Abstract
Background:
It is unclear whether diabetes mellitus (DM) is an adverse prognostic factor in chronic heart failure (CHF) of ischaemic and non-ischaemic aetiology managed with contemporary evidence-based care.
Methods:
In total, 1091 outpatients with CHF with reduced ejection fraction were prospectively observed for a mean of 960 days. Total and cardiovascular mortality was quantified after accounting for potential confounders.
Results:
In total, 25.7% of patients had DM; this group was more likely to have CHF of ischaemic aetiology and was more symptomatic. Patients with DM received comparable medical- and device-based therapies, except for greater doses of loop diuretic. DM was associated with approximately doubled crude and adjusted risk of total and cardiovascular mortality. The association of diabetes with these outcomes in patients with ischaemic and non-ischaemic cardiomyopathies was of similar magnitude.
Conclusions:
In spite of advances in the management of CHF, DM remains a major adverse prognostic feature, irrespective of ischaemic/non-ischaemic aetiology.
Introduction
Diabetes mellitus (DM) is an increasingly common co-morbidity in patients with chronic heart failure (CHF), 1 with many studies quoting its prevalence to be in excess of 25% in CHF with reduced left ventricular (LV) ejection fraction. 1 –3 This trend is concerning given the established evidence base linking DM to significantly increased risk of total and cardiovascular death in the setting of CHF. 4 However, the data linking DM with adverse CHF outcomes are derived from patient cohorts receiving therapeutic regimens that would no longer be recognised as optimal. In particular, many of these studies administered beta-adrenoreceptor antagonists (beta-blockers) to fewer than half of the patients and were conducted prior to the publication of landmark cardiac resynchronisation trials; importantly, patients with DM appear to derive benefit from these therapeutic modalities. 5 –7 Our own work has established that the mortality associated with CHF has fallen significantly over the past 15 years, possibly due in part to the introduction of these therapies; 8 furthermore, we showed a reduction in the proportion of deaths due to cardiovascular causes. Such data highlight the evolving nature of CHF outcomes and emphasise the importance of clarifying whether the adverse prognostic impact of DM persists. We therefore aimed to define the current impact of DM on total and cardiovascular mortality and also to address the ongoing debate as to whether DM impacts differentially on outcomes in CHF of ischaemic or non-ischaemic aetiology.
Methods
We conducted a prospective cohort study of 1091 unselected patients with CHF with the specific aim of examining prognosis in patients treated with ‘state-of-the-art’ therapies carried out in cardiology outpatient clinics of four UK hospitals between June 2006 and December 2011. The Leeds West Research Ethics Committee provided ethical approval, and all patients provided written informed consent to participate. Patients were eligible for inclusion if they were ambulant outpatients with stable clinical signs and symptoms of CHF for 3 months, with a LV ejection fraction of ≤45% on transthoracic echocardiography – that is, CHF with reduced ejection fraction (CHF-REF). All patients also underwent resting 12-lead electrocardiographs, chest radiographs and blood testing for measurement of full blood count, urea and electrolytes, plasma creatinine and random glucose. Estimated glomerular filtration rate (eGFR) was calculated using the Modification of Diet in Renal Disease method. 9 Functional status was assessed using the New York Heart Association (NYHA) classification. 10 Two-dimensional transthoracic echocardiography was performed in all patients and reported by local cardiac sonographers, blinded to patient characteristics, according to British Society of Echocardiography recommendations; 11 LV ejection fraction was calculated according to the Simpson’s biplane method. 12 Diabetes was defined at study recruitment on the basis of previous diagnosis and/or treatment with hypoglycaemic agents. Ischaemic aetiology was determined by the recruiting physician on the basis of detailed history (of risk factors for atherosclerosis, anginal symptoms or prior myocardial infarction), electrocardiograph (presence of Q waves), echocardiogram (LV regional wall motional abnormalities conforming to a typical coronary distribution) and non-invasive assessment of functionally significant coronary disease or invasive coronary angiography, as clinically indicated. Specific data on the use of invasive coronary angiography to aid the clinical definition of CHF aetiology in patients with and without DM are presented within the results; this was generally performed when the history or non-invasive investigations suggested a high risk of underlying flow-limiting coronary artery disease.
