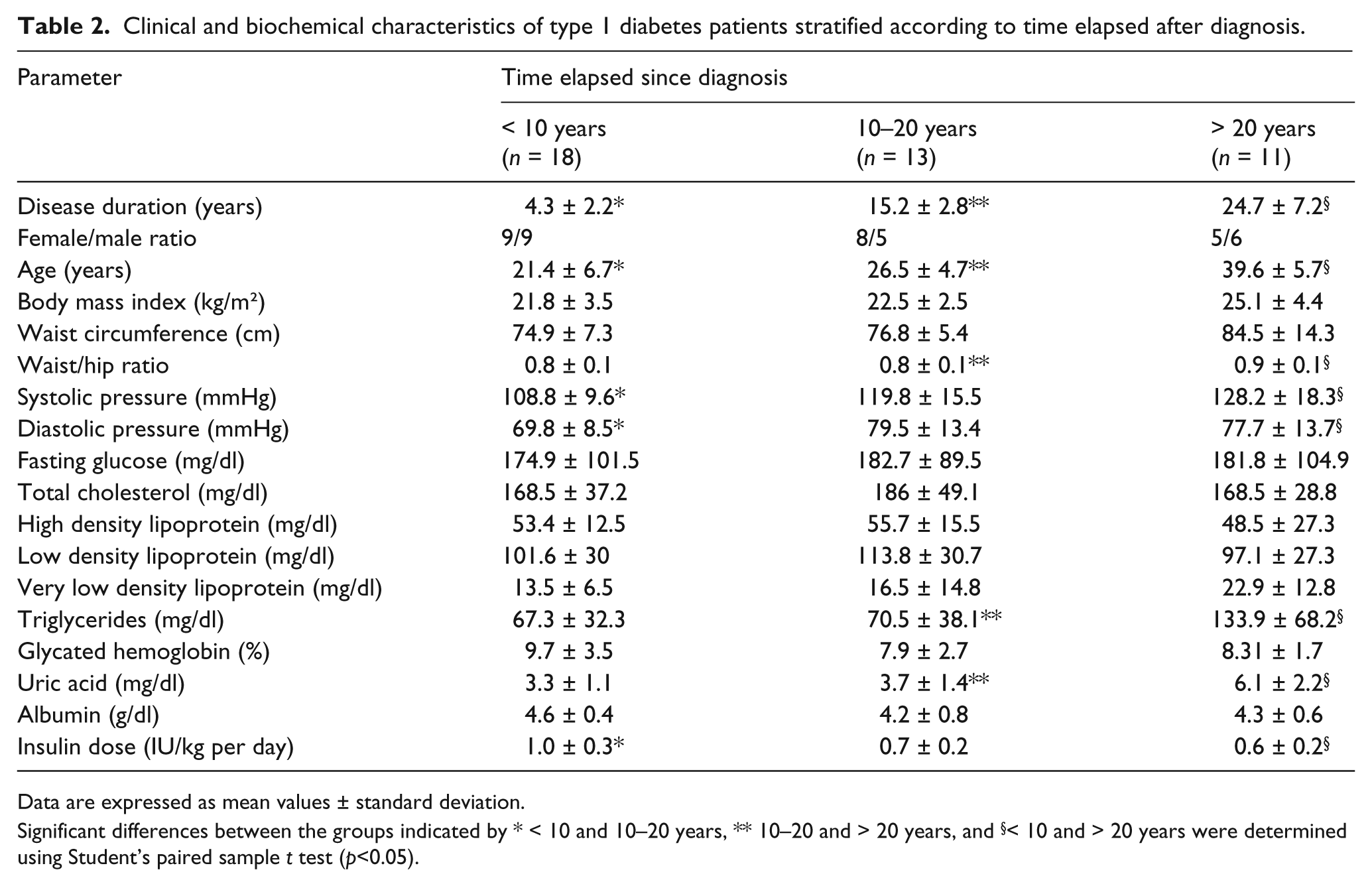
Data are expressed as mean values ± standard deviation.
Significant differences between the groups indicated by * < 10 and 10–20 years, ** 10–20 and > 20 years, and §< 10 and > 20 years were determined using Student’s paired sample t test (p<0.05).
Levels of circulating MDA and sRAGE
No significant difference (p > 0.05) was observed between diabetic patients and healthy subjects with respect to the median values of MDA (5.53 and 4.6 nmol/L, respectively) (Figure 1(a)). In contrast, a significant difference (p = 0.001) was detected between the median values of sRAGE in patients with T1MD and in healthy subjects (2423.75 and 1472.75 pg/ml, respectively) (Figure 1(b)).
