Abstract
Homocysteine suppresses hepatic apolipoprotein AI synthesis in mice. We assessed the relationship between homocysteine and apolipoprotein AI levels in patients with impaired glucose tolerance. A total of 217 participants, including 95 impaired glucose tolerance patients and 122 healthy subjects, were classified as normal control subjects without hyperhomocysteinaemia, control subjects with hyperhomocysteinaemia, impaired glucose tolerance patients without hyperhomocysteinaemia (n-IGT) and impaired glucose tolerance patients with hyperhomocysteinaemia (H-IGT). The impaired glucose tolerance patients had higher plasma levels of homocysteine and homeostasis model assessment index of insulin resistance values, and lower plasma apolipoprotein AI levels than the normal control and control subjects with hyperhomocysteinaemia (all p < 0.01). Decreased plasma apolipoprotein AI levels and increased homeostasis model assessment index of insulin resistance values were observed in the H-IGT group compared with the n-IGT group (p < 0.05). Plasma homocysteine levels were negatively correlated with apolipoprotein AI levels after adjusting for age, gender, body mass index and homeostasis model assessment index of insulin resistance. Plasma homocysteine level independently influenced the apolipoprotein AI levels (β = −0.02, p < 0.05). In conclusion, increased plasma homocysteine levels were associated with decreased apolipoprotein AI levels in impaired glucose tolerance subjects.
Introduction
Hyperhomocysteinaemia (HHcy) is an independent risk factor for cardiovascular disease. 1 Our previous studies showed that patients with HHcy exhibited significant impairments of coronary endothelial function. 2 Homocysteine (Hcy) interrupts the insulin signalling pathway and causes insulin resistance by inducing endoplasmic reticulum stress and up-regulating the expression of resistin.3,4
Impaired glucose tolerance (IGT) is an intermediate category between normal glucose tolerance and type 2 diabetes. 5 IGT patients are at an elevated risk of cardiovascular disease due to their pathophysiologic conditions that include insulin resistance, obesity and dyslipidaemia. 5 IGT is a pre-diabetic condition. Patients with IGT have abnormal lipid profiles, including hypertriglyceridaemia and decreased plasma levels of high-density lipoprotein cholesterol (HDL-C) and apolipoprotein AI (apoAI) that are similar to those of type 2 diabetic patients. 5 Patients with insulin resistance or type 2 diabetes often have HHcy. 6 Animal studies have shown that Hcy suppresses hepatic apoAI synthesis, and Hcy levels are negatively correlated with apoAI levels in male patients with coronary artery disease. 7 However, the associations between Hcy and apoAI levels in early-stage insulin resistance conditions, such as IGT, have not been well-characterized. In this study, we assessed a potential association between Hcy and apoAI levels in IGT patients.
Methods
Subjects
A total of 217 participants, including 95 patients with IGT (IGT group) and 122 subjects with normal glucose tolerance (control group), were recruited from June 2012 through December 2013 at the Endocrinology Department of the Beijing Chao-Yang Hospital Affiliated to Capital Medical University. IGT was diagnosed based on a 75-g oral glucose tolerance test according to the World Health Organization diabetes criteria of 1999. No subject had a history of coronary artery disease, heart failure, liver or renal function impairment, infectious disease, systemic inflammatory disease or cancer. The patients were not taking lipid-lowering agents. The protocol was approved by Ethics Committee of the Beijing Chao-Yang Hospital Affiliated to Capital Medical University. All participants provided written informed consent.
Clinical tests
Blood samples were obtained after an overnight fast and stored at −70°C. Low-density lipoprotein cholesterol (LDL-C), HDL-C, total cholesterol (TC), triglyceride (TG), apoAI and apolipoprotein B (apoB) levels were measured by colorimetric enzymatic assays with an autoanalyzer (Hitachi 7170). Fasting blood glucose (FBG), fasting insulin (FINS), Hcy and haemoglobin A1c (HbA1c) levels were detected at the central chemistry laboratory in the Beijing Chao-Yang Hospital Affiliated to Capital Medical University. The homeostasis model assessment index of insulin resistance (HOMA-IR) was calculated with the following equation: HOMA-IR = glucose (mmol/L) × insulin (mIU/L)/22.5. 2 All subjects completed a questionnaire about their medical history (self-reported as diagnosed by medical doctors).
