Abstract
Background:
Diabetes mellitus is associated with an increased risk of death and hospitalisation in patients with chronic heart failure. Better understanding of potential underlying mechanisms may aid the development of diabetes mellitus–specific chronic heart failure therapeutic strategies.
Methods:
Prospective observational cohort study of 628 patients with chronic heart failure associated with left ventricular systolic dysfunction receiving contemporary evidence-based therapy. Indices of cardiac structure and function, along with symptoms and biochemical parameters, were compared in patients with and without diabetes mellitus at study recruitment and 1 year later.
Results:
Patients with diabetes mellitus (24.2%) experienced higher rates of all-cause [hazard ratio, 2.3 (95% confidence interval, 1.8–3.0)] and chronic heart failure–specific mortality and hospitalisation despite comparable pharmacological and device-based therapies. At study recruitment, patients with diabetes mellitus were more symptomatic, required greater diuretic doses and more frequently had radiologic evidence of pulmonary oedema, despite higher left ventricular ejection fraction. They also exhibited echocardiographic evidence of increased left ventricular wall thickness and pulmonary arterial pressure. Diabetes mellitus was associated with reduced indices of heart rate variability and increased heart rate turbulence. During follow-up, patients with diabetes mellitus experienced less beneficial left ventricular remodelling and greater deterioration in renal function.
Conclusion:
Diabetes mellitus is associated with features of adverse structural and functional cardiac remodelling in patients with chronic heart failure.
Introduction
The prevalence of diabetes mellitus (DM) continues to climb at an exponential rate, with current forecasts projecting more than 500 million sufferers by 2035. 1 Given the substantial cardiovascular risk associated with DM, it is likely that comorbid diabetes will become an increasingly common challenge for all physicians managing cardiovascular disease and vice versa. Our work, 2 and that of others,3,4 has highlighted the significantly increased risk of death and hospitalisation in patients with chronic heart failure (CHF) and DM. Despite this knowledge, our understanding of what mediates the association between DM and adverse CHF outcomes remains somewhat limited. Previous studies have implicated a diverse array of pathophysiological factors, including unfavourable left ventricular (LV) remodeling, 5 cardiac autonomic dysfunction 6 and DM-related comorbidity. 7 However, many studies have addressed these issues in isolation, and so we set out to provide a detailed assessment of these factors, along with their change over time, in patients receiving contemporary CHF therapy.
Methods
We conducted a prospective cohort study of 628 unselected patients with CHF associated with LV systolic dysfunction, with the specific aim of examining prognostic factors in patients receiving contemporary therapy. Recruitment was carried out in the cardiology outpatient clinics of four United Kingdom hospitals between June 2006 and January 2009 with the ethical approval of Leeds West Research Ethics Committee; all patients provided written informed consent. As previously described, 8 patients were eligible for inclusion if they were ambulant outpatients with stable clinical signs and symptoms of CHF for 3 months, with a left ventricular ejection fraction (LVEF) of ⩽45% on transthoracic echocardiography (ECHO).
Baseline and follow-up assessment
During the study recruitment clinic, details of past medical history were recorded and physical examination was conducted. Functional status was assessed using the New York Heart Association (NYHA) classification. Diabetes was defined at study recruitment on the basis of previous diagnosis and/or treatment with hypoglycaemic agents. Ischaemic aetiology of CHF was determined by the recruiting physician on the basis of detailed history (of risk factors for atherosclerosis, anginal symptoms or prior myocardial infarction), electrocardiograph (presence of Q waves), echocardiogram (LV regional wall motional abnormalities conforming to a typical coronary distribution) and non-invasive assessment of functionally significant coronary disease or invasive coronary angiography, as clinically indicated. Doses of diuretic therapy, angiotensin-converting enzyme inhibitors (ACEi), angiotensin receptor blockers (ARB) and beta-blockers were collected at recruitment. As previously reported, 2 doses of loop diuretic, ACEi (or ARB) and beta-adrenoreceptor antagonist were expressed as the equivalent dose of furosemide, ramipril and bisoprolol, respectively. Receipt of cardiac resynchronisation therapy (CRT) or implantable cardioverter-defibrillator (ICD) was assessed during the 6-month period after recruitment.
All patients underwent resting 12-lead electrocardiographs (ECGs), ambulatory 24-h ECGs, ECHO, chest radiographs and blood testing for measurement of full blood count, urea and electrolytes, plasma creatinine and random glucose. All biochemical analyses were conducted in the National Health Service chemical pathology laboratories of the relevant recruiting centres. Estimated glomerular filtration rate (eGFR) was calculated using the Modification of Diet in Renal Disease method. Glycosylated haemoglobin (HbA1c) was measured only in people with known diabetes.
All patients were invited to attend a follow-up clinic approximately 12 months later, where a repeat clinical assessment was made, prior to conducting blood testing, resting and ambulatory electrocardiography and ECHO, as outlined above.
ECHO
Two-dimensional transthoracic ECHO was performed in all patients and reported by local cardiac sonographers, blinded to patient characteristics, according to British Society of Echocardiography recommendations; LVEF was calculated according to Simpson’s biplane method.