Classification of mortality
All patients were registered with the UK Office of Population, Censuses and Surveys, which provided details of death, including location and date. All deaths were evaluated by at least two senior physicians, who reviewed death certificates, autopsy findings and hospital and general practitioners’ records; where these physicians felt insufficient information was available, the mode of death was deemed unclassifiable. Mode of death was classified as cardiovascular or non-cardiovascular, and if cardiovascular, this was further sub-classified as progressive heart failure (HF), sudden death or other cardiovascular death (e.g. cerebrovascular accident, acute myocardial infarction), according to our previously published definitions. 8 Briefly, mode of cardiovascular death was defined as (1) sudden cardiac, if it occurred within 1 h of a change in symptoms or during sleep or while the patient was unobserved; (2) progressive HF, if death occurred after a documented period of symptomatic or haemodynamic deterioration; and (3) other cardiovascular death, if not occurring suddenly or in association with progression of HF. Mortality follow-up data are presented as a mean of 960 days (standard error of mean = 17.5 days) after recruitment.
Pharmacotherapy and device implantation data
Doses of diuretic therapy, angiotensin converting enzyme inhibitors (ACEi), angiotensin receptor blockers (ARB) and beta-blockers were collected at recruitment. For patients receiving bumetanide loop diuretic therapy, an equivalent dose of furosemide was recorded, on the assumption that 1 mg of bumetanide is equivalent to 40 mg of furosemide. Regarding the use of ACEi and ARB, an equivalent dose of ramipril was calculated for each patient based upon the maximum percentage of the individual agent, compared with the maximum licensed dose for the United Kingdom. 8 Similar calculations were performed using beta-blocker data to produce an equivalent dose of bisoprolol for each patient. Receipt of cardiac resynchronisation therapy (CRT) or implantable cardioverter–defibrillator (ICD) was assessed during the 6-month period after recruitment.
Statistical analysis
Continuous group data are given as mean (and standard error of the mean), and categorical data are shown as number (%). All statistical analyses were performed using PASW statistics version 18 (SPSS Inc., Chicago, IL, USA). Groups were compared using two-sided Student’s t-tests for continuous data and two-sided Pearson’s χ 2 tests for categorical data. Survival of groups was compared with log-rank tests, and adjusted mortality (including interaction analysis) was calculated using Cox regression analysis. Adjusted mortality analyses included combinations of age, sex, clinical variables (ischaemic aetiology, NYHA class, LV ejection fraction, haemoglobin, sodium, eGFR) and therapeutic variables (bisoprolol dose, ramipril dose, furosemide dose, CRT and ICD therapy) as indicated in the relevant sections. Statistical significance was defined as p < 0.05.
Results
Demographic, clinical and treatment factors
Diabetes was present in 25.7% of the cohort; these patients had mean glycosylated haemoglobin (HbA1c) of 7.8% (0.1%), with 20% achieving HbA1c ≤ 6.5%. Isolated dietary therapy was required by 26.8%, 33.6% used sulphonylureas, 37.1% used metformin and 24.6% required insulin therapy. Patients with DM exhibited a similar age–sex profile (Table 1) but were more limited by symptoms of HF and more frequently had CHF attributable to ischaemic heart disease (75.7% vs 58.7%; p < 0.001). Invasive coronary angiography was used with similar frequency in patients with and without DM (66.1% vs 62.5%; p = 0.34) in order to aid definition of CHF aetiology.
Cohort characteristics.
