Abstract
Background and aim:
Coronary flow reserve is impaired in asymptomatic patients with aortic stenosis and has a prognostic value. We investigated whether the type II diabetes mellitus additionally impairs microvascular circulation assessed by coronary flow reserve in patients with asymptomatic severe aortic stenosis, normal left ventricular ejection fraction and nonobstructed coronary arteries.
Methods:
A total of 128 patients, mean age of 66.35 ± 10.51 (58.6% males), with severe aortic stenosis and normal left ventricular ejection fraction were enrolled in this study. Patients with diabetes mellitus, those who were treated for diabetes mellitus or had documentation confirming the diagnosis of diabetes mellitus, were considered. All patients underwent coronary angiography and had no obstructive coronary disease (defined as having no stenosis >50% in diameter), standard transthoracic Doppler-echocardiographic study and adenosine stress transthoracic echocardiography for coronary flow reserve measurement.
Results:
Diabetes mellitus was present in 26 patients (20.31%). There was no significant difference in aortic stenosis severity between diabetic and non-diabetic patients [aortic valve area (0.81 ± 0.18 vs 0.85 ± 0.15 cm2) and Vmax (4.20 ± 0.57 vs 4.21 ± 0.48 m/s)]. Mean coronary flow reserve in diabetic patients was 1.98 ± 0.48, while mean coronary flow reserve in non-diabetic patients was 2.64 ± 0.54 (p < 0.01). Diabetes mellitus was independent predictor of coronary flow reserve [B = −0.636, 95% confidence interval (−0.916 to −0.368), p < 0.001].
Conclusion:
Diabetes mellitus additionally impairs coronary microvascular function in asymptomatic patients with severe aortic stenosis and nonobstructed coronary arteries.
Introduction
Coronary flow reserve (CFR) has been shown to be suitable tool to assess microvascular function in patients with aortic stenosis (AS) and without epicardial coronary artery disease. 1 It has been shown that CFR is impaired in asymptomatic patients with AS and normal coronary arteries, and that in these patients impaired CFR has a prognostic value.2,3
The treatment of symptomatic patients with significant AS is well established, whereas the management of asymptomatic patients with severe AS and normal left ventricular ejection fraction (LVEF) is a matter of on-going debate.4–7 There are data4,5 that early aortic valve replacement (AVR) might lead to favourable outcome compared to elective (after symptom onset) surgery. With this respect, there is growing and reasonable interest in adequate risk stratification and identifying the subsets of asymptomatic AS patients who may benefit from early AVR. However, this is not a homogenous group and within these asymptomatic AS patients with normal LVEF, there are those who are at increased risk for adverse event.
Cardiovascular diseases are leading cause of morbidity and mortality in patients with type II diabetes mellitus (DM) and its incidence is increasing worldwide.8–10 In patients with type II DM, microvascular dysfunction has been impaired and also shown as an independent predictor of outcome.11–13 With that regard, asymptomatic patients with severe AS, normal LVEF and type II DM might be especially challenging for risk stratification. Therefore, we investigated whether the type II DM additionally impairs microvascular circulation assessed by CFR in patients with asymptomatic severe AS, normal LVEF and nonobstructed coronary arteries.
Methods
Initially, a total of 128 asymptomatic patients, mean age of 66 ± 10.51 (58.6% males), with severe AS [aortic valve area (AVA)] ⩽ 1 cm2, maximal velocity across the aortic valve (Vmax) > 4 m/s and normal LVEF (>55%) were enrolled in this prospective study. Follow-up was 47 months, median 36 (interquartile range 4). In total of 128 patients, we analysed CFR in patients with type II DM and compared the results to patients without diabetes. In two patients, CFR was not analysed due to the insufficient echocardiographic acoustic window and one patient was excluded because he was being treated for asthma. None of these three patients was included in study analysis.
Patients with type II DM, those who were treated for DM or had documentation confirming the diagnosis of type II DM, were considered. Five patients were not recognized as diabetics at the study inclusion because they did not respond positively to the question whether they have diabetes, but were later included in the diabetic’s group because they reported/documented subsequently during follow-up that they take antidiabetic drugs (including at the time of the study inclusion). All patients underwent coronary angiography and had no obstructive coronary artery disease (defined as having no stenosis >50% in diameter). All patients in the study have signed informed consent. Study was approved by the local ethics committee of Clinical Centre of Serbia.
