Abstract
Background:
Effect of pre-diabetes mellitus on post-myocardial infarction prognosis is unclear.
Methods:
Retrospective cohort analysis of 1056 myocardial infarction survivors with fasting plasma glucose and 2-h post-load plasma glucose measured. Major adverse cardiovascular events included death, non-fatal reinfarction and ischaemic stroke. Cox proportional hazard regression identified predictors of event-free survival. Continuous net reclassification improvement and integrated discrimination improvement determined the added predictive value of glycaemic indices.
Results:
Major adverse cardiovascular events occurred in 25.1% and 16.4% patients with and without pre-diabetes mellitus (hazard ratio with pre-diabetes mellitus: 1.56; 95% confidence interval: 1.17–2.08; p = 0.003) in the whole cohort and in 24.1% and 17.2% patients (hazard ratio with pre-diabetes mellitus, 1.43; 95% confidence interval: 1.03–1.98; p = 0.033) in the matched cohort, respectively. Pre-diabetes mellitus predicted major adverse cardiovascular events–free survival in whole (hazard ratio: 1.39; 95% confidence interval: 1.03–1.89; p = 0.033) and matched cohorts (hazard ratio: 1.42; 95% confidence interval: 1.01–1.99; p = 0.043). The 2-h post-load plasma glucose, but not fasting plasma glucose, predicted major adverse cardiovascular events–free survival in the whole (hazard ratio: 1.16; 95% confidence interval: 1.07–1.26; p < 0.0001) and matched cohorts (hazard ratio: 1.20; 95% confidence interval: 1.09–1.31; p < 0.0001). Adding 2-h post-load plasma glucose to models containing fasting plasma glucose, significantly improved net reclassification improvement and integrated discrimination improvement for both cohorts, but not vice versa.
Conclusion:
Pre-diabetes mellitus predicts major adverse cardiovascular events after myocardial infarction. The 2-h post-load plasma glucose predicts prognosis better than fasting plasma glucose in these patients.
Keywords
Introduction
New diagnosis of pre-diabetes mellitus (pDM), i.e., impaired fasting glucose (IFG) and impaired glucose tolerance (IGT), is common in patients after acute coronary syndrome (ACS) or myocardial infarction (MI).1–11 Some of the studies 1–4,10–14 associating new diagnosed abnormal glucose tolerance (AGT) with adverse post-MI prognosis, report the effect of IGT separately.11,12 IFG adversely affected post-ACS prognosis in some5–9,15 but not other studies.4,12,16–18 pDM diagnosed on HbA1c did not predict post-MI prognosis.18,19
The 2 h post-load glucose (2h-PG) was not measured in studies suggesting adverse post-ACS prognosis in patients with IFG.4–9,12,15–18 It is thus unclear whether the abnormal fasting plasma glucose (FPG) or the associated raised 2h-PG affected the outcomes. Studies suggesting association of adverse outcomes with AGT1,2,4,10–12 do not report the effect of 2h-PG on events either. Some studies suggest that 2h-PG is a better determinant of post-ACS prognosis than FPG, APG and HbA1c, 13,20,21 although not specifically in the pDM patients.
The post-ACS patients are at high risk of cardiovascular events, and pDM may impose additional risk of adverse prognosis. In this study, we explore the relation of pDM with post-MI prognosis in patients without known diabetes mellitus (KDM) and whether FPG or 2h-PG best predicts adverse events in this group.
Methods
Following Glucose tolerance in Acute Myocardial Infarction (GAMI) study 3 and according to some guidelines,22,23 all consecutive post-MI survivors admitted to our unit without KDM underwent pre-discharge oral glucose tolerance test (OGTT) on/after the third day of admission as routine clinical care. Data on age, gender, risk factors for coronary artery disease (CAD), previous history of MI, pre-hospital and discharge medications, vital signs, presence of congestive heart failure and ST-segment depression, troponin I, creatinine, FPG, 2h-PG and revascularisation status were prospectively recorded for the Myocardial Infarction National Audit Project. For this cohort study, we retrospectively analysed these data on patients admitted between November 2005 and October 2008, newly diagnosed with pDM. Patients who died or were transferred to other centres before the OGTT or did not tolerate OGTT were excluded. Permission was sought from the East Yorkshire and North Lincolnshire Research Ethics Committee to analyse the data. As the study retrospectively analysed routinely collected anonymised data on standard clinical practice to contribute to a national audit database, the committee waived the need for formal ethical approval and patient consent. 11
FPG (after fast of ⩾8 h) and 2h-PG (2 h after ingestion of glucose (75 g) in 200 mL of water) were enzymatically determined using the glucose oxidase method. Glycaemic categories: normal glucose tolerance (NGT): FPG <5.6 mmol/L and 2h–PG <7.8 mmol/L, newly diagnosed diabetes (NDM): FPG ⩾7.0 and/or 2h–PG ⩾11.1 mmol/L, ‘isolated’ IFG: FPG = 5.6–6.9 mmol/L and 2h-PG <7.8 mmol/L, ‘isolated’ IGT: FPG <5.6 mmol/L and 2h-PG = 7.8–11 mmol/L and ‘combined’ IFG and IGT: FPG = 5.6–6.9 mmol/L and 2h-PG = 7.8–11 mmol/L. 24 pDM was defined as ‘isolated’ or ‘combined’ IFG and IGT. Pre-existing diabetes mellitus (DM) was diagnosed from history or if patient was on treatment. HbA1c was not done, as it was not routine practice in the unit and was not recommended in guidance at the time when data were collected.22,23
The first occurrence of major adverse cardiovascular event (MACE), that is, all cause death, non-fatal reinfarction (according to the universal definition) or non-haemorrhagic stroke (typical neurological deficit for >24 h with radiological confirmation) during follow-up was obtained from patient records. Mortality data collected from the hospital records for patients who died in hospital and general practitioner medical records for patients, who died in the community, were confirmed by the office of public health intelligence. Completeness of follow up was ensured by manual review of hospital and general practice records.