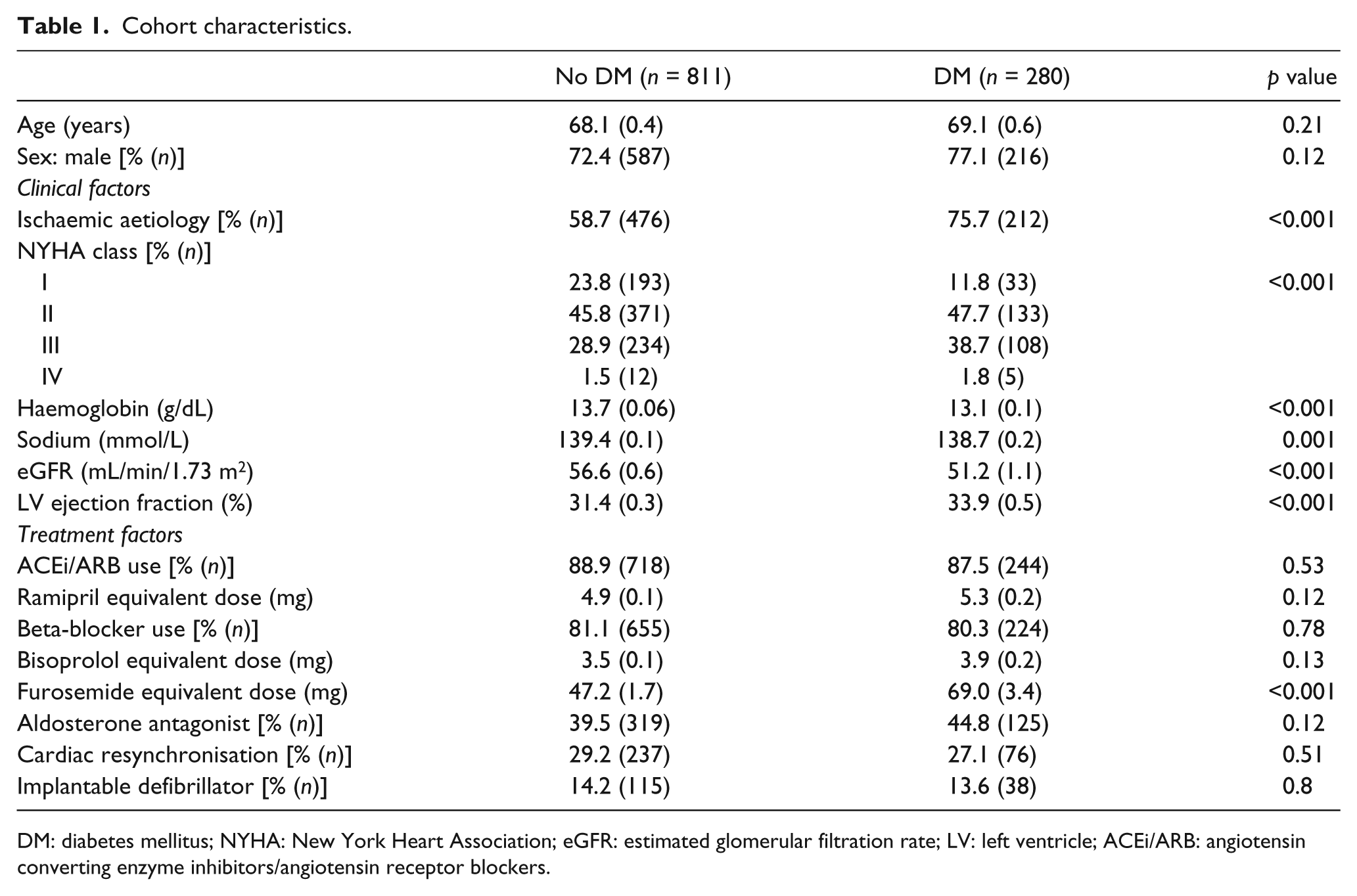
DM: diabetes mellitus; NYHA: New York Heart Association; eGFR: estimated glomerular filtration rate; LV: left ventricle; ACEi/ARB: angiotensin converting enzyme inhibitors/angiotensin receptor blockers.
An adverse biochemical profile was associated with DM, including lower haemoglobin, sodium and eGFR, although LV ejection fraction was greater in patients with diabetes. Medical- and device-based therapy was comparable in patients with and without DM (Table 1), other than for higher furosemide doses in patients with DM [69.0 mg (3.4 mg) vs 47.2 mg (1.7 mg); p < 0.001]. ACEi/ARB (87.5% vs 88.9%) and beta-blocker (80.3% vs 81.1%) administration was comparable in patients with and without DM.