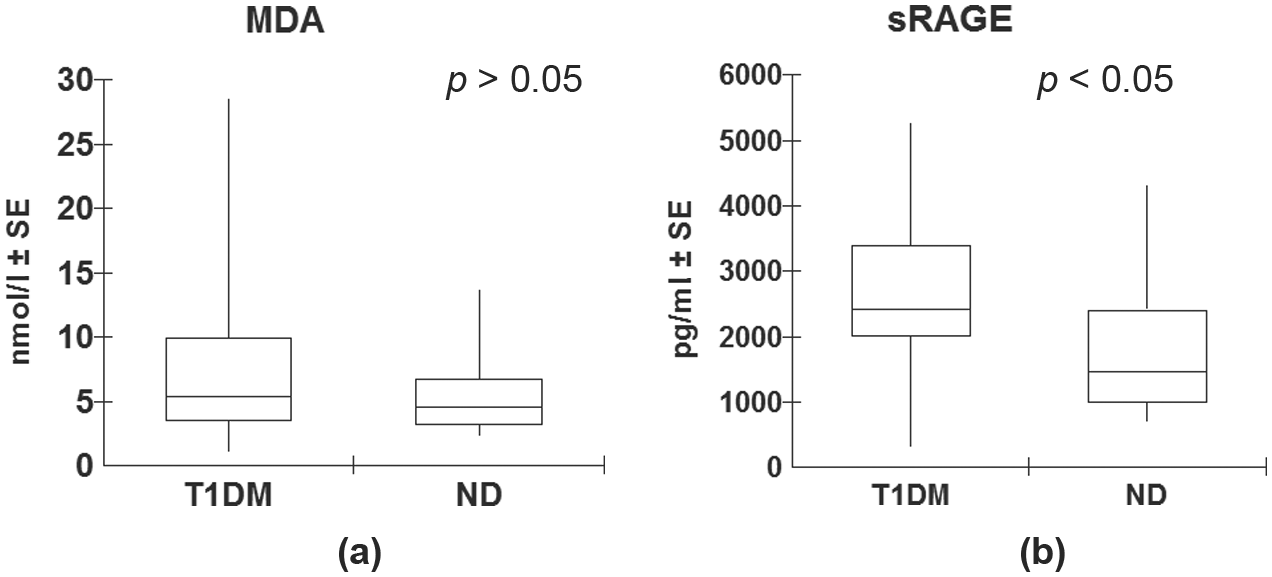
Comparison of levels of circulating MDA (a) and sRAGE (b) between type 1 diabetes patients and healthy controls (non-diabetic).
When the levels of MDA and sRAGE determined in T1DM patients were distributed according to the time elapsed since diagnosis, no significant between-group differences (p > 0.05) were detected with respect to the individual parameters (Figure 2(a) and (b)). Thus, sRAGE and MDA differ of the diabetic population but not when each group was compared separately from non-diabetic control. However, the median values of sRAGE of each of the three separate groups were significantly different (p < 0.05) from the corresponding level recorded in healthy subjects. In contrast, the median levels of MDA of the three individual groups of diabetic patients did not differ significantly (p > 0 .05) from the median value determine in healthy subjects.
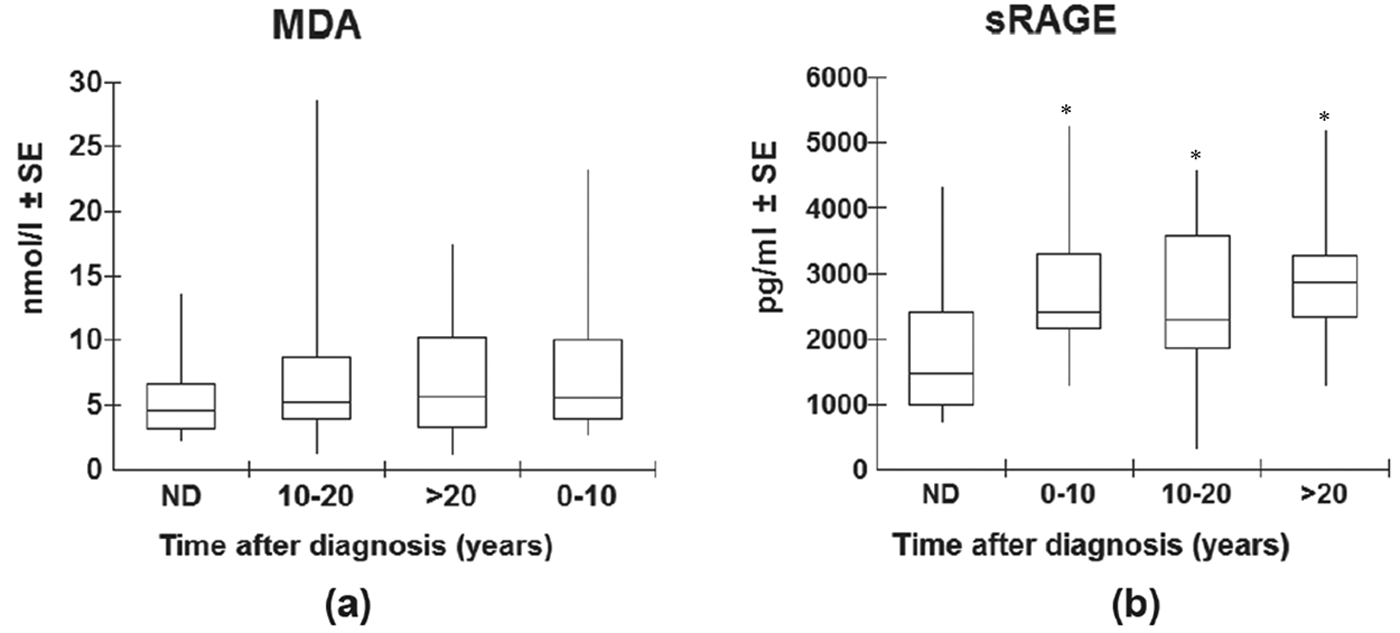
Comparison of circulating MDA (a) and sRAGE (b) between healthy individuals and type 1 diabetes patients stratified according to time after diagnosis.