Statistical analysis
Continuous and categorical variables are presented as medians (inter-quartile range) and counts and proportions (%), respectively. NGT and pDM groups were compared using the analysis of variance (ANOVA) and Kruskal–Wallis test for parametric and non-parametric continuous variables, respectively, and chi-square test for categorical variables. Event-free survival was estimated using the Kaplan–Meier method. Multivariate Cox proportional-hazards regression (MedCalc Statistical Software, version 17.0.4, Ostend, Belgium) was used to analyse the effect of variables on event-free survival. Gender, smoking status, hypercholesterolaemia, hypertension, history of MI and revascularisation, discharge diagnosis, discharge prescription, in-patient revascularisation status, GRACE risk score (GRS), pDM status, FPG and 2h-PG were ‘entered’ into the model. Variables were included in the same model if test for multicollinearity using variance inflation factor (VIF) was negative. Hazard ratios (HRs) and 95% confidence intervals (CIs) are reported. To reduce bias due to confounding variables, a cohort of 348 pairs of pDM and NGT patients matched on gender and the strongest predictors of MACE-free survival in the whole cohort (GRS, hypercholesterolaemia and history of MI) was established. These groups were then used for analysis.
Nested models were compared using chi-square likelihood ratio tests to determine whether the logistic regression models that included FPG or 2h-PG provided a significantly better fit than those without. Nested and non-nested models including 2h-PG and/its FPG were compared using Akaike information criterion (AIC) to estimate the probability that a given model is the ‘best’ fitting model of those studied. Predicted probabilities of MACE were calculated from logistic regression models using GRS, FPG and 2h-PG individually and in combination. The incremental predictive value from adding FPG and 2h-PG to models with GRS was analysed using category-free continuous net reclassification improvement (cNRI>0) and integrated discrimination improvement (IDI). Without pre-defined thresholds of predicted risk for the models, categorical NRI was not used. The event (NRIe) and non-event NRI (NRIne) were calculated.