Total, cardiovascular and mode-specific mortality
DM was associated with significantly greater crude hazard ratios (HRs) for all-cause [2.08 (95% confidence interval, CI: 1.61–2.69); p < 0.001] and cardiovascular mortality [2.34 (95% CI: 1.70–3.22); p < 0.001]; Kaplan–Meier curves are presented in Figure 1. These findings persisted after accounting for age, sex, ischaemic aetiology, clinical variables and treatment variables (listed in Table 1), with only marginal diminution of the magnitude of adverse risk associated with DM (Table 2). Diabetes was also associated with significantly increased HRs for progressive HF [2.02 (95% CI: 1.31–3.13); p = 0.002] and sudden cardiovascular mortality [3.32 (95% CI: 1.85–5.96); p < 0.001] (Figure 1). The proportion of progressive HF, sudden cardiovascular, other cardiovascular, non-cardiovascular and unclassifiable deaths did not differ between cohorts with and without DM (p = 0.43; Table 3).

All-cause, cardiovascular and mode-specific mortality. Kaplan–Meier curves demonstrating increased (a) all-cause (p < 0.001 by log-rank), (b) cardiovascular (p < 0.001), (c) progressive heart failure (p = 0.002) and (d) sudden cardiovascular (p < 0.001) mortality in groups with (red) versus without (blue) diabetes.
Multivariable mortality analyses.
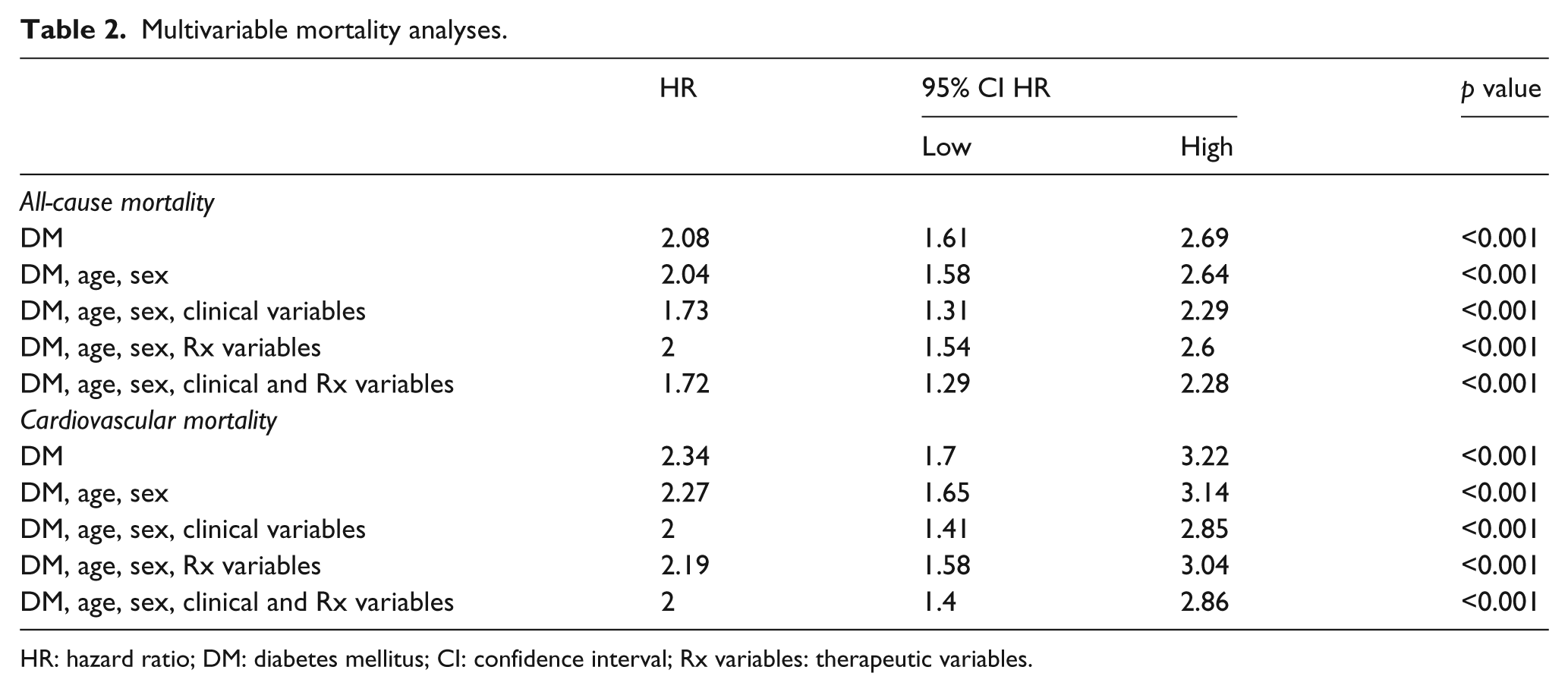
HR: hazard ratio; DM: diabetes mellitus; CI: confidence interval; Rx variables: therapeutic variables.