Transthoracic echocardiography
Transthoracic echocardiography exam was performed on General Electric, Vivid 4 cardiac ultrasound system (BTO6, 1.5–3.6 MHz; GE Healthcare Technologies, Waukesha, WI, USA), whereas CFR was performed on Sequoia Acuson C 256 (Acuson Inc., Mountain View, CA, USA). The subjects were studied in the left lateral decubitus. Left ventricular (LV) internal dimension, posterior wall thickness (PWT) and interventricular septum thickness (IVST) were measured at end-diastole, at a level immediately apical to the mitral valve leaflet tips, in two-dimensional parasternal long-axis view. 14 The LVEF was calculated with biplanar Simpson’s rule from the apical two- and four-chamber views. 15 In addition, considering prognostic importance of indexed LV stroke volume (SVi) in subset of patients of AS with preserved LVEF, we have also calculated SVi as follows: SVi = SV/BSA (SV is stroke volume; BSA is body surface area). LV SV was calculated by the Doppler method (LV outflow tract area × LV outflow tract velocity–time integral measured by pulsed-wave Doppler). 16
The LV mass was calculated using the corrected formula of the American Society of Echocardiography and was indexed for BSA. 17 Calculation of relative wall thickness (RWT) by the formula, (2 PWTd)/LVIDd, permits categorization of an increase in LV mass as either concentric (RWT ⩾ 0.42) or eccentric (RWT ⩽ 0.42) hypertrophy and allows identification of concentric remodelling 18 (PWTd = posterior wall thickness dimension).
Mitral inflow curves were recorded as previously described. 19 Tissue Doppler imaging (TDI) of mitral annular motion has been proposed to correct for the influence of myocardial relaxation on transmitral flows. This has been shown to be an excellent predictor of diastolic filling in subsets of patients. 20 TDI of the mitral annulus was obtained from the apical four-chamber view. A 1.5-mm sample volume was placed sequentially at the lateral and medial mitral annulus. Analysis was performed for the early (E′) and late diastolic velocity (A′). These variables were analysed individually, as the average of the medial and lateral annulus. The ratio of mitral velocity to early diastolic velocity of the mitral annulus (E/E′) is important to calculate as it was demonstrated that this ratio has the best correlation with mean LV diastolic pressure comparing to all other Doppler variables. 21
AS was graded using the continuity equation 22 calculated as severe if AVA is 1.0 cm2 or less. The subaortic diameter was measured from inner edge to inner edge at the level of the base of the aortic cusps in a parasternal long-axis frame frozen in mid-systole. Pulsed-Doppler recordings were made in apical five-chamber view with the sample volume moved axially from the aortic annulus, usually 0.5–1 cm below the valve, recording maximal velocity and velocity–time integral. Continuous-wave recordings were made from the apex and right intercostal positions and the optimal signal was traced to obtain peak velocity, velocity–time integral, systolic ejection time, and peak and mean pressure difference. The mathematically derived parameters that represent LV workload and haemodynamic severity of AS, such as valvulo-arterial impedance [Zva = (SAP + Pmean)/SVi],15,23 where SAP is systolic arterial pressure; stroke work loss [SWL = 100 × (Pmean/(1/3SAP + 2/3DAP))], 24 where DAP is diastolic arterial pressure; energy loss index [ELI = (AVA × Aoasc)/(Aoasc − AVA)/BSA], 25 where Aoasc is ascending aorta diameter and resistance of aortic valve [RVA = (28 × √Pmean)/AVA] 26 were calculated the same way as previously described.
CFR measurement
Using transthoracic Doppler echocardiography, we measured CFR in left anterior descending (LAD) artery. On the day of the CFR measurement, the consumption of caffeine-containing drinks was prohibited. The LAD was searched as a laminar flow towards transducer with colour Doppler in foreshortened apical two-chamber view or in three-chamber view, to reach optimal alignment with the interventricular sulcus. The blood flow velocity was recorded by pulsed-wave Doppler. Due to individual anatomical differences, there were somewhat different locations for sampling volumes. In short, colour Doppler detection of the LAD flow was obtained using a 4-MHz transducer, with Doppler mapping velocity range set between 16 and 24 cm/s. The systolic and diastolic coronary flow velocity spectrum was obtained at baseline and during the peak of hyperaemia induced by intravenous (i.v.) adenosine (0.14 mg/kg/min during 1 min). CFR was measured as a difference ratio between baseline value and peak hyperaemic value in LAD obtained during adenosine infusion [Figure 1(a) and (b)]. All studies were recorded on VHS videotapes, and stop frames and clips were digitally recorded and stored for offline analysis. In each case, three optimal diastolic flow profiles were measured and the results were averaged. Measurements of diastolic basal and hyperaemic flow and CFR were later repeated by an independent observer from our department. Inter-observer agreement regarding CFR measures was 90%.