Results
Of 1056 patients, 206 with NDM were excluded. Of the remaining, 425 with NGT and 425 with pDM (50 with isolated IFG, 86 with IFG and IGT, 289 with isolated IGT) were included in this analysis. We identified 348 matched pairs of patients with NGT and pDM. Table 1 describes the baseline characteristics of patients in the whole and matched cohorts.
Baseline characteristics of the study population.
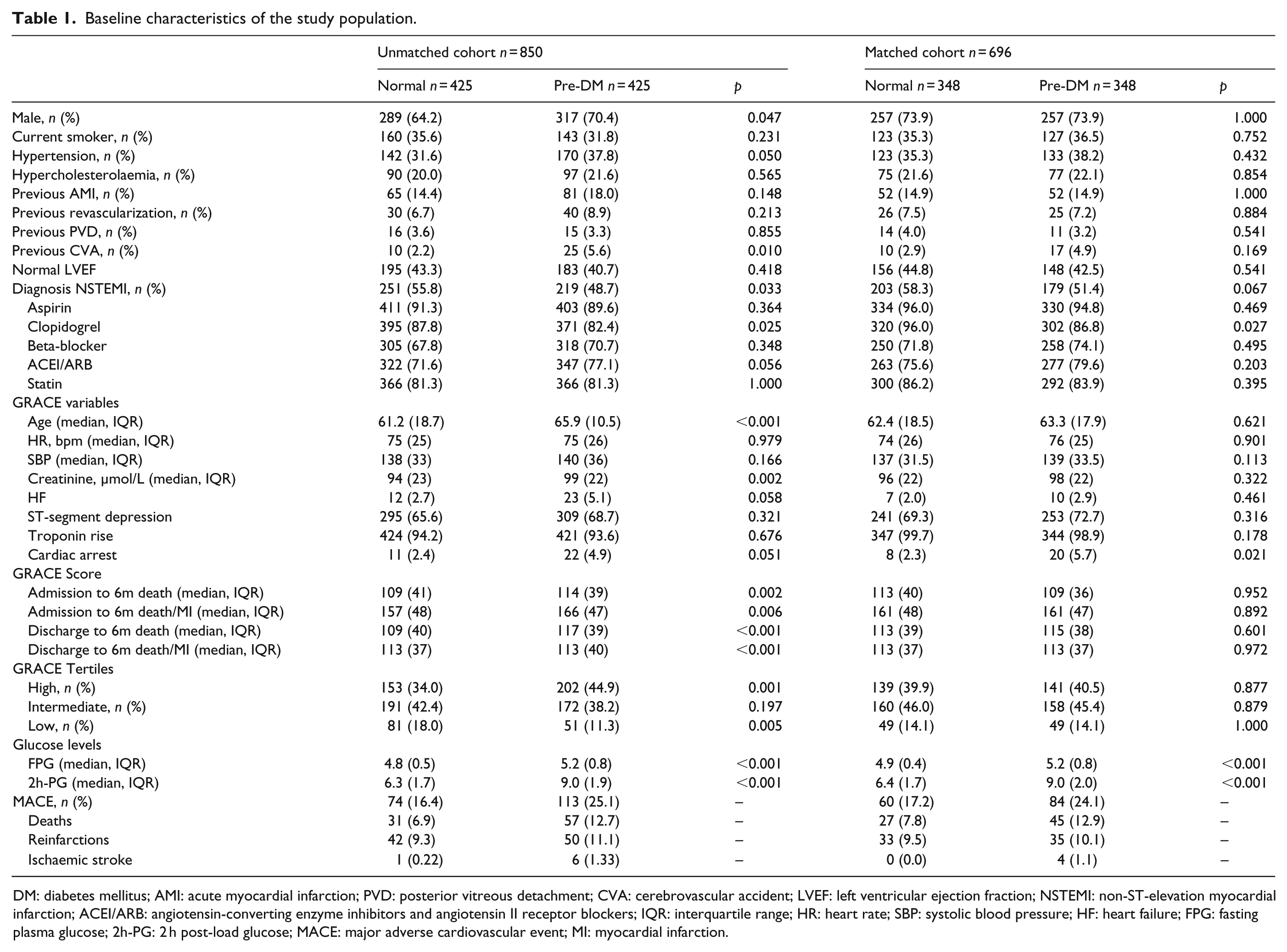
DM: diabetes mellitus; AMI: acute myocardial infarction; PVD: posterior vitreous detachment; CVA: cerebrovascular accident; LVEF: left ventricular ejection fraction; NSTEMI: non-ST-elevation myocardial infarction; ACEI/ARB: angiotensin-converting enzyme inhibitors and angiotensin II receptor blockers; IQR: interquartile range; HR: heart rate; SBP: systolic blood pressure; HF: heart failure; FPG: fasting plasma glucose; 2h-PG: 2 h post-load glucose; MACE: major adverse cardiovascular event; MI: myocardial infarction.
During a median follow up of 2.8 (range: 0.5–5) years, 187 (22.0%) and 144 (20.7%) MACEs occurred in the whole and matched cohorts, respectively. MACE occurred in 113 (25.1%) and 74 (16.4%) subjects with and without pDM (HR with pDM: 1.56; 95% CI: 1.17–2.08; p = 0.003) in the whole cohort and 84 (24.1%) and 60 (17.2%) subjects with and without pDM (HR with pDM: 1.43; 95% CI: 1.03–1.98; p = 0.033) in the matched cohort, respectively. Death occurred in 57 (12.7%) and 31 (6.9%) subjects with and without pDM (HR with pDM: 1.89; 95% CI: 1.24–2.87; p = 0.004) in the whole cohort and 45 (12.9%) and 27 (7.8%) subjects with and without pDM (HR with pDM: 1.71; 95% CI: 1.08–2.71; p = 0.026) in the matched cohort, respectively. pDM did not affect the reinfarction rates. MACE-free survival was worse in the pDM group compared with the NGT group, both in the whole and matched cohort (Figure 1).