Ambulatory electrocardiography
Twenty-four-hour ambulatory ECGs (Lifecard CF, Spacelabs Healthcare Ltd, Hertford, UK) were obtained during normal, unrestricted, out of hospital activity as previously reported. 9 Recordings were analysed with a Spacelabs Sentinel system by independent technical staff blinded to patient characteristics. Non-sustained ventricular tachycardia was defined as the presence of three or more consecutive ventricular complexes at a rate of ⩾120 beats/min, with no evident atrioventricular (AV) concordance. Atrial arrhythmias (flutter or fibrillation) were defined as present if sustained for >30 s.
Each 24-h ECG recording was manually edited to exclude any incorrectly identified R waves and include any unidentified R waves as determined at the automatic processing stage. Ambulatory heart rate range (AHRR) was defined as maximum minus minimum heart rate during the 24-h period of analysis, as previously described. 9 For standard deviation of normal to normal (SDNN) analyses, the standard Delmar Reynolds/Spacelabs RR interval exclusion criteria were then applied to the manually edited records. Specifically, RR intervals were excluded if RR >2.0 s, RR >3 standard deviations of the local 20-min interval, RR intervals <300 ms, RR >120% of previous RR and RR <80% of previous RR. SDNN was then determined using the proprietary Pathfinder software.
Heart rate turbulence (HRT) was assessed using a Quantec data analysis workstation. The turbulence analysis followed the method described by Grimm et al. 10 and Schmidt et al. 11 with the mean HRT slope and mean HRT onset calculated from all valid premature ventricular complexes (PVCs). The HRT onset was defined as the difference between the mean of the first 2 sinus RR intervals preceding the PVC and the mean of the subsequent two sinus RR intervals, expressed as a percentage. The HRT slope is defined as the maximum positive value of the slope of a regression line assessed over any sequence of five subsequent sinus-rhythm RR intervals within the first 20 sinus-rhythm intervals after PVC. If more than one positive slope occurred in this period, the first positive slope was accepted. The value of HRT slope is expressed in milliseconds per RR interval. PVCs were excluded if they failed to satisfy the following criteria: (1) the PVC should occur in isolation with normal sinus beat occurring for at least 12 beats before the PVC and for at least 20 beats after the PVC; (2) the PVC RR maximum and minimum RR intervals should be greater than 20% and less than 20%, respectively, of the mean of the five preceding normal sinus RR intervals; (3) all RR intervals for the 12 beats before the PVC and 20 beats after the PVC should be greater than 300 ms and less than 2000 ms in duration; (4) all normal sinus RR intervals 12 beats before the PVC and 20 beats after the PVC should be within 20% of the mean RR interval of all beats in the 24-h ECG recording; and (5) any change in adjacent normal sinus RR intervals, 12 beats before the PVC and 20 beats after the PVC, should not be greater than 200 ms.
Classification of mortality and hospitalisation
All patients were registered with the United Kingdom Office of Population Censuses and Surveys, which provided details of any participant death, including location and date; data were available for all participants. All deaths were evaluated by at least two senior physicians, who reviewed death certificates, autopsy findings and hospital and general practitioners’ records; where these physicians felt insufficient information was available, the mode of death was deemed unclassifiable. Mode of death was classified according to our previously published definitions. 12 Briefly, these are (1) sudden cardiac, if occurring within 1 h of a change in symptoms or during sleep, or while the patient was unobserved; (2) progressive heart failure (HF), if death occurred after a documented period of symptomatic or haemodynamic deterioration; (3) other cardiovascular death (e.g. stroke or pulmonary embolism), if not occurring suddenly or in association with progression of HF; and (4) non-cardiovascular, if none of the above.
Hospitalisation was assessed with the use of institutional clinical event databases detailing all admissions in recruiting centres. HF-related hospitalisation was defined as a new onset or worsening of signs and symptoms of HF, with evidence of fluid overload, requiring at least 24 h overnight hospitalisation and the use of intravenous diuretics.
Statistical analysis
Continuous data are presented as mean [and standard error of the mean (SEM)]; categorical data are shown as number (percentage). All statistical analyses were performed using IBM SPSS statistics version 21 (IBM Corporation, Armonk, NY, USA). Groups were compared using two-sided Student’s t-tests for continuous data and two-sided Pearson χ2 tests for categorical data. Survival of groups was compared with log-rank tests and 1-year hospitalisation compared using binary logistic regression. Statistical significance was defined as p < 0.05.
Results
Clinical characteristics
A total of 628 unselected patients with CHF associated with LV systolic dysfunction were recruited, of whom 152 (25%) had DM. Of these, 148 (97%) patients had type 2 diabetes, and diabetes treatment was as follows: diet only in 27.6% (n = 42), sulphonylureas in 32.2% (n = 49), metformin in 32.9% (n = 50), insulin in 28.3% (n = 43) and thiazolelidinediones in 3.3% (n = 5). The baseline characteristics of these individuals are shown in Table 1. While the age–sex distribution of patients with diabetes was similar to those without diabetes, it is clear that ischaemic heart disease (IHD), cerebrovascular disease and systemic hypertension were more commonly associated with DM. The systolic blood pressure (SBP) and body mass index of patients with DM were also significantly higher. Notably, renal function was worse in patients with DM, and there was a trend towards a greater prevalence of comorbid chronic obstructive pulmonary disease. An adverse haematological and biochemical profile was evident in those with DM, with an elevated white cell count (WCC), lower haemoglobin and serum sodium concentration compared to those without the condition. Importantly, those with diabetes had a greater symptom burden at baseline with significantly higher NYHA class than those without the condition. Patients with DM were also more likely to have radiographic evidence of pulmonary oedema.
Baseline characteristics.
