Abstract
Aim:
To compare the adhesion, migration and endothelial differentiation potential of peripheral blood-derived mononuclear cells (PBMCs) obtained from drug-naive normal glucose tolerance (NGT) and impaired glucose tolerance (IGT) Asian Indian men.
Methods:
Based on the 75-g oral glucose tolerance test, 30 NGT and 31 IGT subjects were recruited into the study. PBMCs were isolated from fasting blood using histopaque density gradient centrifugation. Isolated PBMCs were analysed for their ability to adhere to extracellular matrices, incorporation into tubular structures formed by matured endothelial cells and differentiation into endothelial cells upon 7-day culture in endothelial-specific growth medium.
Results:
PBMCs obtained from IGT subjects exhibit poor adherence to fibronectin and reduced incorporation into tubular structures. Migration towards stromal cell-derived factor-1α (SDF-1α) in a trans-well filter assembly was also reduced for these cells. Semi-quantitative reverse transcription polymerase chain reaction (RT-PCR) analysis revealed decreased expression of CXCR4 and β2 integrin and increased expression of arginase II in IGT subjects. No differences were observed with regard to endothelial differentiation; however, cultured PBMCs of IGT subjects had decreased intracellular nitric oxide (NO) production.
Conclusion:
In pre-diabetic, Asian Indian men, PBMCs exhibit defective migration and homing potential.
Introduction
Peripheral blood-derived mononuclear cells (PBMCs) exhibit angiogenic properties and are an attractive option for autologous therapeutic neovascularization. Their infusion improves collateral circulation in ischaemic tissue.1,2 The earliest study identifying angiogenic potential in PBMCs was performed by Asahara et al., 3 in which the authors demonstrated that CD34-enriched PBMC fraction promotes neovascularization. Subsequently, other studies identified haematopoietic stem cells, haemangioblasts, endothelial progenitor cells (EPCs) and monocytes present in circulating mononuclear cell pool as the responsible angiogenic mediators.4–6 Additionally, PBMCs mediate endothelial repair by differentiating into endothelial-like cells upon ex vivo culture on fibronectin-coated tissue culture dishes in the presence of endothelial growth medium (EGM). 4 In the physiological setting, upon receipt of appropriate cue such as stromal cell-derived factor-1α (SDF-1α), these mediators mobilize towards the injured site for repair. 7 They also modulate the function of pre-existing endothelial cells through the secretion of paracrine factors. 8
Insulin resistance, which precedes the onset of overt hyperglycaemia in diabetes, is associated with endothelial dysfunction, even in the absence of traditional cardiovascular disease (CVD) risk factors. 9 Intriguingly, Asian Indians are more prone to CVD as they exhibit increased insulin resistance. 10 Although it is known that insulin resistance decreases the circulating counts of EPCs, 11 it is not known whether impaired glucose metabolism observed during pre-diabetes reduces the vasculogenic potential of PBMCs, particularly in Asian Indians. Hence, the objective of this study was to compare the adhesion, migration, tubule incorporation and endothelial differentiation potential of PBMCs obtained from drug-naive healthy and impaired glucose tolerance (IGT) Asian Indian men.
Materials and methods
K2-EDTA vacutainers for blood collection (Cat: 367863), fibronectin for cell culture (Cat: 356008), growth factor-reduced phenol red-free matrigel (Cat: 356231), Anti-human CD31-FITC (Cat: 555445), isotype control mouse IgG1k-FITC (Cat: 555748), Annexin V-PE (Cat: 556422) and 7-amino-actinomycin D (7-AAD, Cat: 559925) were purchased from Becton Dickinson (BD) Biosciences, USA. EGM-2 Bullet kits (Cat: CC-4176) were purchased from Lonza, Switzerland. Human FcR blocking reagent (Cat: 130-059-901), Anti-human CD34-FITC (Cat: 130-081-001) and isotype control mouse IgG2a-FITC antibody (Cat: 130-091-837) were purchased from Miltenyi Biotec, Germany. Anti-PECAM-1 antibody (Cat: 3528) was from Cell Signaling Technology, USA. Other antibodies were from Santa Cruz, USA. Tissue culture dishes and trans-well thin inserts with 8.0 µm pore size were from Costar Corning, USA. All reverse transcription polymerase chain reaction (RT-PCR) primers were purchased from Integrated DNA Technologies (IDT), USA. Reagents required for RNA isolation (Trizol), diamino fluorescein-2 diacetate (DAF-2DA), PKH26,
Study group, inclusion and exclusion criteria
The study was approved by the Indian Institute of Technology Madras (IIT Madras) institutional ethics committee, as required by the Indian Council of Medical Research, Government of India. Informed written consent was obtained from all study subjects. Males aged between 25 and 45 years were recruited for this study through Dr Mohan’s Diabetes Specialties Centre, a tertiary diabetes centre in Chennai, India. They were screened based on the 75-g oral glucose tolerance test (OGTT). Subjects who had their fasting blood glucose values in the range of 5.6–6.9 mmol/L were identified as pre-diabetics with impaired fasting glucose (IFG), and those with post-load glucose levels in the range of 7.8–11 mmol/L were classified as IGT. All the pre-diabetic subjects recruited into the study exhibited IGT, either with or without IFG. The next day, 25 mL of fasting blood was collected from subjects for isolation of PBMCs and other biochemical estimations. The following exclusion criteria were applied: diabetes, established coronary artery disease (CAD) or CVD, any form of medication, smoking, acute infections or immunological disorders, cancer and recent surgery in the past 10 months.