Statistics
The data were analysed with SPSS 15.0 (SPSS, Inc., Chicago, IL). Continuous data were expressed as means ± standard deviation (SD). The TG, FINS and HOMA-IR data were not normally distributed, so these values were expressed as the medians (ranges). The TG, FINS and HOMA-IR data were fitted to a normal distribution following log transformations. The proportions were analysed using chi-square tests. We also used Pearson and Spearman correlation analyses. Multivariate analysis was used to evaluate the correlation. All statistical tests were two-tailed, and p < 0.05 was considered significant.
Results
There were no significant differences between the control and IGT groups in terms of age, gender, FBG, TC, LDL-C or apoB (data not shown). The prevalence of essential hypertension in the IGT group was significantly greater than that among the controls (38.9% vs 17.2%, p < 0.05). The IGT patients had higher body mass index (BMI), FINS, HbA1c and HOMA-IR levels than the control subjects (BMI: 25.89 ± 3.34 vs 24.82 ± 3.06 kg/m2, p < 0.05; FINS: 7.56 (2.00–30.98) vs 6.69 (2.24–25.40) µIU/mL, p < 0.05; HbA1c: 6.34 ± 1.09% vs 5.71 ± 0.62%, p < 0.01; HOMA-IR: 2.14 (1.03–9.03) vs 1.55 (0.53–5.87), p < 0.01). The Hcy levels of the IGT patients were higher than those of the control subjects (16.75 ± 7.63 vs 14.02 ± 3.25 µmol/L, p < 0.01). Increased plasma TG levels and decreased plasma HDL-C and apoAI levels were observed in the IGT patients compared to the control group (TG: 1.65 (0.46–6.31) vs 1.38 (0.44–4.65) mmol/L; p < 0.05; HDL-C: 1.29 ± 0.29 vs 1.47 ± 0.35 mmol/L, p < 0.01; apoAI: 1.31 ± 0.26 vs 1.44 ± 0.27 g/L, p < 0.01).
HHcy was defined by a plasma Hcy level greater than 15 µmol/L. 2 According to the presence of HHcy, the patients were further classified into the following four groups: normal control (n-cont) subjects without hyperhomocysteinaemia (HHcy), control subjects with HHcy (H-cont), IGT patients without HHcy (n-IGT) and IGT patients with HHcy (H-IGT). Table 1 shows the characteristics of the subgroups. There were no significant inter-subgroup differences in age, gender, BMI, FBG, FINS, HbA1c, TC, LDL-C, HDL-C, TG or ApoB compared to the control or IGT groups. There were no differences in HOMA-IR values or apoAI levels between the n-cont and H-cont groups. However, the H-cont group exhibited a significantly higher prevalence of essential hypertension than the n-cont group (23.5% vs 12.8%, p < 0.05). In addition to a higher prevalence of hypertension (46.4% vs 28.2%, p < 0.05), the patients in the H-IGT group also exhibited greater HOMA-IR values than the n-IGT group (2.18(1.21–9.03) vs 1.88 (1.03–5.20), p < 0.05). Moreover, significantly decreased apoAI levels were observed in the H-IGT group compared to the n-IGT group (1.19 ± 0.21 vs 1.37 ± 0.28 g/L, p < 0.01).
Comparison of clinical parameters of subgroups of control subjects and IGT patients, stratified by plasma level of homocysteine.