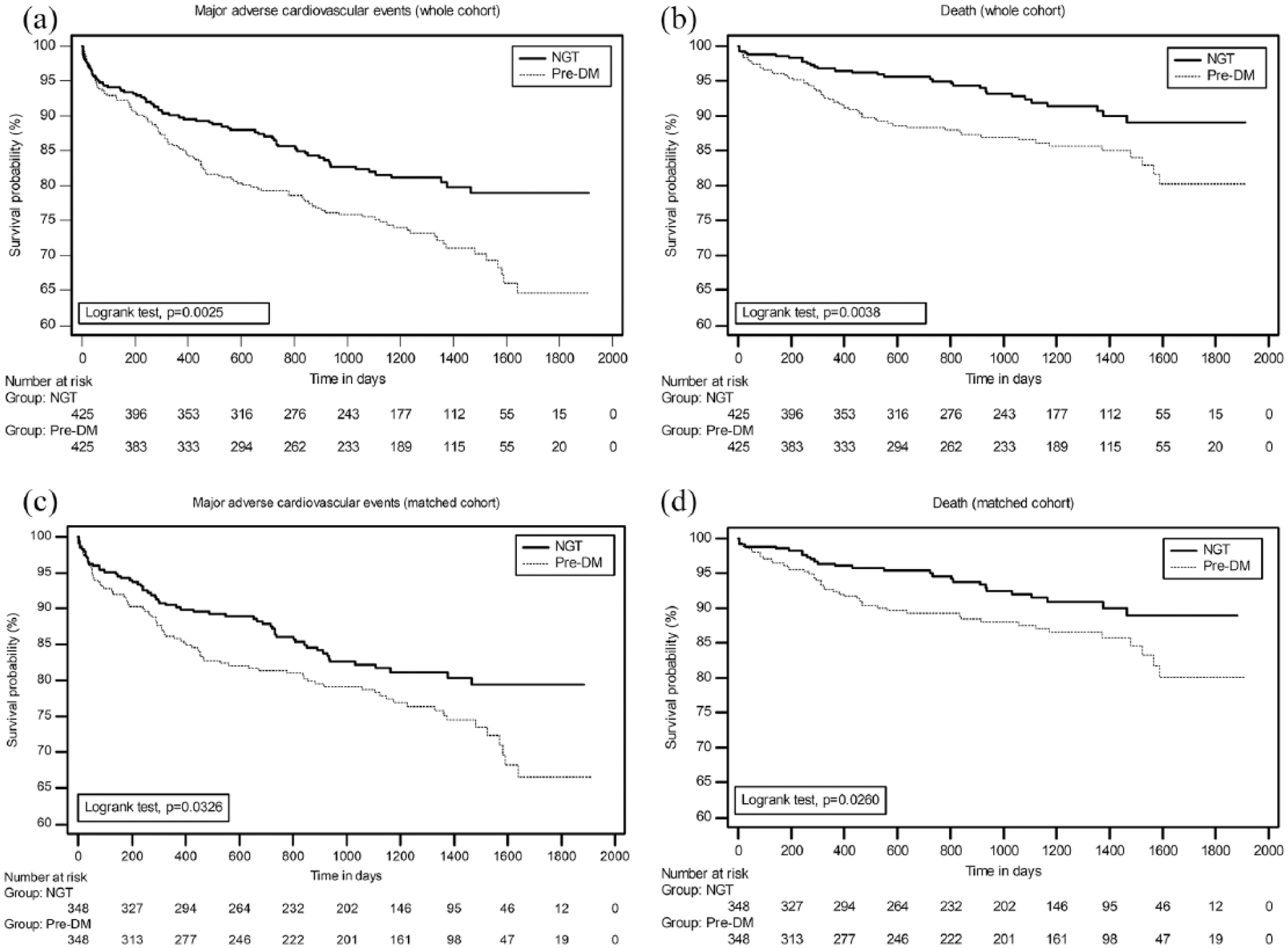
Kaplan–Meier estimates of the composite primary outcome and death.
pDM was an independent predictor of MACE-free survival in both the whole cohort (HR: 1.39; 95% CI: 1.03–1.89; p = 0.033) and the matched groups (HR: 1.42; 95% CI: 1.01–1.9; p = 0.043) along with the GRS, history of MI and hypercholesterolaemia (Supplemental Table 1). pDM also independently predicted death in the whole (HR: 1.59; 95% CI: 1.00–2.52; p = 0.049) and matched (HR: 1.80; 95% CI: 1.10–2.96; p = 0.020) cohort. In the whole cohort, 2h-PG but not FPG was an independent predictor of MACE-free survival along with GRS, previous history of MI, hypercholesterolaemia and low clopidogrel use. This pattern persisted in the matched cohort (Table 2). For every mmol/L rise in the 2h-PG, there was 16%–20% increase in the relative risk of MACE. When pDM categories, that is, isolated IFG, isolated IGT and IFG/IGT were included in the Cox proportional hazard models with other covariates, only the IGT remained an independent predictor of MACE in the whole (HR: 1.62; 95% CI: 1.17–2.24; p = 0.004) and in the matched cohorts (HR: 1.74; 95% CI: 1.21–2.49; p = 0.002) (Supplemental Table 2).
Candidate predictors of MACE-free survival on Cox proportional hazard regression.