Mode-specific mortality analyses.
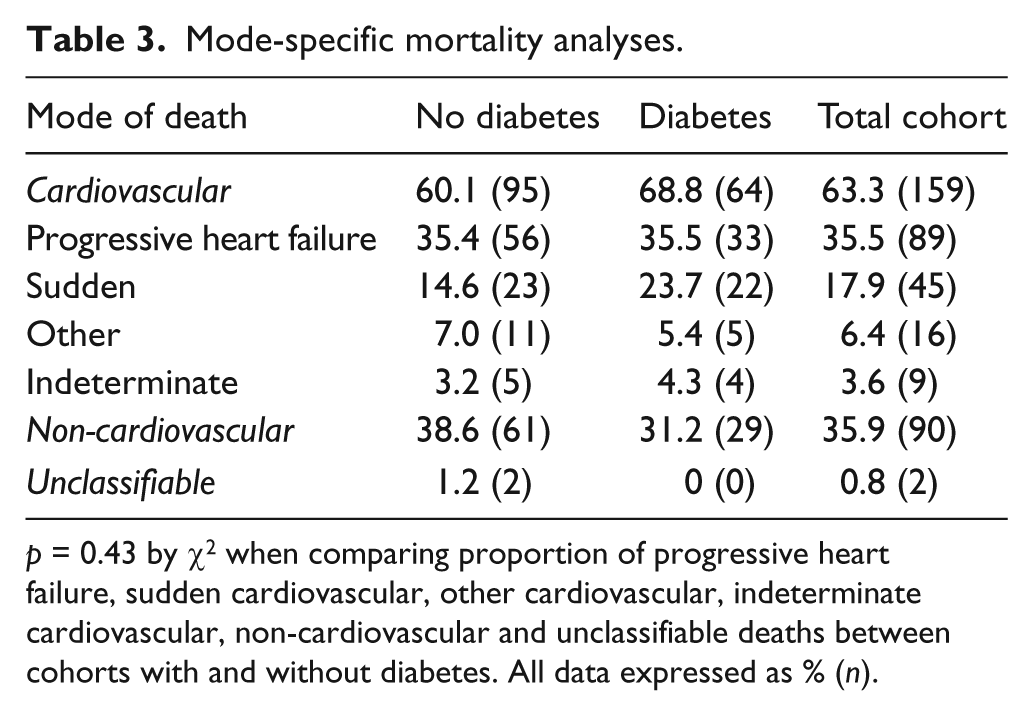
p = 0.43 by χ 2 when comparing proportion of progressive heart failure, sudden cardiovascular, other cardiovascular, indeterminate cardiovascular, non-cardiovascular and unclassifiable deaths between cohorts with and without diabetes. All data expressed as % (n).
Ischaemic and non-ischaemic cardiomyopathy
The proportion of patients with DM was significantly greater in the ischaemic than the non-ischaemic cohort (29.6% vs 15.2%; p < 0.001). Within the ischaemic and non-ischaemic cohorts, DM was associated with broadly similar differences in functional capacity, biochemical and echo parameters (Table 4). Again, DM was not associated with significant differences in demographic or treatment factors (other than diuretic dose) in ischaemic or non-ischaemic cardiomyopathies.
Group characteristics according to DM and aetiology.