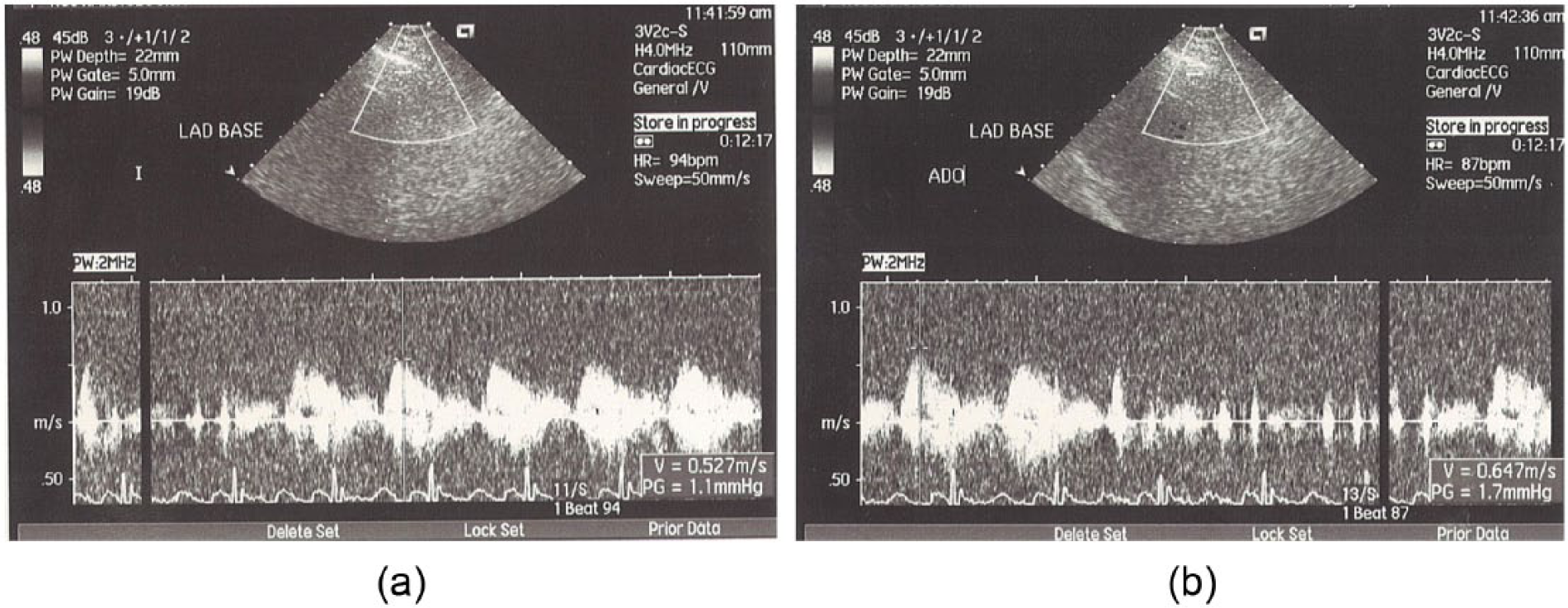
(a) Characteristic flow pattern in LAD artery distribution before adenosine infusion and (b) characteristic flow pattern showing peak hyperaemic value in LAD obtained during adenosine infusion.
Statistical analysis
Considering a Kolmogorov–Smirnov test of normality, continuous variables are presented as mean ± standard deviation (SD) or median (interquartile range). Categorical variables are reported as counts with percentages. Continuous variables were compared using the two-sided Student’s t-test for normal distributed or Mann–Whitney U test for non-normal distributed variables. Fisher and chi-square tests were used to analyse differences between categorical variables. Univariable linear regression analysis was used to evaluate the relation between various clinical and echocardiographic variables with CFR value in the follow-up period. To select covariates independently associated with the CFR, significant univariable predictors were reassessed by a multivariable linear regression analysis, with values for inclusion and elimination set at p < 0.1. Differences in survival between diabetic and non-diabetic patients groups were compared using Kaplan–Meier survival curves and statistical significance was determined by the log rank test.