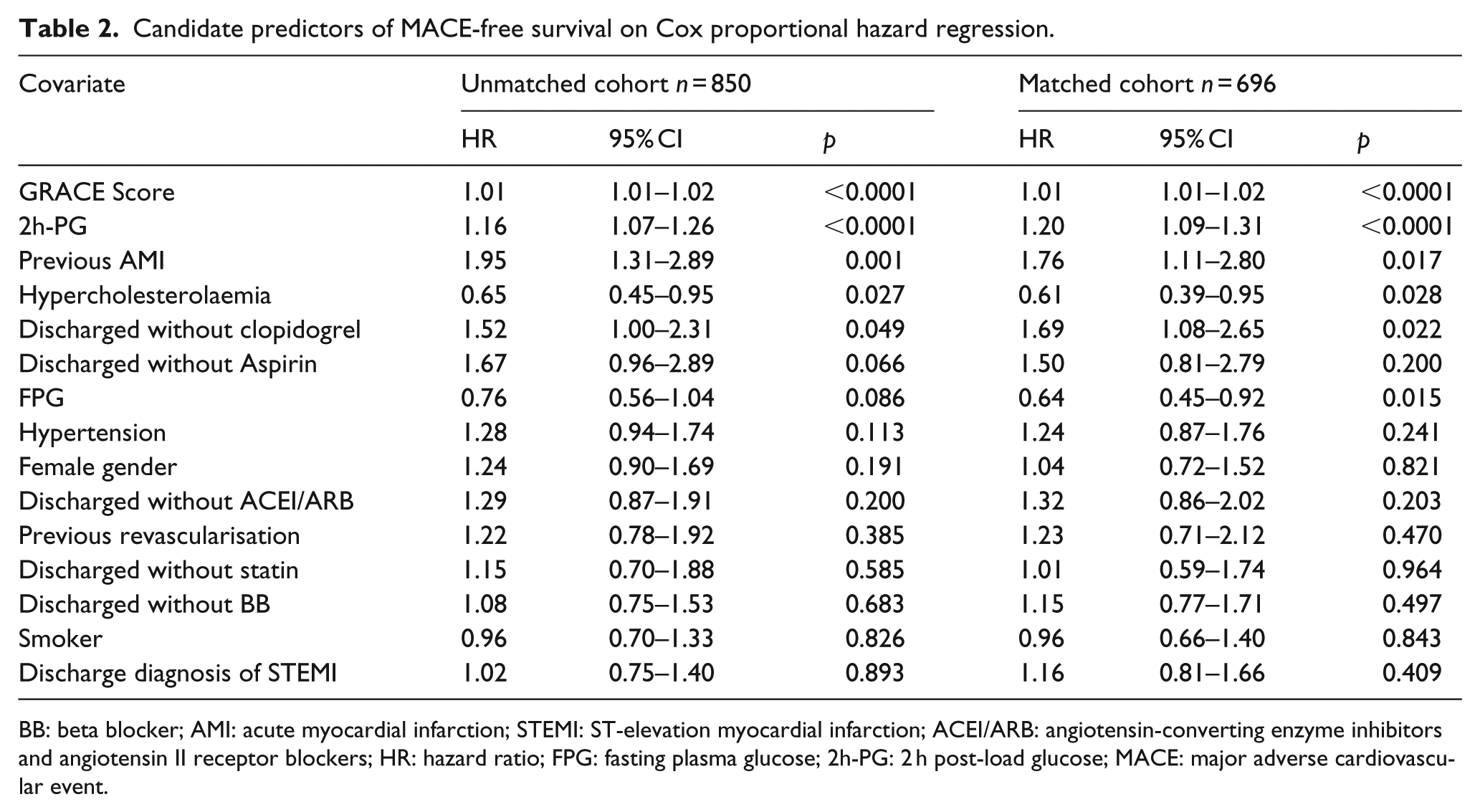
BB: beta blocker; AMI: acute myocardial infarction; STEMI: ST-elevation myocardial infarction; ACEI/ARB: angiotensin-converting enzyme inhibitors and angiotensin II receptor blockers; HR: hazard ratio; FPG: fasting plasma glucose; 2h-PG: 2 h post-load glucose; MACE: major adverse cardiovascular event.
Comparing non-nested models (Table 3) including FPG, 2h-PG or GRS alone and in combination, the models containing 2h-PG yielded the lowest AICc both in the unmatched and matched cohorts with a 96%–99% probability of representing the best model. Comparing non-nested models containing FPG or 2h-PG and both, the model containing 2h-PG alone yielded the lowest AICc in the unmatched cohort. Log-likelihood ratio tests were used to compare the fit of predictive models that were based on GRS (Table 3) combined with FPG or 2h-PG. Comparing nested models derived from the whole cohort, those including 2h-PG significantly improved the prediction of models based on GRS or FPG for MACE. Addition of FPG did not significantly improve the prediction of these models.
Akaike’s information and likelihood ratio test to determine the best fitting model for predicting MACE.