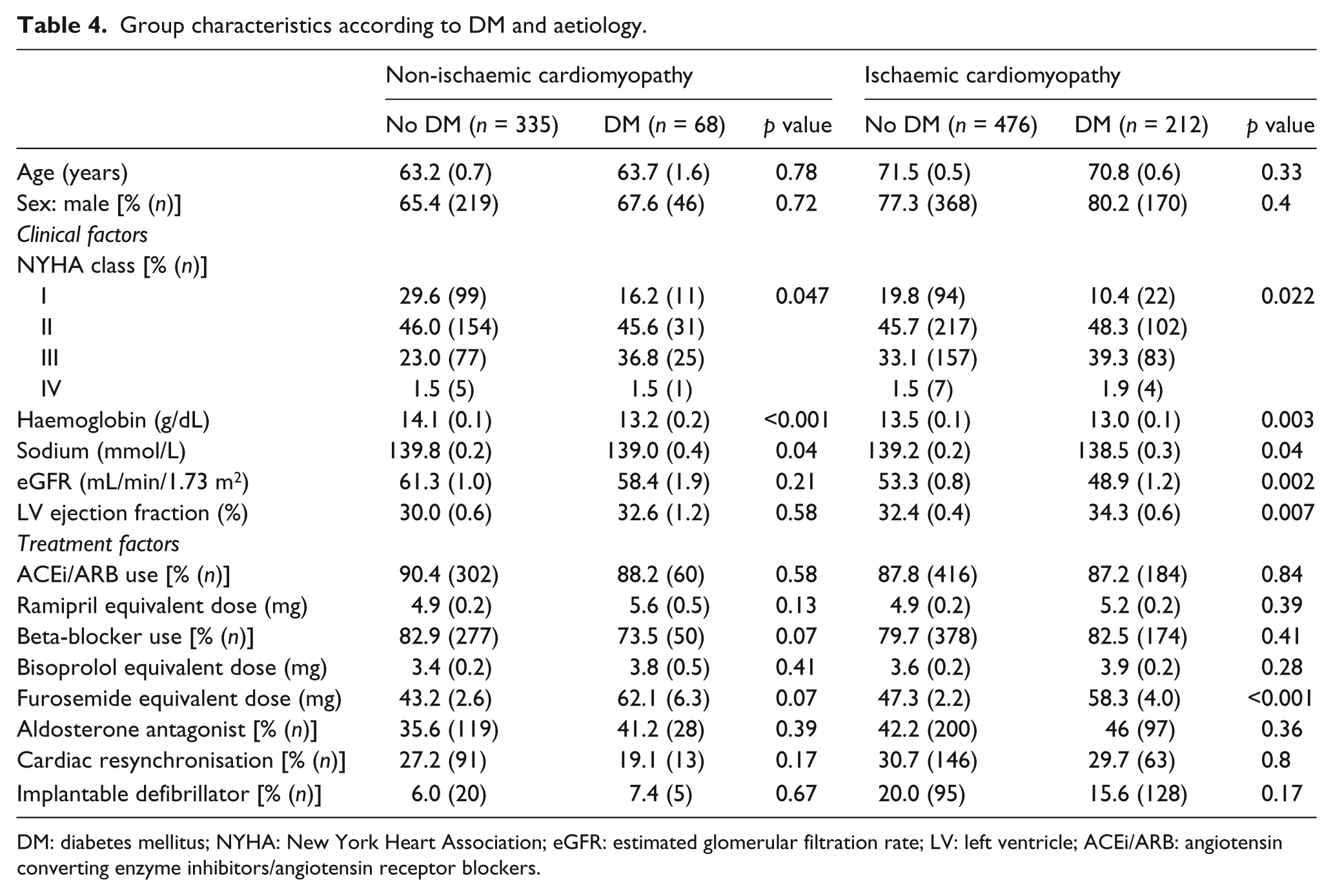
DM: diabetes mellitus; NYHA: New York Heart Association; eGFR: estimated glomerular filtration rate; LV: left ventricle; ACEi/ARB: angiotensin converting enzyme inhibitors/angiotensin receptor blockers.
Kaplan–Meier curves illustrating all-cause and cardiovascular mortality in the four groups are presented in Figure 2. Cox regression analysis revealed no interaction between DM and CHF aetiology (ischaemic versus non-ischaemic) in their association with total or cardiovascular mortality, indicating an equivalent impact of DM on outcomes, irrespective of CHF aetiology. The absence of interaction persisted in multivariable analyses (for total and cardiovascular mortality) accounting for age, sex and the clinical and treatment factors outlined in Table 4.