Statistical analyses were performed using the statistical package for social sciences, version 18 (SPSS, Chicago, IL). Statistical significance was defined as p < 0.05.
Results
The total study population included 53 asymptomatic women and 75 asymptomatic men (58.6% males); mean age was 66.35 ± 10.51. Follow-up was 47 months, median 36 (interquartile range 4). Mean gradient across the valve (Pmean) was 41.88 ± 11.26 mmHg, mean AVA was 0.84 ± 0.15 cm2, mean maximal velocity (Vmax) was 4.20 ± 0.49 m/s, mean LVEF was 70.73% ± 7.93% and mean E/E′ was 12.83 ± 5.20.
A total of 26 patients (20.31%) had type II DM. The two groups of diabetic versus non-diabetic patients were similar age (66 ± 10.65 vs 66 ± 10.29, p = ns), without significant difference in AS severity (Table 1). Hypertension was most frequent comorbidity in both groups, present in 12/26 patients (46.2%) in diabetic group and in 68/102 patients (66.7%) in non-diabetic group (p = 0.064). On the other hand, hyperlipidaemia was more common in non-diabetic patients. The frequencies of the most common risk factors for coronary heart disease in the two groups are shown in Table 2.
Echocardiographic, demographic and laboratory data in the two groups of diabetic and non-diabetic AS patients.
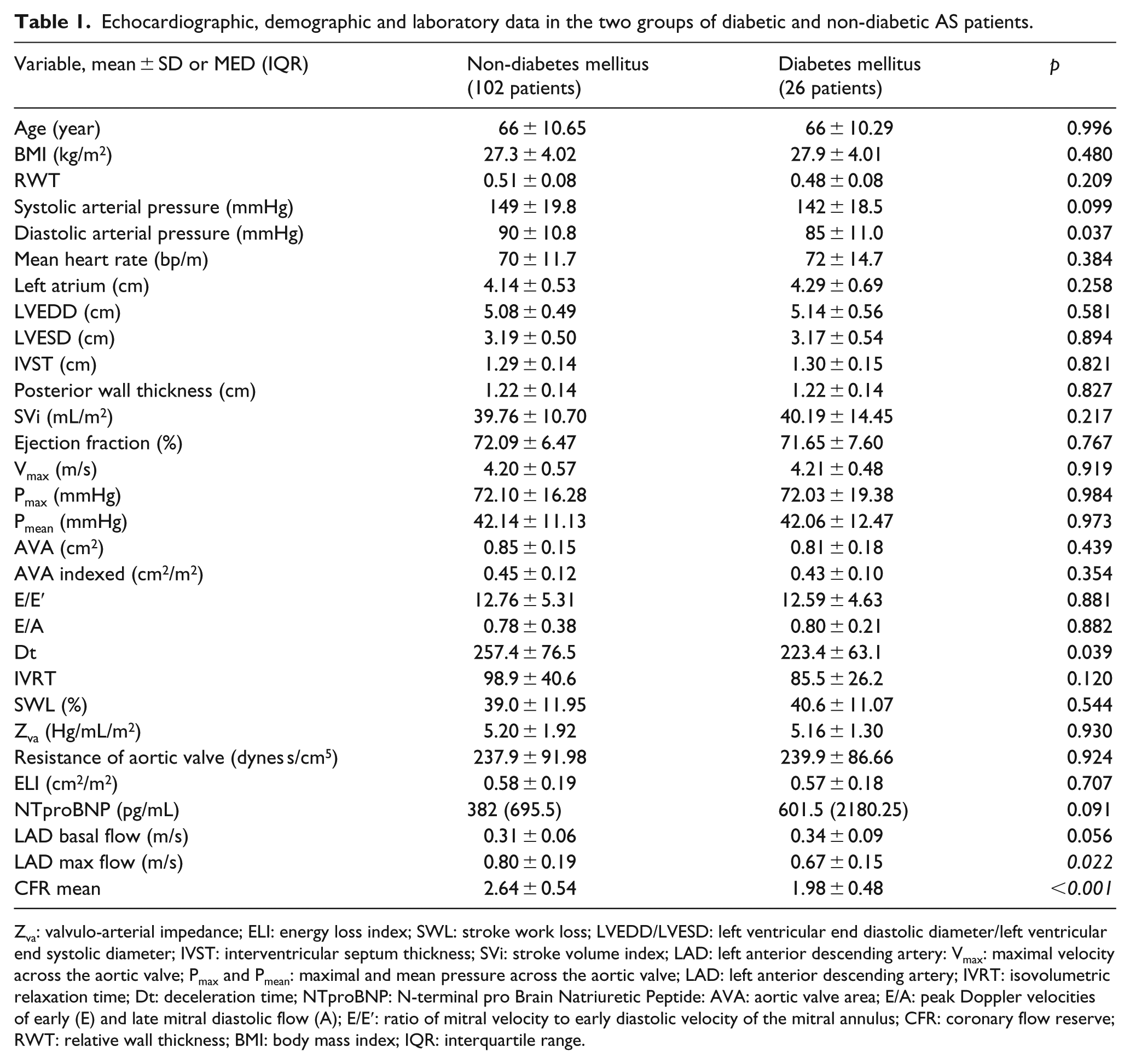