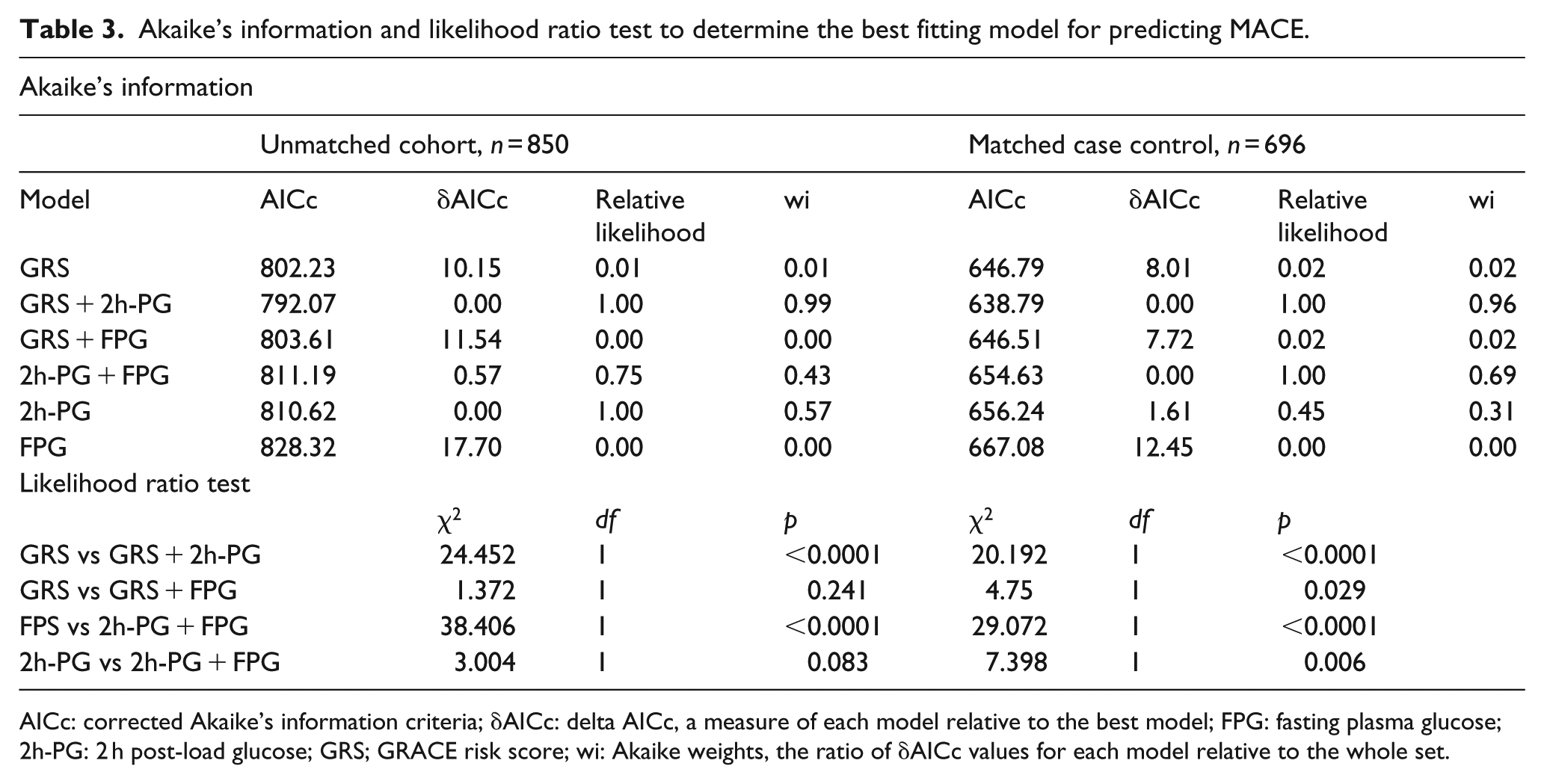
AICc: corrected Akaike’s information criteria; δAICc: delta AICc, a measure of each model relative to the best model; FPG: fasting plasma glucose; 2h-PG: 2 h post-load glucose; GRS; GRACE risk score; wi: Akaike weights, the ratio of δAICc values for each model relative to the whole set.
In the unmatched cohort, addition of 2h-PG to a model containing FPG as the only glycaemic index, correctly reclassified 106 (56.7%) patients who suffered MACE and 398 (60.0%) patients who did not, yielding an NRI>0 of 33.4% (p < 0.001). In the matched cohort, correct reclassification occurred in 80 (55.5%) patients with MACE and 325 (46.7%) without resulting in NRI>0 of 28.9% (p = 0.002). Addition of FPG to a model containing 2h-PG did not significantly change NRI>0 (Table 4). Adding 2h-PG to a model containing FPG, but not vice versa, significantly improved the integrated discrimination in both cohorts. Addition of 2h-PG to models including GRS alone or GRS and FPG resulted in significantly better NRI>0 and IDI in both cohorts.
Continuous net reclassification improvement and integrated discrimination index for model improvement.