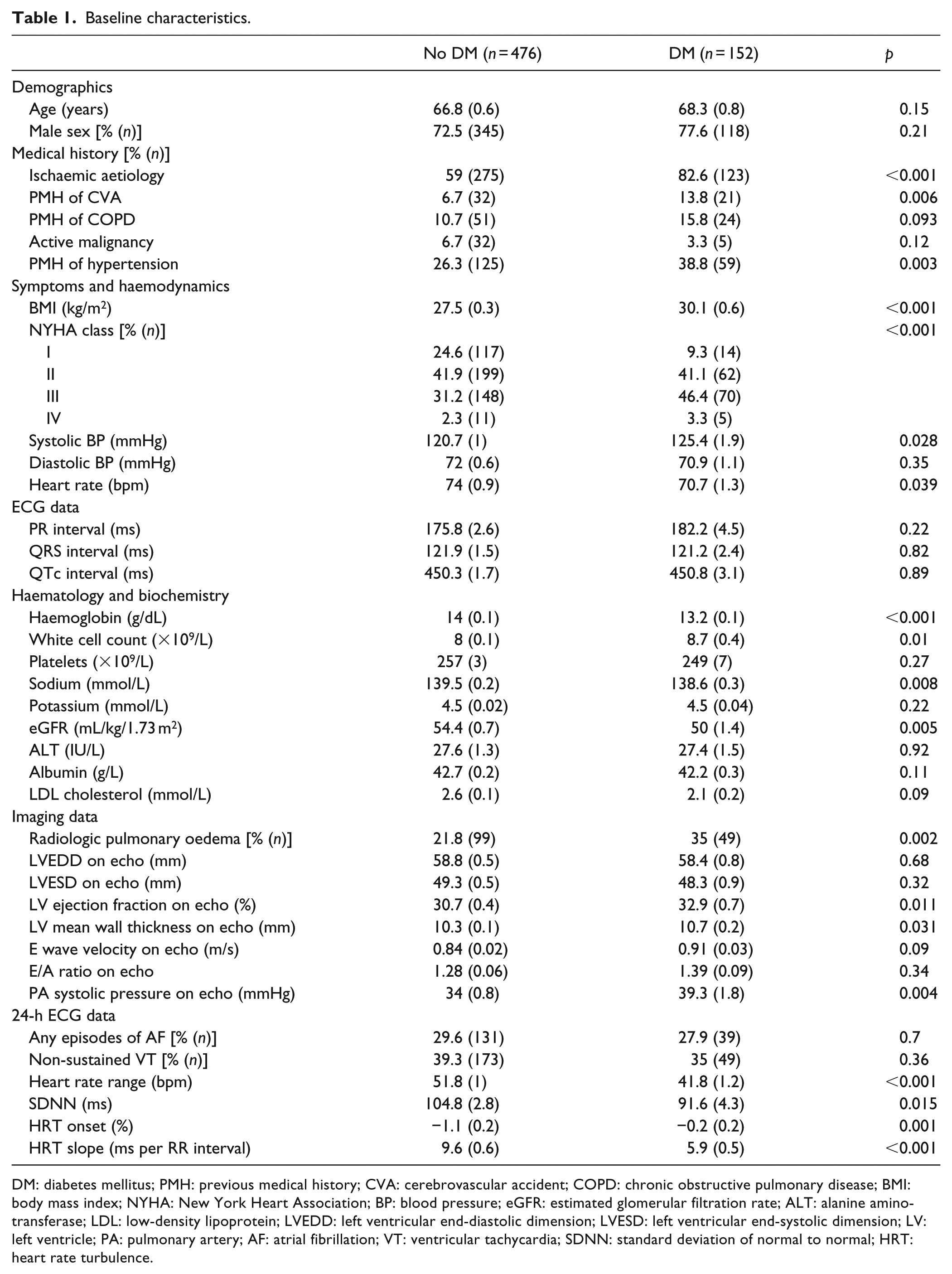
DM: diabetes mellitus; PMH: previous medical history; CVA: cerebrovascular accident; COPD: chronic obstructive pulmonary disease; BMI: body mass index; NYHA: New York Heart Association; BP: blood pressure; eGFR: estimated glomerular filtration rate; ALT: alanine aminotransferase; LDL: low-density lipoprotein; LVEDD: left ventricular end-diastolic dimension; LVESD: left ventricular end-systolic dimension; LV: left ventricle; PA: pulmonary artery; AF: atrial fibrillation; VT: ventricular tachycardia; SDNN: standard deviation of normal to normal; HRT: heart rate turbulence.
Baseline echocardiographic findings
Patients with diabetes had a significantly greater LVEF on transthoracic ECHO at baseline, despite their poorer NYHA status. Structurally, they had significantly increased LV mean wall thickness versus patients without DM, although the left ventricular end-diastolic dimension (LVEDD) and left ventricular end-systolic dimension (LVESD) were similar in both groups. No differences were noted in Doppler measures of LV inflow (E wave velocity and E/A ratio), but as noted in our discussion of limitations, these are suboptimal indices of LV diastolic function. A higher estimated pulmonary artery (PA) systolic pressure was also seen in those with DM. There was a significant, but weak, correlation between baseline LV wall thickness and SBP in patients without DM, although there was no such correlation in those with DM (see Figure 1). Furthermore, HbA1c did not correlate with LV mean wall thickness.