Estimation of anthropometric and biochemical parameters
Anthropometric measurements like age, height, weight, body mass index (BMI) and waist-to-hip circumferences were carried out for all the study subjects. The blood pressure was recorded using a standardized automatic blood pressure monitor in the right arm in the sitting position. All the biochemical estimations were carried out in the Biochemistry laboratory of the associated hospital as described previously. 12 The intra- and inter-assay coefficients of variation for the biochemical assays ranged between 3.1% and 7.6%, respectively. The intra- and inter-assay coefficients of variation of HbA1c were less than 10%.
Isolation and culture of PBMCs
PBMCs were isolated using the histopaque density gradient centrifugation method (Sigma-Aldrich). Following three washes with 1× phosphate buffered saline (PBS) to remove contaminating platelets, the purity of the isolated fraction was confirmed through CD45-PerCP-based (BD Biosciences) flow cytometry, and it was found to be greater than 95% (data not shown). Isolated fractions were then cultured on fibronectin-coated (25 µg/mL) tissue culture dishes in EGM-2 bullet kit medium [supplemented with hydrocortisone, vascular endothelial growth factor (VEGF), insulin-like growth factor 1 (IGF-1), epidermal growth factor (EGF), fibroblast growth factor-basic (FGF-B), ascorbic acid, heparin and serum]. First medium change was done on the fourth day of culture, followed by medium change on the sixth day. On the seventh day of culture, cells were assessed by staining for dil-Ac-LDL (10 µg/mL) and fluorescein-conjugated Ulex-europaeus agglutinin I (10 µg/mL). The expression of endothelial markers – endothelial nitric oxide synthase (eNOS) and von Willebrand factor (vWF) – was confirmed by immunofluorescence.
Immunofluorescence
Cells were washed thrice with 1× PBS, followed by fixing with 4% paraformaldehyde and permeabilization with 0.25% Triton X. Prior to the addition of respective primary antibodies as per manufacturer’s instruction, cells were blocked with 5% serum in PBS. Corresponding fluorescein isothiocyanate (FITC)–conjugated secondary antibodies and 4′,6-diamidino-2-phenylindole (DAPI) (1 µg/mL) were utilized for visualizing stained cells, and appropriate negative controls were employed. Images were analysed using ImageJ software from National Institutes of Health (NIH). For each well, at least six field views were analysed.
Adhesion assay
PBMCs were isolated from normal glucose tolerance (NGT) and IGT subjects using the method described above. Isolated cells were labelled with 4 µmol/L PKH26 Red (Sigma-Aldrich) for 5 min as per manufacturer’s instruction and were washed twice with 1× PBS. For the adhesion assay, 2 × 105 labelled cells were seeded onto wells coated with fibronectin (25 µg/mL), gelatin (0.2% w/v) or collagen (0.01% w/v) in EGM in the absence of serum and were allowed to adhere for 3 h. Non-adherent cells were washed with 1× PBS twice. Images were taken using an Olympus IX51 fluorescence microscope. Each sample was assessed in triplicate, and a minimum of four field views were analysed per well. Data are represented as average number of cells adhered per field view.
Trans-well migration assay
Trans-well migration filters of 8 µm pore size (Corning) were coated with fibronectin on both sides at 37 °C in 5% CO2 for 1 h. Then, 3 × 105 PBMCs were seeded on the upper chamber, and recombinant human SDF-1α (100 ng/mL) was added to the basal medium in the lower chamber. Cells were incubated for 12 h at 37 °C in 5% CO2. Following incubation, the cells on the upper side of the membrane were carefully removed using a sterile cotton swab, and the cells that migrated to the lower side of the membrane were fixed with 4% paraformaldehyde and stained with DAPI (1 µg/mL) for image acquisition. The lower side of the membrane was imaged in four different field views. Data are represented as the average number of cells migrated per field view.
Matrigel assay
Desired wells in a 96-well plate were coated with matrigel (BD Biosciences, USA) and pre-incubated at 37 °C in 5% CO2 for 1 h prior to cell seeding. Freshly isolated PBMCs were stained with PKH26 red dye. Stained PBMCs were mixed in equal proportion with endothelial ECV304 cells; 2 × 104 cells were seeded on matrigel-coated wells and were incubated at 37°C for 18 h in EGM-2 medium supplemented with VEGF (50 ng/mL) prior to visualizing for tube formation in 4× objective. Both phase contrast and fluorescence images were obtained. The number of loops, tubes, length of the tubes and average incorporation of PBMCs per tube were calculated using ImageJ software.