Zva: valvulo-arterial impedance; ELI: energy loss index; SWL: stroke work loss; LVEDD/LVESD: left ventricular end diastolic diameter/left ventricular end systolic diameter; IVST: interventricular septum thickness; SVi: stroke volume index; LAD: left anterior descending artery: Vmax: maximal velocity across the aortic valve; Pmax and Pmean: maximal and mean pressure across the aortic valve; LAD: left anterior descending artery; IVRT: isovolumetric relaxation time; Dt: deceleration time; NTproBNP: N-terminal pro Brain Natriuretic Peptide: AVA: aortic valve area; E/A: peak Doppler velocities of early (E) and late mitral diastolic flow (A); E/E′: ratio of mitral velocity to early diastolic velocity of the mitral annulus; CFR: coronary flow reserve; RWT: relative wall thickness; BMI: body mass index; IQR: interquartile range.
Frequency of other coronary artery disease risk factors between the groups.
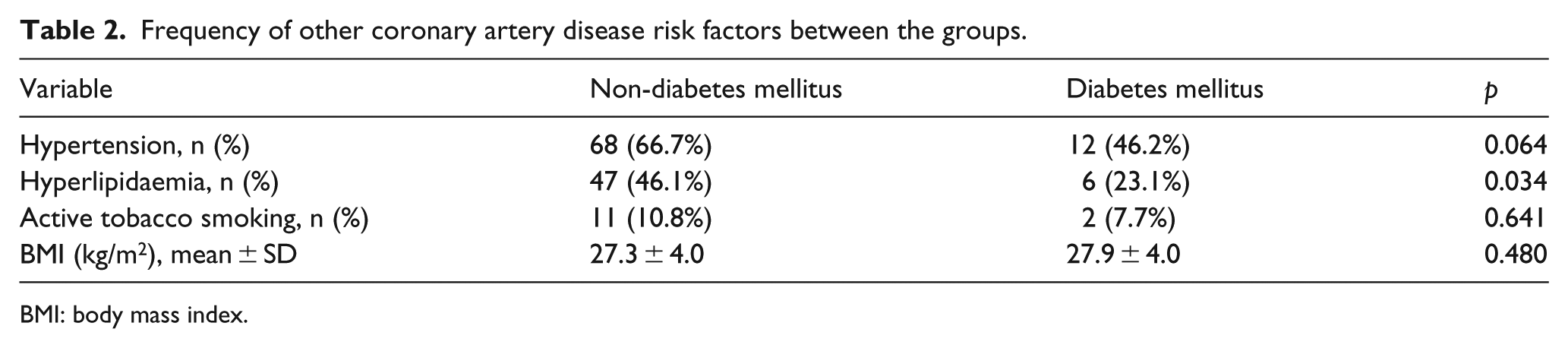
BMI: body mass index.
Mean CFR in diabetic patients was 1.98 ± 0.48, whereas mean CFR in non-diabetic patients was 2.64 ± 0.54 (p < 0.01). LAD basal flow velocity tended to be higher in diabetic patients (0.34 ± 0.09 m/s vs 0.31 ± 0.06 m/s; p = 0.056). Table 3 presents an univariable linear regression analysis of the relation between clinical and echocardiographic variables with CFR. Independent variables related to CFR, assessed in multivariable linear regression analysis, remained age, DM and Vmax. Diabetes was an independent predictor of CFR [B = −0.636, 95% confidence interval (CI) (−0.916 to −0.368), p < 0.001].
A univariable and multivariable linear regression analysis of the relation between clinical and echocardiographic variables with CFR.