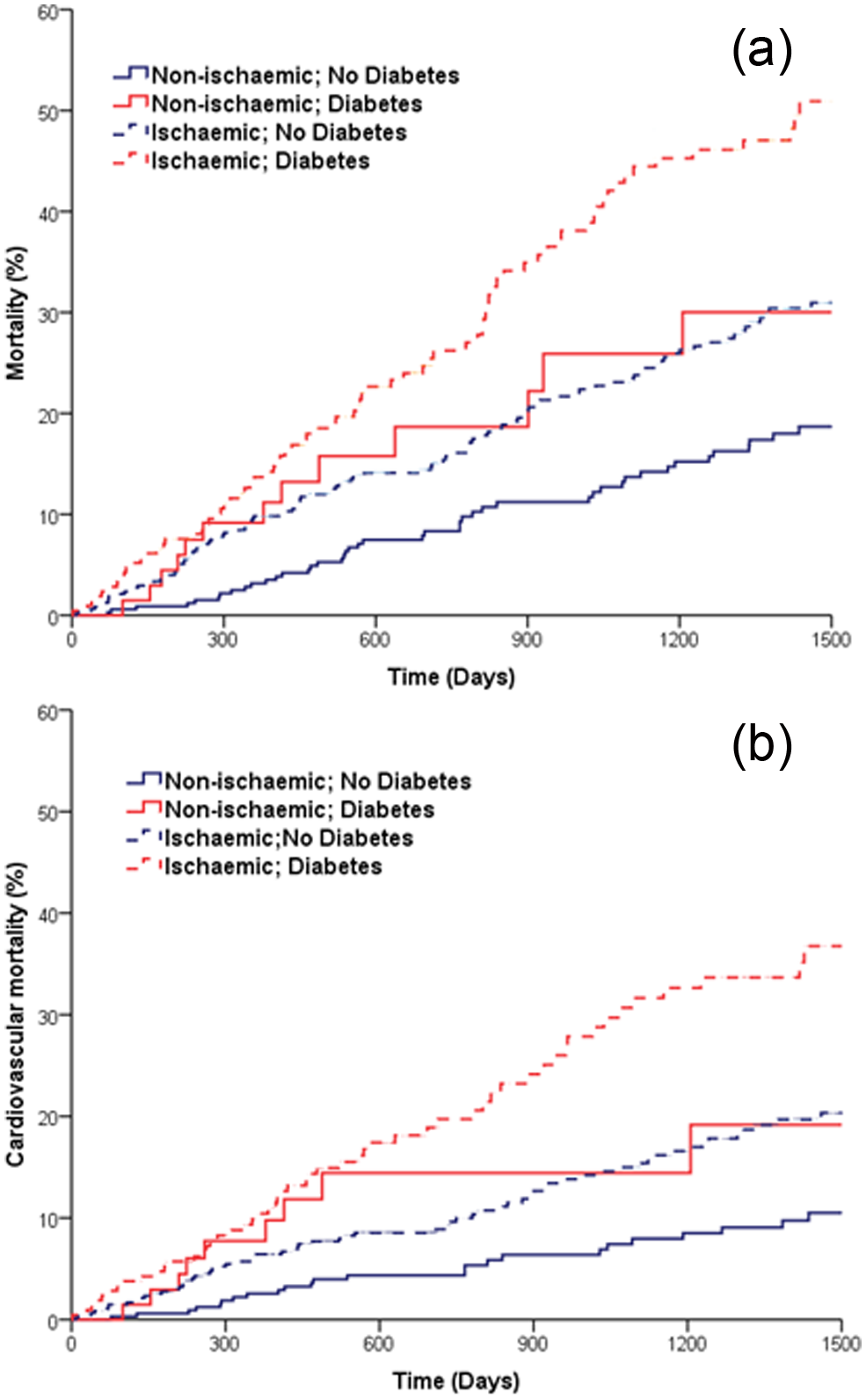
All-cause and cardiovascular mortality according to CHF aetiology. Kaplan–Meier curves demonstrating (a) all-cause and (b) cardiovascular mortality in groups with (red) and without (blue) diabetes, stratified by ischaemic (discontinuous) or non-ischaemic (continuous) aetiology. Diabetes is associated with increased mortality (p < 0.001) in ischaemic and non-ischaemic CHF in both panels.