Detection of nitric oxide
Intracellular nitric oxide (NO) production on seventh day of culture in EGM was analysed using the DAF-2DA method. DAF-2DA is a cell-permeable dye which fluoresces upon binding to NO. Adherent cells were gently washed twice with PBS and then incubated with
Flow cytometry
Following isolation with density gradient centrifugation, PBMCs were washed thrice with 1× PBS and were blocked with 2% foetal bovine serum and FcR blocking reagent for 30 min at 4 °C. Cells were stained with FITC-conjugated test antibodies and their respective isotype controls, as per manufacturer’s instructions, and were fixed with 4% paraformaldehyde. Acquisition was done for 5 × 105 events in FACS Canto, and data were analysed using FlowJo software version 7.2.5 (Tree Star Inc., USA). The FACS operator was blinded for the clinical status of the study subjects.
Semi-quantitative RT-PCR
Reverse transcription was performed with 1 µg DNase-treated RNA for 3 h. The amount of complementary DNA (cDNA) taken for PCR was 200 ng, and the PCR conditions were standardized based on the melting temperature of the primers and the amplicon size. Relative amount of target messenger RNAs (mRNAs) was normalized to Ribosomal Protein L13a (RPL) mRNA as internal control.
Statistical analysis
Clinical data are expressed as mean ± standard error of mean (SEM). For the functional and gene expression studies, data are represented as mean ± SEM for a minimum of 10 subjects in each group unless specified otherwise. Each experiment was done at least six times, with each experiment having at least one NGT and one IGT sample each time. Kolmogorov–Smirnov’s (KS) test was employed to determine the normality of distribution. Comparisons between two groups were performed by the unpaired Student’s t-test or non-parametric analysis of variance (ANOVA) (Kruskal–Wallis test) and Dunn’s multiple comparisons test. Spearman’s rank correlation analysis was performed between biochemical parameters and NO index. Statistical significance was accepted if the null hypothesis could be rejected at p ≤ 0.05. All analyses were done using Windows-based SPSS statistical software (version 15.0).
Results
Characteristics of study population
Based on the 75-g OGTT, 30 NGT and 31 IGT subjects were identified. Anthropometric, biochemical and other clinical profiles of study subjects are listed in Table 1. The average age and weight were higher for IGT compared to the NGT (control) group. Waist circumference and BMI were significantly higher for the IGT group. Fasting and post-load glucose and insulin values were also higher for the IGT group, but these values were not in the clinical range of hyperglycaemia and/or hyperinsulinaemia. Additionally, the subjects in the IGT group were insulin resistant based on the homeostasis model assessment–insulin resistance (HOMA-IR) values. Levels of high-sensitivity C-reactive protein (hs-CRP) for IGT men were above 3.0 mg/L, placing them at high risk of inflammation and peripheral artery disease. 