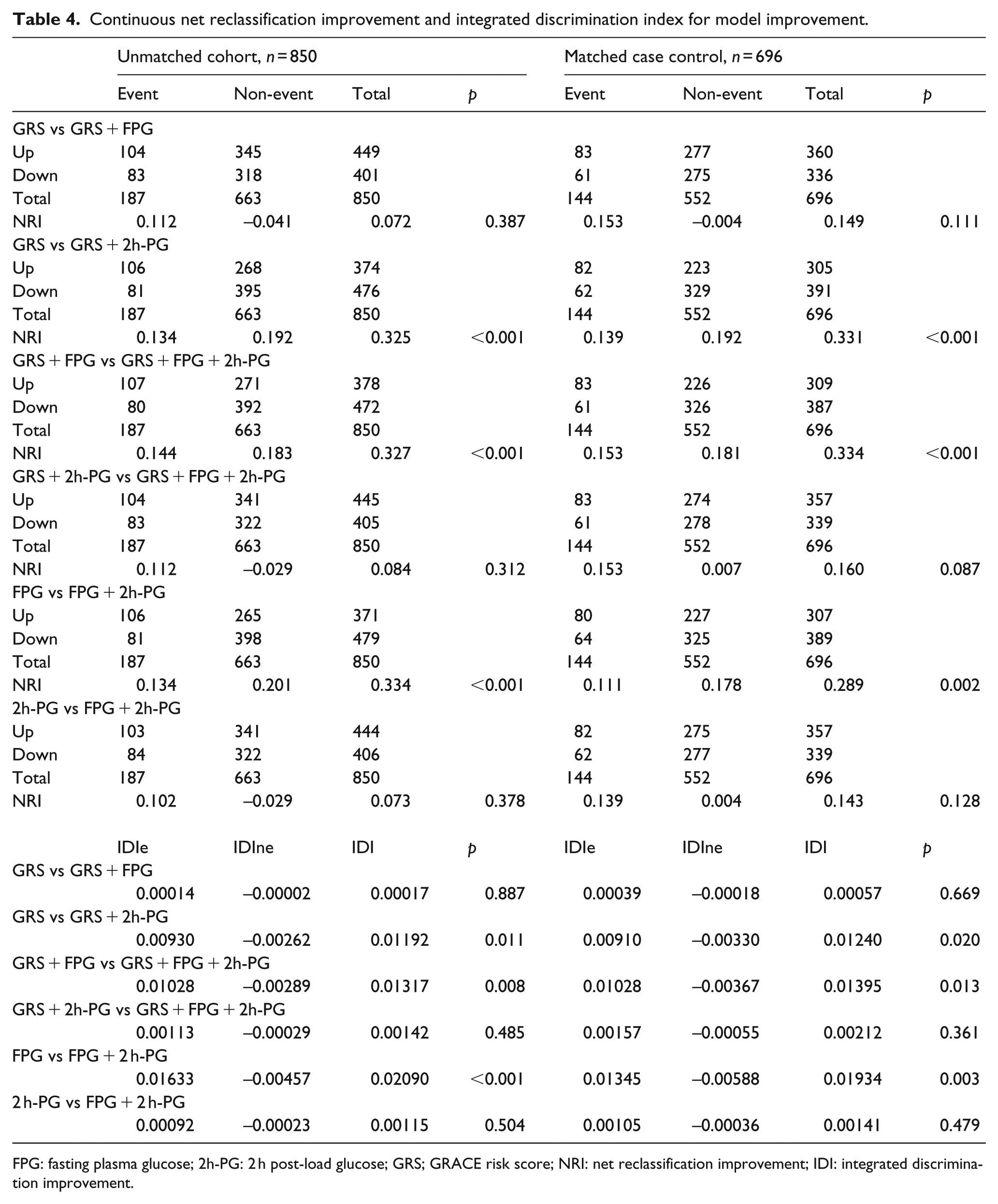
FPG: fasting plasma glucose; 2h-PG: 2 h post-load glucose; GRS; GRACE risk score; NRI: net reclassification improvement; IDI: integrated discrimination improvement.
Discussion
This study confirms that (1) newly diagnosed pDM is an independent predictor of post-MI prognosis and (2) 2h-PG is a better predictor of prognosis in these patients than FPG which does not contain independent predictive information even if impaired.
We included both IFG and IGT in our pDM group as increased risk of DM and cardiovascular disease (CVD) are associated with both the conditions. 23 However, it is unclear whether these adversely affect prognosis in MI survivors without KDM as they do in general population. OGTT in patients with CVD is only recommended if HbA1c and FPG levels are ‘inconclusive’. 22 These tests under-diagnose dysglycaemia. 13 Undiagnosed pDM could add to the already high prognostic risk of this population and would affect the advice given to these patients. Thus, we wanted to explore whether (1) fasting and/or post-load hyperglycaemia below the threshold of DM affected post-MI prognosis, (2) FPG alone could identify this risk and (3) 2h-PG added any prognostic risk over and above FPG.
pDM independently predicted MACE and death both in the whole and matched cohorts. Some post-MI studies11,12,20 have shown an independent effect of IGT on prognosis. Our study differs from previous studies in many ways. This study exclusively includes patients with pDM diagnosed after ACS, the pDM population is larger, may include patients with higher GRSs and has longer follow-up and higher event rates. IFG also adversely affected post-MI prognosis.5–9,15,17,25,26 However, without OGTT, it is unclear whether the undiagnosed IGT or DM, rather than the IFG, affected outcomes in these studies. Without the inclusion of GRS (or all its individual components) in the regression models, it is uncertain if GRS-adjusted IFG would independently predict prognosis. In this study IGT, not IFG, predicted MACE. IFG did not predict prognosis in other post-ACS studies.4,6,12,16,18,20 This is the only study to show that pDM independently predicts post-ACS prognosis after adjusting for the GRS.