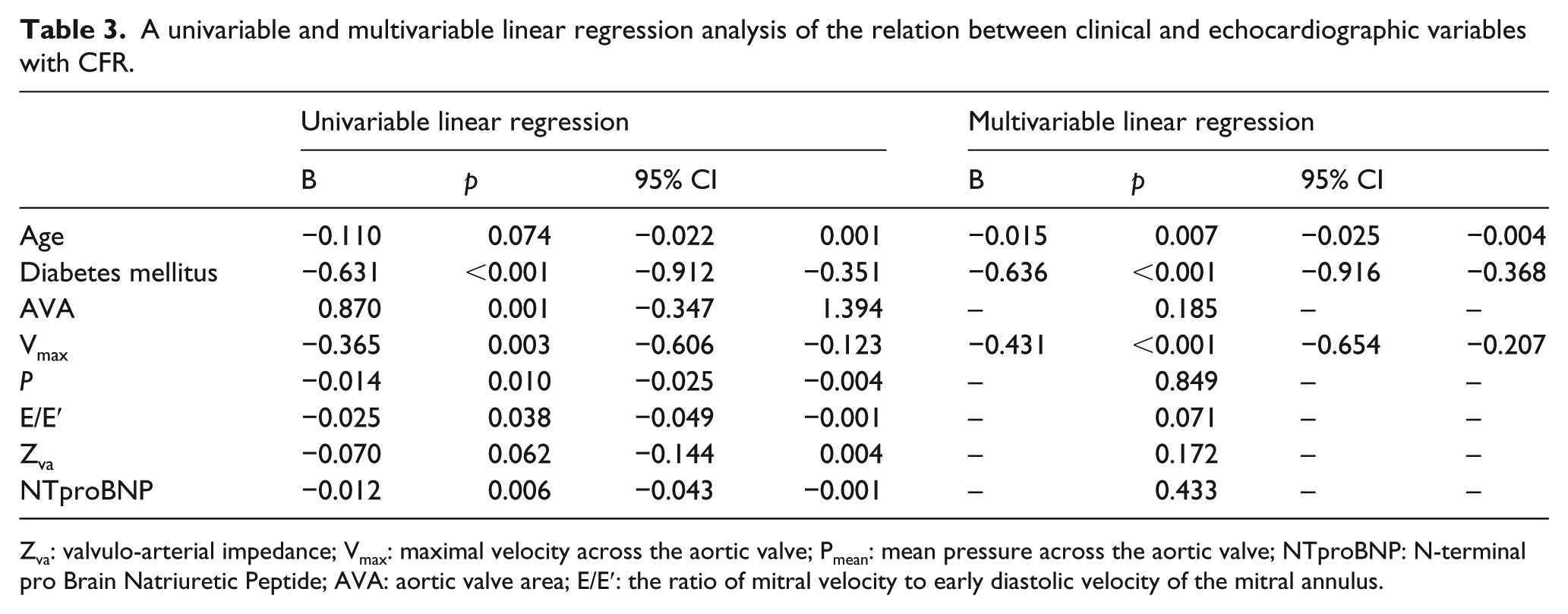
Zva: valvulo-arterial impedance; Vmax: maximal velocity across the aortic valve; Pmean: mean pressure across the aortic valve; NTproBNP: N-terminal pro Brain Natriuretic Peptide; AVA: aortic valve area; E/E′: the ratio of mitral velocity to early diastolic velocity of the mitral annulus.
In all, 12 patients died during follow-up (9.37%). Four patients (15.4%) died in group of AS patients with diabetes and eight (7.9%) patients died in group of AS patients without diabetes. Patients with diabetes had a tendency to a higher mortality during follow-up; however, the statistical significance was not achieved (Figure 2). Out of four patients who died in diabetic group, one patient had a sudden cardiac death, two patients developed AS-related symptoms but refused AVR and one patient died due to the non-cardiac reason. Out of eight patients who have died in non-diabetic group, five patients reported symptom onset but were not operated during follow-up (two of them refused operation recommended by a referring physician), one patient died after AVR due to the postoperative infection, one patient died in a hospital, 2 days before scheduled AVR due to the acute myocardial infarction, and one patient died at home and is assumed to have died due to the sudden cardiac death (based on the description of family members).