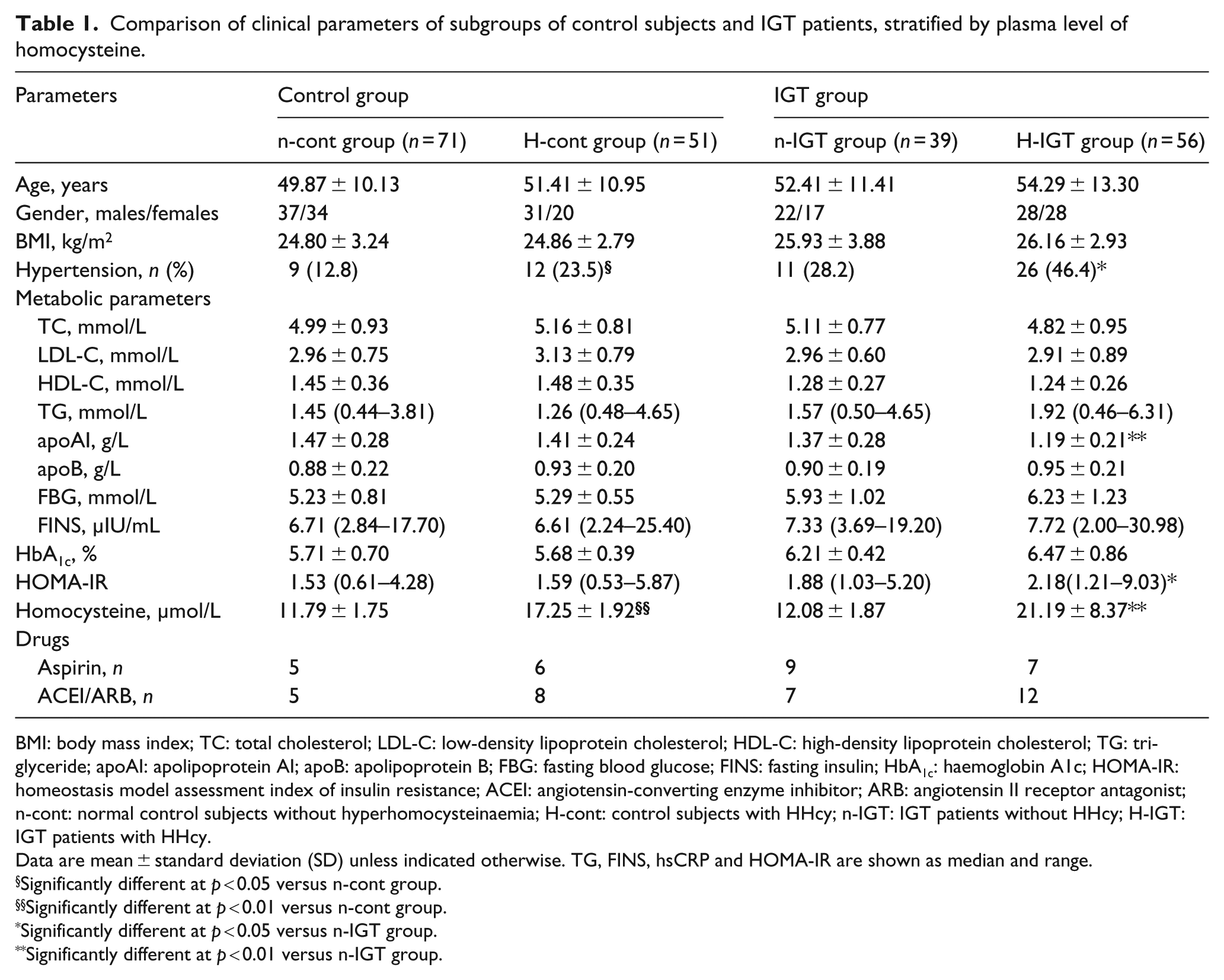
BMI: body mass index; TC: total cholesterol; LDL-C: low-density lipoprotein cholesterol; HDL-C: high-density lipoprotein cholesterol; TG: triglyceride; apoAI: apolipoprotein AI; apoB: apolipoprotein B; FBG: fasting blood glucose; FINS: fasting insulin; HbA1c: haemoglobin A1c; HOMA-IR: homeostasis model assessment index of insulin resistance; ACEI: angiotensin-converting enzyme inhibitor; ARB: angiotensin II receptor antagonist; n-cont: normal control subjects without hyperhomocysteinaemia; H-cont: control subjects with HHcy; n-IGT: IGT patients without HHcy; H-IGT: IGT patients with HHcy.
Data are mean ± standard deviation (SD) unless indicated otherwise. TG, FINS, hsCRP and HOMA-IR are shown as median and range.
Significantly different at p < 0.05 versus n-cont group.
Significantly different at p < 0.01 versus n-cont group.
Significantly different at p < 0.05 versus n-IGT group.
Significantly different at p < 0.01 versus n-IGT group.