13 However, there were no significant differences in the mean blood pressure values and lipid profile (data not shown) between the two groups. Haematological counts were within the normal range for both the groups (data not shown).
Anthropometric, biochemical and clinical characteristics of study subjects.
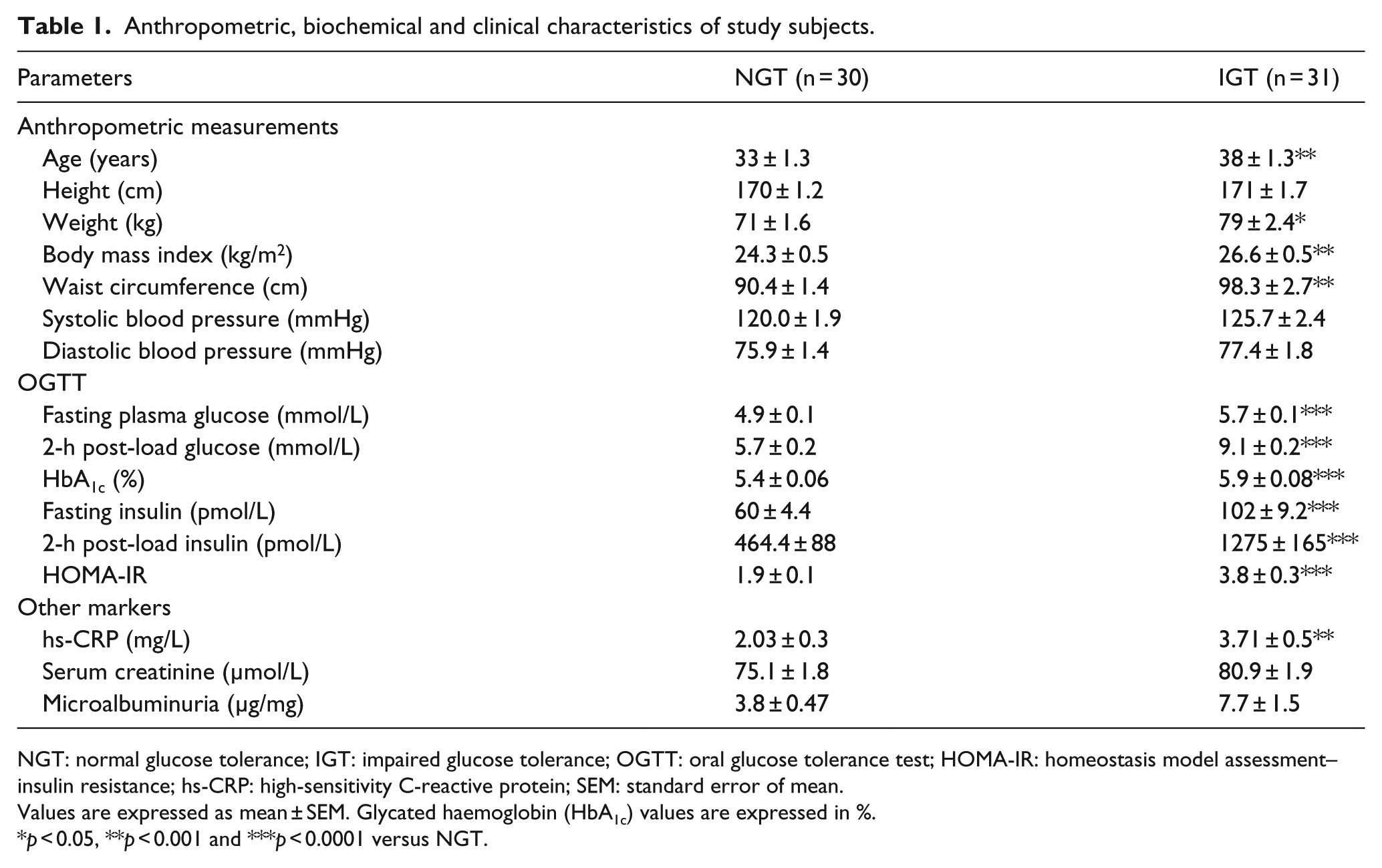
NGT: normal glucose tolerance; IGT: impaired glucose tolerance; OGTT: oral glucose tolerance test; HOMA-IR: homeostasis model assessment–insulin resistance; hs-CRP: high-sensitivity C-reactive protein; SEM: standard error of mean.
Values are expressed as mean ± SEM. Glycated haemoglobin (HbA1c) values are expressed in %.
p < 0.05, **p < 0.001 and ***p < 0.0001 versus NGT.
Matrigel tube formation assay
In order to capture pathological events existent in the study subjects, and not to lose them upon culture, the functional and molecular assays with regard to adhesion, migration, tubule incorporation and gene expression changes were performed with freshly isolated PBMC fraction. On the other hand, to determine the endothelial differential potential of PBMCs, the isolated PBMCs were cultured for 7 days in endothelial differentiation medium followed by assessment for acquisition of endothelial markers.
To determine integration of PBMCs into tubular structures formed by endothelial cells (ECV304), PKH26-labelled PBMCs from either of the study groups were co-cultured with ECV304 cells within matrigel in the presence of VEGF-supplemented (50 ng/mL) EGM. This led to the formation of extensive tubular network for cells obtained from the NGT subjects (Figure 1(a)). Fluorescent tagging with PKH26 enabled us to distinguish incorporated PBMCs from endothelial cells. It should be noted that tubule formation by ECV304 cells was enhanced in the presence of VEGF, while PBMCs on their own failed to form tubules on matrigel (data not shown). As seen in Figure 1(b) and (c), not only were the number and length of tubes decreased for IGT subjects but also the number of PBMCs incorporated per tube length was reduced (Figure 1(d)), thereby implicating defective tubule incorporation or invasion ability. This decrease was not due to apoptosis in freshly isolated PBMCs from IGT subjects as seen in Figure 1(e). Even the number of viable cells between the two groups was similar. Apoptosis and viability were assessed through Annexin V- and 7-AAD-based flow cytometry, respectively.