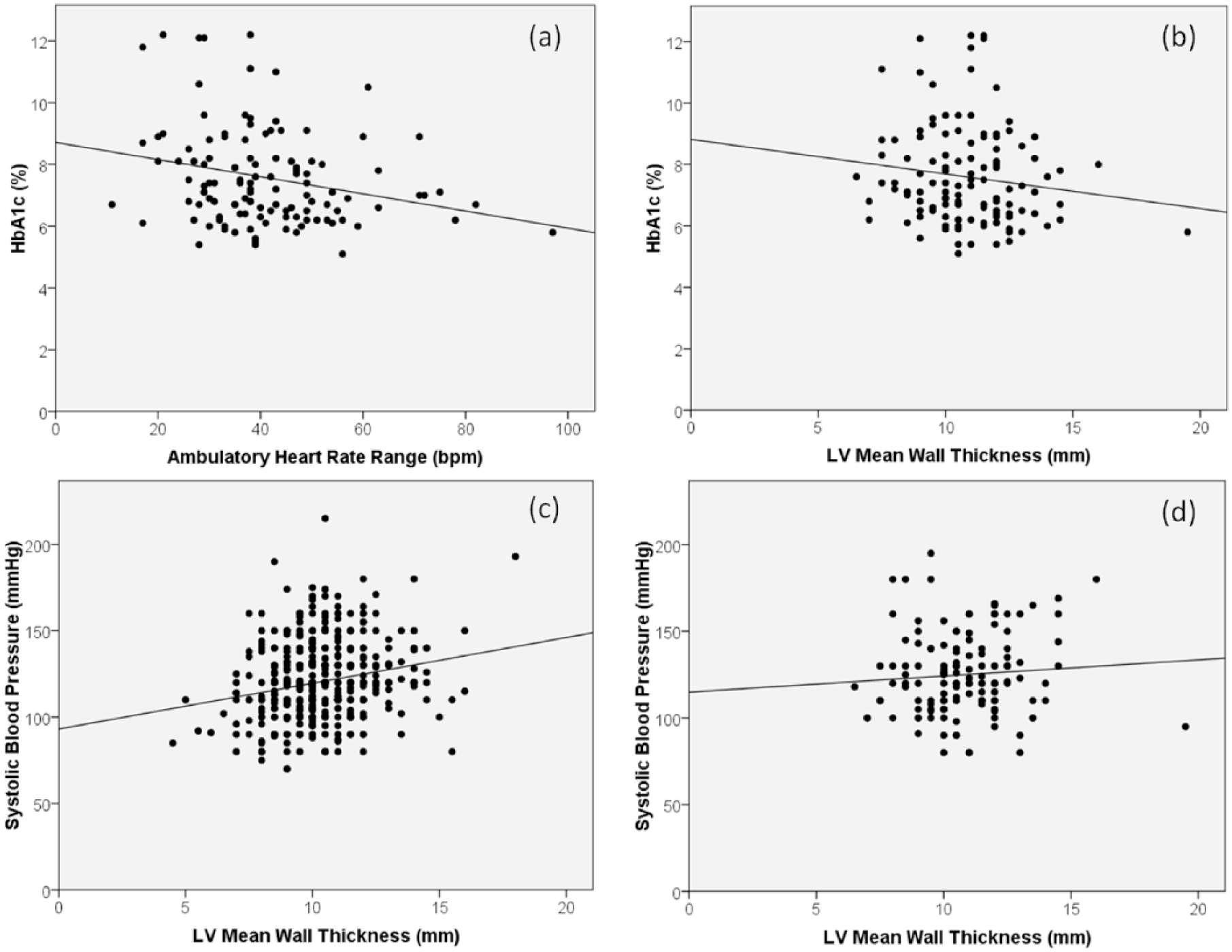
Correlations between (a) ambulatory heart rate range and glycosylated haemoglobin (HbA1c) (R2 = 0.06, p = 0.007), (b) left ventricular (LV) mean wall thickness and HbA1c (R2 = 0.019, p = 0.134), (c) LV mean wall thickness and systolic blood pressure in patients without diabetes (R2 = 0.045, p < 0.0001) and (d) LV mean wall thickness and systolic blood pressure in patients with diabetes (R2 = 0.006, p = 0.379).
Baseline electrocardiographic assessment
Patients with diabetes had a lower resting heart rate at baseline, although other 12-lead ECG parameters did not differ. On 24-h ECG recording, the prevalence of atrial and ventricular arrhythmias was the same in both groups. In contrast, AHRR and SDNN, along with HRT onset and slope, were lower in patients with diabetes (Table 1). Basal AHRR was calculated from all 574 24-h ECGs (137 with DM, 437 without DM) and negatively correlated with HbA1c (see Figure 1). As outlined in the ‘Methods’ section, it is not possible to derive SDNN and HRT from ECG recordings with prolonged periods of non-sinus rhythm. SDNN was derived from 202/574 patients 24-h ECGs in total (52 with DM, 150 without DM) and 221/574 24-h ECGs were analysable for HRT (55 with DM, 166 without DM).
Pharmacological and device therapy
As shown in Table 2, patients with DM were equally likely to receive evidence-based secondary prevention pharmacotherapy for CHF, and the prescribed doses were also similar. Moreover, DM was not associated with altered use of CRT or ICDs. However, patients with DM required significantly larger doses of loop diuretic therapy and were more likely to receive combined loop and thiazide diuretic therapy.
Baseline treatment.

