Abstract
Aims
A retrospective, case–control study to investigate the J wave, a J-point elevation on resting 12-lead electrocardiograms, as a risk factor for sudden cardiac arrest (SCA) in patients diagnosed with hypertrophic cardiomyopathy (HCM).
Methods
Patients with HCM and age- and sex-matched healthy control subjects were recruited, and 12-lead electrocardiograms were performed. The prevalence and related characteristics of J waves were assessed. Patients were followed-up for a mean of 47.9 months.
Results
Patients with HCM (
Conclusion
The J wave may be a risk factor for SCA in patients with HCM.
Keywords
Introduction
Hypertrophic cardiomyopathy (HCM) is a genetic cardiovascular disease with a prevalence of 0.2% in young adults. 1 The majority of patients with HCM live a normal life with mild symptoms, but sudden cardiac death (SCD) occurs in 1% of patients per year, 2 with a higher incidence in younger patients than older patients. 3 Risk stratifications of SCD have primarily focused on clinical factors 4 including: history of ventricular fibrillation (VF), sustained ventricular tachycardia (VT) and/or SCD events 1 ; family history of SCD events 2 ; unexplained syncope 3 ; documented nonsustained VT (three or more beats at ≥120 beats per min [bpm] on ambulatory electrocardiogram [ECG]) 4 ; and maximal left ventricular (LV) wall thickness ≥30 mm. 5 These clinical factors do not predict all SCD events in patients with HCM, however. 4 In addition, ECG abnormalities are present before clinical manifestation of HCM in ∼75–95% of patients, 5 suggesting the need for new ECG predictors of SCD. The J-point elevation (J wave) is an ECG variant that was considered to be a benign phenomenon, 6 but been associated more recently with cardiovascular death in the general population 7 and sudden cardiac arrest (SCA), in patients with VF or SCD events.8,9 The relationship between the J wave and SCA in patients with HCM is currently unknown, however. The aim of the current retrospective case–control study was to investigate the prevalence and characteristics of J waves, and their relationship with SCA in patients with HCM.
Patients and methods
Study population
This retrospective case–control study included patients diagnosed with HCM at seven tertiary teaching hospitals in Beijing (including two of the largest heart centres in China) between January 2001 and December 2010. Inclusion criteria were: (i) aged ≤ 65 years; (ii) confirmed diagnosis of HCM including nonsymmetry, symmetry and apical hypertrophic cardiomyopathy; (iii) diagnosis confirmed by ultrasound cardiogram or magnetic resonance imaging; (iv) ECG with sinus rhythm and QRS duration ≤ 120 ms; (v) clinical and follow-up data available. Exclusion criteria were: (i) heart rate corrected QT interval (QTc) ≤ 340 ms or >440 ms on ECG; (ii) symmetrical LV hypertrophy and other comorbidities including hypertension and aortic stenosis that may contribute to myocardial hypertrophy; (iii) myocardial infarction; (iv) valvular disease, congenital heart disease, pre-excitation syndrome, third degree atrioventricular block or complete/incomplete bundle branch block; (v) permanent pacemaker implantation; (vi) previous interval ablation/surgical excision; (vii) end-stage disease, such as multiple organ failure, respiratory failure or uraemia.
The control group comprised healthy subjects attending Beijing Anzhen Hospital for routine medical checks between March 2011 and September 2011, who were free of any study-related symptoms and structural heart diseases. For each patient, two age- and sex-matched control subjects were recruited.
The study was approved by the Local Human Research Ethics Board, Beijing, China, and all participants provided written informed consent prior to enrolment.
ECG assessment of J waves
Baseline, standard, resting and supine 12-lead ECGs (paper speed 25 mm/s, voltage 10 mm/mV) were performed and all ECG tracings were rendered anonymous. J wave was defined as a J-point (intersection of QRS complex with ST segment) elevation of ≥0.1 mV from baseline in a minimum of two consecutive inferior wall leads (II, III, aVF), lateral wall leads (V4–V6) or high lateral leads (I, aVL).7,9 J wave morphology was categorized as: (i) notched (positive re-entrant waves at the end of QRS complexes); or (ii) slurred (no clear turning points at the QRS complex/ST segment junction, but slow continuous declination from the end of QRS complexes to ST segments) (Figure 1).