Hcy levels were positively correlated with HOMA-IR values (r = 0.33, p < 0.01, 95% confidence interval: 0.19–0.45). However, this positive correlation was not observed after adjustments for age, gender, BMI and TG levels. A negative correlation between Hcy and apoAI levels was observed (r = −0.19, p < 0.05, 95% confidence interval: −0.34 to −0.03), and this negative correlation remained following adjustments for age, gender, BMI and HOMA-IR (r = −0.24, p < 0.05).
To further evaluate the relationship between plasma Hcy and apoAI levels, we performed a multivariate regression analysis. The included variables were age, gender, BMI, history of essential hypertension, HOMA-IR and TG. We found that the plasma Hcy level was a factor that was independently associated with apoAI levels (β = −0.02, p < 0.05).
Discussion
In this study, the IGT patients exhibited higher plasma Hcy levels and HOMA-IR values and significantly lower plasma levels of HDL-C and apoAI than the control subjects. There were differences in HOMA-IR and apoAI levels between the H-IGT and n-IGT groups, but not between the control groups. Furthermore, multivariate regression analysis indicated that plasma Hcy level was a factor that was independently associated with apoAI level.
Many epidemiological studies have demonstrated that IGT patients are at a higher risk of cardiovascular disease. 5 In this study, the IGT patients exhibited significant insulin resistance. Insulin plays a key role in maintaining healthy endothelium by activating phosphorylation in the phosphatidylinositol 3-kinase (PI3-K)/Akt pathway and promoting the activity and expression of endothelial nitric oxide synthase (eNOS). 8 These results suggest that insulin resistance is involved in the pathogenesis of cardiovascular disease in IGT patients.
HHcy is an independent risk factor for cardiovascular disease. 1 The IGT patients exhibited higher plasma Hcy levels than the control subjects. A positive correlation between Hcy and HOMA-IR was observed. Our previous study demonstrated that Hcy interrupts insulin signalling pathway by inducing endoplasmic reticulum stress and up-regulating the expression of resistin.3,4 Hypertensive patients with HHcy exhibit more serious insulin resistance than patients without HHcy. 2 Our study results are consistent with those of previous studies that have supported the association of HHcy with insulin resistance.2–4 Therefore, we suggest that HHcy might be involved in the development of cardiovascular disease in IGT patients via the promotion of insulin resistance.
The subjects with HHcy in both the control and IGT groups exhibited higher prevalences of essential hypertension. HHcy has been reported to disturb the bioavailability of tetrahydrobiopterin, cause endothelial and vascular dysfunction and lead to increased blood pressure. 9 Elevated blood pressure is one factor that is likely to mediate cardiovascular disease in IGT patients.
ApoAI, a major component of HDL, plays a key role in adenosine triphosphate (ATP)–binding cassette transporter A1 (ABCA1)–dependent cholesterol outflow from macrophages and has the capacity to activate lecithin-cholesterol acyltransferase (LCAT) and promote the transformation from cholesterol to cholesterol ester in HDL.10,11 Moreover, apoAI is also involved in the anti-oxidative and anti-inflammatory effects of HDL.10,11 Animal studies have demonstrated that elevated Hcy levels inhibit apoAI synthesis in the livers of mice. 7 In male patients with coronary artery disease, Hcy levels are negatively related to apoAI levels. 7 Low-dose folic acid supplementation for 12 weeks significantly decreases Hcy levels and also increases apoAI levels. 12 Our results are consistent with the results of others and demonstrated the negative correlation between Hcy and apoAI. 7 The inhibition of apoAI synthesis that is induced by Hcy might partially contribute to the cardiovascular damage mediated by HHcy.
In conclusion, the IGT patients exhibited significant insulin resistance and HHcy. Increased plasma Hcy levels were associated with decreased apoAI levels. HHcy induced the development of cardiovascular disease, and the reduction of apoAI levels might contribute to this damage. An effective treatment for HHcy might assist in the prevention and treatment of cardiovascular disease in IGT patients.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This work was supported by grants from the Major National Basic Research Program of China (No. 2011CB503904), the Chinese National Natural Science Foundation (Nos 81270369 and 81070244) and the Beijing Natural Science Foundation (No. 7142060) to Guang Wang.