ACEi: angiotensin-converting enzyme; ARB: angiotensin II receptor blocker; MRA: mineralocorticoid receptor antagonist; CRT: cardiac resynchronisation therapy; ICD: implantable cardioverter-defibrillator.
Up to 6 months after recruitment.
One-year follow-up clinical data and hospitalisation
A total of 408 of the original 628 patients (65%) were reviewed after 1 year [mean (SEM) 354 (7.1) days]; the remainder either died in the interim or failed to attend their appointment. At 1 year following enrolment, patients with diabetes remained more symptomatic, although there was no widening of the gap in NYHA class. The reduction in heart rate and blood pressure was comparable in both groups, and haematological and biochemical parameters were also equivalent (Table 3). This is in keeping with the data on follow-up pharmacological treatment for CHF (see Table 4) which remained comparable between the two groups, although loop diuretic doses were still higher in those with diabetes (mean 73 mg furosemide equivalent dose vs 48 mg for those without DM p < 0.0001). On ECHO, there was significantly less improvement in LVEF in those with diabetes, although the change in other variables was similar.
Change in clinical variables at 1 year.
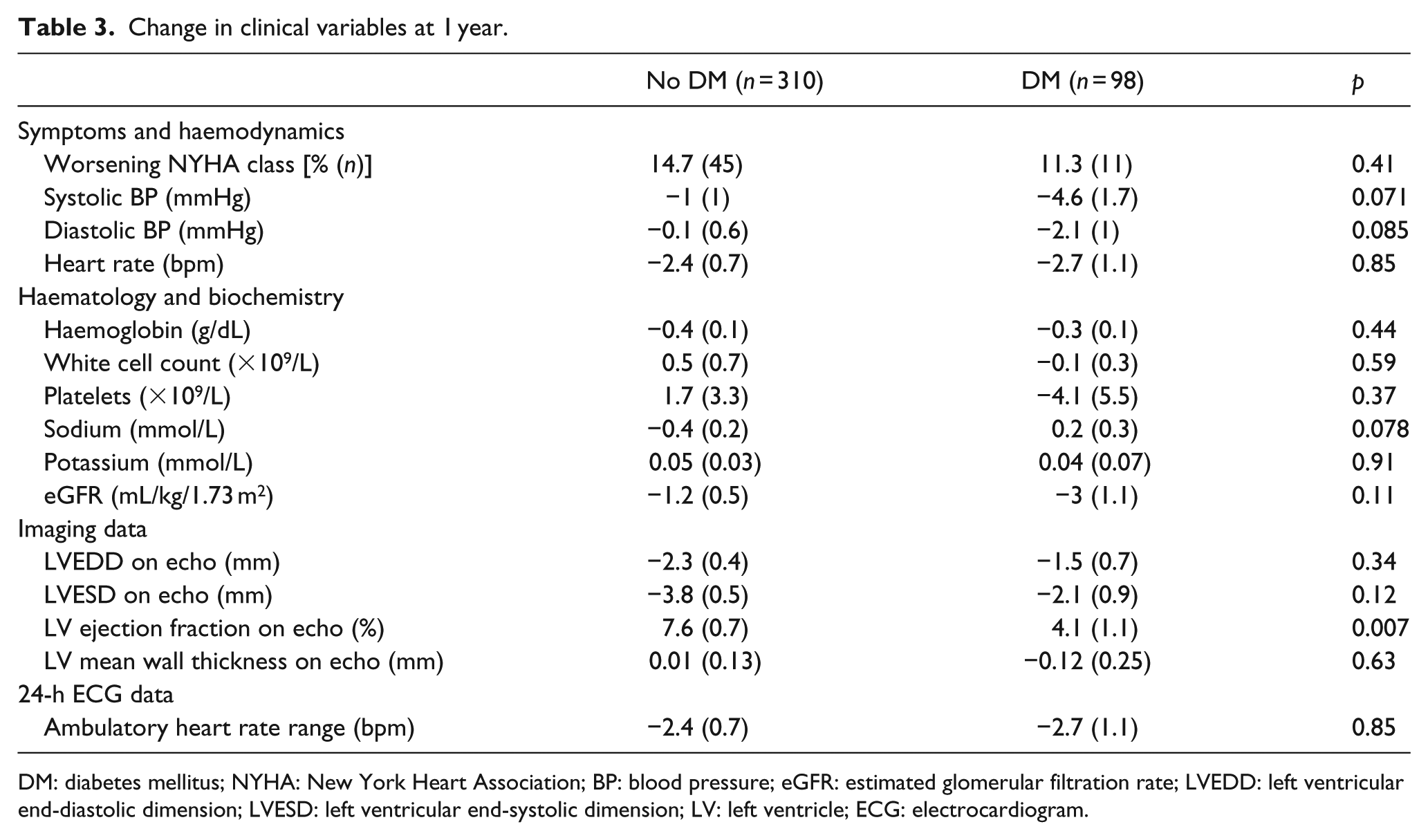
DM: diabetes mellitus; NYHA: New York Heart Association; BP: blood pressure; eGFR: estimated glomerular filtration rate; LVEDD: left ventricular end-diastolic dimension; LVESD: left ventricular end-systolic dimension; LV: left ventricle; ECG: electrocardiogram.
Change in treatment at 1 year.