10
J wave distribution was: (i) monoregional (occurring at one region of the inferior, lateral or high lateral walls); (ii) multiregional (occurring in any two regions of inferior, lateral or high lateral walls); or (iii) global (occurring in all three regions). J wave amplitude was defined as the maximal distances between each regional lead and the highest points on the equipotential lines of the J waves in inferior, lateral or high lateral walls. Any ST segments after J-points were categorized as: (i) horizontal/descending segments with ST segment elevations ≤0.1 mV within 100 ms after J-points; (ii) concave/rapidly ascending segments with ST segment elevations >0.1 mV within 100 ms after J-points; or (iii) sustained elevations >0.1 mV after J-points.
10
The area between the T and P waves was defined as the equipotential lines, and QTc was calculated with Bazett’s formula: QT/(RR)1/2. All ECG parameters were measured digitally (DataInf® Measure, DataInf GmbH, Tübingen, Germany) and analysed by two experienced cardiologists (JM and QY) who were blinded to the study group. In the case of disagreement, a consensus was reached by discussion between the two cardiologists and an additional senior cardiologist (XL).
Sample electrocardiograms indicating J wave morphology (arrows). (A) Slurred and (B) notched J waves in inferior wall leads (II, III, aVF).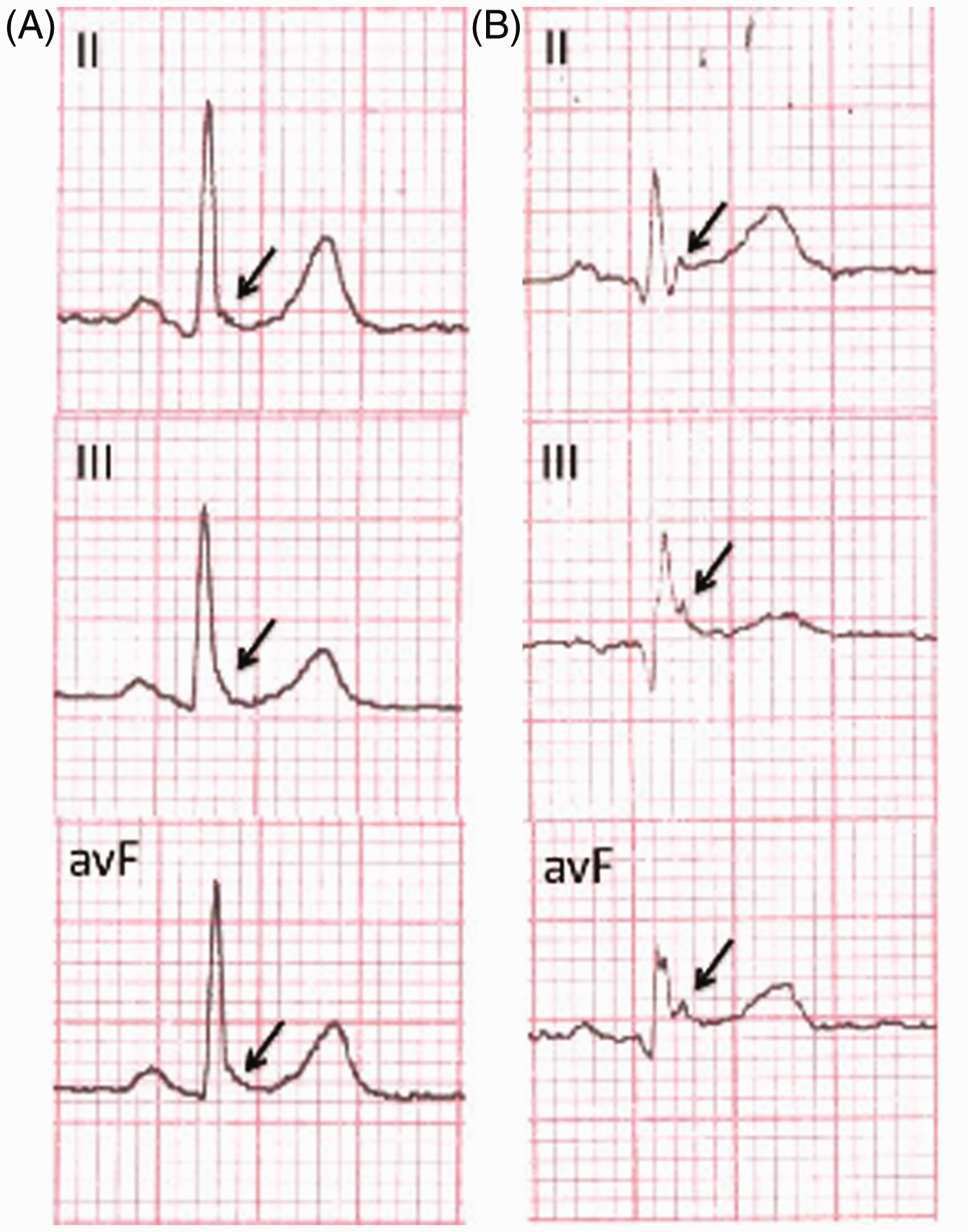
Follow-up and endpoints
Follow-up was carried out via telephone or clinical visit between March 2011 and May 2011. The study endpoint was SCA onset, including SCD (unexpected death due to cardiac cause occurring within 1 h of symptom onset, or witnessed unexpected death), VF, or appropriate discharge of implantable cardioverter-defibrillator (ICD) due to VF or fast VT (≥200 bpm).
Statistical analyses
Continuous data were presented as mean ± SD and categorical variables as
Results
The study identified 1143 patients with HCM, of whom 345 fulfilled the inclusion criteria (270 male/75 female; mean age 49.9 ± 10.7 years; age range 15–73 years) (Figure 2). The control group comprised 690 age- and sex-matched healthy control subjects (540 male/150 female; mean age 49.9 ± 10.7 years; age range 15–73 years).
