Abstract
In patients with diabetes mellitus, around 50% of deaths due to cardiovascular causes are sudden cardiac deaths. The prevalence of diabetes in cohorts with chronic heart failure is increasing, and while sudden cardiac death is an increasingly rare mode of death in chronic heart failure patients as a whole, the risk of this outcome remains high in those with diabetes. This review summarises the current knowledge on the incidence of sudden cardiac death in patients with diabetes and chronic heart failure, before discussing the causes of the excess risk seen in those with these coexistent conditions. We then describe current strategies for risk stratification and prevention of sudden cardiac death in these patients before discussing the priorities for further study in this area.
Introduction
The increasing prevalence of diabetes mellitus (DM) is a major global public health concern. The 2013 International Diabetes Federation Diabetes Atlas estimates 382 million sufferers worldwide, resulting in estimated health care expenditure of US$548 billion per annum. This is projected to soar to US$627 billion by 2035, as 592 million are expected to have the disease across the globe. 1 DM is an established risk factor for heart disease, and doubles the risk of cardiovascular morbidity and mortality. Indeed, 70% of patients with diabetes die from a cardiovascular cause, of which around 50% are sudden cardiac deaths (SCDs). 2 Furthermore, despite diabetes having a prevalence of just below 10% in the European adult population, around 30% of those with chronic heart failure (CHF) have the condition. An adverse cardiovascular outlook is seen even in the early stages of the development of the disease, with prediabetes widely recognised as being a non-benign condition. Indeed, a recent UK registry study quoted the prevalence of prediabetes at 35% in 2011, a more than 200% increase since 2003. 3 Given the growing numbers of those affected by dysglycaemia and cardiovascular disease, it is imperative that we accurately understand the risk of outcomes such as SCD in these individuals.
This review will summarise current knowledge of the incidence of SCD in patients with diabetes and heart failure and then discuss the pathophysiological causes of this outcome. We will then examine current risk stratification strategies before focussing on treatment options to prevent such events. We will conclude by highlighting important areas for further research to identify and treat those at risk of SCD.
The scale of the problem
SCD is defined as that which occurs within 1 hour of a change in symptoms, during sleep or while the patient is unobserved.4,5 Established risk factors for SCD include previous myocardial infarction (MI), systolic heart failure with a left ventricular ejection fraction (LVEF) <35% and previous ventricular arrhythmia, with a cumulative increase in risk seen in those with more than one of these conditions. 6
The incidence of SCD in the general population is estimated at around 1 in 1000 per year. 7 The risk for those with diabetes is substantially increased in both sexes, with large cohort studies reporting a two- to four-fold increase in the incidence of SCD in those with the condition8,9 and establishing diabetes as a key independent risk factor for SCD. 10 Further data confirm that the risk of sudden death increases throughout the prediabetic period prior to the development of frank diabetes, with a Finnish study noting a relative risk of 1.51 [95% confidence interval (CI) 1.07–2.14; p = 0.02] for SCD in non-diabetic men with impaired fasting glucose, when compared with those with normoglycaemia. 11
The observation of large populations over long periods of follow-up in these studies has allowed researchers to detect statistically significant differences between those with and without diabetes, despite the low overall event rates of SCD. In one of the largest cohort studies, there were only 418 SCDs in a population of 161,808 women over a total study duration of 16 years, with 67 of these deaths occurring in patients with diabetes. 7 As such, it is a challenge to estimate accurately the incidence of SCD in individuals with heart failure and diabetes as, despite the substantial increase in risk to those with both conditions, the number of those affected represents a small proportion of the total SCDs in the population as a whole. As a result, there are no large cohort studies published which have specifically investigated the risk in patients with heart failure and diabetes. In lieu of this, the current evidence base is informed by observational studies of cohorts with either diabetes or heart failure, or by subgroup analyses from RCTs studying medical or device treatments for heart failure.