Correlations of parameters within the diabetic groups
There were no significant correlations between the levels of MDA and sRAGE in patients with T1DM considered as a single group or when divided according to time elapsed since diagnosis. Additionally, there were no correlations between MDA and sRAGE levels and the clinical and biochemical parameters evaluated including, specifically, fasting glucose and HbA1c.
Discussion
The results obtained in the present study suggest that in type 1 diabetes patients without chronic complications levels of circulating sRAGE are elevated at all times after disease onset but that concentrations of circulating MDA remain similar throughout disease progression.
Few studies have considered the levels of different AGEs and sRAGE in type 1 diabetes patients compared with their non-diabetic counterparts, and none has evaluated these parameters in relation to the time of diagnosis. Various authors have reported that serum levels of sRAGE are significantly increased in adult patients with diabetes mellitus (DM) in comparison with non-diabetic individuals.6,8,9 Moreover, it has been shown that exogenously administered sRAGE blocked the harmful effects of AGEs in animals by acting as a decoy receptor. 4 Reactive oxygen species (ROS) produced by granulocytes was inhibited by treatment with anti-RAGE antibodies. 10 Considered together, these findings support the concept that levels of circulating sRAGE are elevated in response to serum AGEs and reflect tissue RAGE expression in diabetes, thereby acting as a negative feedback agent against the AGE-elicited vascular injury.6,8,9 The increase in sRAGE expression observed in T1DM patients in the present study can be viewed, therefore, as a possible protective reaction to counterbalance, at least in part, the increase in AGE formation.
The present finding of high levels of sRAGE from the early years of disease onset in the absence of a concomitant increase in MDA is of particular interest. A survey of the literature revealed only one analogous report, and this referred to a group of children with T1DM in which those who had been diagnosed for > 5 years showed increased sRAGE but similar levels of N-carboxymethyl lysine (a major AGE) in comparison with those who had been diagnosed for < 5 years. 11 It is possible that the augmented levels of sRAGE observed in our T1DM patients without diabetic complications may have been sufficient to efficiently eliminate excessive levels of circulating AGEs.
Recent studies have indicated relationships between higher concentrations of sRAGE and the incidence of non-fatal and fatal cardiovascular disease and all-cause mortality in individuals with T1DM.12–14 Thus, Nin et al. have associated an increase in sRAGE with renal dysfunction in type 1 diabetes patients, 12 while the FinnDiane Study Group demonstrated that elevated levels of sRAGE were linked to increased all-cause and cardiovascular mortality in T1DM, and were probably coupled with the production and activation of RAGE during accelerated vascular disease. 13 Other groups have demonstrated that endogenous secretory RAGE, but not sRAGE, is associated with carotid atherosclerosis and albuminuria in type 1 diabetes patients.8,15
The lack of correlation between the levels of MDA and sRAGE and those of fasting glucose or HbA1c may suggest that once the inflammatory response has been triggered, it can be modulated positively or negatively by parameters other than those presently studied including, for example, glucose excursions. 16 If upheld, this hypothesis would represent further evidence of the complexity and multifactorial nature of T1DM.
Taken together, the present observations suggest that serum levels of sRAGE may be increased in type 1 diabetes patients without chronic complications from the early years of the disease as a possible counter-regulatory system against cell damage. Elevated plasma concentrations of this receptor may also reflect enhanced cleavage of RAGE protein by metalloproteinases resulting in the release of sRAGE into plasma. The cohort of patients involved in the present study had not developed any micro- or macro-angiopathies and presented circulating MDA levels that were similar to those of the healthy controls. It is probable that the increased level of sRAGE determined in the diabetic group was sufficient to act as a decoy for pro-inflammatory ligands, including MDA, thus blocking diabetic vascular complications.
The exact functions of sRAGE in plasma are unknown, although it has been proposed that the activity of the receptor may vary according to cellular properties. 17 Additional studies are clearly required in order to investigate possible relationships between levels of AGEs and sRAGE and other parameters, and to elucidate the potential relevance of these markers in the pathogenesis of complications in T1DM.
Footnotes
Funding
This work was supported by the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq); the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) and the Fundação de Amparo à Pesquisa do Estado de Minas Gerais (grant no. Fapemig APQ-00374-10).
Conflict of interest statement
None declared.