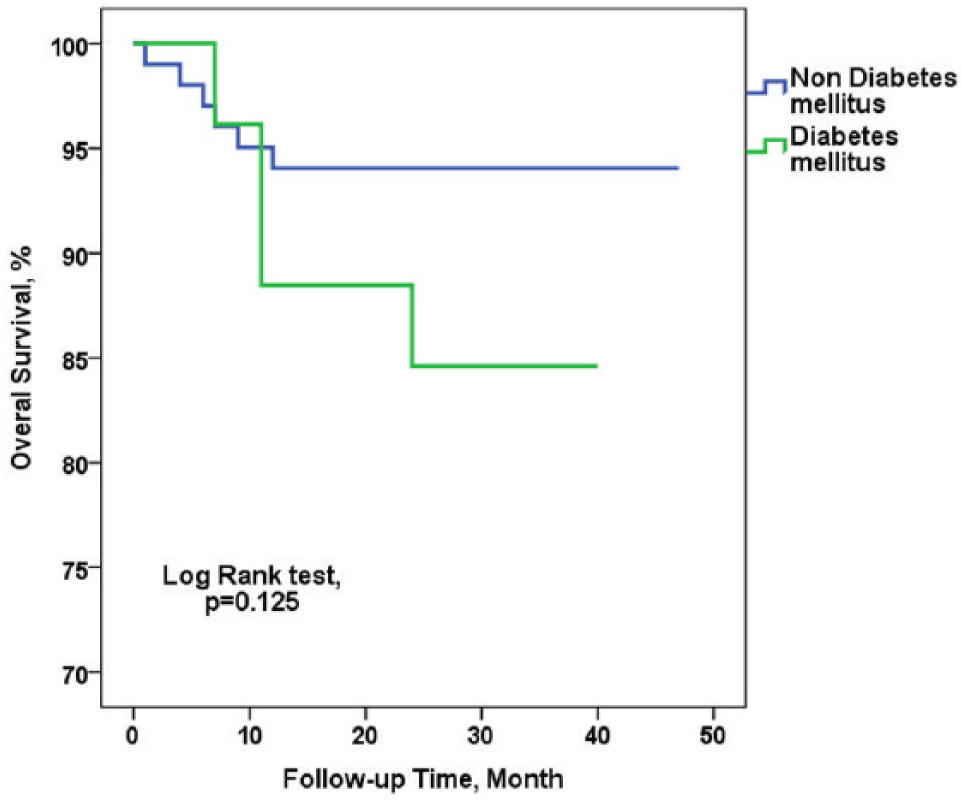
Kaplan–Meier survival curve in diabetic and non-diabetic patients.
Discussion
In a time in which AS has become the most common valvular heart disease and percutaneous aortic valve treatment is becoming increasingly used,27–29 scientists and clinicians worldwide have put special emphasize on risk stratification of asymptomatic patients severe AS and normal LVEF.30–32 Both microvascular dysfunction and type II DM have been shown to significantly contribute to worse prognosis of patients with cardiovascular diseases, including AS.32–34 Also, microvascular complications are major contributors to morbidity and mortality in patients with diabetes. 35 To the best of the authors’ knowledge, this is the first study to demonstrate that type II DM additionally impairs coronary microvascular function in asymptomatic patients with severe AS, normal LVEF and nonobstructed coronary arteries.
Diabetes and coronary microcirculation in severe AS – pathophysiological changes
The fact that the diabetic versus non-diabetic groups were very similar characteristics, and that there was a significant difference in the CFR may indicate that diabetes is solely responsible for additional impair of coronary microvascular function in AS patients. And although non-diabetic patients had more frequently hyperlipidaemia as a comorbidity, this did not influence the difference in coronary microvascular function between the groups. This is not surprising, as it was shown that familiar hypercholesterolaemia much more than secondary hypercholesterolaemia influences coronary microvascular flow, 36 and in our study, the vast majority of patients with hyperlipidaemia were diagnosed with secondary hypercholesterolaemia (type IIa).