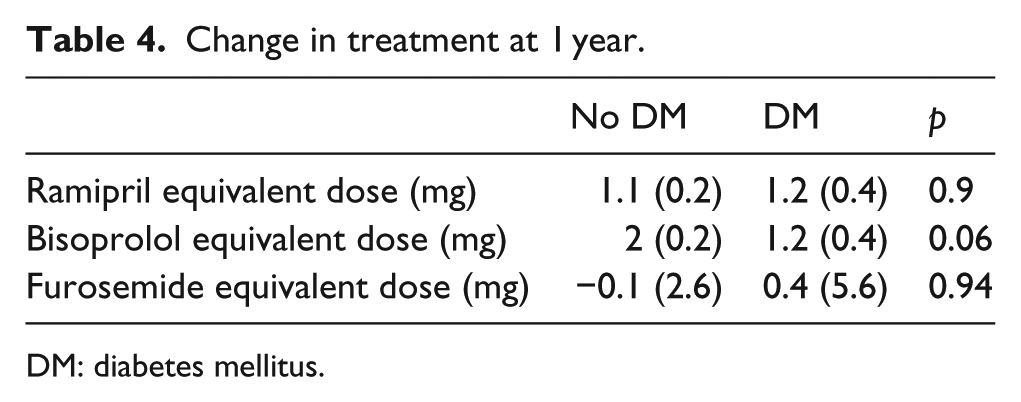
DM: diabetes mellitus.
During the first year of follow-up, 130 patients were hospitalised, of whom 88 had a principal cardiovascular cause of hospitalisation and 44 had a CHF-related hospitalisation. The relative risk associated with DM was 1.68 (95% confidence interval, 1.1−2.57; p = 0.016) for any hospitalisation, 2.12 (1.32−3.42; p = 0.002) for cardiovascular hospitalisation and 2.58 (1.38−4.83; p = 0.003) for CHF-related hospitalisation.
Mortality
After a mean follow-up period of 4.5 (SEM, 0.1) years, 250 (39.8%) deaths occurred; of these, there were 95 progressive CHF-related deaths, 43 sudden deaths, 14 other cardiovascular deaths, 92 non-cardiovascular deaths and 6 non-classifiable deaths (Figure 2). In univariate analysis, the relative risk associated with DM was 2.31 (1.78−2.98; p < 0.001) for overall death, 2.14 (1.4−3.27; p < 0.001) for progressive CHF death and 4.16 (2.28−7.57; p < 0.001) for sudden death. Multivariate analysis (Table 5) showed that DM is an independent risk factor for all-cause mortality in CHF when other established prognostic factors are included, although with a lower hazard ratio than in univariate analysis.
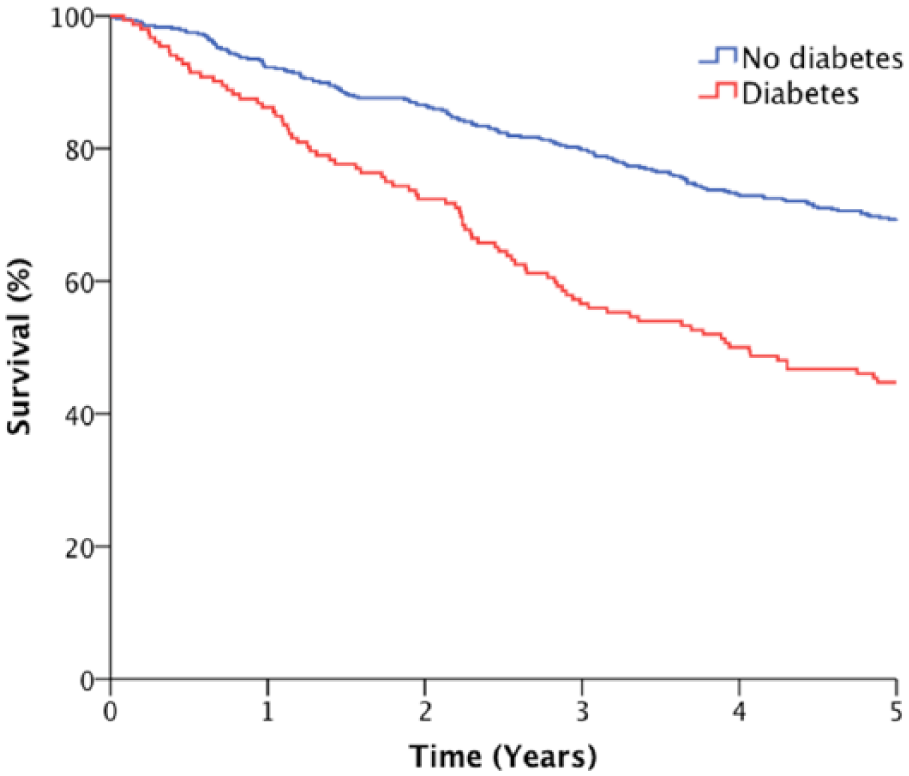
Kaplan–Meier survival analysis demonstrating reduced survival in people with diabetes (p < 0.001 by log-rank test).
Multivariate predictors of all-cause mortality.