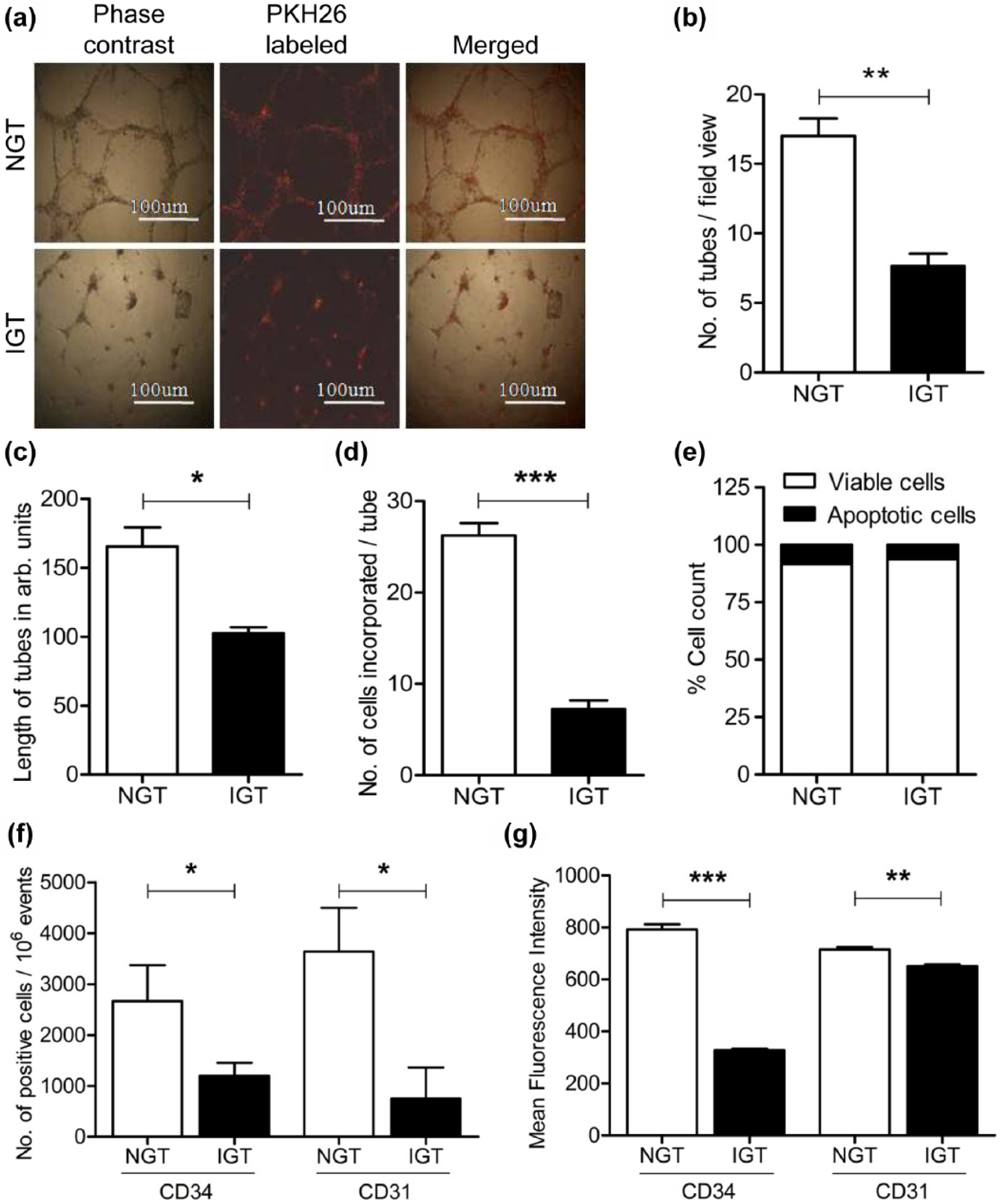
Vasculogenic potential of peripheral blood–derived mononuclear cells (PBMCs). (a) Representative image depicting tube formation by ECV304 cells in the presence of PBMCs from NGT and IGT subjects in response to VEGF (50 ng/mL), with scale bar corresponding to 100 µm. (b–d) Bar graphs summarizing data of number of tubes, length of tubes and number of PBMCs incorporated per tube, respectively, as mean ± SEM. (e) Bar graph representing percentage of viable and apoptotic cells in isolated PBMC fraction from the study groups. (f and g) Bar graphs summarizing cell counts and mean fluorescence intensities for CD34+ and CD31+ cells, respectively, from 10 NGT and 10 IGT subjects (magnification: 10×.
Flow cytometry analysis for CD34 and CD31 expression
Platelet endothelial cell adhesion molecule-1 (PECAM-1) or CD31, expressed constitutively on the surface of mononuclear cells, is necessary for hematosphere formation by isolated PBMCs, 14 for endothelial differentiation of EPCs and for secretion of angiogenic growth factors from them, 15 while the presence of CD34+ cells in circulating blood inversely correlates with Framingham risk score and effectively predicts cardiovascular events in follow-up studies. 16 We hence determined the presence of CD31+ and CD34+ cells in isolated PBMCs from study subjects. In agreement with a previous study, 11 we also observed significant reduction in levels of CD34+ and CD31+ cells in the PBMC fraction of IGTs compared to the control group (Figure 1(f)). Even the mean fluorescence intensity (MFI) for these two markers was reduced for IGT subjects (Figure 1(g)).
Migration in response to SDF-1α (CXCL12)
Chemotaxis of progenitors towards an increasing gradient of SDF-1α in blood promotes mobilization of stem cells from bone marrow into circulation.7,17 The cognate receptor for SDF-1α, namely, CXCR4, is expressed on mononuclear cells and endothelial cells to assist angiogenesis. We next sought to determine the influence of IGT on the migratory potential of isolated PBMCs towards SDF-1α. A trans-well migration assay was performed to assess the same. The number of cells migrating towards SDF-1α was assessed as number of cells reaching the underneath surface of the coated filter in the trans-well assembly. There was a significant increase in the migration of PBMCs from NGT subjects across the membrane in response to human SDF-1α (100 ng/mL) (Figure 2(a) and (b)). This migratory potential was, however, blunted for PBMCs isolated from IGT subjects. Moreover, the expression of CXCR4 was markedly reduced in PBMCs from IGT subjects (Figure 2(c)).