Work from our institute has shown that there has been a near 80% decrease in the contribution of SCD to total mortality in CHF patients without diabetes as medical and device therapies have improved over the last 15 years. 5 Unfortunately, SCD remains a common mode of death in patients with diabetes and heart failure. 12 In our prospective cohort study of ambulant CHF patients, we noted a significantly higher incidence of SCD in patients with diabetes: 22 SCDs from 280 patients versus 23 SCDs from 811 patients without diabetes (p < 0.001) over the mean follow-up period of 960 days. We also noted a non-significant difference in the proportion of deaths due to SCD in our cohort: 24% in those with diabetes as compared with 15% in those without the condition. 12 Research from a group combining the results of two cohort studies which followed up patients after acute MI between 1996 and 2005 provides interesting data highlighting the risk of SCD in patients with DM with different degrees of left ventricular systolic dysfunction (LVSD). 13 The authors identified an extraordinarily high risk of SCD in patients with diabetes and a LVEF ⩽35%: 16% over a mean follow-up duration of 4 years, although the absolute number of events was small, with 15 deaths out of 95 patients with a LVEF ⩽ 35%. This was significantly higher than the equivalent incidence for those without diabetes: 4.9%, with 12 SCDs out of 244 patients with LVEF⩽35% (p < 0.01). Interestingly, the risk of SCD in patients with diabetes with a greater ejection fraction (LVEF > 35%) was nearly identical to that found in patients without diabetes with LVEF ⩽ 35%: 4.1% (22/533) versus 4.9% (12/244). Given that thresholds for primary prevention of SCD with device therapies focus on those with a LVEF <35%, this raises the possibility that many patients with heart failure and diabetes who would currently not qualify for such preventative therapy may be at a similar risk to those currently receiving such interventions. Given the study design and the low overall event rate of SCD, a degree of caution must be exercised when interpreting these findings, although they certainly highlight the need for more detailed study of these patients.
Data from subgroup analyses of RCTs give further insights into the risk of SCD in those with heart failure and diabetes. The Candesartan in Heart Failure: Assessment of Reduction in Mortality and Morbidity (CHARM) study included 7599 patients with heart failure, with 2163 of the total identified as having diabetes. 14 There was a significant increase in the risk of SCD in patients with diabetes: 40 versus 25.9 events per 1000 patient years of follow-up (p < 0.001) although the absolute number of SCDs was not reported. This difference was seen in patients with DM in both heart failure with a reduced ejection fraction (HFREF) and heart failure with a preserved ejection fraction (HFPEF), further highlighting that the condition increases the risk of SCD even in those without significant left ventricular (LV) systolic dysfunction.
Overall, there is a paucity of comprehensive data on the incidence of SCD in patients with diabetes and heart failure. Current observational studies lack adequate power to determine the risk of SCD in these patients accurately. Event rates in RCTs looking at broader populations of patients post-MI or with CHF are also low, exacerbating the inherent difficulties in interpreting the results of subgroup analyses of these cohorts. As such, there is a pressing need for further study of the risk of SCD posed to these patients in order that preventative strategies may be employed promptly and in a cost-effective fashion.
Causes of SCD in DM
The majority of SCDs are due to ventricular tachyarrhythmias, 15 which are often associated with an acute coronary syndrome. 16 As such, the increased coronary atheroma burden seen in patients with diabetes is one mechanism for their increased risk of SCD, and therefore, modification of traditional cardiovascular risk factors such as hypertension and dyslipidaemia may help abrogate the risk of this outcome (Figure 1). 17 Given their high risk of ischaemic heart disease, patients with diabetes are more likely to have had a previous coronary event, potentially leading to myocardial scarring which provides a greater arrhythmogenic substrate for SCD. In addition to an increased risk of macrovascular disease, patients with diabetes are at a greater risk of renal failure, which along with demonstrating a higher general level of microvascular disease is associated with electrolyte abnormalities which may predispose to arrhythmia. 18 Other pathological processes affecting the heart are also important; myocardial fibrosis seen in patients with diabetes leads to both HFPEF and HFREF, 19 therefore contributing to their risk of SCD as mentioned previously.