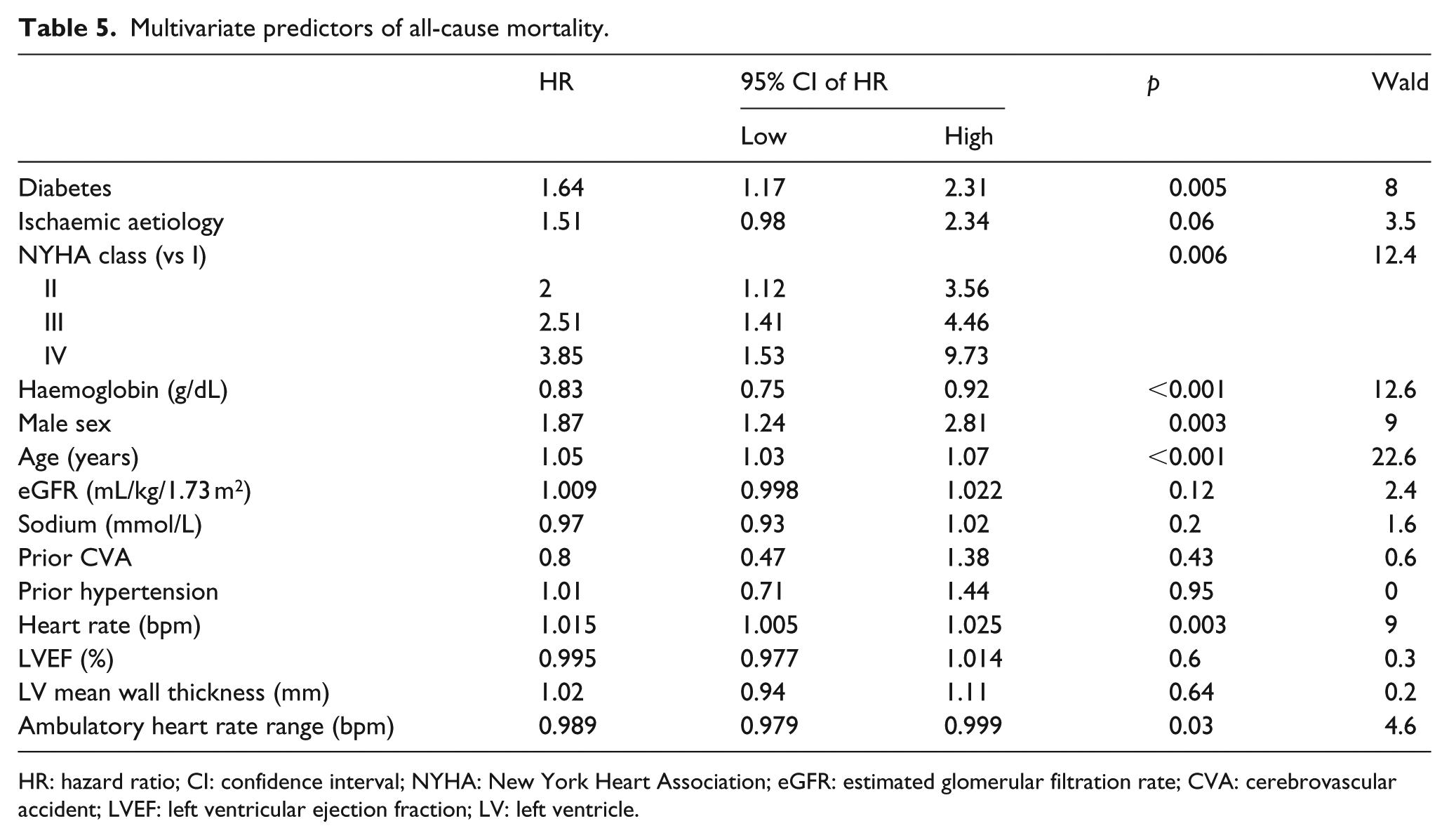
HR: hazard ratio; CI: confidence interval; NYHA: New York Heart Association; eGFR: estimated glomerular filtration rate; CVA: cerebrovascular accident; LVEF: left ventricular ejection fraction; LV: left ventricle.
Discussion
Our investigation provides a number of important and novel insights into how diabetes alters the HF phenotype, potentially offering explanations for the increased symptoms, hospitalisation rates and mortality of this group. First, we have shown that diabetes is more frequently associated with radiologic evidence of pulmonary oedema, in spite of greater LVEF at baseline. This may reflect greater myocardial thickness in those with diabetes, which can increase LV filling pressures, as is supported by greater PA SBP on ECHO. After 1 year of contemporary evidence-based HF therapy, improvements in LVEF were lower in those with diabetes, and they required larger diuretic doses. Second, we have shown that three distinct measures of heart rate variability exhibit unfavourable profiles in people with diabetes. Finally, we explored factors underlying our observations, showing HbA1c correlated with some markers of heart rate variability, although neither HbA1c nor SBP correlated with LV wall thickness in those with diabetes.
CHF phenotype in association with diabetes
Our results provide further evidence that DM has a profound effect on outcomes in patients with CHF. It is associated with a doubling of the risk of cardiovascular and CHF-related hospitalisation and in cardiovascular and all-cause mortality. Moreover, important differences in mode of death were apparent, with a particular excess in the risk of sudden cardiac death (SCD) in patients with diabetes. These findings are consistent with those from other studies, but we also add important complementary data, documenting associated differences in cardiac structure and function, and poorer response to optimal CHF management. Furthermore, our data demonstrate a higher prevalence of comorbidities in those with DM and CHF, and so the influence of multi-morbidity on the prognosis of these patients may be significant, as supported by other studies.7,13 However, our multivariate analysis supports DM as an independent risk factor for mortality in CHF even when other important comorbidities are taken into account.