Flow chart indicating patient recruitment, eligibility and outcomes in a study investigating the J wave as a potential risk factor for sudden cardiac arrest (SCA) in patients with hypertrophic cardiomyopathy. MI, myocardial infarction; WPW, Wolff–Parkinson–White syndrome; AVB, atrioventricular block; ECG, electrocardiogram; SCD, sudden cardiac death; VF, ventricular fibrillation; ICD, implantable cardioverter–defibrillator.
Baseline demographic and clinical characteristics of patients with hypertrophic cardiomyopathy (HCM) included in a study to investigate the association between J wave and sudden cardiac arrest (SCA), stratified according to the occurrence of SCA.

Data presented as mean ± SD or
Student’s
AF, atrial fibrillation; VT, ventricular tachycardia; SCD, sudden cardiac death; CCB, calcium channel blocker; AAD, antiarrhythmic drug; SH, symmetrical septal hypertrophy; ASH, asymmetrical septal hypertrophy; APH, apical hypertrophic cardiomyopathy; Max, maximal; LV, left ventricle; IVS/PW, interventricular septum/posterior wall; LVEDD, left ventricular end-diastolic dimension; LVEF, left ventricular ejection fraction.
Electrocardiogram data for patients with hypertrophic cardiomyopathy (HCM) stratified according to the occurrence of sudden cardiac arrest (SCA), and healthy control subjects.

Data presented as mean ± SD or
bpm, beats per min; LL, limb leads; CHL, precardial lead.
Logistic regression analysis of J wave regional distributions in patients with hypertrophic cardiomyopathy, stratified according to the occurrence of sudden cardiac arrest (SCA).

Data presented as
NS, not statistically significant (
Kaplan–Meier survival curves are shown in Figure 3. The 1-year SCA-free survival rate was 95.0% (38/40) in patients with J waves, and 99.0% (302/305) in those without. After 3 years, SCA-free survival rates were 90.0% (36/40) and 99.0% (302/305), respectively ( (A) Sudden cardiac arrest (SCA)-free survival in patients with hypertrophic cardiomyopathy (HCM), with and without J waves. (B) All cause-free survival in patients with HCM, with and without J waves.