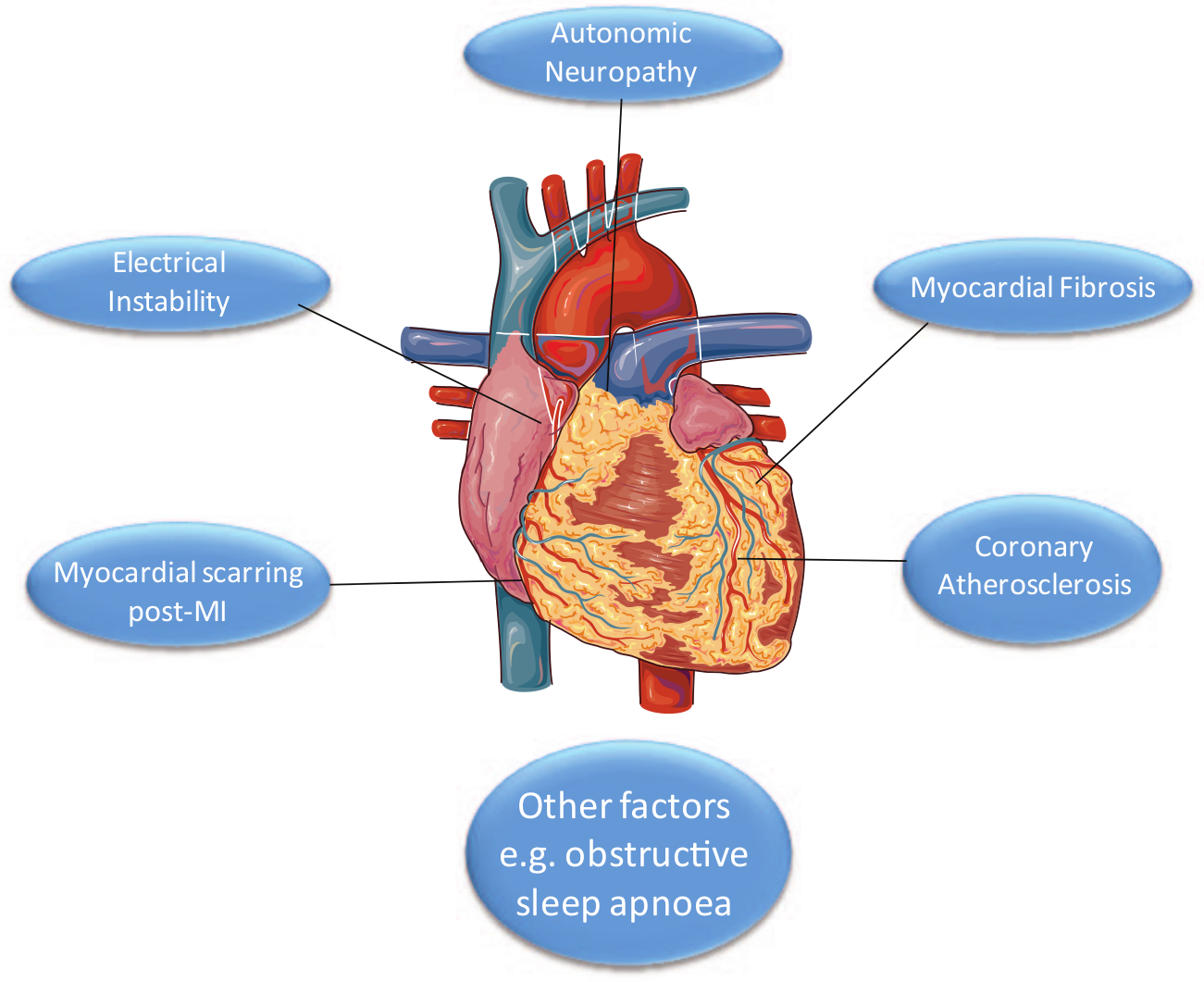
Factors increasing the risk of sudden cardiac death in patients with diabetes and chronic heart failure.
The phenomenon of diabetic autonomic neuropathy is well characterised, with deficient extrinsic stimulation of the heart in this condition being linked with an increased risk of SCD.20,21 Electrocardiograph (ECG) changes associated with DM demonstrate abnormal electrical propagation in the myocardium of sufferers of the condition, further increasing their likelihood of arrhythmic death.20,22 Conditions associated with diabetes such as obstructive sleep apnoea also appear to contribute to the risk of SCD independently, adding to the cumulative risk seen in those with the disease. 23
Dysglycaemia is central to the pathophysiology of diabetes and the metabolic syndrome. Hyperglycaemia has been linked with decreased heart rate variability, implying an increased risk of arrhythmia. 24 However, multiple large RCTs have failed to show any overall cardiovascular benefit for tight glucose control.25–27 Indeed, the Action to Control Cardiovascular Risk in Diabetes (ACCORD) trial was terminated prematurely after noting a higher overall mortality in the group treated to the intensive glycosylated haemoglobin (HbA1c) target. 27 Although hypoglycaemia is associated with a variety of indicators of ischaemia and electrical instability in experimental studies, 28 the relationship with mortality is not straightforward,28,29 and therefore, prevention of this alone is unlikely to improve the risk of SCD in patients with diabetes. Although both hyper- and hypoglycaemia may indeed predispose to arrhythmia and therefore SCD in diabetes, the failure of large-scale trials to improve cardiovascular outcomes by attempting to ameliorate dysglycaemia has shown that such strategies are unlikely to improve the overall macrovascular risk in patients with diabetes, regardless of their potential effects on SCD.