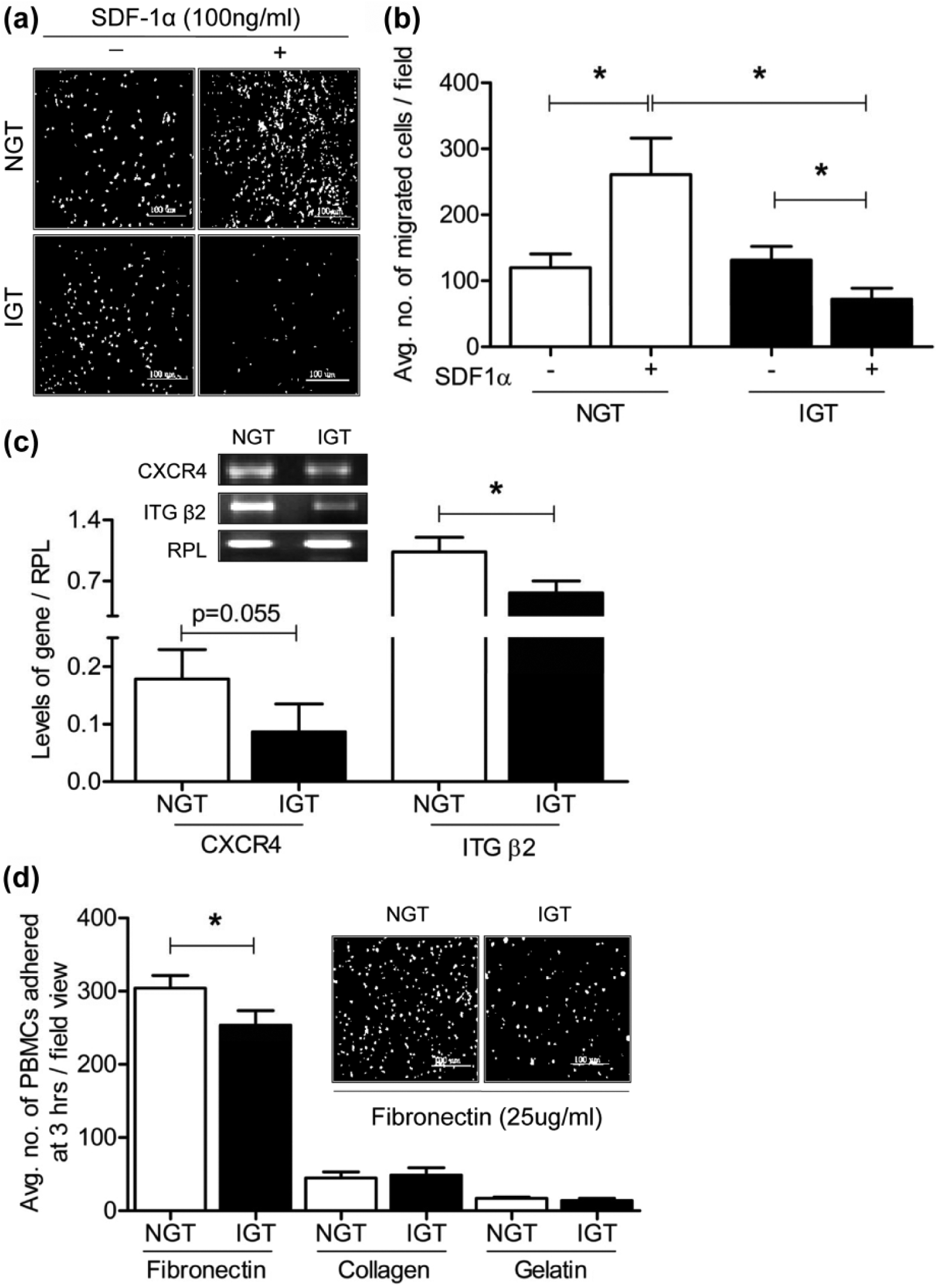
Migration, adhesion and integrin expression in isolated mononuclear cells. (a) Representative image of cells from the two study groups (n = 10 in each group) migrated to the underneath surface of the fibronectin-coated polycarbonate filter in a trans-well filter assembly in response to SDF-1α (100 ng/mL), with scale bar corresponding to 100 µm. (b) Bar graph summarizing data for five independent experiments each performed with a minimum of one NGT and one IGT subject. (c) RT-PCR analysis for the expression of CXCR4 and β2 integrin in PBMCs of NGT (n = 10) and IGT (n = 10) subjects. Data are represented as mean ± SEM. (d) Bar graph summarizing adherence to different matrices for n = 10 subjects in each group. Inset is a representative image depicting adherence of PKH26-labelled PBMCs onto fibronectin (magnification: 10×.