Patients with severe AS and normal coronary angiogram have impaired microvascular function due to the LV hypertrophy with high wall stress and high metabolic demands, endothelial dysfunction, decreased coronary perfusion pressure and extravascular compression.37–39 On the other hand, there is decrease in arterial elasticity and increased arterial stiffness in small arteriolas in diabetic patients40,41 due to the number of reasons, including non-enzymatic glycosylation of collagen and other matrix proteins and formation of reactive oxygen species resulting with fibrosis, direct damage of endothelial cells and their ability to vasodilate. 42 In addition, in diabetic patients, there is an increase in the media cross-sectional area of the vessels, suggesting the presence of hypertrophic remodelling (vascular smooth muscle cells hypertrophy or hyperplasia),40,43 which leads to a more rigid arterial wall, narrowing of the lumen, higher blood flow velocity at rest and insufficient increase in blood flow velocity during vasodilatation. A study from Rizzoni et al. 40 showed correlation between circulating levels of insulin and media to lumen ratio of subcutaneous small arteries in diabetic patients, thus suggesting a possible role of insulin or insulin-like growth factor-1 in the genesis of hypertrophic remodelling in these patients. An important pathophysiological and clinical consequence of the presence of structural alterations in arteriolas may be an impairment of CFR. However, Schofield et al. 43 argue that a stimulus for hypertrophic remodelling in small arteriolas could be the increased wall stress as a consequence of impaired myogenic response due to the pressure-induced vasoconstriction. This pressure-induced vasoconstriction, according to Schofield, might be the key mechanism for impaired blood flow autoregulation in small arterial vessels. Whether these mechanisms are acting alone or in concert with each other, they result in further impairment in coronary microcirculation in patients with both DM and AS comparing with AS alone, which has been demonstrated in our study.
Clinical implication of coronary microvascular dysfunction in patients with DM and AS
The CFR is impaired in asymptomatic severe AS patients.38,44 On the other hand, structural alterations and muscular layer remodelling of small arteries are characteristic for later stages of DM and are per se associated with an increased cardiovascular risk in diabetic patients. 45 In the light of these two facts, the results of our study are somewhat expected.
The mid- and long-term prognostic significance of impaired CFR in AS patients has been already shown.2,3 This is important as we have demonstrated that DM additionally impairs coronary microvascular function in asymptomatic patients with severe AS, normal LVEF and nonobstructed coronary arteries. In addition, the mean CFR value in diabetic patient’s group has been somewhat lower than proposed cut-off for detection of significant epicardial coronary artery stenosis. As a case in point, the cut-off value of <2 of CFR is precise for detecting significant (>75%) LAD stenosis with a sensitivity of 86% and a specificity of 70%. 46 Furthermore, CFR < 2 had 91% sensitivity, 95% specificity, and 96% positive and 97% negative predictive values to detect significant stenosis in patients with LAD stents. 47 Therefore, dysfunction of microvascular coronary flow represented through CFR impairment could be a substrate responsible for the occurrence of angina pectoris symptoms and potentially fatal arrhythmias registered in AS patients with normal epicardial coronary arteries. 48 Angina pectoris is associated with significant increase in the risk of sudden death in AS patients. 49 This fact is even more important if one considers that people with diabetes do not necessarily have to have symptoms despite the existence of angina pectoris. With this regard, the fact that there was a clear tendency towards increased mortality in diabetics in our study is in favour of this, although statistical significance was not achieved, most probably due to the low number of diabetic patients. This indicates that ‘wait and follow’ strategy could be risky in these patients. It should be emphasized that all recommendations regarding patients with asymptomatic severe AS are associated with level of evidence C, which illustrates the low level of evidence on which they rely. 50 We believe the results of this study suggest that CFR testing has a place in an assessment of asymptomatic patients with severe AS, normal LVEF, nonobstructed coronary arteries and type II DM, as a better risk stratification could be made based on its value.
Study limitations
The main limitation of our study is relatively small number of patients with diabetes. However, those patients were extrapolated from broader population of consecutive asymptomatic severe AS patients who were included in the study. Therefore, this might be referred to the percentage of real-life clinical patients with diabetes and severe AS. In addition, percentage of our patients with severe AS and diabetes is very similar to previously reported data 51 which was conducted on population of 523 patients. Nevertheless, our result should be confirmed in a larger population study.
CFR measurements using transthoracic Doppler echocardiography are based on changes in blood flow velocity. Because coronary flow depends not only on flow velocity but also on artery diameter, we can measure changes only in coronary blood flow velocity, but not in coronary blood flow.
Conclusion
DM additionally impairs coronary microvascular function in asymptomatic patients with severe AS and nonobstructed coronary arteries. The reduced CFR in asymptomatic diabetic AS patients suggests these patients should receive special attention as they might have a high risk for subsequent myocardial ischaemia and sudden cardiac death.
Footnotes
Acknowledgements
Abstract of this article is accepted for an oral presentation at ESC congress 2015, London, Great Britain. In addition, the author of this article was winner of 2015 International Medis Awards for medical research in the field of Cardiology based on the presentation of this work on ESC 2015 congress in London.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