Risk stratification
Identifying those at risk of SCD is a significant challenge, given the lack of a broad body of evidence to estimate accurately the incidence of this outcome in different patient groups. Measures of autonomic function and heart rate variability have been studied to assess their predictive value for SCD.30,31 There are indications from studies involving small numbers of SCD events that some of these indices may help identify those at risk from SCD. Measures of heart rate variability such as the standard deviation of all normal RR intervals (SDNN) and heart rate turbulence are cumbersome to derive and are not applicable to those in atrial fibrillation (AF) – which accounts for around a third of all sufferers of CHF. Other measures of electrical instability such as T-wave alternans 32 and the corrected QT interval (QTc) 33 have been linked with SCD in diabetes, but their routine adoption for risk stratification of SCD has not been recommended in international guidelines. 2 In general, there are a large number of electrophysiological indices which are not simple to derive, have relatively little specific predictive value for SCD 34 and, despite having been studied for several years, have not been adopted into routine clinical practice. As such, new methods of risk stratification must be sought.
Clinical risk scoring systems incorporating a broad collection of indices such as demographic details, biochemical test results and radiological findings have been described and offer prognostic prediction in CHF patients, 35 including for SCD. 33 There are no such scores which have been specifically validated in cohorts of patients with diabetes and CHF, and further work must continue in this area to help identify those at risk of SCD with these coexistent conditions.
Prevention of SCD in DM
As previously discussed, contemporary medical therapy for CHF has reduced the incidence of SCD in patients without diabetes. 5 While angiotensin-converting enzyme inhibitors (ACEi), 36 beta-adrenoceptor blocking agents 37 and aldosterone antagonists38,39 improve mortality in those with diabetes and CHF, there are no large-scale trials which have specifically measured their effect on SCD in these patients. Given the significant difference in incidence of SCD seen between CHF patients with and without diabetes with modern CHF treatment, 40 these agents do not appear to offer comprehensive SCD prophylaxis in diabetes, and therefore, other strategies must be sought.
Implantable cardiac defibrillators (ICDs) have decreased mortality in the CHF population as a whole41,42 and are effective in patients with diabetes.43,44 Insights from the Multicenter Automatic Defibrillator Implantation Trial–Reduce Inappropriate Therapy (MADIT-RIT) highlight the differences in cardiac physiology between those with and without diabetes. As might be expected given their likelihood of autonomic neuropathy, inappropriate anti-tachycardia pacing (ATP) due to rapid atrial rhythms (not including AF) was significantly less in those with diabetes. 32 Patients with diabetes did have a significantly higher rate of appropriate therapy, however, with both ATP and electrical defibrillation, thus demonstrating the higher arrhythmogenic potential conferred by the disease. 32 Further understanding of these electrophysiological differences may allow the development of specific programming strategies tailored to those with diabetes to reduce their risk of SCD. International guidelines recommend the use of ICDs for the primary prevention of SCD in patients with diabetes and heart failure of ischaemic aetiology who have a LVEF <35%. 2
How can we prevent SCD in CHF patients with diabetes in the future?
More detailed investigation of the incidence of SCD in patients with CHF and diabetes is required to achieve accurate estimates of the risk of this outcome in these individuals. Large cohort studies designed specifically to investigate this would help characterise the magnitude of risk in these patients. Given the adverse clinical outcomes seen in prediabetes, similar work to study the incidence of SCD in patients across the spectrum of dysglycaemia and insulin resistance will be important to address the risks posed to the growing number of patients with the metabolic syndrome and impaired glucose tolerance. Studies should also examine the degree of risk of SCD in patients with CHF and diabetes who have a LVEF >35%. As mentioned previously, the limited data available indicate a similar incidence of SCD in those with DM and moderate LVSD and non-diabetic patients with LVEF <35%. 13 Patients with diabetes and less severe LVSD may therefore have the potential for benefit with ICD prophylaxis. Further work is required to determine the degree of risk posed to those with diabetes and CHF at all levels of LV impairment and indeed of symptom severity. Insights from further epidemiological investigation should help develop new and existing strategies to risk stratify patients with diabetes and CHF effectively to offer prophylaxis for SCD in a safe and cost-effective manner.
Conclusion
Despite great improvements in the treatment of non-diabetic patients with CHF over the last 15 years, leading to a substantial reduction in the incidence of SCD, 5 the risk of this outcome in patients with diabetes remains unacceptably high. 40 Given the increasing prevalence of DM in those with CHF, there is great potential to improve overall mortality rates should this difference in the rate of SCD be abrogated. In order for this to be achieved, investigators must work to quantify the risk of SCD more accurately in these patients, develop new methods of risk stratification to identify those at greatest risk, fully assess the value of current preventative strategies in patients with diabetes and identify new therapies to reduce the risk of SCD in this group.
Footnotes
Acknowledgements
Figure 1 was produced with Servier Medical Art, available at ![]()
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
A.M.N.W. is supported by a British Heart Foundation Clinical Research Fellowship Grant and R.M.C. is supported by a British Heart Foundation Intermediate Clinical Research Fellowship Grant.