Adhesion to extracellular matrix proteins and integrin expression
Since PBMCs from IGT subjects exhibited reduced migration through fibronectin-coated trans-well filters, as well as incorporation into tubules, we set out to determine adhesion potential of these cells onto different matrices. As seen in Figure 2(d), adhesion to fibronectin and not to gelatin or collagen was significantly attenuated for study subjects. Among the integrins tested for change in gene expression (α4, α5, αV, β1, β2 and β3), the expression of β2 (Figure 2(c)) was consistently and significantly reduced in IGT subjects. Despite our best efforts, we failed to detect the expression of integrins α4, α5 and αV in our study subjects, although the RT-PCR worked for human umbilical vein–derived endothelial cells (HUVECs), which were used as positive control (data not shown).
Assessment of endothelial differentiation and NO index in cultured cells
Endothelial differentiation potential of isolated cells upon culture in EGM-2 was next determined. On the seventh day of culture, they were assessed for Ulex-lectin staining and uptake of dil-Ac-LDL, in addition to assessment for the expression of vWF, PECAM-1 and eNOS. Cells from both the study groups took up dil-Ac-LDL and Ulex-lectin effectively (data not shown). The percentage of cells positive for CD31 and vWF was marginally lower (10%–15%) for IGT subjects (data not shown). The ability of differentiated mononuclear cells to produce NO in the presence and absence of known NOS inhibitor, that is, L-NG-nitro-arginine methyl ester (L-NAME), was determined through the DAF-2DA method. Average NO index per cell at day 7 of culture was significantly lower for IGT subjects (Figure 3(a) and (b)), although all the cultured cells effectively expressed eNOS from both the study groups (Figure 3(c)). It should be noted that the expression of arginase II, the known competitor of eNOS, was significantly increased in PBMCs derived from IGT subjects (Figure 3(d)). Spearman’s rank correlation analysis of the NO index in cultured cells, with observed clinical parameters, indicated negative correlation between NO index and features of metabolic imbalance, that is, waist circumference (ρ = −0.413, p = 0.012), BMI (ρ = −0.350, p = 0.036), HOMA-IR (ρ = −0.681, p = 0.001), HbA1c (ρ = −0.612, p = 0.001), fasting and post-load glucose (ρ = −0.598, p = 0.001; ρ = −0.753, p = 0.001, respectively) and insulin (ρ = −0.536, p = 0.001 for fasting; ρ = −0.592, p = 0.001 for 2 h post-load) values. It also negatively correlated with circulating levels of hs-CRP (ρ = −0.551, p = 0.01).
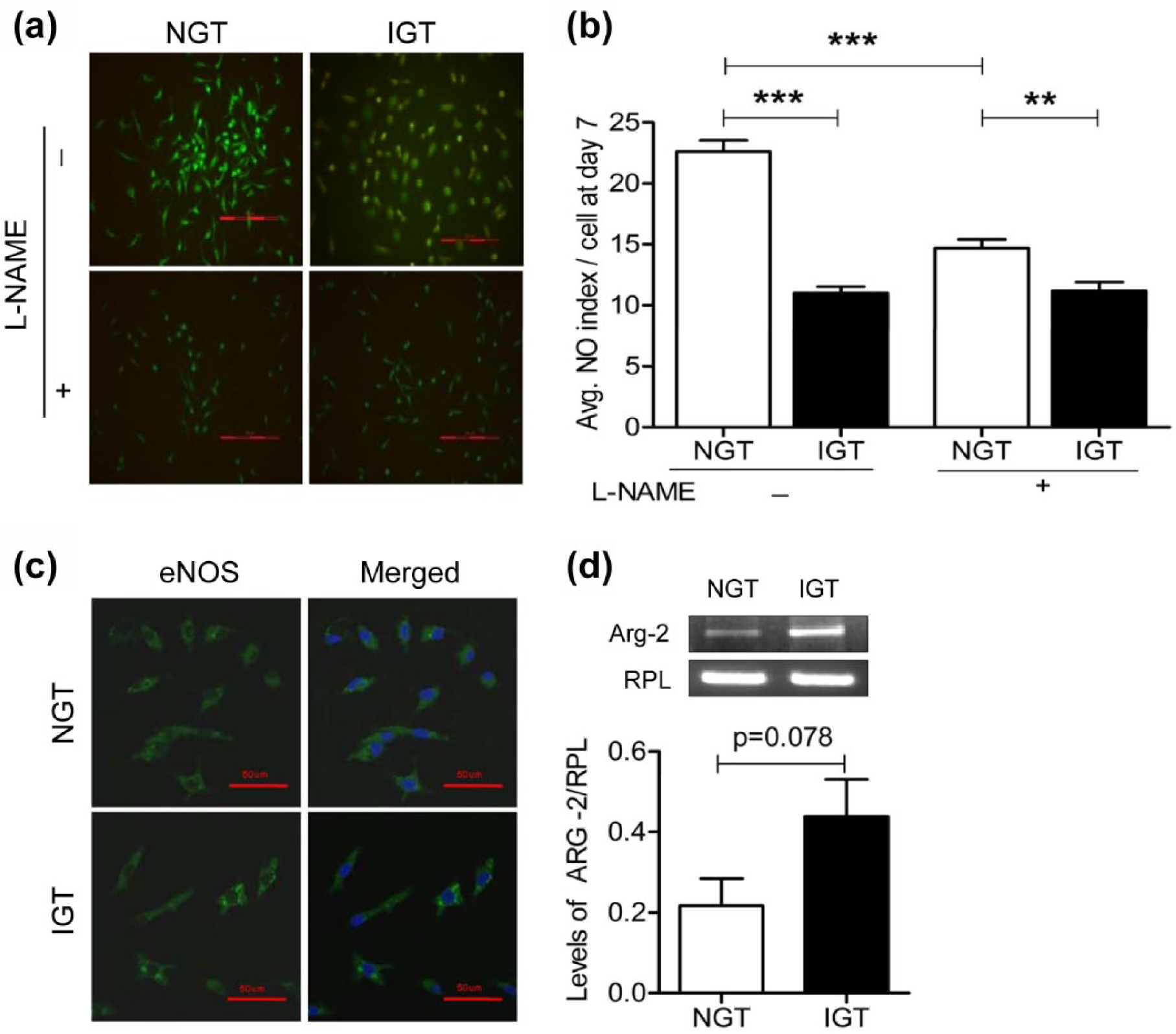
Comparison of NO index, eNOS immunofluorescence and arginase II expression. (a) Representative image depicting NO levels through DAF-2DA fluorescence. (b) Bar graph summarizing average NO index per cell for NGT (n = 18) and IGT (n = 18) subjects. (c) Immunofluorescence staining for eNOS. (d) RT-PCR analysis of arginase II from 10 subjects in each group. Data are represented as mean ± SEM [magnification: 10× with scale bar corresponding to 100 µm for (a) and 20× with scale bar corresponding to 50 µm for (c)].
Discussion