Our data also highlight an adverse haematological and biochemical profile in HF patients with diabetes, including higher WCC, lower haemoglobin, lower serum sodium concentration and lower eGFR compared with non-DM patients; all are established adverse prognostic factors in CHF. 14 The elevated WCC may demonstrate the chronic low-grade inflammation known to occur in insulin-resistant states 15 whereas low haemoglobin may represent anaemia of chronic disease, or in the context of hyponatraemia may indicate fluid overload.
LV structure and function
Interestingly, despite poorer functional capacity at baseline, patients with DM had a significantly greater LVEF on ECHO, indicating that LV systolic impairment is less well tolerated in this group. This may, in part, be explained by their significantly increased mean LV wall thickness, which is often associated with greater filling pressures. Indeed, this assertion is supported by the elevated PA systolic pressures seen in the group with DM. Given the LVEDD and LVESD were similar in both groups of patients, the increased LV mean wall thickness in those with DM indicates concentric remodelling. Somewhat unexpectedly, LV mean wall thickness did not correlate with SBP in people with diabetes, although a weak, but significant, correlation was noted in people without diabetes. Furthermore, HbA1c did not correlate with LV mean wall thickness in patients with diabetes. Importantly, we present the novel finding that DM was associated with less favourable LV remodelling after contemporary evidence-based CHF treatment for 1 year, indicated by significantly less improvement in ejection fraction.
There is established evidence for abnormal LV remodelling in patients with DM after acute myocardial infarction (AMI). 16 Our data further inform the evidence base for adverse structural and functional remodelling as potential contributors to the poor outcomes seen in patients with DM and CHF. Moreover, our data indicate that current evidence-based treatments for CHF appear not to give comparable benefit to patients with DM in terms of improvement in ejection fraction or in clinical outcomes. The development of DM-specific therapies to limit pathological remodelling in these patients is therefore of considerable importance.
Electrocardiographic insights
Resting heart rate gives important prognostic information in people with CHF, and its reduction with pharmacotherapy is associated with improved outcomes.17,18 As such, it is interesting that the heart rate was lower in those with DM at baseline. Our findings from 24-h ECG analyses show important differences in heart rate variability and HRT in patients with diabetes. These indices reflect autonomic tone and have been associated with poorer outcomes in patients with CHF.19–21 The mechanism for this is unclear, although it may indicate a higher predisposition to SCD in these patients. Previous work from our group 2 has shown that SCD remains a common cause of mortality in DM patients with CHF (reviewed in Walker and Cubbon 22 ), in contrast to those without diabetes in whom this outcome has become far less frequent over the last 15 years. 12 In this study, we also note an excess of SCD in patients with diabetes, despite comparable use of beta-blockers and ICDs. The usefulness of HRT and SDNN is limited in CHF cohorts, given they cannot be derived in those with non-sinus rhythm, which may affect over 50% of this population. 23 AHRR, which can be derived irrespective of rhythm, provides complementary data, which is associated with mode-specific death and hospitalisation in those with CHF. 9 Interestingly, there was a weak, but significant, inverse correlation between AHRR and HbA1c in patients with diabetes, although the factors underlying this relationship remain unclear. Overall, our data support the presence of significant neurohumoral and electrophysiological abnormalities in patients with DM and CHF, which are likely to contribute to their excess risk of mortality and hospitalisation. Further understanding of the mechanisms which drive these abnormalities along with improved screening tools to detect those at risk (particularly of SCD) will aid the development of DM-specific treatment strategies for CHF.
Limitations
While our work provides important insights into the factors that may contribute to the disparity in outcomes between CHF patients with and without diabetes, it is important to acknowledge its limitations. The cohort size is relatively small in comparison with large-scale randomised clinical trial populations. However, this has allowed us to phenotype the study population more extensively than would normally be possible in larger trials and include data such as HRT and SDNN in our analyses. The observational nature of our study means we are unable to show causation in the associations we have demonstrated and therefore further work is warranted to define those factors which specifically contribute to poorer outcomes in CHF patients with diabetes. Unfortunately, our data on LV diastolic function are limited as patients were recruited before tissue Doppler imaging techniques were widely available. Although we present data on E wave velocity and E/A ratio, a high proportion of this cohort have atrial fibrillation or pacemakers, meaning these individuals are excluded from this analysis. Moreover, E/A ratio may be misleading in the case of pseudonormalisation noted in some cases of diastolic dysfunction, which may account for the observed lack of difference between patients with and without DM. Thorough characterisation of diastolic function in future studies will be important to investigate its prevalence and impact on mortality in patients with DM and CHF. Finally, as our cohort assessed patients with an ejection fraction of ⩽45%, we have no data for those with CHF with preserved ejection fraction.
Conclusion
DM is associated with important differences in the structural and functional remodelling associated with CHF, which may explain its adverse prognostic impact. Further work is needed to define the mechanisms underlying these observations and guide the development of DM-specific CHF therapies.
Footnotes
Acknowledgements
The authors would like to thank Spacelabs Healthcare Ltd (UK) for the free and unrestricted donation of a development ECG analysis system used to determine the HRV parameters in this project. A.M.N.W. and P.A.P. are supported by British Heart Foundation (BHF) Clinical Research Training Fellowships. R.M.C. is a BHF Intermediate Clinical Research Fellow and M.T.K. is a BHF Professor of Cardiology. K.K.W. is a National Institute for Health Research Clinician Scientist.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was funded by the British Heart Foundation: project grant PG/08/020/24617.