Discussion
The annual prevalence of SCD is estimated to be 1% in patients with HCM, 1 but this may rise to 10% in patients identified as high risk. 12 To the best of our knowledge, the current study is the first to investigate the incidence of J waves and their relationship with SCA, in patients with HCM. Patients with HCM had a significantly higher prevalence of J waves than healthy controls in the present study. In addition, J waves were significantly more common in patients who experienced SCA than in the non-SCA group. The risk of death due to SCA (but not all-cause death) was significantly higher in patients who exhibited J waves than in those who did not. Taken together, the findings of the present study suggest that the J wave may be a risk factor for SCA in patients with HCM.
The prevalence of J waves in the normal population has been shown to be between 1% and 13%,6,9,13,14 with a higher incidence in young men.6,15,16 These data are consistent with the findings of the present study, where the prevalence of J waves in the control group was 7.1%. Patients with HCM showed a higher prevalence of J waves, however (11.6%). Transmural differences in early phases of the action potential have been recognized as the basis for inscription of the electrocardiographic J wave. 17 Patients with HCM have thicker ventricular walls than the general population, which may result in extended myocardial depolarization and repolarization. In addition, it is possible that increased heterozygous myocardial cell repolarization may increase J wave prevalence. Further investigation is required to elucidate the underlying mechanism fully, however.
The J wave was significantly more common in patients who experienced SCA than in those who did not, and patients with J waves had a lower SCA-free survival rate in the present study. The presence of J waves was identified as a risk factor for SCD in patients with idiopathic VF, 9 Brugada syndrome, 18 acute coronary syndrome, 19 and in the general middle-aged population. 20 The current findings suggest that the J wave may be correlated with SCA rather than all-cause mortality, in patients with HCM.
As shown in this study, only a minority of HCM patients with J waves were at risk of SCA. It is therefore important to differentiate between malignant and benign J waves. The risk stratifications of J waves were investigated in several studies, and the reported characteristics of potentially malignant J waves included global distribution, 21 J-point elevation > 0.2 mV 22 and notched morphology. 23 The present study found no significant differences in J wave distribution between patients with and without SCA. In addition, there were no significant differences in J wave amplitude between patients and controls, or between patients with and without SCA.
As well as the regional distribution of J waves, their position may play a role in risk stratifications. J waves in the inferior or inferolateral leads (categorized as type 2 21 ) may be associated with a higher risk for the development of malignant arrhythmias than those in the lateral precordial leads (categorized as type 1). Inconsistent with these findings, the present study found that the prevalence of J waves in the inferior leads was significantly lower in the SCA subgroup than in the non-SCA subgroup, but these findings are limited by the small number of patients with SCA included in the study.
Notched morphology of J waves has been associated with arrhythmic death 10 and idiopathic VT. 23 In addition, J waves combined with horizontal/descending morphology of ST segments after J-points are considered to be associated with idiopathic VF 24 and arrhythmic death. 10 The present study failed to find any morphological differences in J waves between the SCA group and the non-SCA group.
This study has several limitations, the main one being the small sample size of the SCA subgroup. Although 1143 patients were enrolled, only 14 experienced SCA during the follow-up period. This may be due to the low incidence of SCA in the selected patient group and the relatively short follow-up period (47.9 ± 46.6 months). Patients with HCM who underwent myocardial resection or alcohol ablation, or had severe heart failure or bundle branch block, were excluded from the study, greatly reducing the incidence of SCA. It is interesting to note, however, that even though the sample size of the SCA subgroup was small, the J wave prevalence remained significantly higher than in the non-SCA subgroup. Another limitation is related to the dynamic nature and intermittent display of J waves, 9 which may lead to underestimation of their incidence. The results of the present study can only indicate a potential association rather than causality between J waves and SCA in patients with HCM, and the possibility of coincidence cannot be excluded. This issue is encountered by all similar studies because the mechanistic link between J waves and malignant ventricular arrhythmias remains unclear.
In conclusion, this retrospective case–control study reveals a higher prevalence of J waves in patients with HCM compared with control subjects. In addition, J waves are significantly more common in patients with HCM who experienced SCA compared with those who did not. J waves may be a risk factor for SCA in patients with HCM.
Footnotes
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