Numerous studies with type 2 diabetic mice and humans have demonstrated functional aberrations in EPCs;18–20 however, it has not been possible to delineate the contribution of insulin resistance vis-à-vis other associated metabolic abnormalities in this anomaly. Although recent studies do identify decreased circulating levels of EPCs in insulin-resistant men of South Asian Origin 21 and it is known that insulin resistance does corrode the endothelium through decreased bioavailability of NO, how it affects the functionality of EPCs remains largely elusive.
SDF-1α–CXCR4 axis, integrins and NO are indispensable for mobilization, survival, homing and vasculogenic abilities of EPCs.18,19,22 For instance, use of exogenous NO donors or eNOS enhancer AVE9488 improves EPC-mediated neovascularization. 23 Aberrations in SDF-1α–CXCR4 axis attenuate the process of endothelial repair since SDF-1α signalling promotes eNOS activity. 24 Hence, increased expression of arginase II in PBMCs as well as decreased expression of NO in early EPCs, as seen in this study, could delay the process of EPC homing and endothelial repair in insulin-resistant individuals Additionally, integrin-mediated signalling enhances SDF-1α-induced mobilization and homing of progenitors. 25 In fact, ischaemia-induced collateral vessel formation is significantly blocked in β2-deficient mice 26 due to defective adherence of progenitors to extracellular matrix (ECM) proteins. The decreased expression of β2 integrin could thus reduce the recruitment of progenitors to sites desiring endothelial repair during insulin resistance. Unfortunately, the presence of inflammatory mediators such as CRP may further compound this problem as they are reported to decrease proliferation and functionality of EPCs. 27 Intriguingly, we observed that the NO index from cultured cells of IGT subjects negatively correlated with features of metabolic imbalance as well as hs-CRP. In light of these observations, it is imperative to determine molecular mechanisms governing aberrant expression of key players in PBMCs, be it CXCR4, β2 integrin or NO, during glucose intolerance in our future studies.
This study possesses certain limitations. For instance, assessment of endothelial function in study subjects was not performed. Given that IGT is still not a recognized disease state, recruiting sufficient subjects to carry out mechanistic studies was beyond the scope of this work. Additionally, the ethical constraints limited the volume of blood that could be collected to carry out detailed studies. Merits of the study include functional characterization of PBMCs from clinically matched young drug-naive subjects. To the best of our knowledge, this study is the first to address and identify functional impairments in PBMCs of glucose-intolerant Asian Indian men in the absence of other co-morbid conditions such as hypertension and dyslipidaemia. Given that lifestyle and pharmacological interventions which improve insulin resistance also improve endothelial and EPC function,18,19 exploring complex mechanistic links between insulin resistance and EPC-mediated endothelial repair is the need of the hour for effective management of CVDs.
Footnotes
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This work was supported by the Department of Biotechnology (DBT) Government of India sponsored ‘Senior IYBA’ fellowship to Dr Madhulika Dixit.