We explored whether FPG or 2h-PG influenced post-MI prognosis in patients with pDM. Studies suggesting association of MACE with AGT1,2,4,10,12 do not report the effect of 2h-PG on MACE. Kitada et al. 1 report that 2h-PG only above a threshold independently predicted post-MI MACE. In the GAMI study, 2h-PG was not an independent predictor of events.3,14 We have reported that 2h-PG was a better determinant of post-MI prognosis than FPG or APG.13,21 In the EUORASPIRE IV 20 and Silent Diabetes Study, 27 2h-PG, but not FPG or HbA1c, was related to prognosis. The 2h-PG, but not the FPG, was associated with adverse post-MI prognosis in patients with pDM in this. Adding 2h-PG to a model containing FPG improved both NRI and IDI. This is the only study to show that 2h-PG, adjusted for GRS independently predicts post-ACS prognosis and improves the ability of models containing GRS to predict prognosis in pDM patients. In the NAVIGATOR trial, 18 FPG, 2h-PG or HbA1c were not independently associated with prognosis. CVD was present in only 28% of the trial patients though that was the strongest predictor of outcome. Our study population is different from this trial in that all subjects were MI survivors and not drawn from the general population. In the BIO-RESORT trial, 28 pDM was independently associated with worse prognosis in patients undergoing coronary stenting, 70% of whom had ACS. The increased macrovascular morbidity associated with higher 2h-PG rather than FPG seen here may be related to progression of atherosclerosis demonstrated with post-challenge rather than fasting hyperglycaemia. 29 The potential mechanisms for this include (1) more prolonged glycaemic exposure in IGT compared with IFG due to post-load hyperglycaemia, (2) higher prevalence of the metabolic syndrome in IGT patients and (3) greater diurnal fluctuations in plasma glucose due to higher 2h-PG may have a more damaging effect. 30
The limitations of a retrospective observational study have been partially mitigated by using a matched cohort. Although national death register was not consulted directly, a linked general practice database was used. A few patients with reinfarctions admitted to other hospitals may have been missed. Although every effort was made to ensure completeness of the data, information recorded incompletely could not be used in statistical models. Exclusion of small number of patients, albeit for valid reasons, and mainly Caucasian study population could affect the generalizability of the results. Without admission HbA1c, pre-admission hyperglycaemia has not been identified. Thus, some patients with hitherto undiagnosed diabetes may have been included. As OGTT done at or after 5 days seems to reliably predict long-term glucometabolic state, 31 it is unlikely that random fluctuation in glycaemia or stress hyperglycaemia affected our results even though OGTT was not repeated pre- or post-discharge. As pre-discharge 2h-PG predicted post-MI outcomes in pDM patients, the reproducibility of this test and its relation to long-term glucometabolic status may be less relevant when assessing prognostic risk. As IGT is associated frequently with metabolic syndrome, in the absence of anthropometric measurements and other risk factors, it is unclear whether this affected outcomes. The progression of IGT to DM over the follow-up period may also have affected the outcomes.
This study suggests that in patients without KDM (1) newly diagnosed pre-diabetes is associated with worse post-ACS prognosis and (2) 2h-PG is a better predictor of post-MI prognosis in patients with new pre-diabetes than FPG. Thus an appropriately timed OGTT after an MI may be warranted to identify these high-risk patients.
Key messages
Pre-DM is an independent predictor of MACE-free survival in patients without known diabetes after adjusting for the GRACE score.
IGT, but not other categories of pre-DM, is an independent predictor of MACE-free survival after adjusting for the GRACE score.
2h-PG, but not FPG, predicts MACE-free survival after adjusting for the GRACE score.
Addition of 2h-PG to a model containing FPG and GRACE score improved NRI and IDI, suggesting that the 2h-PG is a better predictor of post-MI MACE than FPG in patients with pDM.
Supplemental Material
Supplemental_data – Supplemental material for Pre-diabetes mellitus newly diagnosed after myocardial infarction adversely affects prognosis in patients without known diabetes
Supplemental material, Supplemental_data for Pre-diabetes mellitus newly diagnosed after myocardial infarction adversely affects prognosis in patients without known diabetes by Sudipta Chattopadhyay, Anish George, Joseph John and Thozhukat Sathyapalan in Diabetes & Vascular Disease Research
Footnotes
Acknowledgements
The authors thank all the nurses that helped with the collection of data. They also thank the patients whose data were utilised for this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