Discussion
The principal findings of this study are as follows: (1) DM remains an adverse prognostic factor in patients with CHF receiving contemporary evidence-based therapy and (2) DM impacts similarly upon total and cardiovascular mortality in ischaemic and non-ischaemic cardiomyopathies. In broad terms, our analyses suggest that diabetes continues to double the risk of death in the setting of CHF, in spite of widespread administration of beta-blockers and guideline-based application of aldosterone receptor antagonists and device-based therapy. Given the rising prevalence of DM in CHF cohorts, 1 our data indicate that further improvements in overall CHF survival may be abrogated, or even reverse, if management strategies stagnate. Conversely, the approximate 4-year median survival of our cohort with diabetes and ischaemic CHF suggests that there is scope to dramatically improve overall CHF outcomes if effective diabetes-specific CHF management can be defined.
The question of whether or not DM comparably increases mortality in patients with ischaemic and non-ischaemic cardiomyopathies is important in offering insight into how DM mediates adverse prognosis. However, the published literature presents conflicting conclusions, with some groups suggesting DM increases mortality similarly in both groups, while others suggesting increased risk in only ischaemic or non-ischaemic CHF. For example, De Groote et al. 13 presented an observational study of 1246 patients with CHF-REF recruited in the 1990s; they noted DM to mediate adverse prognosis in ischaemic CHF but a favourable prognosis in non-ischaemic CHF. The use of different patterns of CHF care and their routine use of coronary angiography to define ischaemic aetiology may underlie our differing conclusions, although surprisingly, 54% of their cohort (as compared with 37% of our cohort) had non-ischaemic CHF. Conversely, From et al. 1 suggested that DM was only associated with poor prognosis in non-ischaemic CHF in their study of 665 patients with reduced or preserved LV ejection fraction, recruited between 1979 and 1999. Again, differences in inclusion criteria and clinical practice may offer some explanation for their divergent conclusions. Data from the 7599 patients with reduced and preserved LV ejection fraction recruited to Candesartan in Heart Failure (CHARM) programme support our conclusion that DM impacts equally upon prognosis in ischaemic and non-ischaemic CHF. 3 This is in spite of our differing study design [observational versus randomised controlled trial (RCT)], inclusion criteria and ‘therapeutic eras’.
On the basis of our findings, it is tempting to speculate that factors other than epicardial coronary artery disease mediate the adverse impact of diabetes. Our multivariate analyses offer further insight into the impact of DM upon CHF mortality, suggesting that alterations in recognised prognostic factors (e.g. renal function and age) do not significantly account for its detrimental effect. The greater diuretic requirements, poorer functional capacity and increased risk of cardiovascular death associated with DM in both ischaemic and non-ischaemic CHF suggest that a myocardial process may principally mediate its adverse effect. Other authors have suggested that a ‘diabetic cardiomyopathy’ may be contributory via cardiac glucotoxicity, metabolic perturbation, microvascular dysfunction and altered collagen deposition 14 although we have no data to address this hypothesis.
While our study offers important contemporary insights into how DM impacts upon CHF outcomes, a number of limitations must be acknowledged. First, DM was defined on the basis of previously formulated diagnosis, rather than routine screening of all individuals. This would serve to inadvertently include some patients with DM in the non-diabetic cohort, thereby potentially diminishing the true effect of DM in our analyses. However, the prevalence of DM in our cohort is within the broad range noted in contemporary CHF cohorts, and as such the impact of crossover is likely to be limited. Second, the definition of ischaemic cardiomyopathy is based upon investigations ordered at the recruiting physician’s discretion, rather than systematic use of invasive coronary angiography; however, such a strategy would not be ethically appropriate. Finally, the group of patients with DM and non-ischaemic CHF was relatively small, resulting in some large CIs (although similar effect sizes), but as this group represents only 6% of our total population, a very large study would be required to address this issue comprehensively.
In conclusion, DM remains associated with increased risk of death in patients with ischaemic and non-ischaemic CHF, in spite of recent pharmacological and device-based advances in HF management. If the pattern of improving overall CHF mortality is to persist, strategies specifically targeting the HF phenotype of patients with diabetes appear increasingly necessary.
Footnotes
Conflict of interest
None declared.
Funding
This study was funded by the British Heart Foundation (PG/08/020/24617).
